Assessing Amide Molecule Dynamics for Extended Stability
FEB 28, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Amide Stability Background and Research Objectives
Amide bonds represent one of the most fundamental chemical linkages in biological systems, serving as the backbone of proteins and forming critical structural components in numerous pharmaceutical compounds. These covalent bonds, characterized by the connection between a carbonyl carbon and an amino nitrogen, exhibit unique stability properties that have made them indispensable in drug design and biomolecular engineering. However, the dynamic behavior of amide molecules under various environmental conditions presents significant challenges for maintaining long-term stability in therapeutic applications.
The pharmaceutical industry has increasingly recognized that amide bond stability directly impacts drug efficacy, shelf life, and safety profiles. Traditional approaches to stability assessment have relied primarily on accelerated aging studies and static analytical methods, which often fail to capture the complex molecular dynamics that govern degradation pathways. Recent advances in computational chemistry and molecular simulation techniques have opened new avenues for understanding amide molecule behavior at the atomic level, enabling more precise predictions of stability under extended storage and physiological conditions.
Current research efforts focus on developing comprehensive methodologies to assess amide molecule dynamics through integrated experimental and computational approaches. These investigations aim to establish predictive models that can accurately forecast stability performance across diverse chemical environments, temperature ranges, and time scales. The challenge lies in bridging the gap between molecular-level dynamics occurring on nanosecond timescales and macroscopic stability phenomena that manifest over months or years.
The primary objective of this research initiative centers on establishing robust analytical frameworks for evaluating amide molecule dynamics to predict and enhance extended stability. This encompasses developing advanced simulation protocols that can accurately model conformational changes, hydrolysis susceptibility, and environmental stress responses in amide-containing compounds. Additionally, the research aims to identify key molecular descriptors that correlate with long-term stability, enabling rational design strategies for more stable pharmaceutical formulations.
A secondary objective involves creating standardized testing protocols that integrate real-time molecular dynamics data with traditional stability measurements. This approach seeks to reduce the time and cost associated with conventional stability studies while providing deeper insights into degradation mechanisms. The ultimate goal is to establish predictive capabilities that can guide formulation scientists in optimizing amide-based drug candidates during early development stages, thereby reducing late-stage failures and improving overall success rates in pharmaceutical development.
The pharmaceutical industry has increasingly recognized that amide bond stability directly impacts drug efficacy, shelf life, and safety profiles. Traditional approaches to stability assessment have relied primarily on accelerated aging studies and static analytical methods, which often fail to capture the complex molecular dynamics that govern degradation pathways. Recent advances in computational chemistry and molecular simulation techniques have opened new avenues for understanding amide molecule behavior at the atomic level, enabling more precise predictions of stability under extended storage and physiological conditions.
Current research efforts focus on developing comprehensive methodologies to assess amide molecule dynamics through integrated experimental and computational approaches. These investigations aim to establish predictive models that can accurately forecast stability performance across diverse chemical environments, temperature ranges, and time scales. The challenge lies in bridging the gap between molecular-level dynamics occurring on nanosecond timescales and macroscopic stability phenomena that manifest over months or years.
The primary objective of this research initiative centers on establishing robust analytical frameworks for evaluating amide molecule dynamics to predict and enhance extended stability. This encompasses developing advanced simulation protocols that can accurately model conformational changes, hydrolysis susceptibility, and environmental stress responses in amide-containing compounds. Additionally, the research aims to identify key molecular descriptors that correlate with long-term stability, enabling rational design strategies for more stable pharmaceutical formulations.
A secondary objective involves creating standardized testing protocols that integrate real-time molecular dynamics data with traditional stability measurements. This approach seeks to reduce the time and cost associated with conventional stability studies while providing deeper insights into degradation mechanisms. The ultimate goal is to establish predictive capabilities that can guide formulation scientists in optimizing amide-based drug candidates during early development stages, thereby reducing late-stage failures and improving overall success rates in pharmaceutical development.
Market Demand for Stable Amide-Based Products
The pharmaceutical industry represents the largest market segment for stable amide-based products, driven by the critical need for drug molecules that maintain their therapeutic efficacy throughout extended shelf lives. Amide bonds form the backbone of numerous pharmaceutical compounds, including peptide-based therapeutics, protein drugs, and small molecule pharmaceuticals. The growing emphasis on personalized medicine and biologics has intensified demand for formulations that can withstand various storage conditions while preserving molecular integrity.
Industrial applications constitute another significant demand driver, particularly in the specialty chemicals and materials sectors. Polyamide-based materials, including high-performance polymers and engineering plastics, require enhanced stability to meet stringent performance requirements in automotive, aerospace, and electronics applications. The push toward sustainable manufacturing processes has created additional demand for amide-based catalysts and intermediates that demonstrate superior thermal and chemical stability.
