Iron Oxide and Cementite: Structural Comparison
FEB 12, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Iron Oxide vs Cementite Structure Background and Objectives
Iron-bearing phases represent fundamental constituents in metallurgical systems, with iron oxides and cementite serving as critical structural components in steel production and processing. Iron oxides, primarily existing as wüstite (FeO), magnetite (Fe₃O₄), and hematite (Fe₂O₃), constitute the predominant forms encountered during ore reduction and oxidation processes. Cementite (Fe₃C), conversely, functions as a key hardening phase in carbon steels, profoundly influencing mechanical properties through its distribution and morphology within the iron matrix.
The structural distinction between these phases extends beyond simple compositional differences, encompassing fundamental variations in crystallographic arrangements, bonding characteristics, and thermodynamic stability. Iron oxides exhibit ionic bonding with oxygen atoms occupying interstitial positions within iron lattices, while cementite demonstrates a complex orthorhombic structure where carbon atoms reside in specific crystallographic sites, creating a metastable intermetallic compound. These structural disparities directly correlate with divergent physical properties, including hardness, brittleness, magnetic behavior, and thermal stability.
Understanding the structural relationships between iron oxide and cementite has gained increasing significance in modern metallurgical applications. During steelmaking processes, the transformation between oxidized iron phases and carbide formations critically affects product quality and processing efficiency. The reduction of iron oxides to metallic iron, followed by carbon dissolution and cementite precipitation, represents a fundamental pathway in steel production. Additionally, surface oxidation phenomena and decarburization processes involve structural transitions between these phases, impacting material performance in high-temperature environments.
The primary objective of this structural comparison focuses on elucidating the crystallographic frameworks, atomic arrangements, and bonding mechanisms that differentiate iron oxides from cementite. This investigation aims to establish correlations between structural features and resultant material properties, providing theoretical foundations for optimizing steel processing parameters. Furthermore, understanding these structural distinctions enables prediction of phase transformation behaviors under varying thermal and chemical conditions, supporting development of advanced steel grades and innovative processing technologies. The comparative analysis seeks to bridge fundamental materials science with practical metallurgical applications, facilitating enhanced control over microstructural evolution in iron-based systems.
The structural distinction between these phases extends beyond simple compositional differences, encompassing fundamental variations in crystallographic arrangements, bonding characteristics, and thermodynamic stability. Iron oxides exhibit ionic bonding with oxygen atoms occupying interstitial positions within iron lattices, while cementite demonstrates a complex orthorhombic structure where carbon atoms reside in specific crystallographic sites, creating a metastable intermetallic compound. These structural disparities directly correlate with divergent physical properties, including hardness, brittleness, magnetic behavior, and thermal stability.
Understanding the structural relationships between iron oxide and cementite has gained increasing significance in modern metallurgical applications. During steelmaking processes, the transformation between oxidized iron phases and carbide formations critically affects product quality and processing efficiency. The reduction of iron oxides to metallic iron, followed by carbon dissolution and cementite precipitation, represents a fundamental pathway in steel production. Additionally, surface oxidation phenomena and decarburization processes involve structural transitions between these phases, impacting material performance in high-temperature environments.
The primary objective of this structural comparison focuses on elucidating the crystallographic frameworks, atomic arrangements, and bonding mechanisms that differentiate iron oxides from cementite. This investigation aims to establish correlations between structural features and resultant material properties, providing theoretical foundations for optimizing steel processing parameters. Furthermore, understanding these structural distinctions enables prediction of phase transformation behaviors under varying thermal and chemical conditions, supporting development of advanced steel grades and innovative processing technologies. The comparative analysis seeks to bridge fundamental materials science with practical metallurgical applications, facilitating enhanced control over microstructural evolution in iron-based systems.
Market Demand for Iron-Carbon Phase Materials
The global steel and iron-carbon alloy industry represents one of the largest material markets worldwide, with production volumes exceeding 1.9 billion metric tons annually. Within this vast sector, materials containing iron oxide and cementite phases constitute critical components across multiple industrial segments. The demand for these materials stems primarily from their fundamental roles in steel manufacturing, where understanding and controlling the transformation between iron oxides and iron-carbon phases directly impacts product quality and production efficiency.
Steel production facilities worldwide continuously seek to optimize the reduction process of iron oxides to metallic iron while precisely controlling carbon incorporation to form desired cementite structures. This dual requirement drives substantial market demand for advanced materials characterization and process control technologies. The automotive industry alone consumes significant quantities of high-strength steels where cementite distribution determines mechanical properties, creating persistent demand for materials with optimized iron-carbon phase compositions.
