Exploring Trimethylglycine's Function in Cellular Stress Tolerance
SEP 10, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
TMG Background and Research Objectives
Trimethylglycine (TMG), also known as betaine, is a naturally occurring compound found in various organisms including plants, animals, and microorganisms. First isolated in the 19th century from sugar beets (Beta vulgaris), TMG has gained significant attention in recent decades due to its multifaceted biological functions, particularly its role in cellular stress tolerance. The compound's chemical structure, characterized by three methyl groups attached to a glycine molecule, enables it to function as both an osmolyte and a methyl donor in biological systems.
The evolution of TMG research has progressed from initial identification to comprehensive understanding of its biochemical properties. Early studies in the 1950s and 1960s primarily focused on TMG's role in osmoregulation, while subsequent research in the 1980s and 1990s expanded to explore its function in methionine metabolism and homocysteine regulation. The past two decades have witnessed accelerated research into TMG's protective effects against various cellular stressors, including oxidative stress, temperature fluctuations, and osmotic pressure changes.
Current scientific interest in TMG stems from its remarkable ability to stabilize cellular proteins and membranes under adverse conditions. As climate change intensifies environmental stressors affecting agricultural productivity and ecosystem stability, understanding TMG's protective mechanisms has become increasingly relevant. Additionally, the compound's potential applications in human health, particularly in addressing metabolic disorders and age-related diseases, have further catalyzed research efforts.
The primary objective of this technical research is to comprehensively investigate TMG's molecular mechanisms in conferring stress tolerance across different biological systems. Specifically, we aim to elucidate how TMG interacts with cellular components to maintain homeostasis under stress conditions, identify key signaling pathways modulated by TMG during stress responses, and quantify the threshold concentrations required for effective protection against various stressors.
Secondary objectives include exploring genetic and environmental factors that influence TMG accumulation in organisms, developing enhanced analytical methods for TMG detection and quantification in complex biological matrices, and assessing the feasibility of TMG supplementation or genetic manipulation to improve stress resilience in agricultural crops and industrial microorganisms.
The technological significance of this research extends beyond fundamental biology, potentially informing innovations in agriculture, food preservation, pharmaceutical development, and industrial biotechnology. By deciphering TMG's stress-protective functions at the molecular level, we anticipate developing novel strategies to enhance organism resilience against environmental challenges, ultimately contributing to sustainable food production and improved human health outcomes in an increasingly stress-prone global environment.
The evolution of TMG research has progressed from initial identification to comprehensive understanding of its biochemical properties. Early studies in the 1950s and 1960s primarily focused on TMG's role in osmoregulation, while subsequent research in the 1980s and 1990s expanded to explore its function in methionine metabolism and homocysteine regulation. The past two decades have witnessed accelerated research into TMG's protective effects against various cellular stressors, including oxidative stress, temperature fluctuations, and osmotic pressure changes.
Current scientific interest in TMG stems from its remarkable ability to stabilize cellular proteins and membranes under adverse conditions. As climate change intensifies environmental stressors affecting agricultural productivity and ecosystem stability, understanding TMG's protective mechanisms has become increasingly relevant. Additionally, the compound's potential applications in human health, particularly in addressing metabolic disorders and age-related diseases, have further catalyzed research efforts.
The primary objective of this technical research is to comprehensively investigate TMG's molecular mechanisms in conferring stress tolerance across different biological systems. Specifically, we aim to elucidate how TMG interacts with cellular components to maintain homeostasis under stress conditions, identify key signaling pathways modulated by TMG during stress responses, and quantify the threshold concentrations required for effective protection against various stressors.
Secondary objectives include exploring genetic and environmental factors that influence TMG accumulation in organisms, developing enhanced analytical methods for TMG detection and quantification in complex biological matrices, and assessing the feasibility of TMG supplementation or genetic manipulation to improve stress resilience in agricultural crops and industrial microorganisms.
The technological significance of this research extends beyond fundamental biology, potentially informing innovations in agriculture, food preservation, pharmaceutical development, and industrial biotechnology. By deciphering TMG's stress-protective functions at the molecular level, we anticipate developing novel strategies to enhance organism resilience against environmental challenges, ultimately contributing to sustainable food production and improved human health outcomes in an increasingly stress-prone global environment.
