Comparing Pressure and Temperature Effects on CO2 Capture
MAR 23, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
CO2 Capture Technology Background and Objectives
Carbon dioxide capture technology has emerged as a critical component in global climate change mitigation strategies, representing one of the most promising approaches to reduce atmospheric CO2 concentrations. The technology encompasses various methods designed to separate and capture CO2 from industrial emission sources, power plants, and even directly from ambient air, preventing its release into the atmosphere.
The historical development of CO2 capture can be traced back to the early 20th century when chemical absorption processes were first employed in industrial applications. However, the modern focus on CO2 capture as an environmental solution gained momentum in the 1990s, driven by increasing awareness of greenhouse gas emissions and their impact on global warming. The technology has since evolved from basic chemical absorption methods to sophisticated integrated systems incorporating multiple capture mechanisms.
Current CO2 capture technologies operate on three primary principles: pre-combustion capture, post-combustion capture, and oxy-fuel combustion. Each approach presents unique advantages and challenges, with effectiveness significantly influenced by operational parameters, particularly pressure and temperature conditions. Understanding these thermodynamic effects is crucial for optimizing capture efficiency and economic viability.
The primary objective of advancing CO2 capture technology centers on achieving maximum capture efficiency while minimizing energy consumption and operational costs. Specifically, the comparative analysis of pressure and temperature effects aims to identify optimal operating conditions that enhance CO2 solubility in capture media, improve mass transfer rates, and reduce parasitic energy losses associated with compression and heating processes.
Temperature optimization represents a fundamental challenge, as higher temperatures generally reduce CO2 solubility in liquid solvents while potentially improving reaction kinetics. Conversely, pressure manipulation can significantly enhance capture rates through increased CO2 partial pressure, though this requires substantial compression energy. The interplay between these parameters determines the overall system performance and economic feasibility.
Strategic objectives include developing comprehensive understanding of thermodynamic relationships governing CO2 capture processes, establishing predictive models for performance optimization under varying conditions, and identifying breakthrough approaches that minimize the traditional trade-offs between capture efficiency and energy consumption. These goals ultimately support the broader mission of deploying commercially viable CO2 capture systems at industrial scale.
The historical development of CO2 capture can be traced back to the early 20th century when chemical absorption processes were first employed in industrial applications. However, the modern focus on CO2 capture as an environmental solution gained momentum in the 1990s, driven by increasing awareness of greenhouse gas emissions and their impact on global warming. The technology has since evolved from basic chemical absorption methods to sophisticated integrated systems incorporating multiple capture mechanisms.
Current CO2 capture technologies operate on three primary principles: pre-combustion capture, post-combustion capture, and oxy-fuel combustion. Each approach presents unique advantages and challenges, with effectiveness significantly influenced by operational parameters, particularly pressure and temperature conditions. Understanding these thermodynamic effects is crucial for optimizing capture efficiency and economic viability.
The primary objective of advancing CO2 capture technology centers on achieving maximum capture efficiency while minimizing energy consumption and operational costs. Specifically, the comparative analysis of pressure and temperature effects aims to identify optimal operating conditions that enhance CO2 solubility in capture media, improve mass transfer rates, and reduce parasitic energy losses associated with compression and heating processes.
Temperature optimization represents a fundamental challenge, as higher temperatures generally reduce CO2 solubility in liquid solvents while potentially improving reaction kinetics. Conversely, pressure manipulation can significantly enhance capture rates through increased CO2 partial pressure, though this requires substantial compression energy. The interplay between these parameters determines the overall system performance and economic feasibility.
Strategic objectives include developing comprehensive understanding of thermodynamic relationships governing CO2 capture processes, establishing predictive models for performance optimization under varying conditions, and identifying breakthrough approaches that minimize the traditional trade-offs between capture efficiency and energy consumption. These goals ultimately support the broader mission of deploying commercially viable CO2 capture systems at industrial scale.
Market Demand for Enhanced CO2 Capture Solutions
The global carbon dioxide capture market is experiencing unprecedented growth driven by escalating climate commitments and regulatory pressures worldwide. Industrial sectors including power generation, cement production, steel manufacturing, and petrochemicals are increasingly seeking advanced CO2 capture technologies to meet stringent emission reduction targets. The urgency stems from international climate agreements and national carbon neutrality pledges that mandate substantial emission reductions across multiple decades.