The cosmetics and personal care industry has emerged as a rapidly expanding market for stable amide compounds. Formulation chemists increasingly rely on amide-based emulsifiers, conditioning agents, and active ingredients that must remain stable across diverse climatic conditions and extended product lifecycles. Consumer expectations for longer-lasting products with consistent performance have elevated the importance of molecular stability assessment.
Agricultural applications represent a specialized but growing segment, where amide-based pesticides, herbicides, and plant growth regulators must maintain potency under field conditions. Environmental regulations demanding reduced application frequencies have heightened the need for formulations with extended stability profiles.
The food and beverage industry presents unique challenges for amide stability, particularly in food additives, flavor compounds, and packaging materials. Regulatory requirements for extended shelf life combined with clean label trends have created demand for naturally-derived amide compounds with proven stability characteristics.
Market growth is further accelerated by regulatory frameworks requiring comprehensive stability data for product approvals. Pharmaceutical regulatory agencies worldwide mandate extensive stability testing, creating sustained demand for advanced analytical methods and stability assessment technologies. This regulatory environment has established stability evaluation as a critical component of product development across multiple industries.
Industrial applications constitute another significant demand driver, particularly in the specialty chemicals and materials sectors. Polyamide-based materials, including high-performance polymers and engineering plastics, require enhanced stability to meet stringent performance requirements in automotive, aerospace, and electronics applications. The push toward sustainable manufacturing processes has created additional demand for amide-based catalysts and intermediates that demonstrate superior thermal and chemical stability.
The cosmetics and personal care industry has emerged as a rapidly expanding market for stable amide compounds. Formulation chemists increasingly rely on amide-based emulsifiers, conditioning agents, and active ingredients that must remain stable across diverse climatic conditions and extended product lifecycles. Consumer expectations for longer-lasting products with consistent performance have elevated the importance of molecular stability assessment.
Agricultural applications represent a specialized but growing segment, where amide-based pesticides, herbicides, and plant growth regulators must maintain potency under field conditions. Environmental regulations demanding reduced application frequencies have heightened the need for formulations with extended stability profiles.
The food and beverage industry presents unique challenges for amide stability, particularly in food additives, flavor compounds, and packaging materials. Regulatory requirements for extended shelf life combined with clean label trends have created demand for naturally-derived amide compounds with proven stability characteristics.
Market growth is further accelerated by regulatory frameworks requiring comprehensive stability data for product approvals. Pharmaceutical regulatory agencies worldwide mandate extensive stability testing, creating sustained demand for advanced analytical methods and stability assessment technologies. This regulatory environment has established stability evaluation as a critical component of product development across multiple industries.
Current Amide Dynamics Assessment Challenges
The assessment of amide molecule dynamics for extended stability faces significant methodological limitations that constrain accurate prediction and optimization of pharmaceutical formulations. Traditional analytical approaches often rely on static structural analysis or short-term stability studies, which fail to capture the complex dynamic behaviors that govern long-term molecular stability under various environmental conditions.
Current spectroscopic techniques, while providing valuable insights into molecular vibrations and conformational changes, suffer from temporal resolution constraints that prevent real-time monitoring of rapid dynamic processes. Nuclear magnetic resonance spectroscopy, though powerful for structural elucidation, requires relatively high concentrations and may not accurately reflect dynamics in dilute pharmaceutical formulations where intermolecular interactions differ significantly.
Computational modeling approaches encounter substantial challenges in balancing accuracy with computational feasibility. Molecular dynamics simulations capable of capturing relevant timescales for stability assessment demand enormous computational resources, often limiting studies to nanosecond or microsecond timeframes when pharmaceutical stability concerns span months or years. Force field limitations further compromise the accuracy of predicted amide bond behavior, particularly under stress conditions involving temperature fluctuations, pH variations, or oxidative environments.
Experimental design complexities arise from the multifactorial nature of stability degradation pathways. Amide molecules can undergo hydrolysis, oxidation, deamidation, and cyclization reactions simultaneously, making it difficult to isolate and quantify individual dynamic contributions to overall stability. Traditional accelerated stability testing protocols may not accurately predict real-world storage conditions, as elevated temperatures and humidity levels can alter fundamental molecular dynamics patterns.
Analytical sensitivity represents another critical bottleneck, as early-stage degradation products often exist at trace levels below detection thresholds of conventional analytical methods. This limitation prevents early identification of stability-compromising dynamic behaviors, forcing reliance on endpoint measurements rather than mechanistic understanding of degradation initiation and progression.
Integration challenges between different analytical platforms further complicate comprehensive dynamics assessment. Data from spectroscopic, chromatographic, and computational sources often lack standardized correlation methods, hindering development of unified predictive models for extended stability forecasting.
