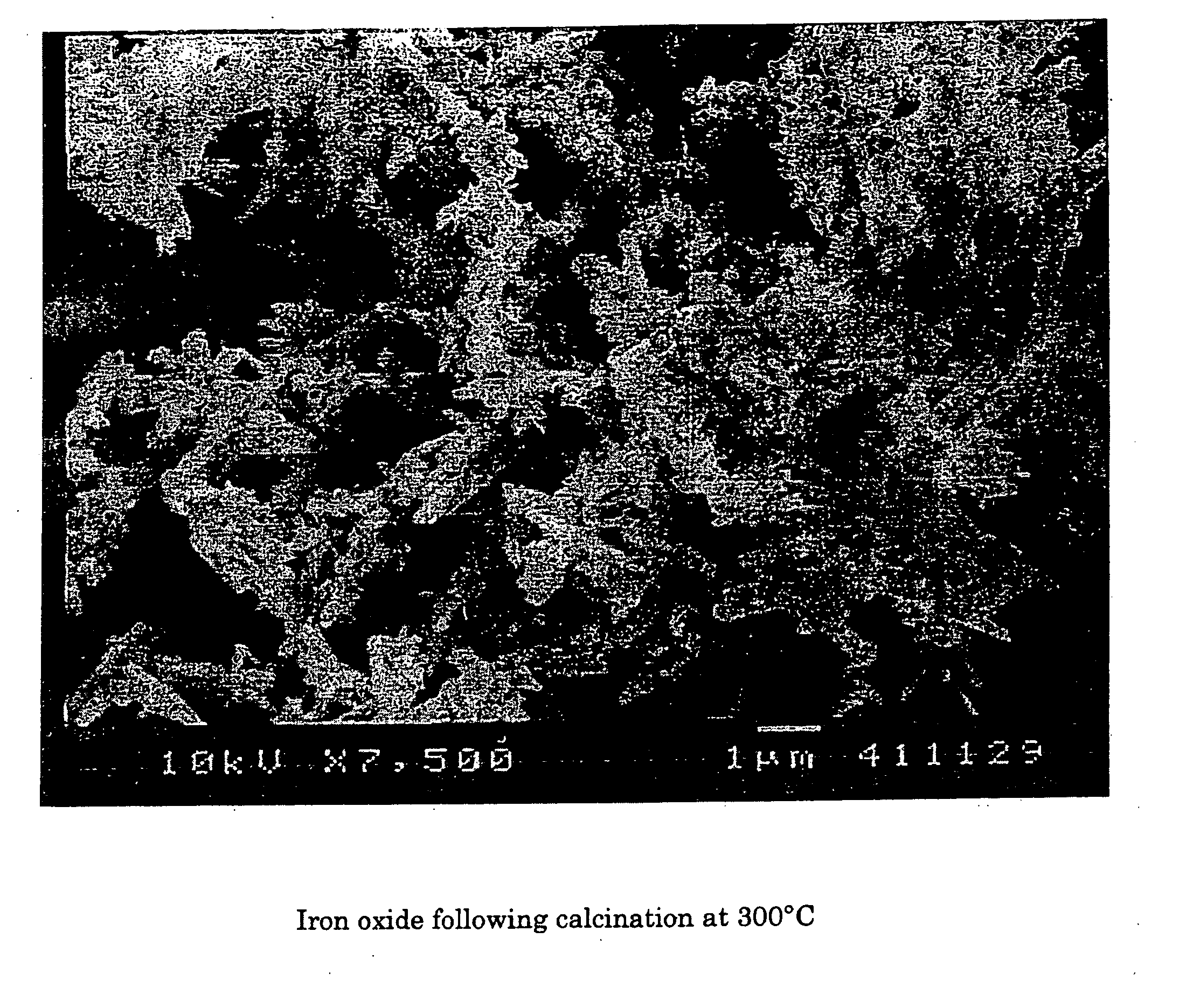
Compare Iron Oxide Functions in Varied Environments
FEB 12, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Iron Oxide Tech Background and Objectives
Iron oxide materials have been fundamental to human civilization for millennia, evolving from simple pigments and construction materials to sophisticated functional compounds in modern technology. The historical trajectory reveals a progression from naturally occurring minerals like hematite and magnetite used in ancient metallurgy to precisely engineered nanostructures designed for specific applications. This evolution reflects deepening understanding of how iron oxide properties respond to environmental variables including pH, temperature, redox potential, and the presence of organic or inorganic ligands.
