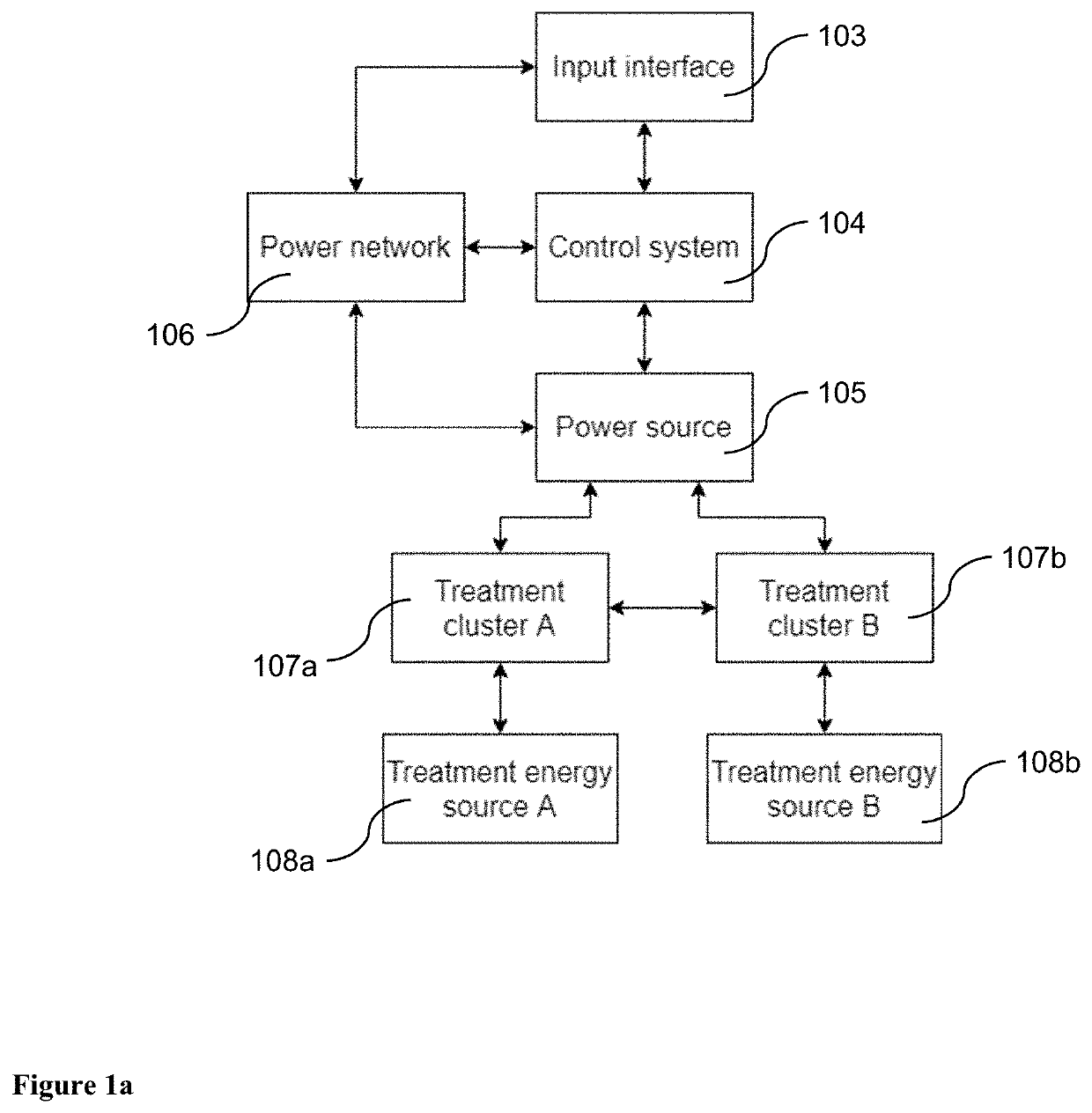
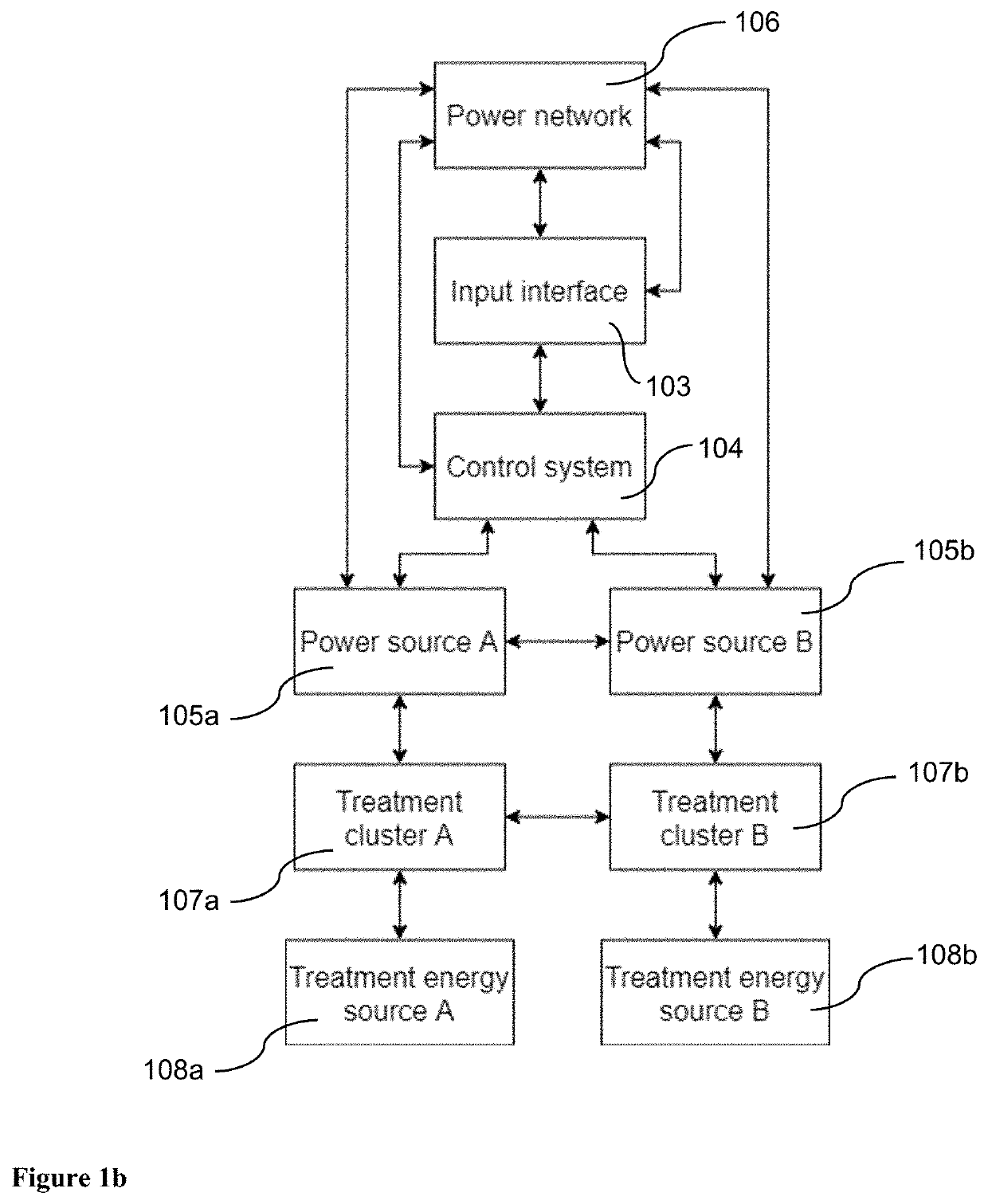
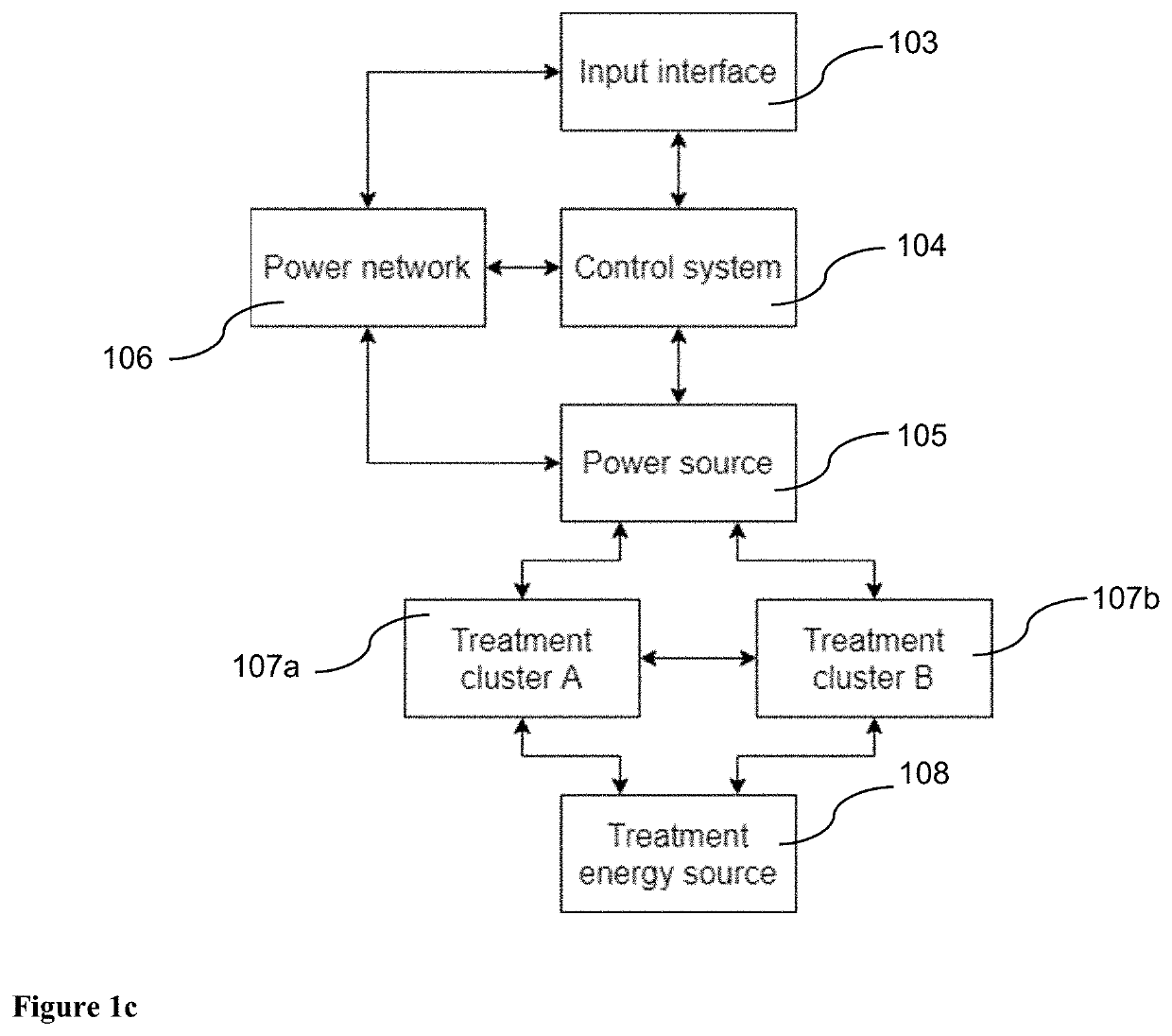
Pulsed Electromagnetic Field vs Microcurrent Therapy: Muscle Stimulation
MAR 7, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
PEMF vs Microcurrent Therapy Background and Objectives
Muscle stimulation therapy has evolved significantly over the past several decades, with two distinct technological approaches emerging as prominent solutions: Pulsed Electromagnetic Field (PEMF) therapy and Microcurrent Therapy. Both modalities represent sophisticated applications of bioelectromagnetic principles, yet they operate through fundamentally different mechanisms to achieve therapeutic muscle stimulation outcomes.
PEMF therapy traces its origins to the early 20th century discoveries in electromagnetic field applications, gaining substantial clinical recognition in the 1970s when the FDA first approved PEMF devices for bone healing. The technology has since expanded into muscle rehabilitation, leveraging time-varying magnetic fields to induce cellular-level electrical currents that promote tissue repair and muscle function enhancement. This non-invasive approach operates at frequencies typically ranging from 1 Hz to 10,000 Hz, with magnetic field intensities measured in Gauss or Tesla units.