Traditional CO2 capture methods face significant limitations in operational efficiency and economic viability, particularly under varying pressure and temperature conditions. Industrial processes operate across diverse thermodynamic environments, creating demand for capture solutions that maintain high performance regardless of operational parameters. Current technologies often exhibit reduced capture efficiency when pressure or temperature conditions deviate from optimal ranges, leading to increased operational costs and suboptimal environmental outcomes.
The market demand for enhanced CO2 capture solutions specifically targets technologies that demonstrate superior performance across variable pressure and temperature conditions. Industries require capture systems that can adapt to fluctuating operational environments while maintaining consistent capture rates and energy efficiency. This demand is particularly pronounced in sectors where process conditions vary significantly throughout operational cycles.
Energy-intensive industries are driving demand for capture technologies that minimize parasitic energy consumption while maximizing CO2 removal efficiency. The economic viability of carbon capture projects heavily depends on the energy penalty associated with capture processes, making pressure and temperature optimization critical for commercial deployment. Enhanced solutions that reduce energy requirements through optimized thermodynamic conditions represent high-value market opportunities.
Emerging carbon utilization markets are creating additional demand for high-purity CO2 streams that require precise capture conditions. Applications in carbon-to-chemicals conversion, enhanced oil recovery, and carbon mineralization necessitate capture technologies capable of delivering consistent product quality under varying operational parameters. This trend expands market opportunities beyond traditional storage-focused applications.
The integration of renewable energy sources with industrial processes is generating demand for flexible CO2 capture systems that can operate efficiently under intermittent power supply conditions. Enhanced capture solutions that maintain performance during variable energy availability periods address critical market needs for sustainable industrial decarbonization strategies.
Traditional CO2 capture methods face significant limitations in operational efficiency and economic viability, particularly under varying pressure and temperature conditions. Industrial processes operate across diverse thermodynamic environments, creating demand for capture solutions that maintain high performance regardless of operational parameters. Current technologies often exhibit reduced capture efficiency when pressure or temperature conditions deviate from optimal ranges, leading to increased operational costs and suboptimal environmental outcomes.
The market demand for enhanced CO2 capture solutions specifically targets technologies that demonstrate superior performance across variable pressure and temperature conditions. Industries require capture systems that can adapt to fluctuating operational environments while maintaining consistent capture rates and energy efficiency. This demand is particularly pronounced in sectors where process conditions vary significantly throughout operational cycles.
Energy-intensive industries are driving demand for capture technologies that minimize parasitic energy consumption while maximizing CO2 removal efficiency. The economic viability of carbon capture projects heavily depends on the energy penalty associated with capture processes, making pressure and temperature optimization critical for commercial deployment. Enhanced solutions that reduce energy requirements through optimized thermodynamic conditions represent high-value market opportunities.
Emerging carbon utilization markets are creating additional demand for high-purity CO2 streams that require precise capture conditions. Applications in carbon-to-chemicals conversion, enhanced oil recovery, and carbon mineralization necessitate capture technologies capable of delivering consistent product quality under varying operational parameters. This trend expands market opportunities beyond traditional storage-focused applications.
The integration of renewable energy sources with industrial processes is generating demand for flexible CO2 capture systems that can operate efficiently under intermittent power supply conditions. Enhanced capture solutions that maintain performance during variable energy availability periods address critical market needs for sustainable industrial decarbonization strategies.
Current Status of Pressure-Temperature CO2 Capture Methods
The current landscape of pressure-temperature CO2 capture methods encompasses several established technologies, each operating under distinct thermodynamic conditions to achieve optimal separation efficiency. Physical absorption processes, primarily utilizing solvents like Selexol and Rectisol, operate at high pressures ranging from 20-70 bar and low temperatures between -40°C to 40°C. These methods leverage the enhanced solubility of CO2 under elevated pressure conditions while maintaining energy-efficient regeneration through pressure reduction.
Chemical absorption remains the most commercially deployed approach, with monoethanolamine (MEA) and other amine-based solvents operating at atmospheric pressure and temperatures between 40-60°C for absorption, followed by thermal regeneration at 100-120°C. Advanced formulations including piperazine-promoted potassium carbonate and novel sterically hindered amines have demonstrated improved performance under varying pressure-temperature profiles, reducing energy penalties associated with solvent regeneration.