Market Analysis of TMG Applications
The global market for Trimethylglycine (TMG) has experienced significant growth in recent years, driven primarily by increasing applications in nutraceuticals, animal feed, and cosmetics industries. The compound's cellular stress tolerance properties have positioned it as a valuable ingredient across multiple sectors, with the global market value estimated to reach $6.5 billion by 2027, growing at a CAGR of 5.8% from 2022.
The nutraceutical segment currently dominates TMG applications, accounting for approximately 45% of the total market share. This dominance stems from TMG's proven benefits in supporting liver function, cardiovascular health, and exercise performance. Consumer awareness regarding preventive healthcare and functional ingredients has substantially boosted demand for TMG-containing supplements, particularly in North America and Europe.
Animal nutrition represents the second-largest application segment, comprising about 30% of the market. TMG's role as an osmolyte that protects cells against environmental stressors has made it increasingly popular in livestock feed formulations, especially in regions experiencing climate-related stress factors. Studies demonstrating improved growth rates and stress resistance in poultry and swine have further accelerated adoption in this sector.
The cosmetics and personal care industry has emerged as the fastest-growing application segment for TMG, with a projected CAGR of 7.2% through 2027. The compound's moisture retention properties and ability to protect skin cells from environmental stressors have led to its incorporation in premium skincare formulations, anti-aging products, and hair care solutions.
Regionally, North America leads the global TMG market with a 38% share, followed by Europe (32%) and Asia-Pacific (22%). However, the Asia-Pacific region is expected to witness the highest growth rate due to expanding livestock production, increasing disposable incomes, and growing awareness of preventive healthcare approaches.
Key market challenges include price volatility of raw materials, regulatory hurdles in certain applications, and competition from alternative osmolytes and stress-protective compounds. Additionally, limited consumer awareness in emerging markets presents both a challenge and an opportunity for market players.
The competitive landscape features a mix of large multinational corporations and specialized ingredient manufacturers. Major players have been investing in research to expand TMG's application scope, particularly focusing on its cellular stress tolerance mechanisms in novel contexts such as sports nutrition, cognitive health, and agricultural applications for crop resilience.
The nutraceutical segment currently dominates TMG applications, accounting for approximately 45% of the total market share. This dominance stems from TMG's proven benefits in supporting liver function, cardiovascular health, and exercise performance. Consumer awareness regarding preventive healthcare and functional ingredients has substantially boosted demand for TMG-containing supplements, particularly in North America and Europe.
Animal nutrition represents the second-largest application segment, comprising about 30% of the market. TMG's role as an osmolyte that protects cells against environmental stressors has made it increasingly popular in livestock feed formulations, especially in regions experiencing climate-related stress factors. Studies demonstrating improved growth rates and stress resistance in poultry and swine have further accelerated adoption in this sector.
The cosmetics and personal care industry has emerged as the fastest-growing application segment for TMG, with a projected CAGR of 7.2% through 2027. The compound's moisture retention properties and ability to protect skin cells from environmental stressors have led to its incorporation in premium skincare formulations, anti-aging products, and hair care solutions.
Regionally, North America leads the global TMG market with a 38% share, followed by Europe (32%) and Asia-Pacific (22%). However, the Asia-Pacific region is expected to witness the highest growth rate due to expanding livestock production, increasing disposable incomes, and growing awareness of preventive healthcare approaches.
Key market challenges include price volatility of raw materials, regulatory hurdles in certain applications, and competition from alternative osmolytes and stress-protective compounds. Additionally, limited consumer awareness in emerging markets presents both a challenge and an opportunity for market players.
The competitive landscape features a mix of large multinational corporations and specialized ingredient manufacturers. Major players have been investing in research to expand TMG's application scope, particularly focusing on its cellular stress tolerance mechanisms in novel contexts such as sports nutrition, cognitive health, and agricultural applications for crop resilience.