Current spectroscopic techniques, while providing valuable insights into molecular vibrations and conformational changes, suffer from temporal resolution constraints that prevent real-time monitoring of rapid dynamic processes. Nuclear magnetic resonance spectroscopy, though powerful for structural elucidation, requires relatively high concentrations and may not accurately reflect dynamics in dilute pharmaceutical formulations where intermolecular interactions differ significantly.
Computational modeling approaches encounter substantial challenges in balancing accuracy with computational feasibility. Molecular dynamics simulations capable of capturing relevant timescales for stability assessment demand enormous computational resources, often limiting studies to nanosecond or microsecond timeframes when pharmaceutical stability concerns span months or years. Force field limitations further compromise the accuracy of predicted amide bond behavior, particularly under stress conditions involving temperature fluctuations, pH variations, or oxidative environments.
Experimental design complexities arise from the multifactorial nature of stability degradation pathways. Amide molecules can undergo hydrolysis, oxidation, deamidation, and cyclization reactions simultaneously, making it difficult to isolate and quantify individual dynamic contributions to overall stability. Traditional accelerated stability testing protocols may not accurately predict real-world storage conditions, as elevated temperatures and humidity levels can alter fundamental molecular dynamics patterns.
Analytical sensitivity represents another critical bottleneck, as early-stage degradation products often exist at trace levels below detection thresholds of conventional analytical methods. This limitation prevents early identification of stability-compromising dynamic behaviors, forcing reliance on endpoint measurements rather than mechanistic understanding of degradation initiation and progression.
Integration challenges between different analytical platforms further complicate comprehensive dynamics assessment. Data from spectroscopic, chromatographic, and computational sources often lack standardized correlation methods, hindering development of unified predictive models for extended stability forecasting.
Current Molecular Dynamics Assessment Solutions
01 Stabilization through molecular structure modification
Amide molecules can be stabilized by modifying their molecular structure, such as introducing specific substituents or functional groups that enhance resistance to hydrolysis and degradation. Structural modifications can include the addition of bulky groups, electron-withdrawing or electron-donating substituents, and the formation of cyclic amide structures. These modifications help protect the amide bond from chemical and enzymatic breakdown, thereby improving overall molecular stability.- Stabilization through molecular structure modification: Amide molecules can be stabilized by modifying their molecular structure, such as introducing specific substituents or functional groups that enhance resistance to hydrolysis and degradation. Structural modifications can include the addition of bulky groups, electron-withdrawing or electron-donating substituents, and the formation of cyclic amide structures. These modifications help protect the amide bond from chemical and enzymatic degradation, thereby improving overall molecular stability.
- Use of stabilizing additives and excipients: The stability of amide molecules can be enhanced through the incorporation of stabilizing additives and excipients in formulations. These additives may include antioxidants, chelating agents, buffering agents, and pH adjusters that prevent oxidative degradation and maintain optimal conditions for amide stability. The selection of appropriate excipients can significantly reduce degradation pathways and extend the shelf life of amide-containing products.
- Encapsulation and delivery system approaches: Encapsulation techniques and specialized delivery systems can protect amide molecules from environmental factors that cause degradation. Methods such as microencapsulation, nanoencapsulation, and liposomal formulations create protective barriers around amide molecules, shielding them from moisture, light, and reactive species. These approaches are particularly useful for pharmaceutical and cosmetic applications where long-term stability is critical.
- Control of environmental storage conditions: Maintaining appropriate environmental conditions during storage and handling is essential for amide molecule stability. Factors such as temperature, humidity, light exposure, and oxygen levels can significantly impact degradation rates. Controlled storage conditions, including refrigeration, use of desiccants, light-protective packaging, and inert atmosphere storage, help preserve amide integrity and prevent premature degradation.
- Analytical methods for stability assessment: Various analytical techniques are employed to assess and monitor the stability of amide molecules over time. These methods include chromatographic techniques, spectroscopic analysis, thermal analysis, and accelerated stability testing protocols. Regular stability testing helps identify degradation products, determine shelf life, and optimize formulation parameters to ensure product quality and efficacy throughout the intended storage period.
