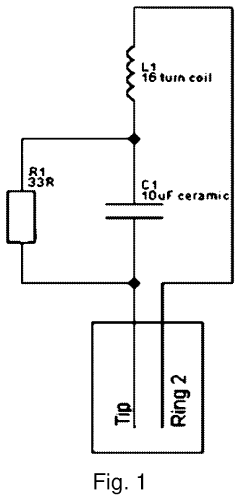
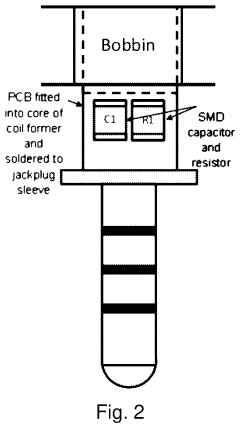
Evaluate Pulsed Electromagnetic Field in Fatigue Science
MAR 7, 20268 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
PEMF Technology Background and Fatigue Research Goals
Pulsed Electromagnetic Field (PEMF) technology represents a non-invasive therapeutic modality that utilizes time-varying electromagnetic fields to influence biological processes at the cellular and tissue levels. The technology emerged from early observations of electromagnetic field effects on biological systems in the mid-20th century, with initial applications focusing on bone healing and fracture repair. Over the past five decades, PEMF has evolved from experimental laboratory techniques to clinically validated therapeutic interventions across multiple medical disciplines.
The fundamental principle underlying PEMF technology involves the generation of controlled electromagnetic pulses that penetrate biological tissues and induce localized electrical currents through electromagnetic induction. These induced currents can modulate cellular membrane potentials, influence ion channel activity, and affect various biochemical pathways involved in cellular metabolism, inflammation response, and tissue repair mechanisms. The therapeutic effects are achieved through specific parameter combinations including frequency, intensity, waveform characteristics, and exposure duration.
In the context of fatigue science, PEMF technology has garnered significant attention due to its potential to address multiple physiological mechanisms underlying fatigue development and recovery. Fatigue, characterized by the decline in physical and cognitive performance capacity, involves complex interactions between metabolic, neurological, and muscular systems. Traditional fatigue management approaches often focus on single-pathway interventions, whereas PEMF offers a multi-modal therapeutic approach that can simultaneously target various fatigue-related mechanisms.
The primary research goals in evaluating PEMF for fatigue applications encompass several critical objectives. First, establishing the optimal electromagnetic field parameters that effectively enhance cellular energy metabolism and mitochondrial function, which are fundamental to fatigue recovery. Second, investigating PEMF's capacity to modulate inflammatory responses and oxidative stress markers that contribute to fatigue progression. Third, determining the technology's effectiveness in improving neuromuscular function and reducing muscle fatigue through enhanced calcium handling and contractile protein efficiency.
Additionally, research objectives include validating PEMF's role in accelerating recovery processes following physical exertion, optimizing treatment protocols for different fatigue types, and establishing evidence-based guidelines for clinical implementation. The ultimate goal involves developing standardized PEMF interventions that can be integrated into comprehensive fatigue management strategies across athletic, occupational, and clinical settings.
The fundamental principle underlying PEMF technology involves the generation of controlled electromagnetic pulses that penetrate biological tissues and induce localized electrical currents through electromagnetic induction. These induced currents can modulate cellular membrane potentials, influence ion channel activity, and affect various biochemical pathways involved in cellular metabolism, inflammation response, and tissue repair mechanisms. The therapeutic effects are achieved through specific parameter combinations including frequency, intensity, waveform characteristics, and exposure duration.
In the context of fatigue science, PEMF technology has garnered significant attention due to its potential to address multiple physiological mechanisms underlying fatigue development and recovery. Fatigue, characterized by the decline in physical and cognitive performance capacity, involves complex interactions between metabolic, neurological, and muscular systems. Traditional fatigue management approaches often focus on single-pathway interventions, whereas PEMF offers a multi-modal therapeutic approach that can simultaneously target various fatigue-related mechanisms.
The primary research goals in evaluating PEMF for fatigue applications encompass several critical objectives. First, establishing the optimal electromagnetic field parameters that effectively enhance cellular energy metabolism and mitochondrial function, which are fundamental to fatigue recovery. Second, investigating PEMF's capacity to modulate inflammatory responses and oxidative stress markers that contribute to fatigue progression. Third, determining the technology's effectiveness in improving neuromuscular function and reducing muscle fatigue through enhanced calcium handling and contractile protein efficiency.