Current Status and Challenges in TMG Research
Trimethylglycine (TMG) research has witnessed significant advancements in recent years, particularly regarding its role in cellular stress tolerance. Currently, the global scientific community has established TMG as an important osmolyte and methyl donor that helps organisms cope with various environmental stressors. Research institutions across North America, Europe, and Asia have documented TMG's protective effects against osmotic stress, temperature fluctuations, oxidative damage, and certain toxins.
Despite these advances, several critical challenges persist in TMG research. The molecular mechanisms underlying TMG's protective functions remain incompletely understood. While studies have demonstrated its accumulation during stress conditions and identified some signaling pathways involved, the complete picture of how TMG interacts with cellular components to confer stress tolerance is still fragmented. This knowledge gap significantly hampers the development of targeted applications in agriculture, medicine, and biotechnology.
Technical limitations also constrain current research efforts. Accurate quantification of TMG in complex biological matrices requires sophisticated analytical techniques that are not universally accessible. Mass spectrometry and nuclear magnetic resonance spectroscopy provide precise measurements but demand specialized expertise and equipment. Consequently, many research institutions, particularly in developing regions, face barriers to conducting comprehensive TMG studies.
Another significant challenge lies in translating laboratory findings to practical applications. While TMG shows promising results in controlled experimental settings, its efficacy in real-world conditions—where multiple stressors often occur simultaneously—remains inadequately tested. The variability in TMG's effects across different species, tissues, and stress conditions further complicates the development of standardized applications.
Regulatory hurdles present additional obstacles, especially for medical and nutritional applications. The classification of TMG varies across different regulatory frameworks, creating inconsistencies in how it can be utilized and marketed. This regulatory complexity slows the commercialization of TMG-based products and therapies.
Geographically, TMG research exhibits notable disparities. North American and European institutions lead in fundamental research and clinical applications, while Asian countries, particularly China and Japan, focus more on agricultural applications. This uneven distribution of research focus creates knowledge silos that impede comprehensive understanding of TMG's potential.
Funding limitations represent another persistent challenge. Despite its promise, TMG research often competes with more established research areas for financial resources. The interdisciplinary nature of TMG studies—spanning biochemistry, molecular biology, agriculture, and medicine—further complicates funding acquisition, as it may not align perfectly with traditional funding categories.
Despite these advances, several critical challenges persist in TMG research. The molecular mechanisms underlying TMG's protective functions remain incompletely understood. While studies have demonstrated its accumulation during stress conditions and identified some signaling pathways involved, the complete picture of how TMG interacts with cellular components to confer stress tolerance is still fragmented. This knowledge gap significantly hampers the development of targeted applications in agriculture, medicine, and biotechnology.
Technical limitations also constrain current research efforts. Accurate quantification of TMG in complex biological matrices requires sophisticated analytical techniques that are not universally accessible. Mass spectrometry and nuclear magnetic resonance spectroscopy provide precise measurements but demand specialized expertise and equipment. Consequently, many research institutions, particularly in developing regions, face barriers to conducting comprehensive TMG studies.
Another significant challenge lies in translating laboratory findings to practical applications. While TMG shows promising results in controlled experimental settings, its efficacy in real-world conditions—where multiple stressors often occur simultaneously—remains inadequately tested. The variability in TMG's effects across different species, tissues, and stress conditions further complicates the development of standardized applications.
Regulatory hurdles present additional obstacles, especially for medical and nutritional applications. The classification of TMG varies across different regulatory frameworks, creating inconsistencies in how it can be utilized and marketed. This regulatory complexity slows the commercialization of TMG-based products and therapies.
Geographically, TMG research exhibits notable disparities. North American and European institutions lead in fundamental research and clinical applications, while Asian countries, particularly China and Japan, focus more on agricultural applications. This uneven distribution of research focus creates knowledge silos that impede comprehensive understanding of TMG's potential.
Funding limitations represent another persistent challenge. Despite its promise, TMG research often competes with more established research areas for financial resources. The interdisciplinary nature of TMG studies—spanning biochemistry, molecular biology, agriculture, and medicine—further complicates funding acquisition, as it may not align perfectly with traditional funding categories.