02 Use of stabilizing additives and excipients
The stability of amide molecules can be enhanced through the incorporation of stabilizing additives and excipients in formulations. These additives may include antioxidants, chelating agents, buffering agents, and pH adjusters that prevent oxidative degradation and maintain optimal conditions for amide stability. The selection of appropriate excipients is crucial for preventing hydrolysis and maintaining the integrity of amide bonds during storage and use.Expand Specific Solutions03 Encapsulation and delivery system design
Amide molecule stability can be improved through advanced encapsulation techniques and specialized delivery systems. These systems protect amide compounds from environmental factors such as moisture, light, and temperature fluctuations. Encapsulation methods may include microencapsulation, nanoencapsulation, and the use of protective coatings or matrices that shield the amide molecules from degradation while allowing controlled release.Expand Specific Solutions04 Control of environmental storage conditions
Maintaining amide molecule stability requires careful control of environmental storage conditions including temperature, humidity, and light exposure. Optimal storage conditions typically involve cool, dry environments with minimal light exposure to prevent photodegradation and hydrolysis. Packaging materials and container designs play a critical role in protecting amide molecules from moisture ingress and atmospheric oxygen, which are primary causes of degradation.Expand Specific Solutions05 Analytical methods for stability assessment
Various analytical methods and testing protocols are employed to assess and monitor the stability of amide molecules over time. These methods include accelerated stability testing, stress testing under various conditions, and the use of analytical techniques to detect degradation products. Stability studies help determine shelf life, optimal storage conditions, and formulation requirements to ensure amide molecules maintain their integrity and efficacy throughout their intended use period.Expand Specific Solutions