Additionally, research objectives include validating PEMF's role in accelerating recovery processes following physical exertion, optimizing treatment protocols for different fatigue types, and establishing evidence-based guidelines for clinical implementation. The ultimate goal involves developing standardized PEMF interventions that can be integrated into comprehensive fatigue management strategies across athletic, occupational, and clinical settings.
Market Demand for PEMF-Based Fatigue Recovery Solutions
The global fatigue recovery market has experienced substantial growth driven by increasing awareness of physical and mental wellness across multiple sectors. Healthcare institutions represent a primary demand source, with hospitals and rehabilitation centers seeking evidence-based solutions for patient recovery protocols. Sports medicine facilities and professional athletic organizations constitute another significant market segment, where rapid recovery from training-induced fatigue directly impacts performance outcomes.
Corporate wellness programs have emerged as a growing market driver, particularly in industries with high physical demands or stress-related fatigue. Companies are increasingly investing in employee health technologies to reduce absenteeism and improve productivity. The military and defense sectors also demonstrate strong interest in fatigue countermeasures, especially for personnel operating in demanding environments where cognitive and physical performance are critical.
Consumer markets show expanding adoption patterns, with fitness enthusiasts and aging populations seeking non-invasive recovery solutions. The rise of home healthcare trends, accelerated by recent global health events, has created opportunities for portable PEMF devices targeting individual consumers. This demographic shift reflects broader healthcare decentralization and preventive care adoption.
Geographic demand varies significantly, with North American and European markets leading adoption due to established healthcare infrastructure and regulatory frameworks supporting electromagnetic therapy research. Asian markets, particularly Japan and South Korea, demonstrate growing interest driven by technological innovation cultures and aging populations experiencing fatigue-related health challenges.
Market barriers include regulatory complexity across different jurisdictions and varying levels of clinical evidence acceptance among healthcare providers. Insurance coverage limitations also constrain market expansion, as many PEMF applications remain classified as complementary rather than primary treatments. Despite these challenges, the convergence of wearable technology trends, personalized medicine approaches, and growing scientific validation of electromagnetic field effects on cellular recovery processes continues to drive market demand expansion across diverse application areas.
Corporate wellness programs have emerged as a growing market driver, particularly in industries with high physical demands or stress-related fatigue. Companies are increasingly investing in employee health technologies to reduce absenteeism and improve productivity. The military and defense sectors also demonstrate strong interest in fatigue countermeasures, especially for personnel operating in demanding environments where cognitive and physical performance are critical.
Consumer markets show expanding adoption patterns, with fitness enthusiasts and aging populations seeking non-invasive recovery solutions. The rise of home healthcare trends, accelerated by recent global health events, has created opportunities for portable PEMF devices targeting individual consumers. This demographic shift reflects broader healthcare decentralization and preventive care adoption.
Geographic demand varies significantly, with North American and European markets leading adoption due to established healthcare infrastructure and regulatory frameworks supporting electromagnetic therapy research. Asian markets, particularly Japan and South Korea, demonstrate growing interest driven by technological innovation cultures and aging populations experiencing fatigue-related health challenges.
Market barriers include regulatory complexity across different jurisdictions and varying levels of clinical evidence acceptance among healthcare providers. Insurance coverage limitations also constrain market expansion, as many PEMF applications remain classified as complementary rather than primary treatments. Despite these challenges, the convergence of wearable technology trends, personalized medicine approaches, and growing scientific validation of electromagnetic field effects on cellular recovery processes continues to drive market demand expansion across diverse application areas.
Current State and Challenges of PEMF in Fatigue Science
The current landscape of Pulsed Electromagnetic Field (PEMF) therapy in fatigue science presents a complex picture of emerging promise coupled with significant methodological and theoretical challenges. While PEMF technology has gained considerable attention as a non-invasive therapeutic intervention for various fatigue-related conditions, the field remains fragmented across different research domains, including chronic fatigue syndrome, cancer-related fatigue, and exercise-induced fatigue recovery.
Research efforts have predominantly focused on low-frequency PEMF applications, typically ranging from 1-100 Hz, with magnetic field intensities varying from 0.1 to 50 mT. However, the lack of standardized protocols represents a fundamental challenge, as studies employ vastly different exposure parameters, treatment durations, and outcome measures. This heterogeneity significantly hampers the ability to draw definitive conclusions about PEMF efficacy and optimal treatment protocols.