Microcurrent therapy emerged from different scientific foundations, building upon the understanding of the body's natural bioelectrical systems. Developed in the 1980s, this approach delivers extremely low-level electrical currents, typically measured in microamperes, directly to targeted muscle tissues. The therapy operates on the principle that these sub-sensory currents can normalize cellular electrical activity and enhance ATP production at the mitochondrial level.
The convergence of these technologies addresses critical challenges in modern muscle rehabilitation and performance enhancement. Traditional muscle stimulation methods often face limitations in terms of patient comfort, treatment depth, and long-term efficacy. Both PEMF and microcurrent therapies emerged to overcome these constraints while providing more targeted, comfortable treatment options.
The primary objective of comparing these technologies centers on establishing their relative effectiveness in muscle stimulation applications. Key performance indicators include muscle strength recovery rates, pain reduction capabilities, treatment duration requirements, and patient compliance factors. Understanding the optimal application scenarios for each technology becomes crucial for healthcare providers and rehabilitation specialists.
Current research objectives focus on determining which technology delivers superior outcomes for specific muscle conditions, including post-surgical recovery, sports injury rehabilitation, chronic pain management, and muscle atrophy prevention. Additionally, investigating potential synergistic effects when combining both modalities represents an emerging area of clinical interest.
The technological evolution continues toward more precise, personalized treatment protocols that can adapt to individual patient responses and specific muscle group requirements, driving innovation in both PEMF and microcurrent therapy platforms.
PEMF therapy traces its origins to the early 20th century discoveries in electromagnetic field applications, gaining substantial clinical recognition in the 1970s when the FDA first approved PEMF devices for bone healing. The technology has since expanded into muscle rehabilitation, leveraging time-varying magnetic fields to induce cellular-level electrical currents that promote tissue repair and muscle function enhancement. This non-invasive approach operates at frequencies typically ranging from 1 Hz to 10,000 Hz, with magnetic field intensities measured in Gauss or Tesla units.