Key Players in Amide Research and Simulation
The amide molecule dynamics assessment field represents an emerging technology sector in early development stages, characterized by significant research activity but limited commercial maturity. The market remains nascent with substantial growth potential as pharmaceutical and chemical industries increasingly recognize the importance of molecular stability for drug development and material science applications. Technology maturity varies considerably across players, with pharmaceutical giants like F. Hoffmann-La Roche Ltd., Pfizer Inc., and Novo Nordisk A/S leading in practical applications through drug development programs, while academic institutions including University of California, Zhejiang University, and Duke University drive fundamental research breakthroughs. Chemical manufacturers such as LG Chem Ltd., Domo Caproleuna GmbH, and Resonac Corp. focus on industrial applications, particularly in polyamide and polymer stability. The competitive landscape shows a clear division between research-intensive academic institutions advancing theoretical understanding and industry players translating findings into commercial applications, indicating the technology is transitioning from laboratory research toward practical implementation phases.
The Regents of the University of California
Technical Solution: UC system researchers have developed cutting-edge computational approaches for amide stability assessment using enhanced sampling methods and free energy calculations. Their research focuses on understanding fundamental mechanisms of amide hydrolysis and developing predictive models for stability under physiological conditions. The university's interdisciplinary teams combine quantum chemistry calculations with classical molecular dynamics to study amide bond stability, solvent effects, and catalytic degradation pathways. Their work includes development of novel force fields specifically parameterized for accurate amide bond representation.
