Aromatic Compounds vs Organometallics: Reaction Efficiency
MAR 5, 20268 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Aromatic vs Organometallic Chemistry Background and Objectives
Aromatic chemistry and organometallic chemistry represent two fundamental pillars of modern synthetic chemistry, each offering distinct advantages and challenges in reaction efficiency. Aromatic compounds, characterized by their stable conjugated π-electron systems, have been the cornerstone of organic synthesis for over a century. These compounds exhibit unique reactivity patterns governed by electrophilic and nucleophilic aromatic substitution mechanisms, providing predictable pathways for functionalization.
Organometallic chemistry emerged as a revolutionary field in the mid-20th century, introducing metal-carbon bonds that enable unprecedented reactivity and selectivity. The incorporation of transition metals, main group metals, and lanthanides has expanded the synthetic toolkit exponentially, allowing for reactions that were previously impossible or highly inefficient using traditional organic methods.
The historical development of these fields reveals a fascinating evolution in reaction efficiency paradigms. Early aromatic chemistry relied heavily on harsh reaction conditions, often requiring elevated temperatures, strong acids, or bases, and lengthy reaction times. The discovery of organometallic catalysts transformed this landscape, enabling milder conditions, higher selectivity, and improved atom economy.
Current technological objectives focus on bridging the efficiency gap between these two approaches. The primary goal involves developing hybrid methodologies that combine the stability and predictability of aromatic systems with the versatility and efficiency of organometallic catalysis. This convergence aims to achieve superior reaction rates, enhanced selectivity, and reduced environmental impact.
Key performance metrics driving this field include turnover frequency, turnover number, substrate scope, functional group tolerance, and stereoselectivity. Modern research emphasizes developing catalytic systems that can efficiently activate aromatic substrates while maintaining high selectivity and minimizing side reactions.
The integration of computational chemistry and machine learning has become increasingly important in predicting and optimizing reaction efficiency. These tools enable researchers to understand the fundamental differences in activation energies, transition state geometries, and electronic effects that govern the efficiency of aromatic versus organometallic transformations.
Sustainability considerations have also shaped current objectives, with emphasis on developing earth-abundant metal catalysts, reducing waste generation, and improving energy efficiency. The ultimate goal is creating reaction systems that combine the best attributes of both aromatic and organometallic chemistry while addressing environmental and economic constraints.
Organometallic chemistry emerged as a revolutionary field in the mid-20th century, introducing metal-carbon bonds that enable unprecedented reactivity and selectivity. The incorporation of transition metals, main group metals, and lanthanides has expanded the synthetic toolkit exponentially, allowing for reactions that were previously impossible or highly inefficient using traditional organic methods.
The historical development of these fields reveals a fascinating evolution in reaction efficiency paradigms. Early aromatic chemistry relied heavily on harsh reaction conditions, often requiring elevated temperatures, strong acids, or bases, and lengthy reaction times. The discovery of organometallic catalysts transformed this landscape, enabling milder conditions, higher selectivity, and improved atom economy.
Current technological objectives focus on bridging the efficiency gap between these two approaches. The primary goal involves developing hybrid methodologies that combine the stability and predictability of aromatic systems with the versatility and efficiency of organometallic catalysis. This convergence aims to achieve superior reaction rates, enhanced selectivity, and reduced environmental impact.
Key performance metrics driving this field include turnover frequency, turnover number, substrate scope, functional group tolerance, and stereoselectivity. Modern research emphasizes developing catalytic systems that can efficiently activate aromatic substrates while maintaining high selectivity and minimizing side reactions.
The integration of computational chemistry and machine learning has become increasingly important in predicting and optimizing reaction efficiency. These tools enable researchers to understand the fundamental differences in activation energies, transition state geometries, and electronic effects that govern the efficiency of aromatic versus organometallic transformations.
Sustainability considerations have also shaped current objectives, with emphasis on developing earth-abundant metal catalysts, reducing waste generation, and improving energy efficiency. The ultimate goal is creating reaction systems that combine the best attributes of both aromatic and organometallic chemistry while addressing environmental and economic constraints.
Market Demand for High-Efficiency Catalytic Reactions
The global chemical industry is experiencing unprecedented demand for high-efficiency catalytic reactions, driven by the urgent need for sustainable manufacturing processes and environmental compliance. Traditional catalytic systems face mounting pressure to deliver superior performance while minimizing energy consumption and waste generation. This market shift has intensified focus on optimizing reaction efficiency between aromatic compounds and organometallic catalysts.