Emerging applications in advanced manufacturing sectors further amplify market requirements. The renewable energy sector, particularly wind turbine production, demands specialized steels with controlled cementite morphologies to ensure structural integrity under cyclic loading conditions. Similarly, the construction industry's shift toward high-performance structural materials necessitates precise control over iron-carbon phase distributions to achieve superior strength-to-weight ratios.
The market landscape also reflects growing demand from the powder metallurgy sector, where iron oxide powders serve as precursors for producing components with tailored cementite contents. This application area has experienced notable expansion due to additive manufacturing technologies requiring feedstock materials with specific phase compositions. Additionally, the magnetic materials industry utilizes iron oxides and controlled iron-carbon phases for producing soft magnetic components in electrical applications.
Regional demand patterns show concentration in industrialized economies with established steel industries, while emerging markets demonstrate accelerating consumption driven by infrastructure development. The ongoing transition toward sustainable manufacturing practices has intensified interest in optimizing iron oxide reduction processes and achieving precise cementite formation with reduced energy consumption, thereby creating new market opportunities for innovative material solutions and processing technologies.
Steel production facilities worldwide continuously seek to optimize the reduction process of iron oxides to metallic iron while precisely controlling carbon incorporation to form desired cementite structures. This dual requirement drives substantial market demand for advanced materials characterization and process control technologies. The automotive industry alone consumes significant quantities of high-strength steels where cementite distribution determines mechanical properties, creating persistent demand for materials with optimized iron-carbon phase compositions.
Emerging applications in advanced manufacturing sectors further amplify market requirements. The renewable energy sector, particularly wind turbine production, demands specialized steels with controlled cementite morphologies to ensure structural integrity under cyclic loading conditions. Similarly, the construction industry's shift toward high-performance structural materials necessitates precise control over iron-carbon phase distributions to achieve superior strength-to-weight ratios.
The market landscape also reflects growing demand from the powder metallurgy sector, where iron oxide powders serve as precursors for producing components with tailored cementite contents. This application area has experienced notable expansion due to additive manufacturing technologies requiring feedstock materials with specific phase compositions. Additionally, the magnetic materials industry utilizes iron oxides and controlled iron-carbon phases for producing soft magnetic components in electrical applications.
Regional demand patterns show concentration in industrialized economies with established steel industries, while emerging markets demonstrate accelerating consumption driven by infrastructure development. The ongoing transition toward sustainable manufacturing practices has intensified interest in optimizing iron oxide reduction processes and achieving precise cementite formation with reduced energy consumption, thereby creating new market opportunities for innovative material solutions and processing technologies.
Current Understanding and Challenges in Phase Structure Analysis
The structural analysis of iron oxide and cementite represents a fundamental challenge in materials science, particularly within the context of steel metallurgy and iron-based material development. Current understanding relies heavily on crystallographic characterization techniques, yet significant gaps remain in comprehensively mapping the structural relationships between these phases under various thermodynamic conditions.
Iron oxides exist in multiple polymorphs, including wüstite, magnetite, and hematite, each exhibiting distinct crystal structures ranging from cubic to rhombohedral symmetries. Cementite, with its orthorhombic structure and complex iron-carbon arrangement, presents a contrasting framework. The primary challenge lies in establishing precise structural correlations during phase transformations, particularly at interfaces where these phases coexist during oxidation or reduction processes.
Advanced characterization methods such as high-resolution transmission electron microscopy and synchrotron X-ray diffraction have enhanced our ability to probe atomic arrangements. However, limitations persist in capturing dynamic structural evolution during real-time phase transitions. The metastable nature of certain iron oxide phases and the sensitivity of cementite to decomposition under observation conditions complicate direct comparative studies.
A critical technical obstacle involves accurately determining lattice parameter variations and atomic position shifts during the transformation between oxidized and carbide phases. Computational approaches, including density functional theory calculations, have provided theoretical insights into energy landscapes and structural stability. Nevertheless, discrepancies between theoretical predictions and experimental observations highlight the need for improved modeling frameworks that account for defect structures, grain boundaries, and compositional gradients.
The interface structure between iron oxide scales and underlying cementite-containing substrates remains poorly understood, particularly regarding coherency relationships and strain accommodation mechanisms. This knowledge gap directly impacts the development of oxidation-resistant steels and the optimization of reduction processes in ironmaking. Furthermore, the influence of alloying elements on the structural characteristics of both phases introduces additional complexity that current analytical methods struggle to fully resolve.
Addressing these challenges requires integrated approaches combining in-situ characterization, advanced computational modeling, and systematic experimental validation across multiple length scales. The development of more sophisticated analytical protocols capable of operating under industrially relevant conditions represents a crucial step toward comprehensive structural understanding.