Strengths: Leading-edge research capabilities and strong theoretical foundations in molecular modeling. Weaknesses: Limited industrial validation and longer development timelines for practical applications.
F. Hoffmann-La Roche Ltd.
Technical Solution: Roche has developed comprehensive amide stability assessment platforms utilizing advanced molecular dynamics simulations combined with accelerated stability testing protocols. Their approach integrates computational modeling with experimental validation to predict long-term amide degradation pathways, particularly focusing on hydrolysis mechanisms and conformational changes under various environmental conditions. The company employs machine learning algorithms to analyze molecular trajectories and identify critical stability markers, enabling prediction of shelf-life and storage requirements for amide-containing pharmaceutical compounds.
Strengths: Extensive pharmaceutical expertise and validated regulatory pathways for stability testing. Weaknesses: High computational costs and limited applicability to novel amide structures outside their database.
Core Technologies in Amide Stability Prediction
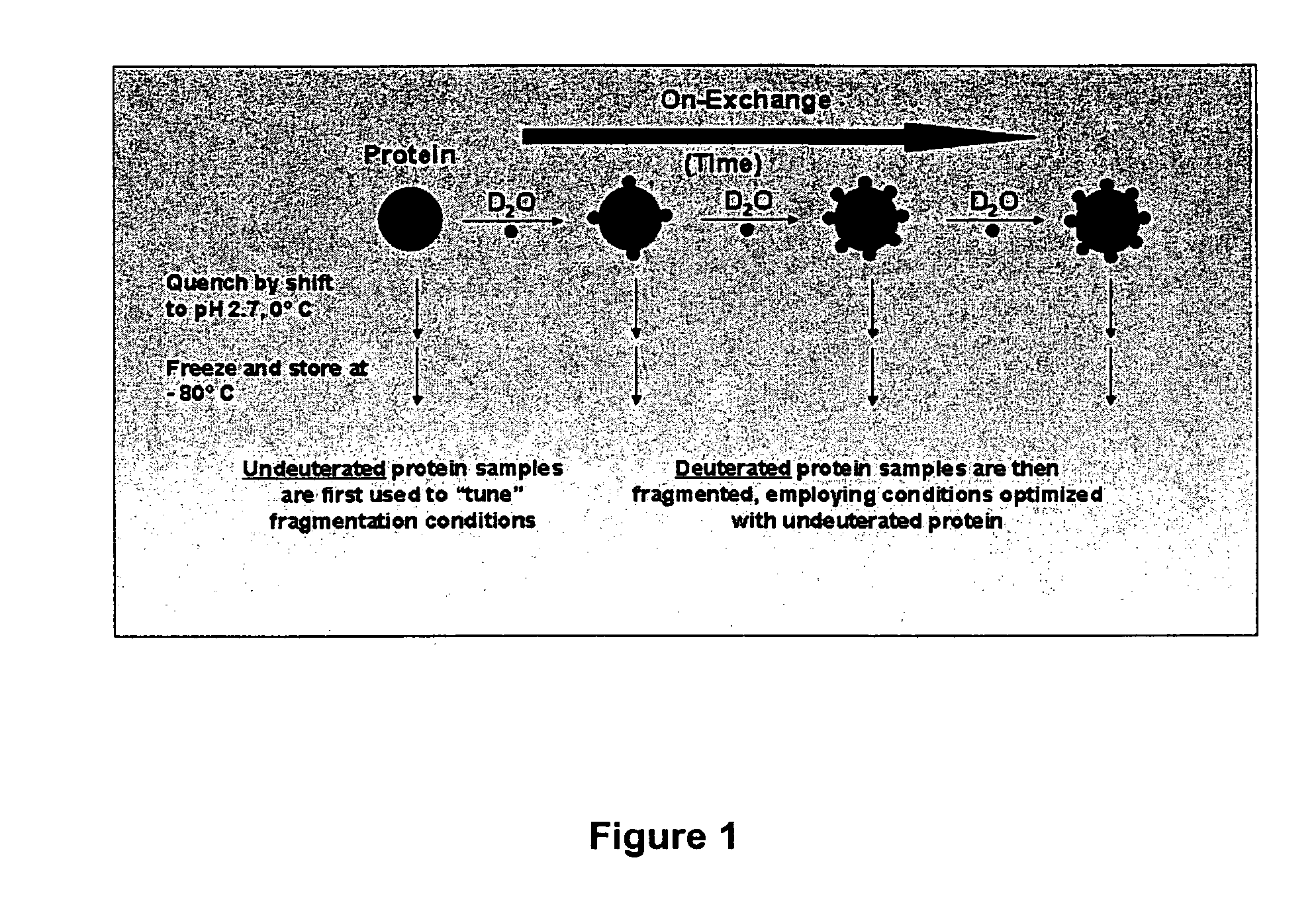
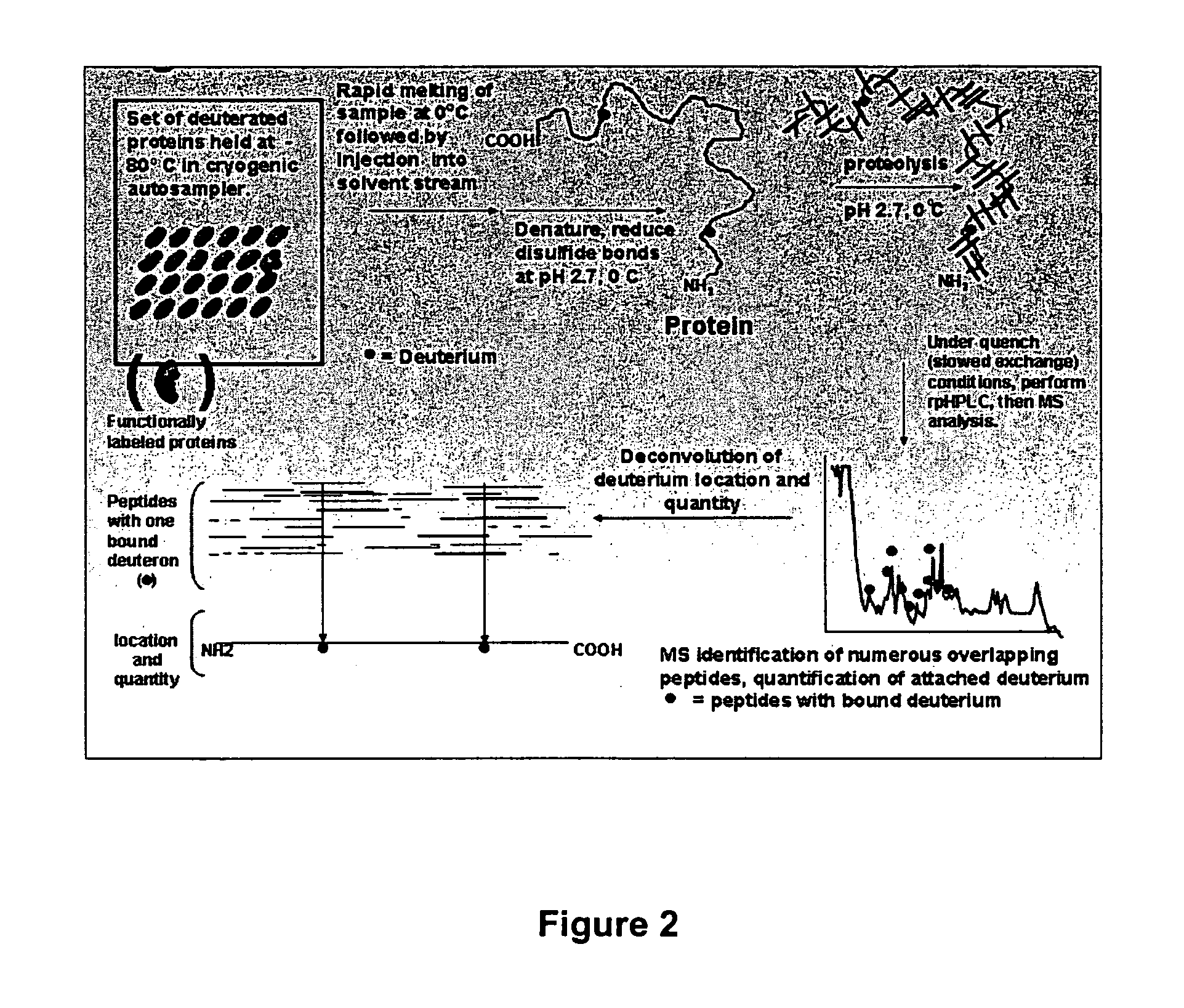
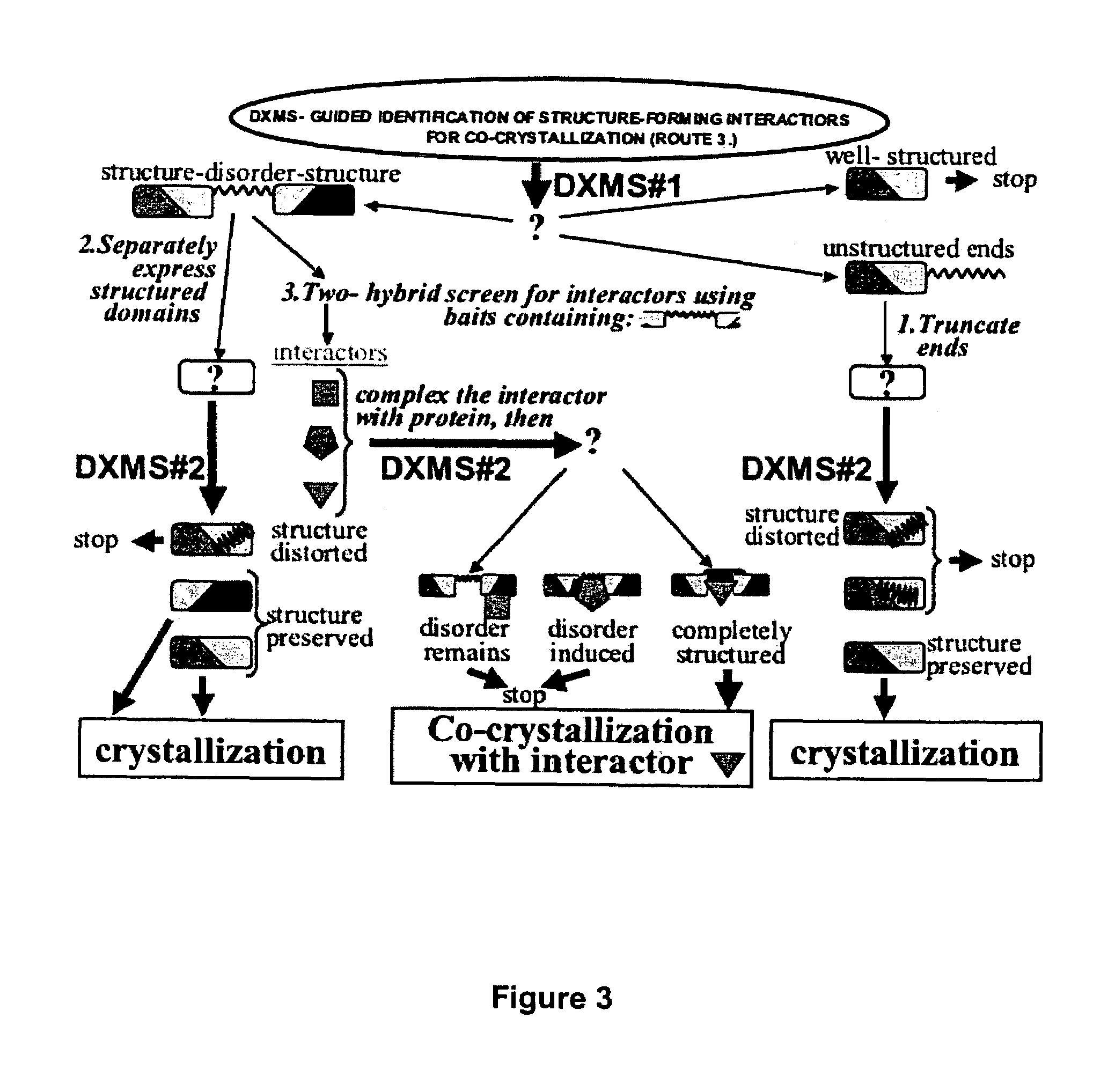
Enhanced methods for crystallographic structure determination employing hydrogen exchange analysis
PatentInactiveUS7363171B2
Innovation
- Employing hydrogen exchange analysis, particularly through deuterium exchange mass spectrometry (DXMS), to identify and stabilize unstructured regions within proteins by identifying structure-forming agents and conditions, enabling co-crystallization with binding partners to enhance crystallization success.