The contemporary significance of iron oxides stems from their polymorphic nature, with distinct phases such as α-Fe2O3, γ-Fe2O3, Fe3O4, and FeOOH exhibiting dramatically different behaviors under varying conditions. Environmental context fundamentally alters their catalytic activity, magnetic properties, electron transfer capabilities, and structural stability. In aqueous environments, iron oxides function as adsorbents and redox mediators; in atmospheric conditions, they influence aerosol chemistry and climate processes; within biological systems, they participate in metabolic pathways and can exhibit toxicity or therapeutic effects.
Recent technological advances have intensified interest in environment-dependent iron oxide functionality. Applications spanning environmental remediation, energy storage, biomedical devices, and catalysis demand precise control over performance across diverse operational conditions. However, the complexity of iron oxide behavior in heterogeneous environments presents significant challenges for predictable application design.
The primary objective of this technical investigation is to systematically compare and characterize how iron oxide functions transform across varied environmental contexts. This encompasses understanding phase stability transitions, surface chemistry modifications, reactivity pattern changes, and performance degradation mechanisms under different conditions. A secondary objective involves identifying environmental parameters that most critically influence functional outcomes, enabling predictive modeling and rational material design. Ultimately, this research aims to establish comprehensive frameworks for selecting appropriate iron oxide phases and formulations for specific environmental applications, while revealing opportunities for novel functionalities through controlled environmental manipulation.
The contemporary significance of iron oxides stems from their polymorphic nature, with distinct phases such as α-Fe2O3, γ-Fe2O3, Fe3O4, and FeOOH exhibiting dramatically different behaviors under varying conditions. Environmental context fundamentally alters their catalytic activity, magnetic properties, electron transfer capabilities, and structural stability. In aqueous environments, iron oxides function as adsorbents and redox mediators; in atmospheric conditions, they influence aerosol chemistry and climate processes; within biological systems, they participate in metabolic pathways and can exhibit toxicity or therapeutic effects.
Recent technological advances have intensified interest in environment-dependent iron oxide functionality. Applications spanning environmental remediation, energy storage, biomedical devices, and catalysis demand precise control over performance across diverse operational conditions. However, the complexity of iron oxide behavior in heterogeneous environments presents significant challenges for predictable application design.
The primary objective of this technical investigation is to systematically compare and characterize how iron oxide functions transform across varied environmental contexts. This encompasses understanding phase stability transitions, surface chemistry modifications, reactivity pattern changes, and performance degradation mechanisms under different conditions. A secondary objective involves identifying environmental parameters that most critically influence functional outcomes, enabling predictive modeling and rational material design. Ultimately, this research aims to establish comprehensive frameworks for selecting appropriate iron oxide phases and formulations for specific environmental applications, while revealing opportunities for novel functionalities through controlled environmental manipulation.
Market Demand for Iron Oxide Applications
The global market for iron oxide spans multiple high-value sectors, driven by diverse functional requirements across varied environmental conditions. Construction and coatings industries represent the largest consumption segments, where iron oxides serve as pigments and protective agents. The demand in these sectors is propelled by urbanization trends and infrastructure development, particularly in emerging economies where construction activities continue to expand rapidly. Environmental stability and color retention properties make iron oxides indispensable in exterior architectural applications.
The electronics and data storage sector demonstrates accelerating demand for high-purity iron oxide nanoparticles. Magnetic recording media, sensors, and semiconductor applications require iron oxides with precisely controlled particle sizes and crystalline structures. This segment values performance in controlled environments where electromagnetic properties must remain consistent under varying temperature and humidity conditions.
Biomedical applications constitute a rapidly growing niche market with premium pricing potential. Iron oxide nanoparticles are increasingly utilized in magnetic resonance imaging contrast agents, targeted drug delivery systems, and hyperthermia cancer treatments. The biomedical sector demands iron oxides that maintain stability and biocompatibility in physiological environments, driving research into surface modifications and functionalization techniques.
Environmental remediation represents an emerging application area with substantial growth prospects. Iron oxides demonstrate effectiveness in water treatment, soil decontamination, and catalytic degradation of pollutants. Regulatory pressures regarding environmental protection and water quality standards are expanding market opportunities for iron oxide-based remediation technologies, particularly in industrial wastewater treatment facilities.