Microcurrent therapy emerged from different scientific foundations, building upon the understanding of the body's natural bioelectrical systems. Developed in the 1980s, this approach delivers extremely low-level electrical currents, typically measured in microamperes, directly to targeted muscle tissues. The therapy operates on the principle that these sub-sensory currents can normalize cellular electrical activity and enhance ATP production at the mitochondrial level.
The convergence of these technologies addresses critical challenges in modern muscle rehabilitation and performance enhancement. Traditional muscle stimulation methods often face limitations in terms of patient comfort, treatment depth, and long-term efficacy. Both PEMF and microcurrent therapies emerged to overcome these constraints while providing more targeted, comfortable treatment options.
The primary objective of comparing these technologies centers on establishing their relative effectiveness in muscle stimulation applications. Key performance indicators include muscle strength recovery rates, pain reduction capabilities, treatment duration requirements, and patient compliance factors. Understanding the optimal application scenarios for each technology becomes crucial for healthcare providers and rehabilitation specialists.
Current research objectives focus on determining which technology delivers superior outcomes for specific muscle conditions, including post-surgical recovery, sports injury rehabilitation, chronic pain management, and muscle atrophy prevention. Additionally, investigating potential synergistic effects when combining both modalities represents an emerging area of clinical interest.
The technological evolution continues toward more precise, personalized treatment protocols that can adapt to individual patient responses and specific muscle group requirements, driving innovation in both PEMF and microcurrent therapy platforms.
Market Demand for Muscle Stimulation Technologies
The global muscle stimulation technology market demonstrates robust growth driven by multiple converging factors across healthcare, sports, and wellness sectors. Aging populations worldwide increasingly seek non-invasive therapeutic solutions for muscle weakness, pain management, and rehabilitation, creating substantial demand for both pulsed electromagnetic field and microcurrent therapy devices.
Healthcare institutions represent the largest market segment, with hospitals, rehabilitation centers, and physical therapy clinics adopting these technologies for post-surgical recovery, neurological rehabilitation, and chronic pain management. The growing prevalence of musculoskeletal disorders, estimated to affect hundreds of millions globally, drives consistent demand for effective muscle stimulation solutions.
The sports medicine and athletic performance market exhibits particularly strong growth momentum. Professional sports teams, fitness centers, and individual athletes increasingly recognize the benefits of muscle stimulation for performance enhancement, injury prevention, and accelerated recovery. This segment values both the deep tissue penetration capabilities of pulsed electromagnetic fields and the precise, localized treatment offered by microcurrent therapy.
Consumer wellness markets show expanding adoption patterns, with home-use devices gaining popularity among health-conscious individuals. The convenience of portable muscle stimulation devices appeals to users seeking proactive muscle health management and pain relief without frequent clinical visits. This trend accelerates demand for user-friendly, FDA-cleared devices suitable for personal use.
Demographic shifts significantly influence market dynamics. Younger populations gravitate toward technologically advanced solutions with smartphone connectivity and customizable treatment protocols, while older demographics prioritize ease of use and proven clinical efficacy. Both pulsed electromagnetic field and microcurrent technologies address these diverse preferences through different device configurations and treatment approaches.
Geographic market distribution reveals strong demand in developed regions with established healthcare infrastructure, while emerging markets show increasing adoption as healthcare access expands. Regulatory approvals and clinical evidence supporting both technologies continue to drive market acceptance and professional adoption across various therapeutic applications.
Healthcare institutions represent the largest market segment, with hospitals, rehabilitation centers, and physical therapy clinics adopting these technologies for post-surgical recovery, neurological rehabilitation, and chronic pain management. The growing prevalence of musculoskeletal disorders, estimated to affect hundreds of millions globally, drives consistent demand for effective muscle stimulation solutions.
The sports medicine and athletic performance market exhibits particularly strong growth momentum. Professional sports teams, fitness centers, and individual athletes increasingly recognize the benefits of muscle stimulation for performance enhancement, injury prevention, and accelerated recovery. This segment values both the deep tissue penetration capabilities of pulsed electromagnetic fields and the precise, localized treatment offered by microcurrent therapy.
Consumer wellness markets show expanding adoption patterns, with home-use devices gaining popularity among health-conscious individuals. The convenience of portable muscle stimulation devices appeals to users seeking proactive muscle health management and pain relief without frequent clinical visits. This trend accelerates demand for user-friendly, FDA-cleared devices suitable for personal use.
Demographic shifts significantly influence market dynamics. Younger populations gravitate toward technologically advanced solutions with smartphone connectivity and customizable treatment protocols, while older demographics prioritize ease of use and proven clinical efficacy. Both pulsed electromagnetic field and microcurrent technologies address these diverse preferences through different device configurations and treatment approaches.