Iron oxides exist in multiple polymorphs, including wüstite, magnetite, and hematite, each exhibiting distinct crystal structures ranging from cubic to rhombohedral symmetries. Cementite, with its orthorhombic structure and complex iron-carbon arrangement, presents a contrasting framework. The primary challenge lies in establishing precise structural correlations during phase transformations, particularly at interfaces where these phases coexist during oxidation or reduction processes.
Advanced characterization methods such as high-resolution transmission electron microscopy and synchrotron X-ray diffraction have enhanced our ability to probe atomic arrangements. However, limitations persist in capturing dynamic structural evolution during real-time phase transitions. The metastable nature of certain iron oxide phases and the sensitivity of cementite to decomposition under observation conditions complicate direct comparative studies.
A critical technical obstacle involves accurately determining lattice parameter variations and atomic position shifts during the transformation between oxidized and carbide phases. Computational approaches, including density functional theory calculations, have provided theoretical insights into energy landscapes and structural stability. Nevertheless, discrepancies between theoretical predictions and experimental observations highlight the need for improved modeling frameworks that account for defect structures, grain boundaries, and compositional gradients.
The interface structure between iron oxide scales and underlying cementite-containing substrates remains poorly understood, particularly regarding coherency relationships and strain accommodation mechanisms. This knowledge gap directly impacts the development of oxidation-resistant steels and the optimization of reduction processes in ironmaking. Furthermore, the influence of alloying elements on the structural characteristics of both phases introduces additional complexity that current analytical methods struggle to fully resolve.
Addressing these challenges requires integrated approaches combining in-situ characterization, advanced computational modeling, and systematic experimental validation across multiple length scales. The development of more sophisticated analytical protocols capable of operating under industrially relevant conditions represents a crucial step toward comprehensive structural understanding.
Existing Characterization Methods for Crystal Structures
01 Microstructure control of iron oxide and cementite in steel
Methods for controlling the microstructure of steel materials by regulating the distribution and morphology of iron oxide and cementite phases. This involves heat treatment processes and cooling rate control to achieve desired mechanical properties. The microstructure optimization focuses on the balance between ferrite, cementite, and oxide phases to enhance strength and toughness.- Microstructure control of iron oxide and cementite in steel: Methods for controlling the microstructure of steel materials by regulating the distribution and morphology of iron oxide and cementite phases. This involves heat treatment processes and cooling rate control to achieve desired mechanical properties. The microstructure optimization focuses on the size, shape, and distribution of cementite particles within the iron oxide matrix to enhance strength and toughness.