Novel non-natural C-linked carbo-beta-peptides with robust secondary structures
PatentInactiveUS20040192885A1
Innovation
- The synthesis of novel C-linked carbo-.beta.-peptides using C-linked carbo-.beta.-amino acids, which incorporate carbohydrate moieties into the peptide backbone, allowing for the formation of stable helical structures and enhanced biological activity through chirality control and the use of commercial reagents for large-scale production.
Computational Infrastructure for MD Simulations
The computational infrastructure for molecular dynamics simulations of amide molecules requires sophisticated hardware and software architectures capable of handling the complex calculations involved in assessing molecular stability over extended timeframes. High-performance computing clusters with parallel processing capabilities form the backbone of these computational systems, typically featuring multi-core processors, substantial memory allocation, and high-speed interconnects to manage the intensive calculations required for long-duration simulations.
Modern MD simulation infrastructure relies heavily on GPU acceleration, particularly NVIDIA's CUDA-enabled graphics cards, which can significantly reduce computation time for force field calculations and trajectory analysis. The parallel nature of MD algorithms makes them well-suited for GPU architectures, where thousands of cores can simultaneously process particle interactions. This hardware acceleration is essential when simulating amide molecules over microsecond to millisecond timescales necessary for stability assessment.
Software frameworks constitute another critical component of the computational infrastructure. Leading MD simulation packages such as GROMACS, AMBER, NAMD, and LAMMPS provide optimized algorithms specifically designed for biomolecular systems. These platforms incorporate advanced force fields like CHARMM, AMBER ff14SB, and OPLS-AA that accurately model amide bond characteristics, hydrogen bonding patterns, and conformational dynamics essential for stability predictions.
Storage infrastructure presents unique challenges for extended MD simulations, as trajectory files can reach terabyte scales when monitoring amide molecule dynamics over extended periods. High-throughput storage systems with parallel file systems like Lustre or GPFS are typically employed to handle the continuous data streams generated during simulations. Additionally, data compression algorithms and selective trajectory saving protocols help manage storage requirements while preserving critical conformational information.
Cloud computing platforms have emerged as viable alternatives to traditional on-premise clusters, offering scalable resources for MD simulations. Services like AWS ParallelCluster, Google Cloud HPC, and Microsoft Azure CycleCloud provide access to specialized instances optimized for computational chemistry workloads, enabling researchers to scale computational resources based on simulation complexity and duration requirements.
Workflow management systems integrate these infrastructure components, automating simulation setup, execution monitoring, and result analysis. Tools like Nextflow, Snakemake, and custom pipeline solutions ensure reproducible simulation protocols while managing resource allocation and job scheduling across distributed computing environments, ultimately enabling systematic assessment of amide molecule stability across diverse chemical spaces.
Modern MD simulation infrastructure relies heavily on GPU acceleration, particularly NVIDIA's CUDA-enabled graphics cards, which can significantly reduce computation time for force field calculations and trajectory analysis. The parallel nature of MD algorithms makes them well-suited for GPU architectures, where thousands of cores can simultaneously process particle interactions. This hardware acceleration is essential when simulating amide molecules over microsecond to millisecond timescales necessary for stability assessment.
Software frameworks constitute another critical component of the computational infrastructure. Leading MD simulation packages such as GROMACS, AMBER, NAMD, and LAMMPS provide optimized algorithms specifically designed for biomolecular systems. These platforms incorporate advanced force fields like CHARMM, AMBER ff14SB, and OPLS-AA that accurately model amide bond characteristics, hydrogen bonding patterns, and conformational dynamics essential for stability predictions.