The energy storage sector shows increasing interest in iron oxide materials for battery electrodes and catalysts. Lithium-ion batteries and fuel cells benefit from iron oxide's electrochemical properties, especially when optimized for specific operating environments. Cost advantages compared to precious metal catalysts position iron oxides favorably for large-scale energy applications.
Agricultural applications leverage iron oxide's nutrient delivery capabilities and antimicrobial properties. Controlled-release fertilizers and crop protection formulations incorporate iron oxides to enhance efficacy across diverse soil and climate conditions. Growing emphasis on sustainable agriculture practices supports demand for iron oxide-based agricultural solutions that minimize environmental impact while maintaining effectiveness.
The electronics and data storage sector demonstrates accelerating demand for high-purity iron oxide nanoparticles. Magnetic recording media, sensors, and semiconductor applications require iron oxides with precisely controlled particle sizes and crystalline structures. This segment values performance in controlled environments where electromagnetic properties must remain consistent under varying temperature and humidity conditions.
Biomedical applications constitute a rapidly growing niche market with premium pricing potential. Iron oxide nanoparticles are increasingly utilized in magnetic resonance imaging contrast agents, targeted drug delivery systems, and hyperthermia cancer treatments. The biomedical sector demands iron oxides that maintain stability and biocompatibility in physiological environments, driving research into surface modifications and functionalization techniques.
Environmental remediation represents an emerging application area with substantial growth prospects. Iron oxides demonstrate effectiveness in water treatment, soil decontamination, and catalytic degradation of pollutants. Regulatory pressures regarding environmental protection and water quality standards are expanding market opportunities for iron oxide-based remediation technologies, particularly in industrial wastewater treatment facilities.
The energy storage sector shows increasing interest in iron oxide materials for battery electrodes and catalysts. Lithium-ion batteries and fuel cells benefit from iron oxide's electrochemical properties, especially when optimized for specific operating environments. Cost advantages compared to precious metal catalysts position iron oxides favorably for large-scale energy applications.
Agricultural applications leverage iron oxide's nutrient delivery capabilities and antimicrobial properties. Controlled-release fertilizers and crop protection formulations incorporate iron oxides to enhance efficacy across diverse soil and climate conditions. Growing emphasis on sustainable agriculture practices supports demand for iron oxide-based agricultural solutions that minimize environmental impact while maintaining effectiveness.
Current Status of Iron Oxide in Different Environments
Iron oxide materials have demonstrated remarkable versatility across diverse environmental conditions, establishing themselves as critical functional materials in multiple industrial and scientific domains. The current deployment of iron oxides spans aqueous systems, atmospheric environments, biological matrices, and extreme conditions, each leveraging distinct physicochemical properties of various iron oxide phases including magnetite, hematite, and maghemite.
In aqueous environments, iron oxides function primarily as adsorbents and catalysts for water treatment applications. Their high surface area and reactive hydroxyl groups enable effective removal of heavy metals, arsenic, and organic pollutants from contaminated water sources. Recent implementations have shown iron oxide nanoparticles achieving removal efficiencies exceeding 90% for chromium and lead ions. Additionally, their photocatalytic properties under visible light irradiation facilitate degradation of persistent organic compounds in wastewater treatment facilities.
Atmospheric applications of iron oxides center on air purification and catalytic conversion processes. Iron oxide-based catalysts demonstrate significant activity in selective catalytic reduction of nitrogen oxides, with industrial installations reporting NOx conversion rates above 85% at operating temperatures between 300-400°C. The material's stability under oxidative conditions and resistance to sulfur poisoning make it particularly suitable for flue gas treatment in power generation facilities.
Within biological environments, iron oxide nanoparticles have gained prominence in biomedical applications including magnetic resonance imaging contrast enhancement, targeted drug delivery, and hyperthermia cancer treatment. Their biocompatibility and magnetic responsiveness enable precise control in physiological conditions, with current clinical trials demonstrating tumor targeting efficiencies of 60-75% using surface-functionalized magnetite nanoparticles.
In extreme environments such as high-temperature industrial processes and corrosive chemical reactors, iron oxides serve as protective coatings and structural materials. Their thermal stability up to 1400°C and chemical inertness provide essential protection in metallurgical operations and petrochemical refining units. However, performance limitations emerge under highly acidic conditions below pH 2, where dissolution rates increase significantly, and in strongly reducing atmospheres where phase transformations compromise structural integrity. These environmental constraints currently define the operational boundaries for iron oxide deployment across different application sectors.