- Phase transformation between iron oxide and cementite: Techniques for controlling phase transformation processes between different iron oxide phases and cementite formation during steel manufacturing. This includes understanding the thermodynamic conditions and kinetics that govern the transformation from austenite to ferrite with cementite precipitation. The control of transformation temperature and time is critical for achieving specific microstructural characteristics.
- Spheroidization of cementite in iron-based materials: Methods for spheroidizing cementite structures in iron-based alloys to improve machinability and mechanical properties. The process involves specific heat treatment cycles that transform lamellar or network cementite into spheroidal forms. This structural modification enhances ductility and reduces brittleness in the final product.
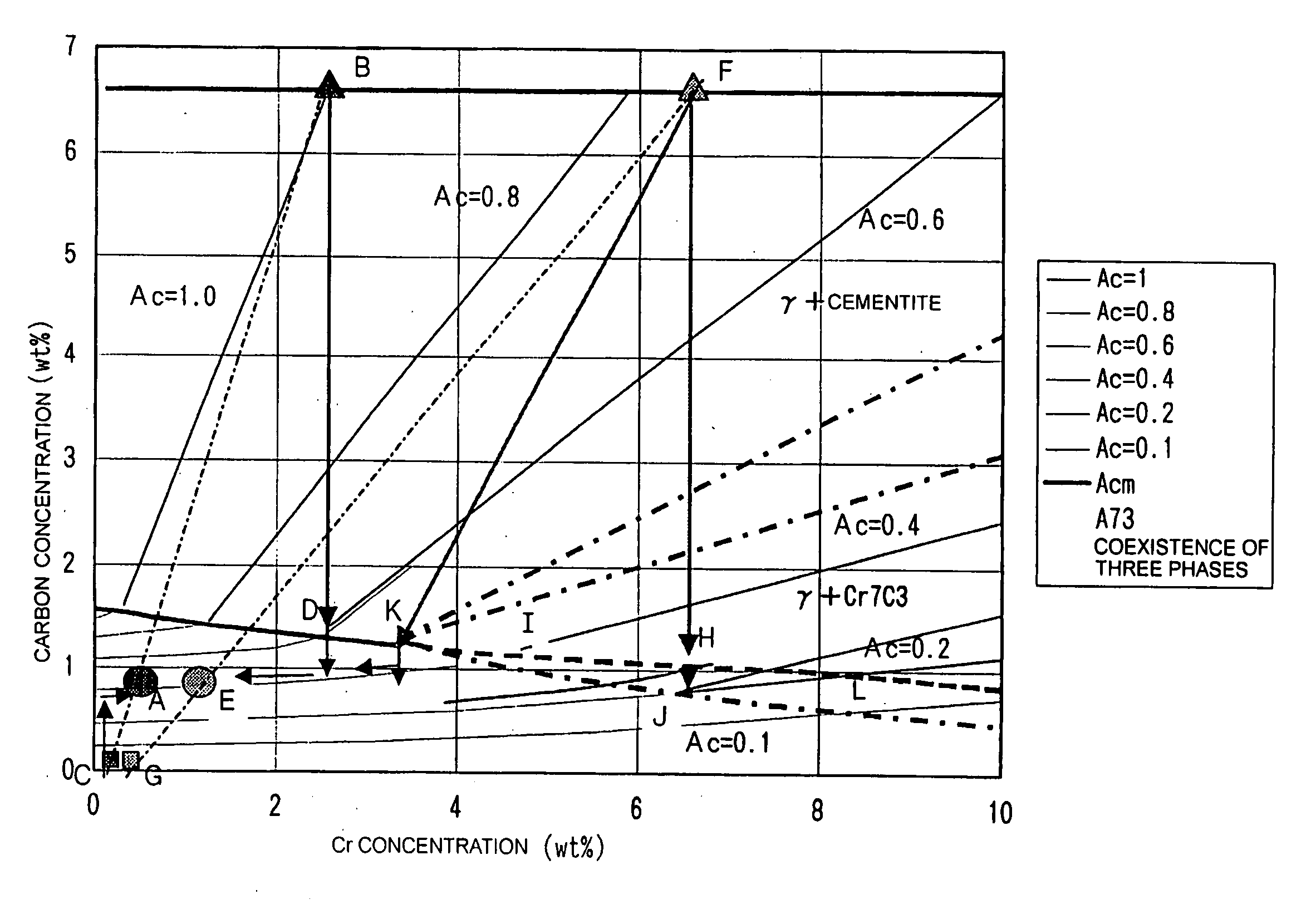
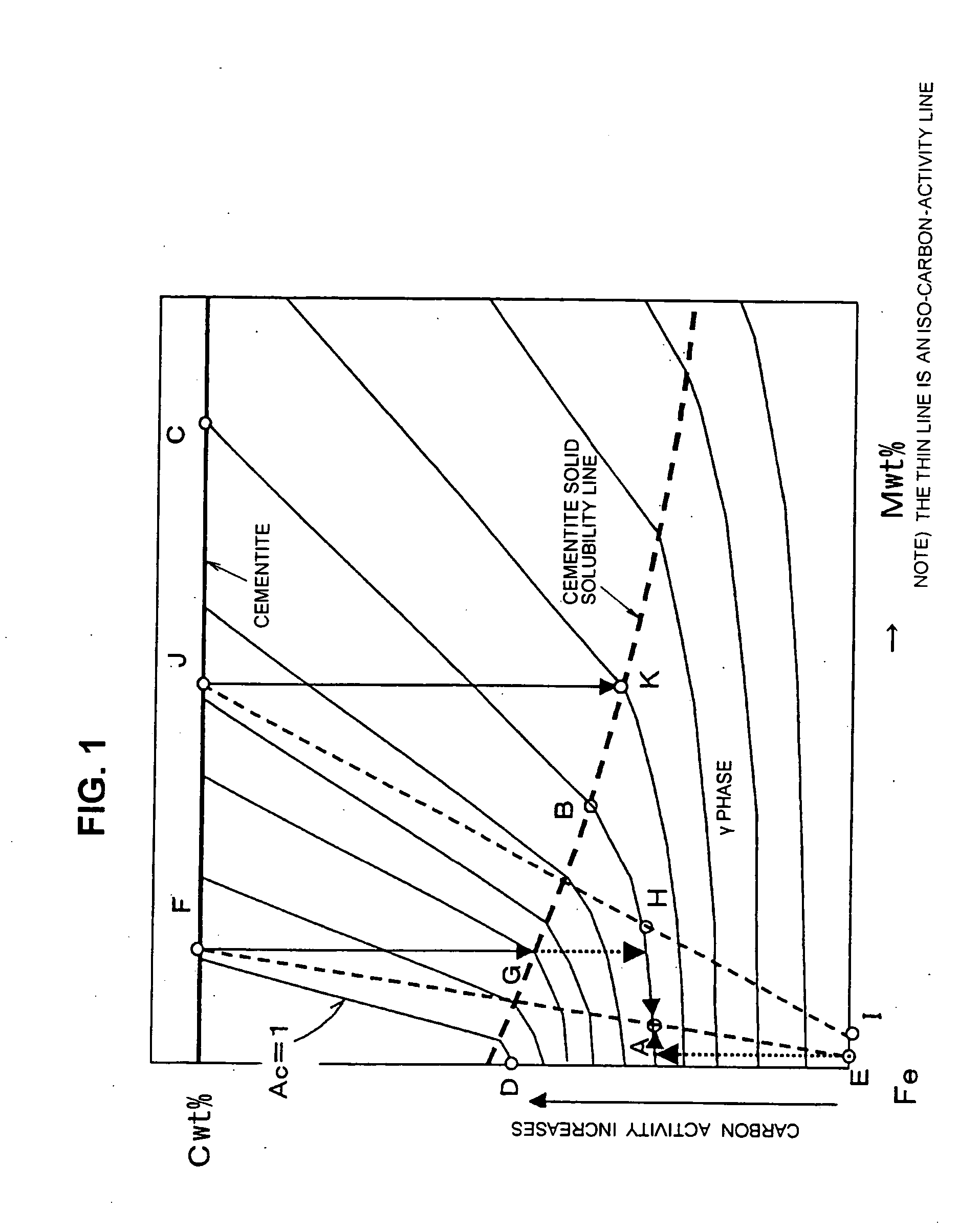
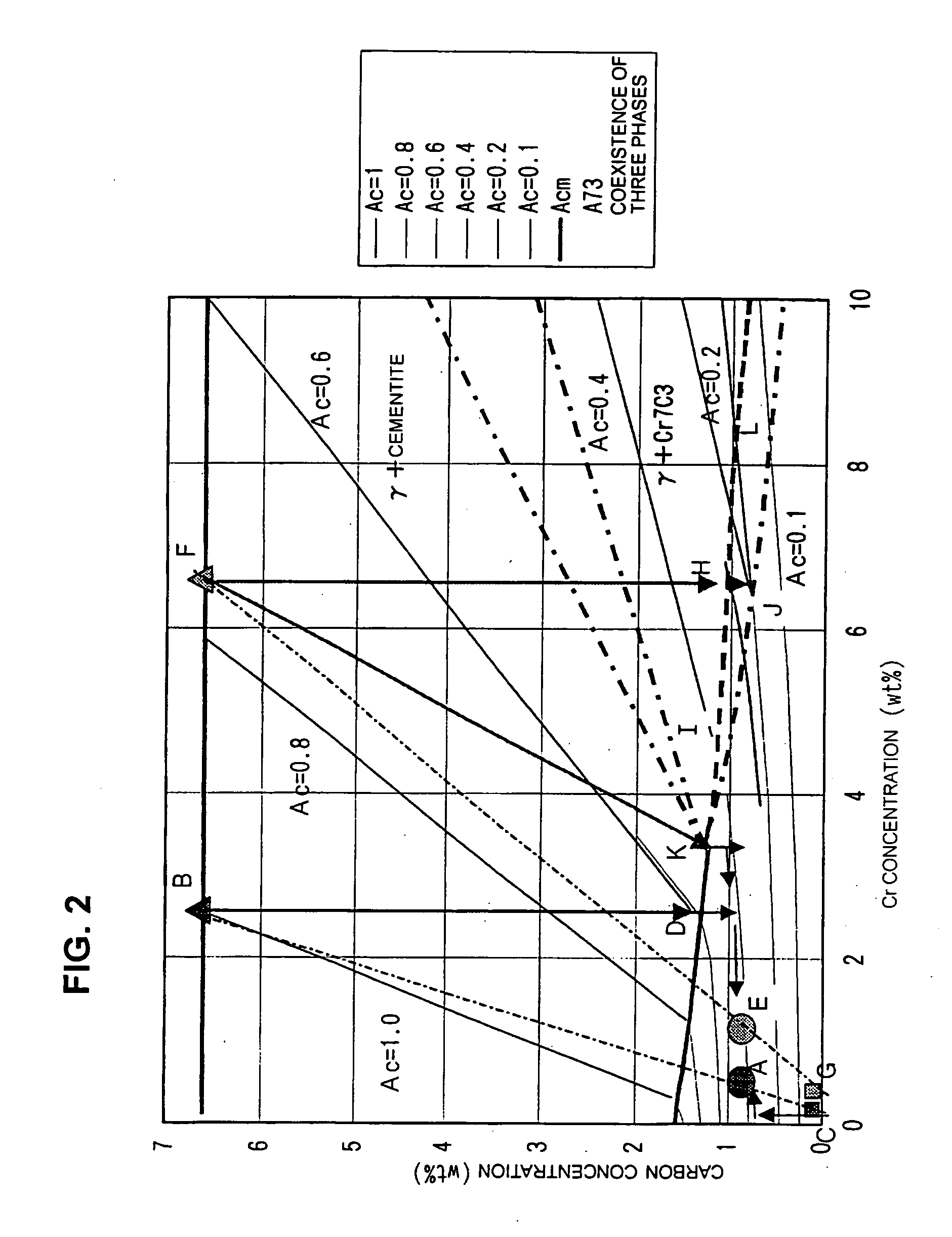
- Composition and crystal structure analysis of iron oxide-cementite systems: Analytical methods and characterization techniques for determining the crystal structure and chemical composition of iron oxide and cementite phases. This includes X-ray diffraction analysis, electron microscopy, and spectroscopic methods to identify phase boundaries and lattice parameters. Understanding the atomic arrangement and bonding characteristics is essential for predicting material behavior.