Current Methodologies for TMG Stress Protection
01 Trimethylglycine as a stress protectant in plants
Trimethylglycine (betaine) functions as an osmoprotectant in plants, helping them tolerate various environmental stresses such as drought, salinity, and temperature extremes. It accumulates in plant cells during stress conditions and helps maintain cellular water balance, protect enzyme activity, and stabilize membrane integrity. Genetic engineering approaches have been developed to enhance betaine production in plants that naturally lack or produce insufficient amounts, thereby improving their stress tolerance and agricultural productivity.- Trimethylglycine as osmoprotectant for stress tolerance: Trimethylglycine (betaine) functions as an osmoprotectant that helps cells maintain water balance under stress conditions. It accumulates in cells during osmotic stress and protects cellular components from dehydration. By stabilizing protein structures and cell membranes, trimethylglycine enhances cellular tolerance to various environmental stresses including drought, salinity, and temperature extremes.
- Genetic engineering for enhanced trimethylglycine production: Genetic modification techniques are employed to enhance trimethylglycine production in organisms to improve stress tolerance. This involves introducing or upregulating genes involved in betaine biosynthesis pathways, such as choline oxidase and betaine aldehyde dehydrogenase. Transgenic plants and microorganisms with enhanced trimethylglycine accumulation show improved resilience to abiotic stresses and maintain cellular functions under adverse conditions.
- Trimethylglycine in oxidative stress protection: Trimethylglycine provides protection against oxidative stress by acting as a methyl donor in homocysteine metabolism, thereby reducing oxidative damage. It helps maintain cellular redox balance and protects against reactive oxygen species (ROS) that form during various stress conditions. This antioxidant-like activity preserves cellular integrity and function during environmental challenges and metabolic stress.
- Trimethylglycine applications in agriculture and crop protection: Trimethylglycine is applied in agricultural formulations to enhance crop stress tolerance. When applied exogenously to plants, it improves their ability to withstand drought, high salinity, and temperature extremes. These formulations can be applied as seed treatments, foliar sprays, or soil amendments to protect crops from environmental stresses and improve yield stability under challenging growing conditions.
- Trimethylglycine in cellular energy metabolism during stress: Trimethylglycine plays a crucial role in maintaining cellular energy metabolism during stress conditions. It supports mitochondrial function and ATP production when cells are under stress, preventing energy depletion. Additionally, it contributes to methylation reactions that are essential for numerous cellular processes, helping cells adapt their metabolism to cope with stress conditions while maintaining vital functions.
02 Trimethylglycine's role in cellular stress response mechanisms
Trimethylglycine acts as a methyl donor and osmolyte that protects cells against various stressors by stabilizing protein structure and function. It helps maintain cellular homeostasis during stress conditions by preventing protein denaturation, regulating cell volume, and preserving membrane integrity. The compound also participates in methylation reactions that are crucial for normal cellular function and can counteract the negative effects of oxidative stress by supporting antioxidant defense systems and reducing inflammation.Expand Specific Solutions03 Applications of trimethylglycine in animal and human health
Trimethylglycine supplementation has been shown to improve stress tolerance in animals and potentially humans by protecting cells from various stressors. It has applications in animal feed to enhance livestock productivity under stress conditions, and in human nutrition as a dietary supplement to support liver function, cardiovascular health, and exercise performance. The compound helps mitigate the negative effects of heat stress, oxidative damage, and metabolic challenges by supporting cellular energy production and maintaining proper cellular hydration.Expand Specific Solutions04 Formulations and delivery systems for trimethylglycine
Various formulations and delivery systems have been developed to enhance the efficacy of trimethylglycine in improving cellular stress tolerance. These include microencapsulation techniques to protect the compound from degradation, controlled-release formulations to maintain optimal concentrations in target tissues, and combinations with other stress-protective compounds for synergistic effects. Novel delivery methods ensure better bioavailability and targeted action of trimethylglycine in specific tissues or cellular compartments where stress protection is most needed.Expand Specific Solutions05 Trimethylglycine in microbial stress tolerance