The mechanistic understanding of PEMF effects on fatigue remains incomplete, though several hypotheses have emerged. Current theories suggest PEMF may influence cellular energy metabolism through mitochondrial function enhancement, modulate inflammatory responses, and affect neurotransmitter systems involved in fatigue perception. However, the precise biological pathways through which PEMF exerts its effects are not fully elucidated, creating challenges for rational treatment design.
Clinical evidence presents mixed results, with some studies demonstrating significant improvements in fatigue scores while others show minimal or no effects. The quality of available research varies considerably, with many studies suffering from small sample sizes, inadequate control groups, and subjective outcome measures. Double-blinding remains particularly challenging due to the difficulty in creating truly inert sham devices that replicate the sensory experience of active PEMF treatment.
Regulatory frameworks for PEMF devices in fatigue applications remain underdeveloped in many jurisdictions. While some devices have received regulatory approval for specific indications, the evidence base supporting their use in fatigue management often relies on limited clinical data. This regulatory uncertainty creates barriers for both research advancement and clinical adoption.
The field also faces significant challenges in establishing biomarkers for fatigue assessment and treatment response monitoring. Current reliance on subjective fatigue scales limits the ability to objectively measure treatment effects and understand individual response variability. The development of objective, quantifiable measures of fatigue and recovery remains a critical unmet need that constrains progress in PEMF research and clinical application.
Research efforts have predominantly focused on low-frequency PEMF applications, typically ranging from 1-100 Hz, with magnetic field intensities varying from 0.1 to 50 mT. However, the lack of standardized protocols represents a fundamental challenge, as studies employ vastly different exposure parameters, treatment durations, and outcome measures. This heterogeneity significantly hampers the ability to draw definitive conclusions about PEMF efficacy and optimal treatment protocols.
The mechanistic understanding of PEMF effects on fatigue remains incomplete, though several hypotheses have emerged. Current theories suggest PEMF may influence cellular energy metabolism through mitochondrial function enhancement, modulate inflammatory responses, and affect neurotransmitter systems involved in fatigue perception. However, the precise biological pathways through which PEMF exerts its effects are not fully elucidated, creating challenges for rational treatment design.
Clinical evidence presents mixed results, with some studies demonstrating significant improvements in fatigue scores while others show minimal or no effects. The quality of available research varies considerably, with many studies suffering from small sample sizes, inadequate control groups, and subjective outcome measures. Double-blinding remains particularly challenging due to the difficulty in creating truly inert sham devices that replicate the sensory experience of active PEMF treatment.
Regulatory frameworks for PEMF devices in fatigue applications remain underdeveloped in many jurisdictions. While some devices have received regulatory approval for specific indications, the evidence base supporting their use in fatigue management often relies on limited clinical data. This regulatory uncertainty creates barriers for both research advancement and clinical adoption.
The field also faces significant challenges in establishing biomarkers for fatigue assessment and treatment response monitoring. Current reliance on subjective fatigue scales limits the ability to objectively measure treatment effects and understand individual response variability. The development of objective, quantifiable measures of fatigue and recovery remains a critical unmet need that constrains progress in PEMF research and clinical application.
Existing PEMF Solutions for Fatigue Management
01 PEMF therapy devices for treating chronic fatigue and related conditions
Pulsed electromagnetic field therapy devices are designed to address chronic fatigue syndrome and fatigue-related conditions through controlled electromagnetic stimulation. These devices utilize specific frequency ranges and pulse patterns to target cellular energy production and reduce fatigue symptoms. The therapeutic approach involves non-invasive application of electromagnetic fields to stimulate biological processes that combat fatigue at the cellular level.- PEMF therapy devices for treating chronic fatigue and related conditions: Pulsed electromagnetic field therapy devices are designed to address chronic fatigue syndrome and fatigue-related conditions through controlled electromagnetic stimulation. These devices utilize specific frequency ranges and pulse patterns to target cellular energy production and reduce fatigue symptoms. The therapeutic approach involves non-invasive application of electromagnetic fields to stimulate biological processes that combat fatigue at the cellular level.