Storage infrastructure presents unique challenges for extended MD simulations, as trajectory files can reach terabyte scales when monitoring amide molecule dynamics over extended periods. High-throughput storage systems with parallel file systems like Lustre or GPFS are typically employed to handle the continuous data streams generated during simulations. Additionally, data compression algorithms and selective trajectory saving protocols help manage storage requirements while preserving critical conformational information.
Cloud computing platforms have emerged as viable alternatives to traditional on-premise clusters, offering scalable resources for MD simulations. Services like AWS ParallelCluster, Google Cloud HPC, and Microsoft Azure CycleCloud provide access to specialized instances optimized for computational chemistry workloads, enabling researchers to scale computational resources based on simulation complexity and duration requirements.
Workflow management systems integrate these infrastructure components, automating simulation setup, execution monitoring, and result analysis. Tools like Nextflow, Snakemake, and custom pipeline solutions ensure reproducible simulation protocols while managing resource allocation and job scheduling across distributed computing environments, ultimately enabling systematic assessment of amide molecule stability across diverse chemical spaces.
Quality Standards for Pharmaceutical Amides
The establishment of comprehensive quality standards for pharmaceutical amides represents a critical foundation for ensuring drug safety, efficacy, and regulatory compliance. These standards encompass multiple analytical dimensions that collectively define the acceptable parameters for amide-containing pharmaceutical compounds throughout their lifecycle.
Purity specifications constitute the primary quality criterion, typically requiring amide compounds to maintain purity levels exceeding 98% for active pharmaceutical ingredients. Impurity profiling must identify and quantify degradation products, synthetic byproducts, and potential genotoxic impurities below established threshold limits. Residual solvent content, particularly for amide synthesis involving DMF or NMP, must comply with ICH Q3C guidelines.
Stability-indicating analytical methods form the backbone of quality assessment, employing techniques such as high-performance liquid chromatography with mass spectrometry detection to monitor amide bond integrity over time. These methods must demonstrate specificity for the parent compound while detecting hydrolysis products, oxidative degradation, and other transformation pathways that compromise molecular stability.
Physical characterization standards address critical material properties including polymorphic form identification, particle size distribution, and moisture content. Amide compounds often exhibit hygroscopic behavior, necessitating strict water activity limits typically below 0.6 aw to prevent accelerated degradation. Crystalline form control ensures consistent bioavailability and manufacturing performance.
Microbiological quality standards mandate sterility testing for injectable formulations and bioburden limits for oral dosage forms. Endotoxin levels must remain below pharmacopeial limits, particularly crucial for amide compounds intended for parenteral administration where pyrogenic responses pose significant safety risks.
Container-closure integrity testing ensures packaging systems maintain product quality throughout shelf life. This includes extractables and leachables studies to identify potential interactions between amide compounds and packaging materials, which may introduce impurities or affect stability profiles.
Analytical method validation follows ICH Q2 guidelines, demonstrating accuracy, precision, linearity, range, specificity, and robustness for all quality control procedures. Method transfer protocols ensure consistent implementation across manufacturing sites and contract testing laboratories.
Purity specifications constitute the primary quality criterion, typically requiring amide compounds to maintain purity levels exceeding 98% for active pharmaceutical ingredients. Impurity profiling must identify and quantify degradation products, synthetic byproducts, and potential genotoxic impurities below established threshold limits. Residual solvent content, particularly for amide synthesis involving DMF or NMP, must comply with ICH Q3C guidelines.
Stability-indicating analytical methods form the backbone of quality assessment, employing techniques such as high-performance liquid chromatography with mass spectrometry detection to monitor amide bond integrity over time. These methods must demonstrate specificity for the parent compound while detecting hydrolysis products, oxidative degradation, and other transformation pathways that compromise molecular stability.
Physical characterization standards address critical material properties including polymorphic form identification, particle size distribution, and moisture content. Amide compounds often exhibit hygroscopic behavior, necessitating strict water activity limits typically below 0.6 aw to prevent accelerated degradation. Crystalline form control ensures consistent bioavailability and manufacturing performance.
Microbiological quality standards mandate sterility testing for injectable formulations and bioburden limits for oral dosage forms. Endotoxin levels must remain below pharmacopeial limits, particularly crucial for amide compounds intended for parenteral administration where pyrogenic responses pose significant safety risks.
Container-closure integrity testing ensures packaging systems maintain product quality throughout shelf life. This includes extractables and leachables studies to identify potential interactions between amide compounds and packaging materials, which may introduce impurities or affect stability profiles.
Analytical method validation follows ICH Q2 guidelines, demonstrating accuracy, precision, linearity, range, specificity, and robustness for all quality control procedures. Method transfer protocols ensure consistent implementation across manufacturing sites and contract testing laboratories.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!