In aqueous environments, iron oxides function primarily as adsorbents and catalysts for water treatment applications. Their high surface area and reactive hydroxyl groups enable effective removal of heavy metals, arsenic, and organic pollutants from contaminated water sources. Recent implementations have shown iron oxide nanoparticles achieving removal efficiencies exceeding 90% for chromium and lead ions. Additionally, their photocatalytic properties under visible light irradiation facilitate degradation of persistent organic compounds in wastewater treatment facilities.
Atmospheric applications of iron oxides center on air purification and catalytic conversion processes. Iron oxide-based catalysts demonstrate significant activity in selective catalytic reduction of nitrogen oxides, with industrial installations reporting NOx conversion rates above 85% at operating temperatures between 300-400°C. The material's stability under oxidative conditions and resistance to sulfur poisoning make it particularly suitable for flue gas treatment in power generation facilities.
Within biological environments, iron oxide nanoparticles have gained prominence in biomedical applications including magnetic resonance imaging contrast enhancement, targeted drug delivery, and hyperthermia cancer treatment. Their biocompatibility and magnetic responsiveness enable precise control in physiological conditions, with current clinical trials demonstrating tumor targeting efficiencies of 60-75% using surface-functionalized magnetite nanoparticles.
In extreme environments such as high-temperature industrial processes and corrosive chemical reactors, iron oxides serve as protective coatings and structural materials. Their thermal stability up to 1400°C and chemical inertness provide essential protection in metallurgical operations and petrochemical refining units. However, performance limitations emerge under highly acidic conditions below pH 2, where dissolution rates increase significantly, and in strongly reducing atmospheres where phase transformations compromise structural integrity. These environmental constraints currently define the operational boundaries for iron oxide deployment across different application sectors.
Existing Iron Oxide Solutions Across Environments
01 Iron oxide as pigment and colorant in cosmetic formulations
Iron oxides are widely used as pigments and colorants in various cosmetic and personal care products. They provide color stability, opacity, and coverage in formulations such as foundations, lipsticks, eye shadows, and other makeup products. Iron oxides are available in different shades including red, yellow, and black, allowing for versatile color customization in cosmetic applications.- Iron oxide as a pigment and colorant: Iron oxides are widely used as pigments and colorants in various applications due to their excellent color properties and stability. They provide a range of colors from yellow to red to black depending on their chemical composition and particle size. These pigments are commonly incorporated into cosmetic formulations, coatings, and other products where color stability and safety are important considerations.
- Iron oxide for UV protection and sun screening: Iron oxides function as physical UV blockers and are incorporated into sunscreen and cosmetic formulations to provide protection against harmful ultraviolet radiation. Their ability to reflect and scatter UV light makes them effective sun protection agents. These materials are particularly valued for their photostability and broad-spectrum protection capabilities in topical applications.
- Iron oxide as a catalyst or catalytic component: Iron oxides serve as catalysts or catalytic supports in various chemical reactions and industrial processes. Their catalytic properties can be utilized in oxidation reactions, synthesis processes, and environmental applications. The specific surface area, crystal structure, and oxidation state of iron oxides influence their catalytic activity and selectivity in different reaction systems.
- Iron oxide for magnetic properties and applications: Iron oxides exhibit magnetic properties that make them useful in various technological applications. These materials can be utilized in magnetic recording media, magnetic separation processes, and biomedical applications. The magnetic characteristics of iron oxides depend on their crystal structure, particle size, and morphology, which can be tailored for specific functional requirements.
- Iron oxide as a functional additive in pharmaceutical and cosmetic formulations: Iron oxides are incorporated into pharmaceutical and cosmetic products as functional additives to improve product performance and stability. They can enhance texture, provide opacity, improve adhesion properties, and contribute to the overall aesthetic quality of formulations. These materials are valued for their biocompatibility and regulatory acceptance in personal care and pharmaceutical applications.