Pharmaceutical manufacturing represents the largest market segment demanding enhanced catalytic efficiency. The sector requires precise control over reaction selectivity and yield, particularly in complex multi-step syntheses where aromatic substrates interact with organometallic catalysts. Rising drug development costs and stringent regulatory requirements have created substantial market pressure for catalytic systems that can achieve higher conversion rates with reduced byproduct formation.
The fine chemicals industry demonstrates equally compelling demand for improved catalytic performance. Specialty chemical manufacturers increasingly seek catalytic solutions that can handle diverse aromatic feedstocks while maintaining consistent organometallic catalyst activity. Market competition has intensified the need for processes that can achieve target products with minimal purification requirements and reduced processing time.
Environmental regulations across major industrial regions have created additional market drivers for high-efficiency catalytic reactions. Stricter emission standards and waste disposal costs have made reaction efficiency a critical economic factor. Companies face significant financial incentives to adopt catalytic systems that maximize atom economy and minimize environmental impact through improved aromatic compound conversion rates.
The petrochemical sector presents substantial market opportunities for enhanced catalytic efficiency, particularly in aromatic compound processing and functionalization reactions. Growing demand for high-value aromatic derivatives has created market pressure for catalytic systems capable of achieving superior selectivity and productivity. Organometallic catalyst systems that can operate under milder conditions while maintaining high turnover frequencies command premium market positioning.
Emerging markets in Asia-Pacific regions show accelerating demand for efficient catalytic technologies, driven by rapid industrialization and increasing environmental awareness. Local manufacturers seek catalytic solutions that can compete with established technologies while meeting international quality standards. This geographic expansion of demand has created new market segments focused specifically on cost-effective, high-efficiency catalytic processes involving aromatic substrates and organometallic systems.
Pharmaceutical manufacturing represents the largest market segment demanding enhanced catalytic efficiency. The sector requires precise control over reaction selectivity and yield, particularly in complex multi-step syntheses where aromatic substrates interact with organometallic catalysts. Rising drug development costs and stringent regulatory requirements have created substantial market pressure for catalytic systems that can achieve higher conversion rates with reduced byproduct formation.
The fine chemicals industry demonstrates equally compelling demand for improved catalytic performance. Specialty chemical manufacturers increasingly seek catalytic solutions that can handle diverse aromatic feedstocks while maintaining consistent organometallic catalyst activity. Market competition has intensified the need for processes that can achieve target products with minimal purification requirements and reduced processing time.
Environmental regulations across major industrial regions have created additional market drivers for high-efficiency catalytic reactions. Stricter emission standards and waste disposal costs have made reaction efficiency a critical economic factor. Companies face significant financial incentives to adopt catalytic systems that maximize atom economy and minimize environmental impact through improved aromatic compound conversion rates.
The petrochemical sector presents substantial market opportunities for enhanced catalytic efficiency, particularly in aromatic compound processing and functionalization reactions. Growing demand for high-value aromatic derivatives has created market pressure for catalytic systems capable of achieving superior selectivity and productivity. Organometallic catalyst systems that can operate under milder conditions while maintaining high turnover frequencies command premium market positioning.
Emerging markets in Asia-Pacific regions show accelerating demand for efficient catalytic technologies, driven by rapid industrialization and increasing environmental awareness. Local manufacturers seek catalytic solutions that can compete with established technologies while meeting international quality standards. This geographic expansion of demand has created new market segments focused specifically on cost-effective, high-efficiency catalytic processes involving aromatic substrates and organometallic systems.
Current Challenges in Aromatic-Organometallic Reaction Systems
The integration of aromatic compounds with organometallic systems faces significant thermodynamic barriers that fundamentally limit reaction efficiency. The electron-rich nature of aromatic rings creates substantial activation energies for initial coordination steps, particularly in cross-coupling reactions where C-H or C-X bond activation is required. These high energy barriers often necessitate elevated temperatures and extended reaction times, leading to decreased selectivity and increased side product formation.
Catalyst deactivation represents a persistent challenge across aromatic-organometallic reaction systems. Organometallic catalysts frequently undergo decomposition through β-hydride elimination, reductive elimination, or ligand dissociation when exposed to the harsh conditions required for aromatic substrate activation. The formation of catalytically inactive metal clusters or precipitation of metallic species significantly reduces turnover numbers and compromises reaction reproducibility.