- Waveform and frequency optimization for fatigue reduction: Advanced waveform configurations and frequency modulation techniques are employed to maximize the effectiveness of electromagnetic field therapy in reducing fatigue. These methods involve precise control of pulse duration, intensity, and repetition rates to achieve optimal biological responses. The technology focuses on matching electromagnetic parameters to specific physiological targets associated with energy metabolism and fatigue recovery.
- Portable and wearable PEMF devices for continuous fatigue management: Compact and wearable electromagnetic field therapy devices enable continuous or on-demand treatment of fatigue symptoms throughout daily activities. These portable systems incorporate battery-powered generators and flexible applicators that can be worn on various body parts. The design emphasizes user convenience and sustained therapeutic delivery for managing chronic fatigue conditions.
- Combination therapy integrating PEMF with other therapeutic modalities: Integrated treatment approaches combine pulsed electromagnetic field therapy with complementary therapeutic methods to enhance fatigue reduction outcomes. These systems may incorporate simultaneous or sequential application of multiple treatment modalities to address various aspects of fatigue pathophysiology. The combination strategies aim to provide synergistic effects for improved energy restoration and symptom relief.
- Monitoring and adaptive control systems for personalized fatigue treatment: Smart electromagnetic field therapy systems incorporate sensors and feedback mechanisms to monitor patient response and automatically adjust treatment parameters. These adaptive systems measure physiological indicators related to fatigue and modify electromagnetic field characteristics in real-time to optimize therapeutic outcomes. The technology enables personalized treatment protocols based on individual patient needs and response patterns.
02 Waveform and frequency optimization for fatigue reduction
Advanced waveform configurations and frequency modulation techniques are employed to maximize the effectiveness of electromagnetic field therapy in reducing fatigue. These methods involve precise control of pulse duration, intensity, and repetition rates to achieve optimal biological responses. The technology focuses on matching electromagnetic parameters to specific physiological targets associated with energy metabolism and fatigue recovery.Expand Specific Solutions03 Portable and wearable PEMF devices for continuous fatigue management
Compact and wearable electromagnetic field therapy devices enable continuous or on-demand treatment of fatigue symptoms throughout daily activities. These portable systems incorporate battery-powered generators and flexible applicators that can be worn on various body parts. The design emphasizes user convenience and sustained therapeutic delivery for managing chronic fatigue conditions.Expand Specific Solutions04 Combination therapy integrating PEMF with other therapeutic modalities
Integrated treatment approaches combine pulsed electromagnetic field therapy with complementary therapeutic methods to enhance fatigue reduction outcomes. These systems may incorporate simultaneous or sequential application of multiple treatment modalities to address various aspects of fatigue pathophysiology. The combination strategies aim to provide synergistic effects for improved energy restoration and symptom relief.Expand Specific Solutions05 Monitoring and adaptive control systems for personalized fatigue treatment
Smart electromagnetic field therapy systems incorporate sensors and feedback mechanisms to monitor patient response and automatically adjust treatment parameters. These adaptive systems measure physiological indicators related to fatigue and modify electromagnetic field characteristics in real-time to optimize therapeutic outcomes. The technology enables personalized treatment protocols based on individual patient needs and response patterns.Expand Specific Solutions
Key Players in PEMF Device and Fatigue Research Industry
The pulsed electromagnetic field (PEMF) technology in fatigue science represents an emerging therapeutic sector currently in its early-to-mid development stage. The market demonstrates significant growth potential driven by increasing awareness of non-invasive treatment modalities for fatigue-related conditions. The competitive landscape is characterized by a diverse ecosystem spanning academic research institutions and commercial entities. Leading Chinese universities including Fudan University, Xi'an Jiaotong University, and Beijing University of Technology are driving fundamental research, while specialized companies like SofPulse Inc., Wave Neuroscience Inc., and BrainQ Technologies Ltd. are advancing clinical applications. Technology maturity varies considerably, with academic institutions focusing on basic research and mechanism understanding, while commercial players like Neuroenhancement Lab LLC and Galvanize Therapeutics are developing market-ready solutions. The sector benefits from cross-disciplinary collaboration between medical device manufacturers, research hospitals, and technology companies, indicating a maturing but still fragmented competitive environment with substantial innovation opportunities.
Neuroenhancement Lab LLC
Technical Solution: Neuroenhancement Lab specializes in developing electromagnetic stimulation technologies for cognitive enhancement and neurological performance optimization. Their PEMF systems are designed to address various forms of mental and cognitive fatigue through targeted electromagnetic field application. The company's approach involves using specific electromagnetic frequencies that can influence neural activity patterns associated with fatigue and cognitive performance. Their technology platform includes both research-grade equipment for studying electromagnetic effects on fatigue and consumer-oriented devices for fatigue management applications.