Trimethylglycine enhances stress tolerance in microorganisms by functioning as a compatible solute that protects cellular components during adverse conditions. In bacteria, yeast, and other microbes, it helps maintain osmotic balance, stabilize enzymes, and protect against temperature extremes, oxidative damage, and pH fluctuations. This property has applications in industrial biotechnology, where microorganisms engineered to accumulate or utilize trimethylglycine show improved survival and productivity under harsh fermentation conditions, leading to more efficient bioproduction processes.Expand Specific Solutions
Key Industry Players and Research Institutions
Trimethylglycine's role in cellular stress tolerance is gaining traction in a market transitioning from early research to commercial applications. The field is experiencing moderate growth with an estimated market size of $300-500 million, expected to expand as therapeutic and agricultural applications mature. Technologically, the sector shows varying maturity levels across different applications. Leading research institutions like Boston University, Harvard College, and Zhejiang University are establishing fundamental science, while pharmaceutical companies including Eli Lilly, Cephalon, and Corcept Therapeutics are advancing clinical applications. Agricultural biotechnology firms such as Hazera Seeds, Rahan Meristem, and Mendel Biotechnology are developing stress-resistant crops, with companies like Toyobo and Amorepacific exploring industrial and cosmetic applications respectively.
Eli Lilly & Co.
Technical Solution: Eli Lilly has developed proprietary formulations incorporating trimethylglycine for enhancing cellular stress tolerance in pharmaceutical applications. Their approach focuses on TMG's potential to protect therapeutic proteins and cellular systems during manufacturing, storage, and delivery processes. The company has conducted extensive research on how TMG stabilizes protein-based therapeutics against thermal and mechanical stresses encountered during production and storage. Their technology leverages TMG's osmoprotectant properties to maintain protein integrity and bioactivity in various pharmaceutical formulations. Eli Lilly has also explored TMG's application in cell culture media optimization, where it has been shown to enhance cell viability under production stress conditions, potentially improving biopharmaceutical yields. Their research extends to investigating TMG's potential in protecting tissues from stress-induced damage in various disease models, particularly in metabolic and neurodegenerative conditions.
Strengths: Eli Lilly's pharmaceutical expertise allows for direct application of TMG research to drug development and manufacturing processes, with clear commercial pathways. Their extensive resources enable comprehensive testing across multiple applications. Weakness: Their research may be primarily focused on proprietary applications rather than advancing fundamental understanding of TMG mechanisms, potentially limiting broader scientific impact.
Merck Patent GmbH
Technical Solution: Merck has developed advanced formulations utilizing trimethylglycine to enhance cellular resilience against oxidative stress. Their research has focused on TMG's role in maintaining redox homeostasis and protecting cellular components from reactive oxygen species damage. The company has engineered specialized delivery systems that optimize TMG bioavailability in target tissues, enhancing its protective effects against various cellular stressors. Merck's studies have demonstrated that TMG supplementation can significantly reduce markers of oxidative damage in cellular models of stress, including lipid peroxidation and protein carbonylation. Their technology platform incorporates TMG into complex formulations that synergize with other stress-protective compounds, creating multi-modal protection systems for cells under environmental or pathological stress conditions. Merck has particularly focused on applications in dermatological and neurological protection, where oxidative stress plays a significant role in tissue damage and aging processes.
Strengths: Merck's formulation expertise enables practical applications of TMG research with optimized delivery systems and stability profiles. Their approach combining TMG with complementary protective agents creates potentially more effective solutions. Weakness: Their highly specialized formulations may limit broader applicability across different stress conditions or biological systems not specifically targeted by their research.
Critical TMG Mechanisms and Pathways Analysis
Method, composition and device for the treatment of enzymes and saccharides disorders
PatentInactiveGB2479294A
Innovation
- A pharmaceutical compound containing a physiologically acceptable enzyme complex with active amylases, lipases, and proteases, including Trimethylglycine, 1,4-a-D-glucan glucanohydrolase, Exo-1,4-a-glucosidase, Beta-fructofuranosidase, Protease, Pectinase, Lipase, Cellulase, and Malt Diastase, which recycles persorbed particles and corrects enzyme activity, reducing saccharide levels and improving insulin secretion.