- Manufacturing processes for iron oxide-cementite composite materials: Industrial production methods for creating composite materials containing controlled amounts of iron oxide and cementite phases. These processes include powder metallurgy, casting, and thermomechanical processing techniques. The manufacturing approach aims to achieve uniform distribution of phases and optimize the interface characteristics between iron oxide and cementite for enhanced performance properties.
02 Cementite decomposition and spheroidization treatment
Techniques for decomposing lamellar cementite structures and promoting spheroidization to improve material ductility and machinability. The process involves specific temperature ranges and holding times to transform the cementite morphology from lamellar to spherical particles. This treatment is particularly important for high carbon steel applications.Expand Specific Solutions03 Iron oxide scale formation and removal in steel processing
Methods addressing the formation, characteristics, and removal of iron oxide scales during steel manufacturing processes. The techniques include descaling processes, oxide layer control during hot rolling, and prevention of excessive oxidation. Understanding the oxide structure is crucial for surface quality control.Expand Specific Solutions04 Phase transformation between iron oxide and cementite
Processes involving the phase transformation mechanisms between different iron oxide forms and cementite during carburization, decarburization, or reduction reactions. These transformations are controlled through atmosphere composition, temperature, and time parameters. The understanding of these transformations is essential for producing materials with specific properties.Expand Specific Solutions05 Composite structures containing iron oxide and cementite phases
Development of composite materials or coatings that intentionally incorporate both iron oxide and cementite phases to achieve specific functional properties. These structures may be designed for wear resistance, corrosion protection, or specialized mechanical characteristics. The controlled distribution of these phases creates synergistic effects for enhanced performance.Expand Specific Solutions
Major Players in Steel and Iron Oxide Industries
The structural comparison between iron oxide and cementite represents a mature research area within metallurgical science, currently in an advanced development stage with established industrial applications. The global steel industry, valued at over $900 billion annually, drives continuous investigation into these fundamental iron phases. Major steel manufacturers including NIPPON STEEL CORP., JFE Steel Corp., Baoshan Iron & Steel Co., Ltd., and Kobe Steel, Ltd. demonstrate high technical maturity through their sophisticated production processes and quality control systems. Academic institutions like Taiyuan University of Technology and University of California contribute fundamental research advancing phase transformation understanding. Industrial players such as Komatsu Ltd. and equipment manufacturers like Dongfang Boiler Group apply this knowledge in materials engineering. The competitive landscape shows strong integration between basic research and industrial implementation, with Japanese and Chinese steel producers leading technological refinement while research institutions provide theoretical foundations for optimizing microstructural properties.
NIPPON STEEL CORP.
Technical Solution: Nippon Steel has developed advanced analytical techniques for comparing iron oxide and cementite structures in steel materials. Their approach utilizes high-resolution transmission electron microscopy (TEM) combined with electron diffraction analysis to characterize the crystallographic differences between these phases. The company focuses on understanding how iron oxide (Fe3O4, Fe2O3) with cubic/hexagonal structures differs from cementite (Fe3C) with orthorhombic structure in terms of lattice parameters, atomic arrangements, and interfacial characteristics. Their research emphasizes the phase transformation mechanisms during steel processing, particularly examining how oxygen content and carbon distribution affect the formation and stability of these competing phases. This structural comparison is critical for optimizing steel properties, as iron oxides represent detrimental inclusions while cementite contributes to hardness and wear resistance.