Selectivity control remains problematic due to the multiple reactive sites present in both aromatic and organometallic components. Regioselectivity issues arise when aromatic substrates contain multiple potential reaction sites, while chemoselectivity challenges emerge from competing reaction pathways such as homocoupling versus cross-coupling. The electronic and steric properties of both reaction partners must be carefully balanced to achieve desired selectivity profiles.
Ligand design limitations constrain the optimization of aromatic-organometallic reactions. Current ligand systems often fail to provide simultaneous control over catalyst stability, substrate binding affinity, and product release rates. The development of ligands that can effectively modulate the electronic properties of metal centers while maintaining structural integrity under reaction conditions remains an ongoing challenge.
Substrate scope restrictions limit the broad applicability of existing methodologies. Electron-deficient aromatic compounds often exhibit poor reactivity due to reduced nucleophilicity, while electron-rich aromatics may undergo undesired side reactions such as oxidative coupling or polymerization. Similarly, organometallic reagents with sensitive functional groups frequently decompose under the reaction conditions required for aromatic activation.
Mass transfer limitations in heterogeneous aromatic-organometallic systems create additional efficiency barriers. Poor solubility of aromatic substrates in compatible solvents, combined with the air and moisture sensitivity of organometallic reagents, restricts reaction medium selection and often leads to biphasic systems with limited interfacial contact. These transport phenomena significantly impact reaction rates and overall process efficiency.
Catalyst deactivation represents a persistent challenge across aromatic-organometallic reaction systems. Organometallic catalysts frequently undergo decomposition through β-hydride elimination, reductive elimination, or ligand dissociation when exposed to the harsh conditions required for aromatic substrate activation. The formation of catalytically inactive metal clusters or precipitation of metallic species significantly reduces turnover numbers and compromises reaction reproducibility.
Selectivity control remains problematic due to the multiple reactive sites present in both aromatic and organometallic components. Regioselectivity issues arise when aromatic substrates contain multiple potential reaction sites, while chemoselectivity challenges emerge from competing reaction pathways such as homocoupling versus cross-coupling. The electronic and steric properties of both reaction partners must be carefully balanced to achieve desired selectivity profiles.
Ligand design limitations constrain the optimization of aromatic-organometallic reactions. Current ligand systems often fail to provide simultaneous control over catalyst stability, substrate binding affinity, and product release rates. The development of ligands that can effectively modulate the electronic properties of metal centers while maintaining structural integrity under reaction conditions remains an ongoing challenge.
Substrate scope restrictions limit the broad applicability of existing methodologies. Electron-deficient aromatic compounds often exhibit poor reactivity due to reduced nucleophilicity, while electron-rich aromatics may undergo undesired side reactions such as oxidative coupling or polymerization. Similarly, organometallic reagents with sensitive functional groups frequently decompose under the reaction conditions required for aromatic activation.
Mass transfer limitations in heterogeneous aromatic-organometallic systems create additional efficiency barriers. Poor solubility of aromatic substrates in compatible solvents, combined with the air and moisture sensitivity of organometallic reagents, restricts reaction medium selection and often leads to biphasic systems with limited interfacial contact. These transport phenomena significantly impact reaction rates and overall process efficiency.
Current Solutions for Optimizing Reaction Efficiency
01 Use of organometallic catalysts in aromatic substitution reactions
Organometallic compounds, particularly transition metal complexes, can serve as effective catalysts for aromatic substitution reactions. These catalysts facilitate the activation of aromatic rings and improve reaction selectivity and yield. The use of palladium, nickel, and other transition metal-based catalysts has shown significant improvements in reaction efficiency for various aromatic transformations including coupling reactions and functionalization processes.- Use of organometallic catalysts in aromatic substitution reactions: Organometallic compounds, particularly transition metal complexes, can serve as effective catalysts for aromatic substitution reactions. These catalysts facilitate the activation of aromatic rings and improve reaction selectivity and yield. The use of palladium, nickel, and other transition metal-based catalysts has shown significant improvements in reaction efficiency for various aromatic transformations including coupling reactions and functionalization processes.