Solid sorbent technologies present diverse pressure-temperature operating windows depending on material composition. Metal-organic frameworks (MOFs) exhibit optimal CO2 uptake at pressures of 1-30 bar and temperatures ranging from ambient to 200°C, with materials like MOF-177 and HKUST-1 showing exceptional selectivity under specific conditions. Zeolites and activated carbons demonstrate robust performance across broader temperature ranges, typically operating between 25-400°C at pressures up to 50 bar.
Membrane separation technologies operate under moderate pressure differentials of 10-40 bar, with polymeric membranes functioning optimally at temperatures below 100°C, while ceramic and metallic membranes can withstand temperatures exceeding 500°C. Recent developments in mixed-matrix membranes have expanded operational flexibility by incorporating MOF particles into polymer matrices, enabling enhanced performance across wider pressure-temperature ranges.
Cryogenic separation represents the high-pressure, low-temperature extreme, operating at pressures above 30 bar and temperatures below -50°C to achieve CO2 liquefaction. This approach demonstrates exceptional purity levels but requires significant energy input for refrigeration. Emerging hybrid systems combine multiple capture mechanisms, such as pressure swing adsorption coupled with temperature swing processes, to optimize energy efficiency while maintaining high capture rates across variable operating conditions.
Chemical absorption remains the most commercially deployed approach, with monoethanolamine (MEA) and other amine-based solvents operating at atmospheric pressure and temperatures between 40-60°C for absorption, followed by thermal regeneration at 100-120°C. Advanced formulations including piperazine-promoted potassium carbonate and novel sterically hindered amines have demonstrated improved performance under varying pressure-temperature profiles, reducing energy penalties associated with solvent regeneration.
Solid sorbent technologies present diverse pressure-temperature operating windows depending on material composition. Metal-organic frameworks (MOFs) exhibit optimal CO2 uptake at pressures of 1-30 bar and temperatures ranging from ambient to 200°C, with materials like MOF-177 and HKUST-1 showing exceptional selectivity under specific conditions. Zeolites and activated carbons demonstrate robust performance across broader temperature ranges, typically operating between 25-400°C at pressures up to 50 bar.
Membrane separation technologies operate under moderate pressure differentials of 10-40 bar, with polymeric membranes functioning optimally at temperatures below 100°C, while ceramic and metallic membranes can withstand temperatures exceeding 500°C. Recent developments in mixed-matrix membranes have expanded operational flexibility by incorporating MOF particles into polymer matrices, enabling enhanced performance across wider pressure-temperature ranges.
Cryogenic separation represents the high-pressure, low-temperature extreme, operating at pressures above 30 bar and temperatures below -50°C to achieve CO2 liquefaction. This approach demonstrates exceptional purity levels but requires significant energy input for refrigeration. Emerging hybrid systems combine multiple capture mechanisms, such as pressure swing adsorption coupled with temperature swing processes, to optimize energy efficiency while maintaining high capture rates across variable operating conditions.
Existing Pressure-Temperature CO2 Capture Solutions
01 Amine-based CO2 capture systems and efficiency optimization
Amine-based solvents are widely used for CO2 capture due to their high reactivity with carbon dioxide. The capture efficiency can be optimized through solvent composition adjustments, temperature control, and process parameter modifications. Advanced amine formulations and blended solvents can enhance absorption rates while reducing energy consumption during regeneration cycles. System design improvements including contactor configuration and flow rate optimization contribute to higher overall capture efficiency.- Amine-based CO2 capture systems and efficiency optimization: Amine-based solvents are widely used for CO2 capture due to their high reactivity with carbon dioxide. The capture efficiency can be optimized through solvent composition adjustments, temperature control, and process parameter modifications. Advanced amine formulations and blended solvents can enhance absorption rates while reducing energy consumption during regeneration cycles.
- Solid sorbent materials for enhanced CO2 capture: Solid sorbent materials including metal-organic frameworks, zeolites, and activated carbons offer advantages in CO2 capture efficiency through high surface area and selective adsorption properties. These materials can be engineered with specific pore structures and surface chemistries to maximize capture capacity and enable lower energy regeneration compared to liquid solvents.
- Membrane-based separation technologies: Membrane separation systems provide continuous CO2 capture through selective permeation mechanisms. The efficiency depends on membrane material properties, operating pressure, temperature conditions, and gas flow configurations. Advanced polymer and ceramic membranes can achieve high selectivity and permeability for improved capture performance.
- Process integration and energy recovery systems: Integration of CO2 capture units with industrial processes and implementation of heat recovery systems significantly improve overall capture efficiency. Waste heat utilization, pressure swing optimization, and multi-stage capture configurations reduce parasitic energy losses and enhance the economic viability of capture operations.