Geographic market distribution reveals strong demand in developed regions with established healthcare infrastructure, while emerging markets show increasing adoption as healthcare access expands. Regulatory approvals and clinical evidence supporting both technologies continue to drive market acceptance and professional adoption across various therapeutic applications.
Current State of PEMF and Microcurrent Therapy Challenges
Both PEMF and microcurrent therapy have established themselves as viable muscle stimulation modalities, yet each faces distinct technological and clinical challenges that limit their broader adoption and optimization. The current landscape reveals significant disparities in standardization, efficacy validation, and practical implementation across different therapeutic contexts.
PEMF technology confronts substantial challenges in parameter standardization, with frequency ranges varying dramatically from 1 Hz to several thousand Hz across different devices and applications. The lack of consensus on optimal magnetic field intensity, pulse duration, and treatment protocols creates confusion among practitioners and inconsistent clinical outcomes. Current PEMF systems struggle with precise field penetration control, as magnetic field distribution becomes increasingly heterogeneous with tissue depth, making it difficult to target specific muscle groups effectively.
Device portability and power consumption represent additional hurdles for PEMF systems. Most effective PEMF units require substantial power infrastructure, limiting their use in portable or home-care settings. The electromagnetic interference potential of PEMF devices also restricts their application in clinical environments with sensitive electronic equipment, creating operational constraints for healthcare facilities.
Microcurrent therapy faces different but equally significant challenges, primarily centered around electrode placement precision and current distribution uniformity. The therapy's effectiveness heavily depends on optimal electrode positioning, yet current guidance systems lack sophistication, often relying on practitioner experience rather than objective measurement tools. Skin impedance variations significantly affect current delivery, with factors such as hydration levels, skin thickness, and electrode contact quality creating unpredictable treatment variables.
Patient compliance represents a critical challenge for microcurrent therapy, as treatment protocols typically require extended session durations and frequent applications. The subtle nature of microcurrent stimulation, often imperceptible to patients, raises questions about treatment adherence and psychological placebo effects, complicating clinical outcome assessments.
Both modalities struggle with limited real-time monitoring capabilities and objective outcome measurement tools. Current assessment methods rely heavily on subjective patient reporting and basic functional tests, lacking sophisticated biomarkers or imaging techniques that could provide quantitative treatment efficacy data. This measurement gap impedes evidence-based protocol optimization and comparative effectiveness research between the two therapeutic approaches.
PEMF technology confronts substantial challenges in parameter standardization, with frequency ranges varying dramatically from 1 Hz to several thousand Hz across different devices and applications. The lack of consensus on optimal magnetic field intensity, pulse duration, and treatment protocols creates confusion among practitioners and inconsistent clinical outcomes. Current PEMF systems struggle with precise field penetration control, as magnetic field distribution becomes increasingly heterogeneous with tissue depth, making it difficult to target specific muscle groups effectively.
Device portability and power consumption represent additional hurdles for PEMF systems. Most effective PEMF units require substantial power infrastructure, limiting their use in portable or home-care settings. The electromagnetic interference potential of PEMF devices also restricts their application in clinical environments with sensitive electronic equipment, creating operational constraints for healthcare facilities.
Microcurrent therapy faces different but equally significant challenges, primarily centered around electrode placement precision and current distribution uniformity. The therapy's effectiveness heavily depends on optimal electrode positioning, yet current guidance systems lack sophistication, often relying on practitioner experience rather than objective measurement tools. Skin impedance variations significantly affect current delivery, with factors such as hydration levels, skin thickness, and electrode contact quality creating unpredictable treatment variables.
Patient compliance represents a critical challenge for microcurrent therapy, as treatment protocols typically require extended session durations and frequent applications. The subtle nature of microcurrent stimulation, often imperceptible to patients, raises questions about treatment adherence and psychological placebo effects, complicating clinical outcome assessments.
Both modalities struggle with limited real-time monitoring capabilities and objective outcome measurement tools. Current assessment methods rely heavily on subjective patient reporting and basic functional tests, lacking sophisticated biomarkers or imaging techniques that could provide quantitative treatment efficacy data. This measurement gap impedes evidence-based protocol optimization and comparative effectiveness research between the two therapeutic approaches.
Existing PEMF and Microcurrent Therapy Solutions
01 Pulsed electromagnetic field therapy devices for muscle stimulation and tissue healing
Devices utilizing pulsed electromagnetic fields (PEMF) are designed to stimulate muscle tissue and promote healing through controlled electromagnetic pulses. These systems generate specific frequency ranges and pulse patterns to penetrate tissue and induce cellular responses. The technology can be applied for muscle rehabilitation, pain management, and accelerating recovery processes by influencing cellular metabolism and ion transport mechanisms.- Pulsed electromagnetic field therapy devices for muscle stimulation and tissue healing: Devices utilizing pulsed electromagnetic fields (PEMF) are designed to stimulate muscle tissue and promote healing through controlled electromagnetic pulses. These systems generate specific frequency ranges and pulse patterns to penetrate tissue and induce cellular responses. The technology can be applied for muscle rehabilitation, pain management, and accelerating recovery processes by influencing cellular metabolism and ion transport mechanisms.