- Optimization of reaction conditions for aromatic-organometallic coupling: The efficiency of reactions between aromatic compounds and organometallic reagents can be enhanced through careful control of reaction parameters such as temperature, pressure, solvent selection, and reagent ratios. Specific reaction conditions including the use of appropriate bases, ligands, and additives can significantly improve conversion rates and product selectivity in cross-coupling and other organometallic-mediated aromatic transformations.
- Novel organometallic reagents for aromatic functionalization: Development of new organometallic reagents with improved reactivity profiles enables more efficient aromatic compound transformations. These reagents include modified Grignard reagents, organolithium compounds, and organoboron species that exhibit enhanced stability and selectivity. The design of these reagents focuses on achieving better functional group tolerance and milder reaction conditions while maintaining high reaction efficiency.
- Ligand design for enhanced catalytic performance: The development of specialized ligands plays a crucial role in improving the efficiency of organometallic-catalyzed aromatic reactions. Phosphine-based ligands, N-heterocyclic carbenes, and other coordinating molecules can modulate the electronic and steric properties of metal centers, leading to improved catalyst activity, stability, and selectivity. Proper ligand selection enables better control over reaction pathways and reduces side reactions.
- Process intensification through continuous flow systems: Implementation of continuous flow technology for aromatic-organometallic reactions offers advantages in terms of reaction efficiency, safety, and scalability. Flow reactors provide better heat and mass transfer, enable precise control of residence time, and allow for the safe handling of reactive organometallic intermediates. This approach can lead to improved yields, reduced reaction times, and enhanced reproducibility compared to traditional batch processes.
02 Enhancement of reaction efficiency through ligand modification
The modification of ligands in organometallic complexes plays a crucial role in improving the efficiency of reactions involving aromatic compounds. By adjusting the electronic and steric properties of ligands, the reactivity and selectivity of organometallic catalysts can be optimized. This approach allows for better control over reaction pathways and can significantly reduce reaction times while improving product yields in aromatic compound transformations.Expand Specific Solutions03 Application of organometallic reagents in aromatic coupling reactions