02 Iron oxide as UV protection agent in sunscreen products
Iron oxides function as physical UV blockers in sunscreen and sun protection formulations. They provide broad-spectrum protection by reflecting and scattering both UVA and UVB radiation. As mineral-based ingredients, iron oxides offer photostable protection without chemical degradation, making them suitable for sensitive skin formulations and enhancing the sun protection factor of products.Expand Specific Solutions03 Iron oxide in pharmaceutical and drug delivery applications
Iron oxides serve multiple functions in pharmaceutical formulations including use as coating agents for tablets and capsules, contrast agents for medical imaging, and carriers in drug delivery systems. They provide controlled release properties, improve bioavailability, and can be functionalized for targeted delivery applications. Iron oxide nanoparticles are particularly useful in advanced drug delivery systems.Expand Specific Solutions04 Iron oxide as catalyst and functional material in industrial processes
Iron oxides function as catalysts and functional materials in various industrial and chemical processes. They exhibit catalytic activity in oxidation and reduction reactions, serve as magnetic materials in separation processes, and act as adsorbents for pollutant removal. Their magnetic properties and high surface area make them valuable in environmental remediation and chemical synthesis applications.Expand Specific Solutions05 Iron oxide in food and nutritional supplement applications
Iron oxides are utilized in food products and nutritional supplements as iron fortification agents and colorants. They serve as a source of dietary iron to address iron deficiency and anemia, while also providing coloring properties in food applications. Iron oxides used in food applications must meet specific purity and safety standards for human consumption.Expand Specific Solutions
Major Players in Iron Oxide Industry
The iron oxide functionality research field demonstrates a mature yet evolving competitive landscape, characterized by diverse applications spanning industrial manufacturing, environmental remediation, and advanced materials development. The market exhibits steady growth driven by increasing demands in catalysis, water treatment, and energy storage sectors. Major industrial players including JFE Steel Corp., Umicore SA, BASF Corp., and Lam Research Corp. lead commercialization efforts, while chemical manufacturers like ISHIHARA SANGYO KAISHA Ltd. and Mitsubishi Gas Chemical Co. focus on production optimization. Academic institutions such as Nanyang Technological University, Johns Hopkins University, and Tianjin University contribute fundamental research breakthroughs. The technology maturity varies significantly across applications: traditional uses in steel manufacturing and pigments represent established markets, whereas emerging applications in semiconductor processing, electrochemistry, and environmental catalysis remain in development stages, indicating substantial innovation potential and market expansion opportunities ahead.
JFE Steel Corp.
Technical Solution: JFE Steel has developed iron oxide technologies primarily focused on metallurgical and industrial process environments. Their iron oxide products function as sintering additives in steelmaking processes, operating effectively in high-temperature oxidizing atmospheres (1000-1300°C) to improve ore agglomeration and reduce energy consumption. JFE's iron oxide formulations for blast furnace operations demonstrate controlled reduction behavior across temperature gradients, optimizing iron production efficiency. In environmental control applications, JFE produces iron oxide-based materials for industrial wastewater treatment, particularly for removing phosphates and heavy metals in neutral to alkaline conditions (pH 7-10). Their iron oxide dust collection and recycling systems function in harsh industrial atmospheres containing particulates, acidic gases, and elevated temperatures. JFE has also developed iron oxide coatings for corrosion protection in marine and industrial environments, demonstrating durability in chloride-rich atmospheres and cyclic wet-dry conditions typical of coastal steel structures.
Strengths: Extensive experience in high-temperature industrial applications, robust materials designed for harsh process environments, integrated approach combining iron oxide production with steel manufacturing expertise. Weaknesses: Primary focus on heavy industrial applications with limited diversification into emerging markets, less emphasis on nanoscale or advanced functional materials, environmental footprint concerns associated with traditional steel industry operations.
Umicore SA
Technical Solution: Umicore specializes in functional iron oxide materials for catalytic and environmental applications across multiple operational environments. Their iron oxide catalyst systems are engineered for automotive emission control, functioning effectively in high-temperature exhaust environments (400-600°C) with resistance to thermal degradation and poisoning. In aqueous systems, Umicore's iron oxide materials serve as active components in electrochemical processes, including water electrolysis and fuel cell applications, demonstrating stable performance in both acidic and alkaline electrolytes. Their iron oxide formulations for battery applications function across wide temperature ranges (-30°C to 60°C) with maintained electrochemical activity. Umicore has developed surface-engineered iron oxide particles that exhibit controlled reactivity in atmospheric, aqueous, and soil environments, with particular emphasis on recyclability and circular economy principles. Their materials demonstrate consistent performance in humid conditions (up to 95% relative humidity) and corrosive atmospheres containing sulfur and nitrogen compounds.