Strengths: Extensive industrial experience in steel metallurgy with state-of-the-art characterization facilities; deep understanding of phase relationships in Fe-C-O systems. Weaknesses: Research primarily focused on practical steel applications rather than fundamental structural science; limited public disclosure of proprietary analytical methods.
JFE Steel Corp.
Technical Solution: JFE Steel has established comprehensive structural comparison methodologies for iron oxide and cementite phases encountered in steelmaking processes. Their technical approach combines X-ray diffraction (XRD) analysis with scanning electron microscopy (SEM) to distinguish between the face-centered cubic structure of magnetite (Fe3O4), the rhombohedral structure of hematite (Fe2O3), and the complex orthorhombic structure of cementite (Fe3C). The company has developed proprietary databases correlating crystal structure parameters with mechanical properties, focusing on how the metallic bonding in cementite differs from the mixed ionic-covalent bonding in iron oxides. Their research addresses the thermodynamic stability ranges of these phases and their formation kinetics during cooling and heat treatment processes, which is essential for controlling steel microstructures and preventing oxide-related defects.
Strengths: Strong integration of structural analysis with process control systems; extensive pilot-scale testing capabilities for validating structural models. Weaknesses: Technology development centered on conventional steelmaking rather than emerging applications; limited collaboration with academic institutions on fundamental crystallography.
Key Research on Fe3O4 and Fe3C Structural Differences
Rolling element and method of producing the same
PatentInactiveUS20060021679A1
Innovation
- The development of a rolling element with a steel material containing high Al and/or Si content, dispersed fine cementite particles, and specific alloy elements like Ti, Zr, Nb, Ta, and Hf carbides, nitrides, and carbonitrides to enhance temper hardness and seizure resistance, achieved through induction hardening and tempering processes.
Method for thermal processing of articles consisting of alloys on the basis of iron (variants)
PatentInactiveIN1793DELNP2013A
Innovation
- A method involving heating to form austenite and subsequent cooling schedules that create marinite regions, followed by the formation of pearlite with various degrees of dispersion and/or quenched structures, using calculated or experimental approaches to determine optimal cooling parameters and schedules.
Material Property Optimization Strategies
Optimizing material properties in iron-based systems requires strategic manipulation of phase composition and microstructural features. The fundamental structural differences between iron oxide and cementite provide distinct leverage points for property enhancement. Iron oxides, with their ionic bonding and defect-rich structures, offer opportunities for tuning electronic properties, magnetic behavior, and catalytic activity through controlled stoichiometry variations and dopant incorporation. Conversely, cementite's metastable nature and orthorhombic structure enable mechanical property optimization through morphology control and interfacial engineering.
Grain refinement represents a primary optimization strategy applicable to both phases. In cementite-containing steels, reducing cementite particle size and achieving uniform distribution significantly enhances toughness while maintaining strength. This can be accomplished through controlled cooling rates, thermomechanical processing, or microalloying additions that modify nucleation kinetics. For iron oxide systems, nanostructuring enhances surface-to-volume ratios, improving reactivity and enabling novel functionalities in energy storage and conversion applications.
Compositional engineering offers another critical pathway. Substitutional alloying in cementite, though limited by its complex crystal structure, can modify lattice parameters and bonding characteristics. Transition metal additions may alter electronic structure and magnetic properties. In iron oxides, cation substitution provides extensive tunability of band gaps, magnetic ordering temperatures, and redox behavior, enabling tailored performance for specific applications.
Interface optimization between iron oxide and metallic iron phases, or between cementite and ferrite matrices, critically influences overall material performance. Coherent or semi-coherent interfaces minimize energy barriers for load transfer and enhance mechanical integrity. Surface treatments, coating technologies, and controlled oxidation processes can engineer beneficial interfacial structures that improve corrosion resistance, wear properties, or adhesion characteristics.
Thermal treatment strategies enable phase transformation control and residual stress management. Tempering processes in cementite-containing steels optimize the balance between hardness and ductility. For iron oxide systems, calcination temperature and atmosphere control determine phase purity, crystallinity, and defect concentrations, directly impacting functional properties. Advanced processing techniques including spark plasma sintering and additive manufacturing open new possibilities for achieving non-equilibrium structures with superior property combinations.
Grain refinement represents a primary optimization strategy applicable to both phases. In cementite-containing steels, reducing cementite particle size and achieving uniform distribution significantly enhances toughness while maintaining strength. This can be accomplished through controlled cooling rates, thermomechanical processing, or microalloying additions that modify nucleation kinetics. For iron oxide systems, nanostructuring enhances surface-to-volume ratios, improving reactivity and enabling novel functionalities in energy storage and conversion applications.