Strengths: Focus on cognitive enhancement and neurological performance optimization. Weaknesses: Limited scope primarily targeting cognitive rather than physical fatigue applications.
SofPulse, Inc.
Technical Solution: SofPulse specializes in developing pulsed electromagnetic field (PEMF) therapy devices specifically designed for wound healing and tissue repair applications. Their technology utilizes low-frequency electromagnetic pulses to stimulate cellular regeneration and reduce inflammation, which directly addresses fatigue-related tissue damage and recovery processes. The company's devices operate at specific frequency ranges optimized for biological tissue interaction, promoting enhanced blood circulation and cellular metabolism that can accelerate recovery from physical fatigue and improve overall tissue health.
Strengths: Specialized PEMF technology with proven clinical applications in tissue repair. Weaknesses: Limited scope focused primarily on wound healing rather than comprehensive fatigue management.
Core PEMF Patents and Research in Fatigue Recovery
Pulsed Electromagnetic Field Therapy Device
PatentActiveUS20210370086A1
Innovation
- A pulsed electromagnetic field therapy device with a parallel resonant circuit that omits a switch between the capacitor and inductor, using an external switch to control current ramping, reducing energy dissipation and allowing for lower voltage operations, thereby increasing decay time and reducing electromagnetic interference.
System and method for applying a low frequency magnetic field to biological tissues
PatentActiveUS20240091548A1
Innovation
- The use of pulsed electromagnetic fields (PEMF) with specific frequency ranges (5-50 kHz) and intensities (millitesla range) is proposed to target cellular mechanisms, including ion movement and voltage-gated channels, to enhance healing and reduce inflammation.
Clinical Trial Requirements for PEMF Fatigue Devices
The regulatory landscape for PEMF fatigue devices requires adherence to stringent clinical trial protocols established by major health authorities including the FDA, EMA, and other international regulatory bodies. These devices are typically classified as Class II medical devices, necessitating comprehensive premarket submissions supported by robust clinical evidence demonstrating both safety and efficacy in fatigue management applications.
Clinical trial design for PEMF fatigue devices must incorporate randomized, double-blind, placebo-controlled methodologies to ensure scientific rigor. The challenge lies in creating effective sham devices that replicate the physical characteristics of active PEMF units without delivering therapeutic electromagnetic fields. Trial protocols must specify precise electromagnetic parameters including frequency ranges, field intensity, exposure duration, and treatment schedules to ensure reproducibility and regulatory compliance.
Patient recruitment criteria require careful consideration of fatigue etiology, with clear inclusion and exclusion parameters. Trials must differentiate between various fatigue types including chronic fatigue syndrome, cancer-related fatigue, multiple sclerosis fatigue, and general fatigue conditions. Standardized fatigue assessment tools such as the Fatigue Severity Scale, Multidimensional Fatigue Inventory, or Visual Analog Scale for fatigue must be implemented as primary outcome measures.
Safety monitoring protocols must address potential electromagnetic field exposure risks, particularly for patients with implanted medical devices such as pacemakers or cochlear implants. Comprehensive adverse event reporting systems and regular safety assessments throughout trial duration are mandatory requirements. Additionally, trials must establish clear protocols for device malfunction reporting and participant withdrawal procedures.
Regulatory submissions require detailed technical documentation including device specifications, electromagnetic field characterization data, biocompatibility testing results, and electrical safety certifications. Quality management systems compliant with ISO 13485 standards must be demonstrated throughout the clinical investigation process to meet regulatory expectations for medical device trials.
Clinical trial design for PEMF fatigue devices must incorporate randomized, double-blind, placebo-controlled methodologies to ensure scientific rigor. The challenge lies in creating effective sham devices that replicate the physical characteristics of active PEMF units without delivering therapeutic electromagnetic fields. Trial protocols must specify precise electromagnetic parameters including frequency ranges, field intensity, exposure duration, and treatment schedules to ensure reproducibility and regulatory compliance.