- Microcurrent electrical stimulation systems for therapeutic applications: Microcurrent therapy devices deliver low-level electrical currents to target tissues for therapeutic purposes. These systems operate at subsensory levels, typically in the microampere range, to stimulate cellular activity and promote tissue repair. The technology is used for muscle conditioning, pain relief, and enhancing muscle function through gentle electrical stimulation that mimics natural bioelectrical processes.
- Combined electromagnetic and electrical stimulation therapy systems: Integrated therapeutic devices that combine both electromagnetic field generation and electrical current delivery for enhanced muscle stimulation effects. These hybrid systems leverage the synergistic benefits of multiple stimulation modalities to achieve improved therapeutic outcomes. The combination approach allows for simultaneous or sequential application of different energy forms to optimize muscle activation and recovery.
- Wearable and portable muscle stimulation devices: Compact, user-friendly devices designed for convenient application of electromagnetic or microcurrent therapy in various settings. These portable systems enable patients to receive treatment outside clinical environments, featuring ergonomic designs and adjustable parameters. The devices often incorporate wireless control, battery operation, and flexible electrode configurations for targeted muscle group stimulation.
- Control systems and parameter optimization for muscle stimulation therapy: Advanced control mechanisms and algorithms for optimizing stimulation parameters in electromagnetic and microcurrent therapy applications. These systems feature programmable pulse patterns, frequency modulation, and intensity adjustment capabilities to customize treatment protocols. The technology includes feedback mechanisms and monitoring systems to ensure safe and effective delivery of therapeutic stimulation based on individual patient responses.
02 Microcurrent electrical stimulation systems for therapeutic applications
Microcurrent therapy systems deliver low-level electrical currents to target tissues for therapeutic purposes. These devices operate at subsensory levels, typically in the microampere range, to stimulate cellular repair mechanisms and enhance muscle function. The technology is used for muscle conditioning, pain relief, and promoting tissue regeneration through bioelectrical stimulation that mimics natural cellular electrical activity.Expand Specific Solutions03 Combined electromagnetic and electrical stimulation therapy systems
Integrated therapeutic systems that combine both electromagnetic field generation and electrical current delivery for enhanced muscle stimulation effects. These hybrid approaches utilize synergistic mechanisms to optimize therapeutic outcomes by simultaneously applying different energy modalities. The combined therapy can provide improved muscle activation, enhanced blood circulation, and accelerated healing compared to single-modality treatments.Expand Specific Solutions04 Wearable and portable muscle stimulation devices
Compact, portable therapeutic devices designed for convenient application of electromagnetic or microcurrent stimulation to muscles. These systems feature ergonomic designs, battery operation, and user-friendly interfaces for home or clinical use. The devices often include adjustable parameters, multiple electrode configurations, and programmable treatment protocols to accommodate various therapeutic needs and target different muscle groups.Expand Specific Solutions05 Control systems and treatment protocols for optimized muscle stimulation
Advanced control mechanisms and treatment methodologies for regulating electromagnetic field and microcurrent parameters during muscle stimulation therapy. These systems incorporate feedback mechanisms, programmable pulse sequences, and adaptive algorithms to customize treatment based on patient response. The technology enables precise control of stimulation intensity, frequency, duration, and waveform characteristics to maximize therapeutic efficacy while ensuring patient safety.Expand Specific Solutions
Key Players in PEMF and Microcurrent Device Industry
The muscle stimulation therapy market comparing Pulsed Electromagnetic Field (PEMF) and Microcurrent technologies is experiencing rapid growth, driven by increasing demand for non-invasive rehabilitation solutions. The industry is in a mature expansion phase with established players like Medtronic, BTL Medical Solutions, and DJO LLC dominating through comprehensive product portfolios. Technology maturity varies significantly across segments - PEMF therapy shows high maturity with companies like STORZ MEDICAL and Re5 ApS offering proven clinical applications, while microcurrent therapy demonstrates emerging potential through innovators like Highland Instruments and Pulse Biosciences developing next-generation stimulation platforms. The competitive landscape features a mix of established medical device manufacturers, specialized therapy equipment providers, and emerging technology companies, indicating a dynamic market with substantial growth opportunities across both therapeutic modalities.