- Monitoring and control systems for capture optimization: Advanced monitoring systems and automated control strategies enable real-time optimization of CO2 capture efficiency. Sensor networks, predictive algorithms, and feedback control mechanisms adjust operating parameters dynamically to maintain optimal capture rates under varying feed gas conditions and process disturbances.
02 Solid sorbent materials for enhanced CO2 capture
Solid sorbent materials including metal-organic frameworks, zeolites, and activated carbons offer advantages in CO2 capture applications. These materials can be engineered with specific pore structures and surface chemistries to maximize adsorption capacity and selectivity. The capture efficiency is influenced by sorbent regeneration methods, operating temperature ranges, and material stability over multiple cycles. Novel composite materials and functionalized sorbents demonstrate improved performance metrics.Expand Specific Solutions03 Membrane-based separation technologies
Membrane separation systems provide an alternative approach to CO2 capture with potential for lower energy requirements. The efficiency depends on membrane material properties including permeability, selectivity, and mechanical stability. Polymeric, ceramic, and mixed-matrix membranes can be tailored for specific gas separation applications. Multi-stage membrane configurations and hybrid systems combining membranes with other capture methods can achieve higher overall capture rates.Expand Specific Solutions04 Process integration and energy recovery systems
Integration of CO2 capture systems with industrial processes and power generation facilities requires careful optimization to maintain high capture efficiency while minimizing energy penalties. Heat integration strategies, waste heat recovery, and process intensification techniques can significantly improve overall system performance. Advanced control systems and real-time monitoring enable dynamic optimization of capture operations under varying feed conditions and operational requirements.Expand Specific Solutions05 Novel capture methods and emerging technologies
Emerging CO2 capture technologies include electrochemical separation, cryogenic distillation, and bio-inspired capture systems. These innovative approaches aim to overcome limitations of conventional methods by offering improved efficiency, reduced costs, or enhanced scalability. Hybrid systems combining multiple capture mechanisms can leverage synergistic effects to achieve superior performance. Research focuses on developing materials and processes that enable high capture rates with minimal environmental impact and operational complexity.Expand Specific Solutions
Major Players in CO2 Capture Technology Sector
The CO2 capture technology sector is experiencing rapid evolution across multiple development stages, with the market expanding significantly driven by global decarbonization mandates and carbon pricing mechanisms. The competitive landscape spans from mature industrial gas companies like Air Liquide SA, Praxair Technology, and Siemens AG leveraging established infrastructure, to specialized innovators such as Climeworks AG and Neustark AG pioneering direct air capture and concrete carbonation technologies. Technology maturity varies considerably, with traditional post-combustion capture reaching commercial deployment through energy giants like CHN Energy Investment Group and PetroChina Co., while advanced pressure-temperature optimization remains largely in R&D phases led by academic institutions including ETH Zurich, Texas A&M University, and Beijing University of Chemical Technology, alongside corporate research arms like Huaneng Clean Energy Research Institute, creating a dynamic ecosystem balancing proven solutions with breakthrough innovations.
Climeworks AG
Technical Solution: Climeworks employs direct air capture (DAC) technology using temperature swing adsorption with solid amine sorbents. Their process operates at ambient pressure and utilizes temperature cycling between 80-100°C for CO2 desorption. The company's modular systems can capture CO2 directly from atmospheric air with concentrations around 400 ppm. Their technology demonstrates that lower pressure operations at ambient conditions can be effective when combined with optimized temperature management, achieving energy consumption of approximately 1500-2000 kWh per ton of CO2 captured. The pressure-temperature relationship in their systems shows that atmospheric pressure operation reduces compression costs while controlled heating enables efficient sorbent regeneration.
Strengths: Proven commercial-scale operation, modular design flexibility, ambient pressure operation reduces energy costs. Weaknesses: High energy consumption for heating, limited to atmospheric CO2 concentrations, high capital costs.
Air Liquide SA
Technical Solution: Air Liquide implements cryogenic CO2 capture technology that leverages extreme pressure and temperature conditions for separation and purification. Their systems operate at pressures up to 30 bar and temperatures as low as -50°C to achieve phase separation of CO2 from gas mixtures. The company's technology demonstrates how high-pressure, low-temperature conditions can achieve near-complete CO2 separation with purities exceeding 99.5%. Their process optimization shows that increased pressure enhances separation efficiency while controlled cooling enables selective condensation. The integrated cryogenic systems combine compression, cooling, and separation stages to maximize capture rates while minimizing energy consumption through advanced heat integration and process optimization techniques.