Patient recruitment criteria require careful consideration of fatigue etiology, with clear inclusion and exclusion parameters. Trials must differentiate between various fatigue types including chronic fatigue syndrome, cancer-related fatigue, multiple sclerosis fatigue, and general fatigue conditions. Standardized fatigue assessment tools such as the Fatigue Severity Scale, Multidimensional Fatigue Inventory, or Visual Analog Scale for fatigue must be implemented as primary outcome measures.
Safety monitoring protocols must address potential electromagnetic field exposure risks, particularly for patients with implanted medical devices such as pacemakers or cochlear implants. Comprehensive adverse event reporting systems and regular safety assessments throughout trial duration are mandatory requirements. Additionally, trials must establish clear protocols for device malfunction reporting and participant withdrawal procedures.
Regulatory submissions require detailed technical documentation including device specifications, electromagnetic field characterization data, biocompatibility testing results, and electrical safety certifications. Quality management systems compliant with ISO 13485 standards must be demonstrated throughout the clinical investigation process to meet regulatory expectations for medical device trials.
Safety Standards and Biocompatibility of PEMF Systems
The safety standards and biocompatibility of PEMF systems represent critical considerations for their application in fatigue science research and clinical practice. Current regulatory frameworks primarily follow guidelines established by the International Electrotechnical Commission (IEC) and the Food and Drug Administration (FDA), which classify PEMF devices based on their intended use and electromagnetic field characteristics. These standards mandate specific absorption rate (SAR) limits, typically not exceeding 2 watts per kilogram for localized exposure, ensuring thermal effects remain within safe physiological boundaries.
Biocompatibility assessments for PEMF systems encompass multiple evaluation criteria, including electromagnetic field intensity, frequency ranges, and exposure duration protocols. Research demonstrates that low-frequency PEMF applications, typically operating between 1-100 Hz with magnetic flux densities below 10 millitesla, exhibit excellent biocompatibility profiles with minimal adverse effects reported in clinical studies. The non-invasive nature of PEMF therapy eliminates concerns related to material biocompatibility that affect implantable devices.
International safety standards require comprehensive electromagnetic compatibility (EMC) testing to prevent interference with other medical devices, particularly pacemakers and implantable cardioverter defibrillators. PEMF systems must demonstrate compliance with IEC 60601-1-2 standards, ensuring safe operation in healthcare environments without compromising the functionality of adjacent electronic equipment.
Long-term safety data from fatigue-related PEMF applications indicate favorable safety profiles, with reported side effects limited to mild skin irritation in less than 2% of users. Contraindications primarily include pregnancy, active malignancies, and the presence of metallic implants in treatment areas. Quality assurance protocols mandate regular calibration of magnetic field generators and continuous monitoring of output parameters to maintain consistent therapeutic delivery.
Emerging biocompatibility research focuses on cellular-level interactions, investigating potential effects on gene expression and protein synthesis pathways. Current evidence suggests that therapeutic PEMF parameters do not induce genotoxic effects or cellular damage, supporting their continued application in fatigue management protocols while maintaining rigorous safety oversight.
Biocompatibility assessments for PEMF systems encompass multiple evaluation criteria, including electromagnetic field intensity, frequency ranges, and exposure duration protocols. Research demonstrates that low-frequency PEMF applications, typically operating between 1-100 Hz with magnetic flux densities below 10 millitesla, exhibit excellent biocompatibility profiles with minimal adverse effects reported in clinical studies. The non-invasive nature of PEMF therapy eliminates concerns related to material biocompatibility that affect implantable devices.
International safety standards require comprehensive electromagnetic compatibility (EMC) testing to prevent interference with other medical devices, particularly pacemakers and implantable cardioverter defibrillators. PEMF systems must demonstrate compliance with IEC 60601-1-2 standards, ensuring safe operation in healthcare environments without compromising the functionality of adjacent electronic equipment.
Long-term safety data from fatigue-related PEMF applications indicate favorable safety profiles, with reported side effects limited to mild skin irritation in less than 2% of users. Contraindications primarily include pregnancy, active malignancies, and the presence of metallic implants in treatment areas. Quality assurance protocols mandate regular calibration of magnetic field generators and continuous monitoring of output parameters to maintain consistent therapeutic delivery.
Emerging biocompatibility research focuses on cellular-level interactions, investigating potential effects on gene expression and protein synthesis pathways. Current evidence suggests that therapeutic PEMF parameters do not induce genotoxic effects or cellular damage, supporting their continued application in fatigue management protocols while maintaining rigorous safety oversight.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!