Organometallic reagents are widely employed in cross-coupling reactions of aromatic compounds to form carbon-carbon bonds. These reagents enable efficient coupling between aromatic halides and various nucleophiles, facilitating the synthesis of complex aromatic structures. The methodology has proven particularly valuable in pharmaceutical and materials chemistry applications where precise aromatic functionalization is required.Expand Specific Solutions04 Optimization of reaction conditions for aromatic organometallic transformations
The efficiency of reactions between aromatic compounds and organometallics can be significantly enhanced through careful optimization of reaction parameters such as temperature, pressure, solvent systems, and reaction time. Specific reaction conditions including the use of appropriate bases, additives, and controlled atmospheres have been developed to maximize conversion rates and minimize side reactions in aromatic organometallic chemistry.Expand Specific Solutions05 Development of novel organometallic complexes for aromatic functionalization
New organometallic complexes with improved stability and reactivity have been developed specifically for aromatic compound functionalization. These novel complexes often incorporate specialized metal centers and unique coordination environments that enable previously challenging transformations. The development of such complexes has expanded the scope of accessible aromatic derivatives and improved overall reaction efficiency in synthetic applications.Expand Specific Solutions
Core Innovations in Aromatic-Organometallic Interactions
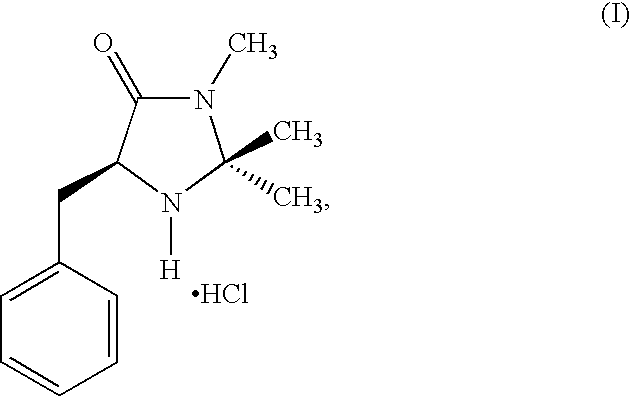
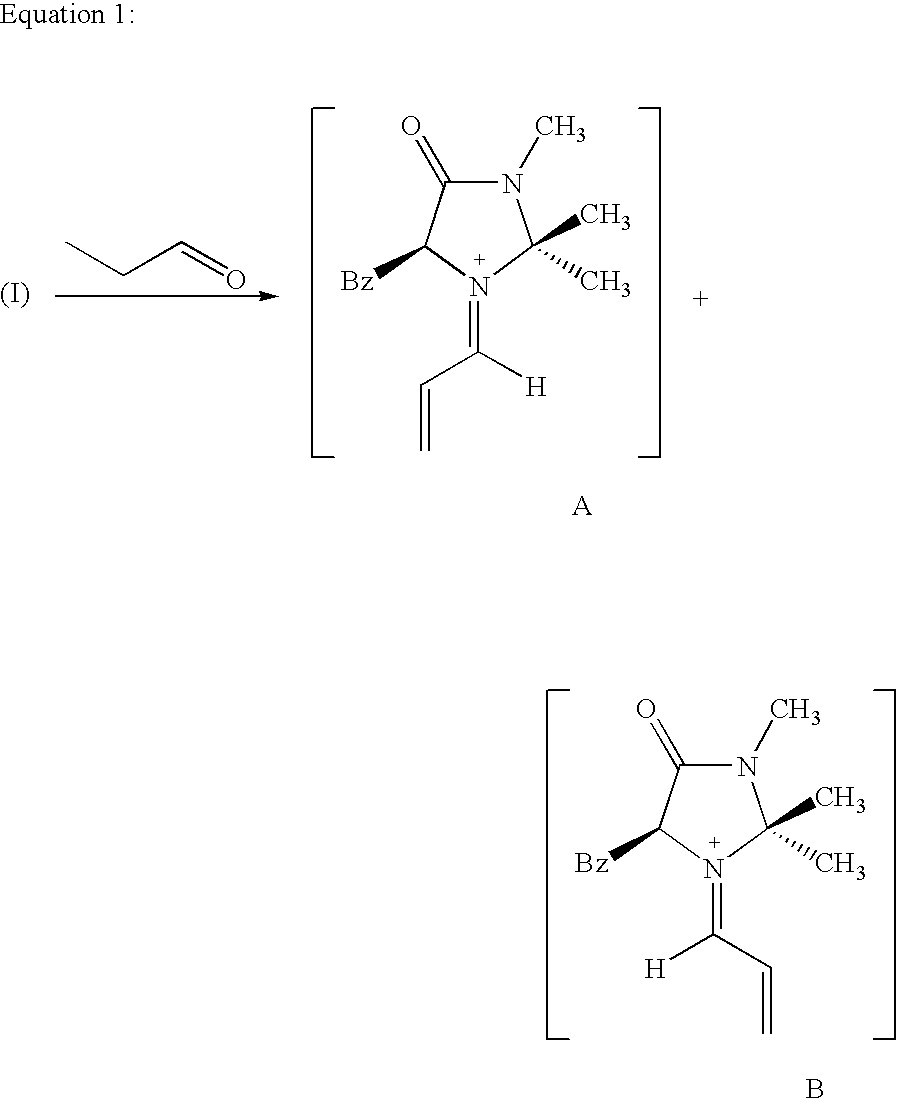
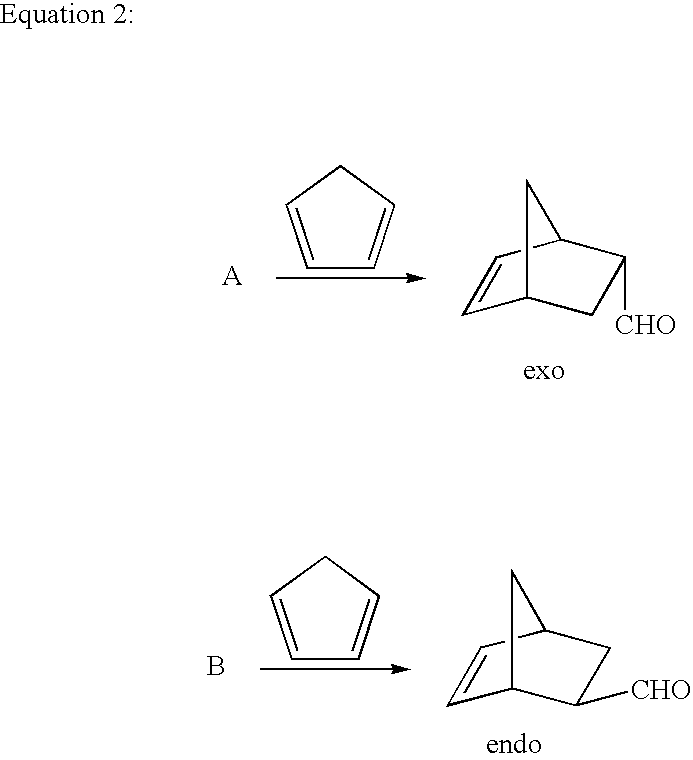
Enantioselective 1,4-addition of aromatic nucleophiles to alpha,beta-unsaturated aldehydes using chiral organic catalysts
PatentInactiveUS7173139B2
Innovation
- The use of nonmetallic, organic imidazolidinone compounds as catalysts, specifically in the form of acid addition salts, to lower the LUMO of α,β-unsaturated aldehydes, enabling enantioselective nucleophilic additions with high enantiofacial discrimination and compatibility with aerobic conditions.