Strengths: Very high purity CO2 output, excellent separation efficiency, proven cryogenic expertise, suitable for large-scale operations. Weaknesses: High energy consumption for compression and cooling, complex equipment requirements, significant capital investment needed.
Core Innovations in Thermodynamic CO2 Capture
Systems and methods for optimizing carbon dioxide capture using temperature management
PatentWO2024232890A1
Innovation
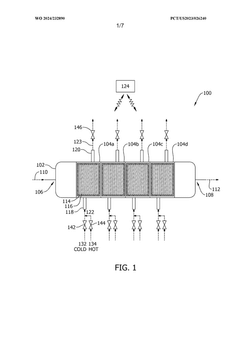
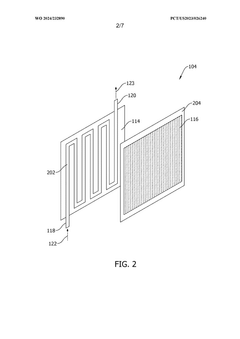
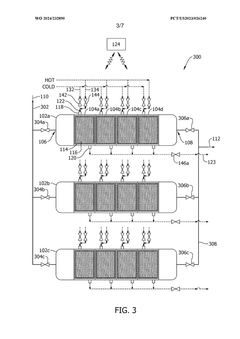
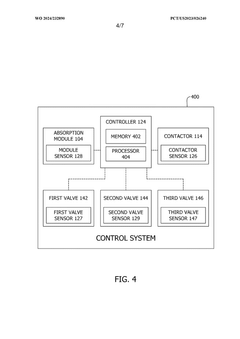
- A capture system utilizing a contactor with a regulating fluid stream comprising a cold and hot stream, managed by a controller to modulate the temperature of adsorption modules, allowing for indirect heating and cooling to optimize carbon dioxide adsorption and desorption efficiency.
Method for capturing carbon dioxide
PatentWO2025247878A1
Innovation
- A carbon dioxide capture process involving multiple desorbers operating at varying temperatures and pressures, with latent heat from water vapor condensation used to supply heat to boilers, optimizing the energy distribution and reducing the overall energy demand.
Environmental Policy Impact on CO2 Capture Development
Environmental policies have emerged as the primary catalyst driving innovation and investment in CO2 capture technologies, fundamentally reshaping the research landscape around pressure and temperature optimization studies. The implementation of carbon pricing mechanisms, emissions trading systems, and mandatory carbon reduction targets has created unprecedented economic incentives for developing more efficient capture processes that can operate effectively under varying thermodynamic conditions.
The Paris Agreement and subsequent national climate commitments have established stringent emission reduction timelines, compelling researchers to accelerate investigations into optimal pressure and temperature parameters for different capture technologies. These policy frameworks have particularly influenced the prioritization of research funding toward understanding how environmental conditions affect capture efficiency, leading to more comprehensive comparative studies between pressure-swing and temperature-swing adsorption processes.
Regulatory standards for industrial emissions have directly impacted the technical specifications required for CO2 capture systems, driving research toward identifying optimal operating conditions that balance energy consumption with capture rates. Government mandates for carbon capture and storage deployment have necessitated detailed analysis of how pressure and temperature variations affect system reliability and long-term performance in real-world applications.
Financial incentives such as carbon credits and tax benefits have accelerated private sector investment in comparative studies examining the economic viability of different pressure and temperature operating regimes. These policy-driven investments have enabled large-scale pilot projects that provide crucial data on how varying thermodynamic conditions impact capture costs and energy requirements across different industrial sectors.
International cooperation frameworks established through environmental policies have facilitated knowledge sharing regarding optimal pressure and temperature conditions for CO2 capture across diverse geographical and industrial contexts. This collaborative approach has enhanced the quality and scope of comparative research, enabling more robust conclusions about the relative effectiveness of different thermodynamic approaches under varying environmental and operational constraints.
The Paris Agreement and subsequent national climate commitments have established stringent emission reduction timelines, compelling researchers to accelerate investigations into optimal pressure and temperature parameters for different capture technologies. These policy frameworks have particularly influenced the prioritization of research funding toward understanding how environmental conditions affect capture efficiency, leading to more comprehensive comparative studies between pressure-swing and temperature-swing adsorption processes.