Strengths: Advanced surface engineering capabilities, excellent thermal and chemical stability, strong focus on sustainable and recyclable materials, proven performance in harsh industrial environments. Weaknesses: Premium pricing structure, limited availability for small-scale applications, specialized handling requirements for some formulations.
Core Technologies in Iron Oxide Functionality
A method for facile, rapid and industrially scalable preparation of metal hydroxide composition
PatentActiveIN201941054546A
Innovation
- A rapid and cost-effective method involving the hydrolysis of metal ions below 40°C with a non-ionic dispersant and ionic settling agent, followed by immediate filtration and drying, to produce monodispersed, poorly crystalline metal hydroxides that are easily filterable and stable, allowing for scalable production and enhanced filtration capacity.
Low sulfur red iron oxide useful as a catalyst or catalyst component and a process for making low sulfur red iron oxide
PatentInactiveUS20070014716A1
Innovation
- A process involving careful washing and low-temperature calcination of yellow iron oxide to reduce sulfur content below 0.15% without deforming the iron oxide, allowing for the preservation of acicular shape and enhanced catalyst properties.
Environmental Impact Assessment of Iron Oxide
Iron oxide compounds exhibit distinct environmental behaviors across different ecological settings, necessitating comprehensive impact assessments that account for their multifaceted interactions with natural systems. The environmental implications of iron oxide deployment vary significantly depending on atmospheric conditions, aquatic environments, soil compositions, and industrial contexts, each presenting unique pathways for ecological influence.
In aquatic ecosystems, iron oxide nanoparticles demonstrate complex bioavailability patterns that affect both water quality and aquatic organisms. Studies indicate that pH levels, dissolved organic matter, and ionic strength substantially modify iron oxide solubility and aggregation behavior. These particles can adsorb heavy metals and organic pollutants, potentially serving remediation functions, yet simultaneously may accumulate in sediments and enter food chains through bioaccumulation processes. The persistence of iron oxide in marine environments raises concerns about long-term ecological disruption, particularly affecting benthic communities and filter-feeding organisms.
Terrestrial environments present different assessment challenges, where iron oxide interactions with soil microbiomes and plant root systems require careful evaluation. Agricultural applications of iron oxide as fertilizer supplements or soil amendments demonstrate both beneficial nutrient delivery and potential phytotoxicity at elevated concentrations. Soil pH buffering capacity and organic content significantly influence iron oxide mobility and plant uptake efficiency, affecting crop productivity and soil health sustainability.
Atmospheric dispersion of iron oxide particulates from industrial sources introduces air quality considerations, with particle size distribution determining respiratory exposure risks and deposition patterns. Fine particulate iron oxides contribute to PM2.5 pollution levels, while larger particles settle more rapidly, affecting local soil chemistry. The photocatalytic properties of certain iron oxide forms may influence atmospheric chemistry through interactions with volatile organic compounds and nitrogen oxides.
Industrial wastewater containing iron oxide requires specialized treatment protocols to prevent ecosystem contamination. Discharge standards must address both dissolved iron species and suspended particulate forms, considering downstream ecological receptors and cumulative loading effects. Life cycle assessments increasingly incorporate iron oxide production energy requirements, raw material extraction impacts, and end-of-life disposal scenarios to provide holistic environmental footprint evaluations across varied application contexts.
In aquatic ecosystems, iron oxide nanoparticles demonstrate complex bioavailability patterns that affect both water quality and aquatic organisms. Studies indicate that pH levels, dissolved organic matter, and ionic strength substantially modify iron oxide solubility and aggregation behavior. These particles can adsorb heavy metals and organic pollutants, potentially serving remediation functions, yet simultaneously may accumulate in sediments and enter food chains through bioaccumulation processes. The persistence of iron oxide in marine environments raises concerns about long-term ecological disruption, particularly affecting benthic communities and filter-feeding organisms.
Terrestrial environments present different assessment challenges, where iron oxide interactions with soil microbiomes and plant root systems require careful evaluation. Agricultural applications of iron oxide as fertilizer supplements or soil amendments demonstrate both beneficial nutrient delivery and potential phytotoxicity at elevated concentrations. Soil pH buffering capacity and organic content significantly influence iron oxide mobility and plant uptake efficiency, affecting crop productivity and soil health sustainability.