Compositional engineering offers another critical pathway. Substitutional alloying in cementite, though limited by its complex crystal structure, can modify lattice parameters and bonding characteristics. Transition metal additions may alter electronic structure and magnetic properties. In iron oxides, cation substitution provides extensive tunability of band gaps, magnetic ordering temperatures, and redox behavior, enabling tailored performance for specific applications.
Interface optimization between iron oxide and metallic iron phases, or between cementite and ferrite matrices, critically influences overall material performance. Coherent or semi-coherent interfaces minimize energy barriers for load transfer and enhance mechanical integrity. Surface treatments, coating technologies, and controlled oxidation processes can engineer beneficial interfacial structures that improve corrosion resistance, wear properties, or adhesion characteristics.
Thermal treatment strategies enable phase transformation control and residual stress management. Tempering processes in cementite-containing steels optimize the balance between hardness and ductility. For iron oxide systems, calcination temperature and atmosphere control determine phase purity, crystallinity, and defect concentrations, directly impacting functional properties. Advanced processing techniques including spark plasma sintering and additive manufacturing open new possibilities for achieving non-equilibrium structures with superior property combinations.
Applications in Advanced Steel Manufacturing
The structural distinctions between iron oxide and cementite fundamentally influence their respective roles in advanced steel manufacturing processes. Iron oxides, primarily existing as scale formations during hot rolling and heat treatment, must be carefully managed or removed to ensure surface quality and dimensional accuracy. Conversely, cementite serves as a critical strengthening phase within the steel matrix, where its morphology, distribution, and volume fraction directly determine mechanical properties. Understanding these structural differences enables manufacturers to optimize processing parameters and achieve superior product performance.
In modern steel production, the controlled transformation between iron oxide and cementite phases represents a cornerstone of quality control. During continuous casting and subsequent hot working operations, surface decarburization and oxidation can compromise the intended microstructure. Advanced manufacturing facilities employ protective atmospheres and precise temperature control to minimize unwanted iron oxide formation while promoting desired cementite precipitation patterns. This dual approach ensures that the final microstructure contains appropriately distributed cementite particles without detrimental oxide inclusions that could initiate fatigue cracks or corrosion.
The application of this structural knowledge extends to emerging steel grades designed for automotive lightweighting and high-performance applications. Third-generation advanced high-strength steels leverage fine cementite dispersions within complex multiphase microstructures to achieve exceptional strength-ductility combinations. Simultaneously, manufacturers implement innovative descaling technologies and surface treatment methods to eliminate residual iron oxides that would otherwise interfere with coating adhesion or welding quality. These developments demonstrate how fundamental understanding of phase structures translates directly into manufacturing capabilities.
Furthermore, additive manufacturing of steel components introduces new considerations regarding oxide formation and carbide precipitation. Rapid solidification and thermal cycling inherent to powder bed fusion processes create unique opportunities to engineer cementite distributions while managing oxide contamination from powder feedstock. Process optimization requires precise control over oxygen partial pressure and cooling rates to achieve the desired balance between these competing phases, ultimately enabling production of components with tailored mechanical properties previously unattainable through conventional manufacturing routes.
In modern steel production, the controlled transformation between iron oxide and cementite phases represents a cornerstone of quality control. During continuous casting and subsequent hot working operations, surface decarburization and oxidation can compromise the intended microstructure. Advanced manufacturing facilities employ protective atmospheres and precise temperature control to minimize unwanted iron oxide formation while promoting desired cementite precipitation patterns. This dual approach ensures that the final microstructure contains appropriately distributed cementite particles without detrimental oxide inclusions that could initiate fatigue cracks or corrosion.
The application of this structural knowledge extends to emerging steel grades designed for automotive lightweighting and high-performance applications. Third-generation advanced high-strength steels leverage fine cementite dispersions within complex multiphase microstructures to achieve exceptional strength-ductility combinations. Simultaneously, manufacturers implement innovative descaling technologies and surface treatment methods to eliminate residual iron oxides that would otherwise interfere with coating adhesion or welding quality. These developments demonstrate how fundamental understanding of phase structures translates directly into manufacturing capabilities.
Furthermore, additive manufacturing of steel components introduces new considerations regarding oxide formation and carbide precipitation. Rapid solidification and thermal cycling inherent to powder bed fusion processes create unique opportunities to engineer cementite distributions while managing oxide contamination from powder feedstock. Process optimization requires precise control over oxygen partial pressure and cooling rates to achieve the desired balance between these competing phases, ultimately enabling production of components with tailored mechanical properties previously unattainable through conventional manufacturing routes.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!