Regulatory standards for industrial emissions have directly impacted the technical specifications required for CO2 capture systems, driving research toward identifying optimal operating conditions that balance energy consumption with capture rates. Government mandates for carbon capture and storage deployment have necessitated detailed analysis of how pressure and temperature variations affect system reliability and long-term performance in real-world applications.
Financial incentives such as carbon credits and tax benefits have accelerated private sector investment in comparative studies examining the economic viability of different pressure and temperature operating regimes. These policy-driven investments have enabled large-scale pilot projects that provide crucial data on how varying thermodynamic conditions impact capture costs and energy requirements across different industrial sectors.
International cooperation frameworks established through environmental policies have facilitated knowledge sharing regarding optimal pressure and temperature conditions for CO2 capture across diverse geographical and industrial contexts. This collaborative approach has enhanced the quality and scope of comparative research, enabling more robust conclusions about the relative effectiveness of different thermodynamic approaches under varying environmental and operational constraints.
Economic Feasibility of Advanced CO2 Capture Systems
The economic feasibility of advanced CO2 capture systems is fundamentally influenced by the operational parameters of pressure and temperature, which directly impact both capital expenditures and operational costs. Higher pressure operations typically require more robust equipment designs, specialized materials, and enhanced safety systems, leading to increased initial investment requirements. Temperature variations affect energy consumption patterns, with higher temperatures generally demanding more sophisticated heat management systems and thermal-resistant components.
Energy consumption represents the largest operational cost component in CO2 capture systems, with pressure and temperature optimization playing crucial roles in overall system efficiency. Elevated pressure conditions can improve capture rates but require additional compression energy, creating a trade-off between capture efficiency and energy costs. Temperature management affects solvent regeneration energy requirements in chemical absorption processes, where optimal temperature ranges can significantly reduce steam consumption and associated operational expenses.
The choice of capture technology becomes economically critical when considering pressure and temperature effects. Physical absorption processes favor high-pressure, low-temperature conditions, while chemical absorption systems operate effectively across broader temperature ranges but require energy-intensive regeneration cycles. Solid sorbent technologies demonstrate varying economic performance depending on operating conditions, with temperature swing adsorption showing different cost profiles compared to pressure swing systems.
Scale economics significantly influence the cost-effectiveness of pressure and temperature optimization strategies. Large-scale industrial applications can justify sophisticated pressure and temperature control systems that would be economically prohibitive for smaller installations. The integration of waste heat recovery systems becomes more economically attractive at higher operational scales, potentially offsetting increased temperature management costs.
Long-term economic viability depends on the durability and maintenance requirements of systems operating under specific pressure and temperature conditions. Extreme operating conditions may reduce equipment lifespan and increase maintenance frequencies, affecting total cost of ownership calculations. The development of materials and technologies capable of efficient operation under moderate conditions presents opportunities for improved economic performance while maintaining acceptable capture rates.
Energy consumption represents the largest operational cost component in CO2 capture systems, with pressure and temperature optimization playing crucial roles in overall system efficiency. Elevated pressure conditions can improve capture rates but require additional compression energy, creating a trade-off between capture efficiency and energy costs. Temperature management affects solvent regeneration energy requirements in chemical absorption processes, where optimal temperature ranges can significantly reduce steam consumption and associated operational expenses.
The choice of capture technology becomes economically critical when considering pressure and temperature effects. Physical absorption processes favor high-pressure, low-temperature conditions, while chemical absorption systems operate effectively across broader temperature ranges but require energy-intensive regeneration cycles. Solid sorbent technologies demonstrate varying economic performance depending on operating conditions, with temperature swing adsorption showing different cost profiles compared to pressure swing systems.
Scale economics significantly influence the cost-effectiveness of pressure and temperature optimization strategies. Large-scale industrial applications can justify sophisticated pressure and temperature control systems that would be economically prohibitive for smaller installations. The integration of waste heat recovery systems becomes more economically attractive at higher operational scales, potentially offsetting increased temperature management costs.
Long-term economic viability depends on the durability and maintenance requirements of systems operating under specific pressure and temperature conditions. Extreme operating conditions may reduce equipment lifespan and increase maintenance frequencies, affecting total cost of ownership calculations. The development of materials and technologies capable of efficient operation under moderate conditions presents opportunities for improved economic performance while maintaining acceptable capture rates.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!