BTL Medical Solutions as
Technical Solution: BTL Medical Solutions specializes in advanced electromagnetic field therapy systems for muscle stimulation and rehabilitation. Their technology utilizes high-intensity focused electromagnetic (HIFEM) fields that penetrate deep into muscle tissue, generating supramaximal contractions that are impossible to achieve through voluntary muscle action. The company's PEMF systems operate at specific frequencies ranging from 1-100 Hz, delivering targeted electromagnetic pulses that enhance cellular metabolism, improve blood circulation, and accelerate muscle recovery. Their devices feature precise control algorithms that adjust field intensity and pulse duration based on treatment protocols, ensuring optimal therapeutic outcomes while maintaining patient safety through real-time monitoring systems.
Strengths: Deep tissue penetration capability, non-invasive treatment approach, proven clinical efficacy in muscle strengthening. Weaknesses: Higher equipment costs, requires specialized training for operators, limited portability of devices.
DJO LLC
Technical Solution: DJO LLC develops comprehensive electrotherapy solutions combining both PEMF and microcurrent technologies for muscle stimulation applications. Their microcurrent therapy systems deliver low-amplitude electrical currents (typically 50-1000 microamperes) that mimic the body's natural bioelectrical processes, promoting cellular repair and reducing inflammation. The company's PEMF devices generate pulsed magnetic fields at therapeutic frequencies, creating induced electrical currents within tissues to stimulate muscle fibers and enhance neuromuscular function. Their integrated approach allows clinicians to select optimal treatment modalities based on patient-specific conditions, with programmable protocols that can combine both technologies for synergistic therapeutic effects in rehabilitation and performance enhancement applications.
Strengths: Dual-technology platform offering treatment flexibility, established clinical validation, comprehensive training and support programs. Weaknesses: Complex device operation requiring extensive user education, higher maintenance requirements for dual-system devices.
Core Patents in Electromagnetic Muscle Stimulation
Apparatus and method for microcurrent-stimulation therapy
PatentActiveUS20240216687A1
Innovation
- A system comprising a headset with individually controlled electrodes and sensors that deliver bio-electric microcurrent, light, and heat stimulation therapies, connected to a controller that adjusts therapy parameters based on real-time impedance feedback to ensure consistent treatment delivery, targeting specific areas around the eye with precision.
Methods and devices for aesthetic treatment of biological structures by radiofrequency and magnetic energy
PatentActiveUS20220096142A1
Innovation
- A treatment device and method utilizing segmented RF electrodes with apertures, cutouts, or protrusions to minimize eddy currents and overheating, combined with an energy storage device for magnetic field generation, allowing precise targeting and synergistic effects of RF and magnetic treatments for muscle contraction and heating, while preventing interaction between electric and magnetic signals.
FDA Regulatory Framework for Muscle Stimulation Devices
The FDA regulatory framework for muscle stimulation devices establishes a comprehensive classification system that directly impacts both pulsed electromagnetic field (PEMF) and microcurrent therapy technologies. These devices are primarily categorized under Class II medical devices, requiring 510(k) premarket notification to demonstrate substantial equivalence to predicate devices already cleared by the FDA.
PEMF devices typically fall under the FDA's classification for electromagnetic therapy equipment, specifically regulated under 21 CFR 890.5850 for stimulator devices. The regulatory pathway requires manufacturers to provide clinical evidence demonstrating safety and efficacy for intended muscle stimulation applications. Key regulatory considerations include electromagnetic compatibility testing, biocompatibility assessments, and electrical safety standards compliance.
Microcurrent therapy devices are regulated under similar provisions but often require additional scrutiny regarding current delivery mechanisms and patient safety protocols. The FDA mandates specific labeling requirements, including contraindications, warnings, and proper usage instructions. Manufacturers must demonstrate that microcurrent levels remain within safe therapeutic ranges while achieving intended muscle stimulation effects.
Both technology categories must comply with FDA Quality System Regulation (QSR) requirements, encompassing design controls, manufacturing processes, and post-market surveillance obligations. The regulatory framework emphasizes risk management throughout the device lifecycle, requiring comprehensive risk analysis documentation and mitigation strategies for potential adverse events.
Recent FDA guidance documents have clarified expectations for clinical trial design and endpoint selection for muscle stimulation devices. The agency requires robust clinical data demonstrating measurable improvements in muscle function, strength, or rehabilitation outcomes. Comparative effectiveness studies between PEMF and microcurrent technologies may be necessary when making superiority claims.
Post-market requirements include adverse event reporting through the Medical Device Reporting (MDR) system and compliance with FDA's unique device identification (UDI) regulations. Manufacturers must maintain comprehensive quality management systems and be prepared for FDA inspections to ensure ongoing regulatory compliance and patient safety standards.
PEMF devices typically fall under the FDA's classification for electromagnetic therapy equipment, specifically regulated under 21 CFR 890.5850 for stimulator devices. The regulatory pathway requires manufacturers to provide clinical evidence demonstrating safety and efficacy for intended muscle stimulation applications. Key regulatory considerations include electromagnetic compatibility testing, biocompatibility assessments, and electrical safety standards compliance.