Atmospheric dispersion of iron oxide particulates from industrial sources introduces air quality considerations, with particle size distribution determining respiratory exposure risks and deposition patterns. Fine particulate iron oxides contribute to PM2.5 pollution levels, while larger particles settle more rapidly, affecting local soil chemistry. The photocatalytic properties of certain iron oxide forms may influence atmospheric chemistry through interactions with volatile organic compounds and nitrogen oxides.
Industrial wastewater containing iron oxide requires specialized treatment protocols to prevent ecosystem contamination. Discharge standards must address both dissolved iron species and suspended particulate forms, considering downstream ecological receptors and cumulative loading effects. Life cycle assessments increasingly incorporate iron oxide production energy requirements, raw material extraction impacts, and end-of-life disposal scenarios to provide holistic environmental footprint evaluations across varied application contexts.
Performance Testing Standards for Iron Oxide
Establishing robust performance testing standards for iron oxide materials is essential to ensure their reliability and effectiveness across diverse environmental applications. These standards provide a systematic framework for evaluating key properties including magnetic characteristics, thermal stability, chemical reactivity, and mechanical durability. International organizations such as ISO and ASTM have developed baseline protocols, yet specific testing methodologies must be adapted to address the unique functional requirements of iron oxide in different operational contexts.
Physical characterization forms the foundation of performance evaluation, encompassing particle size distribution analysis through laser diffraction or electron microscopy, surface area measurement via BET methods, and crystallographic phase identification using X-ray diffraction. These fundamental assessments establish baseline material properties that directly influence performance outcomes. Magnetic property testing requires specialized equipment including vibrating sample magnetometers and superconducting quantum interference devices to measure saturation magnetization, coercivity, and remanence under controlled temperature and field conditions.
Chemical stability testing protocols evaluate iron oxide resistance to oxidation, reduction, and dissolution under simulated environmental conditions. Accelerated aging tests expose materials to elevated temperatures, humidity cycles, and corrosive atmospheres to predict long-term performance degradation. For catalytic applications, activity measurements must quantify conversion rates, selectivity, and deactivation kinetics under standardized reaction conditions. Electrochemical testing standards assess charge-discharge characteristics, cycling stability, and rate capability for energy storage applications.
Environmental simulation chambers replicate specific operational conditions including temperature extremes, pH variations, and atmospheric compositions. Comparative testing across multiple environmental scenarios requires standardized sample preparation, consistent measurement protocols, and statistical validation methods. Quality control procedures must incorporate batch-to-batch reproducibility assessments and inter-laboratory comparison studies to ensure data reliability.
Emerging testing standards increasingly incorporate real-time monitoring techniques and in-situ characterization methods that capture dynamic performance changes during actual operational conditions. Digital documentation systems and data management protocols facilitate comprehensive performance tracking and enable predictive modeling of material behavior across varied environmental parameters.
Physical characterization forms the foundation of performance evaluation, encompassing particle size distribution analysis through laser diffraction or electron microscopy, surface area measurement via BET methods, and crystallographic phase identification using X-ray diffraction. These fundamental assessments establish baseline material properties that directly influence performance outcomes. Magnetic property testing requires specialized equipment including vibrating sample magnetometers and superconducting quantum interference devices to measure saturation magnetization, coercivity, and remanence under controlled temperature and field conditions.
Chemical stability testing protocols evaluate iron oxide resistance to oxidation, reduction, and dissolution under simulated environmental conditions. Accelerated aging tests expose materials to elevated temperatures, humidity cycles, and corrosive atmospheres to predict long-term performance degradation. For catalytic applications, activity measurements must quantify conversion rates, selectivity, and deactivation kinetics under standardized reaction conditions. Electrochemical testing standards assess charge-discharge characteristics, cycling stability, and rate capability for energy storage applications.
Environmental simulation chambers replicate specific operational conditions including temperature extremes, pH variations, and atmospheric compositions. Comparative testing across multiple environmental scenarios requires standardized sample preparation, consistent measurement protocols, and statistical validation methods. Quality control procedures must incorporate batch-to-batch reproducibility assessments and inter-laboratory comparison studies to ensure data reliability.
Emerging testing standards increasingly incorporate real-time monitoring techniques and in-situ characterization methods that capture dynamic performance changes during actual operational conditions. Digital documentation systems and data management protocols facilitate comprehensive performance tracking and enable predictive modeling of material behavior across varied environmental parameters.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!