Use of N-oxide compounds in coupling reactions
PatentInactiveUS20080132698A1
Innovation
- The use of N-oxides, such as pyridine N-oxides, as bench-stable replacements in metal-catalyzed coupling reactions, allowing for direct arylation with aryl bromides and providing a cost-effective and stable alternative to traditional organometallics, with specific conditions optimizing yields and selectivity.
Environmental Impact of Organometallic Processes
The environmental implications of organometallic processes present significant challenges that must be carefully evaluated when comparing reaction efficiency between aromatic compounds and organometallic systems. Traditional organometallic catalysis often relies on precious metals such as palladium, platinum, and rhodium, which carry substantial environmental burdens throughout their lifecycle from mining to disposal.
Metal extraction and purification processes generate considerable carbon footprints and toxic waste streams. Mining operations for precious metals typically involve energy-intensive procedures and can result in habitat destruction and water contamination. The scarcity of these metals also raises sustainability concerns, as their continued use in large-scale industrial processes may not be viable long-term without effective recycling strategies.
Organometallic reactions frequently require harsh reaction conditions, including high temperatures, inert atmospheres, and anhydrous solvents. These conditions contribute to increased energy consumption and generate volatile organic compound emissions. Additionally, many organometallic processes utilize halogenated solvents and phosphine ligands, which pose toxicity risks and create challenging waste disposal requirements.
The catalyst recovery and recycling efficiency significantly impacts the overall environmental footprint. While homogeneous organometallic catalysts often demonstrate superior selectivity, their separation from reaction products typically requires additional purification steps, generating secondary waste streams. Heterogeneous alternatives may offer easier recovery but often sacrifice reaction efficiency.
Emerging green chemistry approaches are addressing these environmental concerns through the development of earth-abundant metal catalysts, aqueous reaction media, and flow chemistry techniques. Iron, copper, and nickel-based systems are gaining attention as environmentally benign alternatives, though they may require optimization to match the efficiency of precious metal catalysts.
The regulatory landscape increasingly emphasizes environmental impact assessment, with stricter guidelines on metal emissions and waste management. This trend is driving innovation toward more sustainable organometallic processes that balance reaction efficiency with environmental responsibility, ultimately influencing the comparative advantages between aromatic and organometallic reaction pathways.
Metal extraction and purification processes generate considerable carbon footprints and toxic waste streams. Mining operations for precious metals typically involve energy-intensive procedures and can result in habitat destruction and water contamination. The scarcity of these metals also raises sustainability concerns, as their continued use in large-scale industrial processes may not be viable long-term without effective recycling strategies.
Organometallic reactions frequently require harsh reaction conditions, including high temperatures, inert atmospheres, and anhydrous solvents. These conditions contribute to increased energy consumption and generate volatile organic compound emissions. Additionally, many organometallic processes utilize halogenated solvents and phosphine ligands, which pose toxicity risks and create challenging waste disposal requirements.
The catalyst recovery and recycling efficiency significantly impacts the overall environmental footprint. While homogeneous organometallic catalysts often demonstrate superior selectivity, their separation from reaction products typically requires additional purification steps, generating secondary waste streams. Heterogeneous alternatives may offer easier recovery but often sacrifice reaction efficiency.
Emerging green chemistry approaches are addressing these environmental concerns through the development of earth-abundant metal catalysts, aqueous reaction media, and flow chemistry techniques. Iron, copper, and nickel-based systems are gaining attention as environmentally benign alternatives, though they may require optimization to match the efficiency of precious metal catalysts.