Microcurrent therapy devices are regulated under similar provisions but often require additional scrutiny regarding current delivery mechanisms and patient safety protocols. The FDA mandates specific labeling requirements, including contraindications, warnings, and proper usage instructions. Manufacturers must demonstrate that microcurrent levels remain within safe therapeutic ranges while achieving intended muscle stimulation effects.
Both technology categories must comply with FDA Quality System Regulation (QSR) requirements, encompassing design controls, manufacturing processes, and post-market surveillance obligations. The regulatory framework emphasizes risk management throughout the device lifecycle, requiring comprehensive risk analysis documentation and mitigation strategies for potential adverse events.
Recent FDA guidance documents have clarified expectations for clinical trial design and endpoint selection for muscle stimulation devices. The agency requires robust clinical data demonstrating measurable improvements in muscle function, strength, or rehabilitation outcomes. Comparative effectiveness studies between PEMF and microcurrent technologies may be necessary when making superiority claims.
Post-market requirements include adverse event reporting through the Medical Device Reporting (MDR) system and compliance with FDA's unique device identification (UDI) regulations. Manufacturers must maintain comprehensive quality management systems and be prepared for FDA inspections to ensure ongoing regulatory compliance and patient safety standards.
Clinical Efficacy Comparison Studies and Standards
The establishment of standardized clinical efficacy comparison protocols for pulsed electromagnetic field therapy and microcurrent therapy in muscle stimulation applications represents a critical gap in current therapeutic evaluation frameworks. Existing clinical trial methodologies often lack consistency in outcome measurement parameters, treatment duration protocols, and patient selection criteria, leading to fragmented evidence bases that complicate direct therapeutic comparisons.
Current clinical efficacy standards primarily rely on subjective pain assessment scales, functional mobility measurements, and electromyographic readings to evaluate muscle stimulation effectiveness. However, these metrics demonstrate significant variability across different research institutions and clinical settings. The absence of universally accepted biomarkers for muscle recovery and stimulation response creates challenges in establishing definitive therapeutic superiority between electromagnetic field and microcurrent modalities.
Comparative clinical studies reveal substantial methodological inconsistencies in treatment parameter standardization. Pulsed electromagnetic field studies typically employ frequency ranges from 1-100 Hz with varying magnetic field intensities, while microcurrent therapy investigations utilize current amplitudes between 1-1000 microamperes. This parameter diversity significantly impacts study reproducibility and cross-study comparison validity, necessitating more rigorous standardization protocols.
Recent meta-analyses highlight the need for standardized outcome measurement tools specifically designed for muscle stimulation therapies. Proposed evaluation frameworks should incorporate quantitative muscle strength assessments, tissue oxygenation measurements, inflammatory biomarker analysis, and patient-reported outcome measures using validated instruments. These comprehensive assessment protocols would enable more accurate therapeutic efficacy comparisons.
The development of evidence-based clinical practice guidelines requires establishing minimum study duration requirements, standardized control group protocols, and consistent follow-up assessment timelines. Current literature suggests optimal study periods of 6-12 weeks for acute muscle conditions and 3-6 months for chronic applications, with standardized assessment intervals at 2, 4, 8, and 12-week markers to capture both immediate and sustained therapeutic effects across both treatment modalities.
Current clinical efficacy standards primarily rely on subjective pain assessment scales, functional mobility measurements, and electromyographic readings to evaluate muscle stimulation effectiveness. However, these metrics demonstrate significant variability across different research institutions and clinical settings. The absence of universally accepted biomarkers for muscle recovery and stimulation response creates challenges in establishing definitive therapeutic superiority between electromagnetic field and microcurrent modalities.
Comparative clinical studies reveal substantial methodological inconsistencies in treatment parameter standardization. Pulsed electromagnetic field studies typically employ frequency ranges from 1-100 Hz with varying magnetic field intensities, while microcurrent therapy investigations utilize current amplitudes between 1-1000 microamperes. This parameter diversity significantly impacts study reproducibility and cross-study comparison validity, necessitating more rigorous standardization protocols.
Recent meta-analyses highlight the need for standardized outcome measurement tools specifically designed for muscle stimulation therapies. Proposed evaluation frameworks should incorporate quantitative muscle strength assessments, tissue oxygenation measurements, inflammatory biomarker analysis, and patient-reported outcome measures using validated instruments. These comprehensive assessment protocols would enable more accurate therapeutic efficacy comparisons.
The development of evidence-based clinical practice guidelines requires establishing minimum study duration requirements, standardized control group protocols, and consistent follow-up assessment timelines. Current literature suggests optimal study periods of 6-12 weeks for acute muscle conditions and 3-6 months for chronic applications, with standardized assessment intervals at 2, 4, 8, and 12-week markers to capture both immediate and sustained therapeutic effects across both treatment modalities.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!