The regulatory landscape increasingly emphasizes environmental impact assessment, with stricter guidelines on metal emissions and waste management. This trend is driving innovation toward more sustainable organometallic processes that balance reaction efficiency with environmental responsibility, ultimately influencing the comparative advantages between aromatic and organometallic reaction pathways.
Safety Protocols for Organometallic Compound Handling
Organometallic compounds present unique safety challenges due to their inherent reactivity, air and moisture sensitivity, and potential toxicity. These compounds often exhibit extreme sensitivity to oxygen and water, leading to violent reactions that can produce toxic gases, fires, or explosions. The pyrophoric nature of many organometallic species, such as alkyllithium reagents and Grignard compounds, necessitates stringent handling protocols to prevent accidental ignition upon exposure to air.
Personal protective equipment forms the cornerstone of safe organometallic handling. Laboratory personnel must utilize appropriate gloves resistant to organic solvents and potential permeation by organometallic compounds. Nitrile or neoprene gloves are typically preferred over latex due to superior chemical resistance. Safety glasses with side shields and laboratory coats made from flame-resistant materials are mandatory. In cases involving highly toxic or volatile organometallic compounds, full face shields and specialized respiratory protection may be required.
Inert atmosphere techniques are essential for maintaining compound integrity and preventing hazardous reactions. Schlenk line systems and glove boxes filled with nitrogen or argon provide controlled environments for manipulation. Proper purging procedures must be established to eliminate trace oxygen and moisture, with oxygen levels maintained below 1 ppm for highly sensitive compounds. Regular monitoring of atmosphere quality using oxygen and moisture analyzers ensures optimal conditions.
Storage protocols require specialized considerations for organometallic compounds. These materials must be stored under inert atmospheres in appropriate containers, often requiring refrigeration or freezing to maintain stability. Segregation from incompatible materials, particularly oxidizers and protic solvents, prevents dangerous reactions. Inventory management systems should track expiration dates and degradation indicators, as aged organometallic compounds may become more hazardous.
Emergency response procedures must address the unique risks associated with organometallic compound incidents. Fire suppression systems should utilize appropriate extinguishing agents, as water-based systems can exacerbate reactions with water-sensitive compounds. Class D fire extinguishers containing dry powder are typically recommended for organometallic fires. Spill response protocols require immediate isolation of the area, elimination of ignition sources, and neutralization using appropriate quenching agents under inert atmosphere conditions.
Training programs for personnel handling organometallic compounds should encompass both theoretical knowledge and practical skills. Regular safety audits and refresher training ensure continued compliance with established protocols and adaptation to evolving safety standards.
Personal protective equipment forms the cornerstone of safe organometallic handling. Laboratory personnel must utilize appropriate gloves resistant to organic solvents and potential permeation by organometallic compounds. Nitrile or neoprene gloves are typically preferred over latex due to superior chemical resistance. Safety glasses with side shields and laboratory coats made from flame-resistant materials are mandatory. In cases involving highly toxic or volatile organometallic compounds, full face shields and specialized respiratory protection may be required.
Inert atmosphere techniques are essential for maintaining compound integrity and preventing hazardous reactions. Schlenk line systems and glove boxes filled with nitrogen or argon provide controlled environments for manipulation. Proper purging procedures must be established to eliminate trace oxygen and moisture, with oxygen levels maintained below 1 ppm for highly sensitive compounds. Regular monitoring of atmosphere quality using oxygen and moisture analyzers ensures optimal conditions.
Storage protocols require specialized considerations for organometallic compounds. These materials must be stored under inert atmospheres in appropriate containers, often requiring refrigeration or freezing to maintain stability. Segregation from incompatible materials, particularly oxidizers and protic solvents, prevents dangerous reactions. Inventory management systems should track expiration dates and degradation indicators, as aged organometallic compounds may become more hazardous.
Emergency response procedures must address the unique risks associated with organometallic compound incidents. Fire suppression systems should utilize appropriate extinguishing agents, as water-based systems can exacerbate reactions with water-sensitive compounds. Class D fire extinguishers containing dry powder are typically recommended for organometallic fires. Spill response protocols require immediate isolation of the area, elimination of ignition sources, and neutralization using appropriate quenching agents under inert atmosphere conditions.
Training programs for personnel handling organometallic compounds should encompass both theoretical knowledge and practical skills. Regular safety audits and refresher training ensure continued compliance with established protocols and adaptation to evolving safety standards.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!