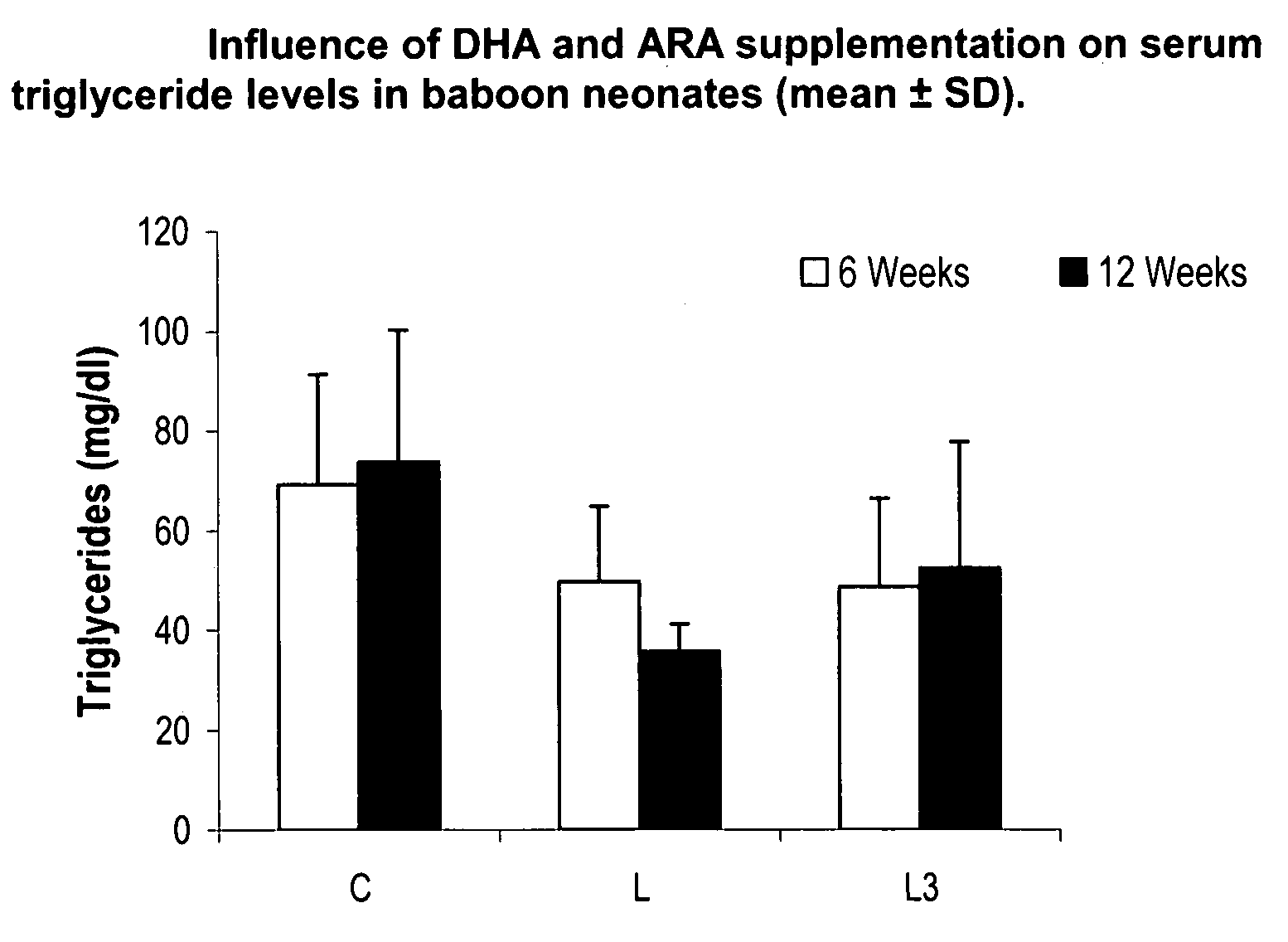
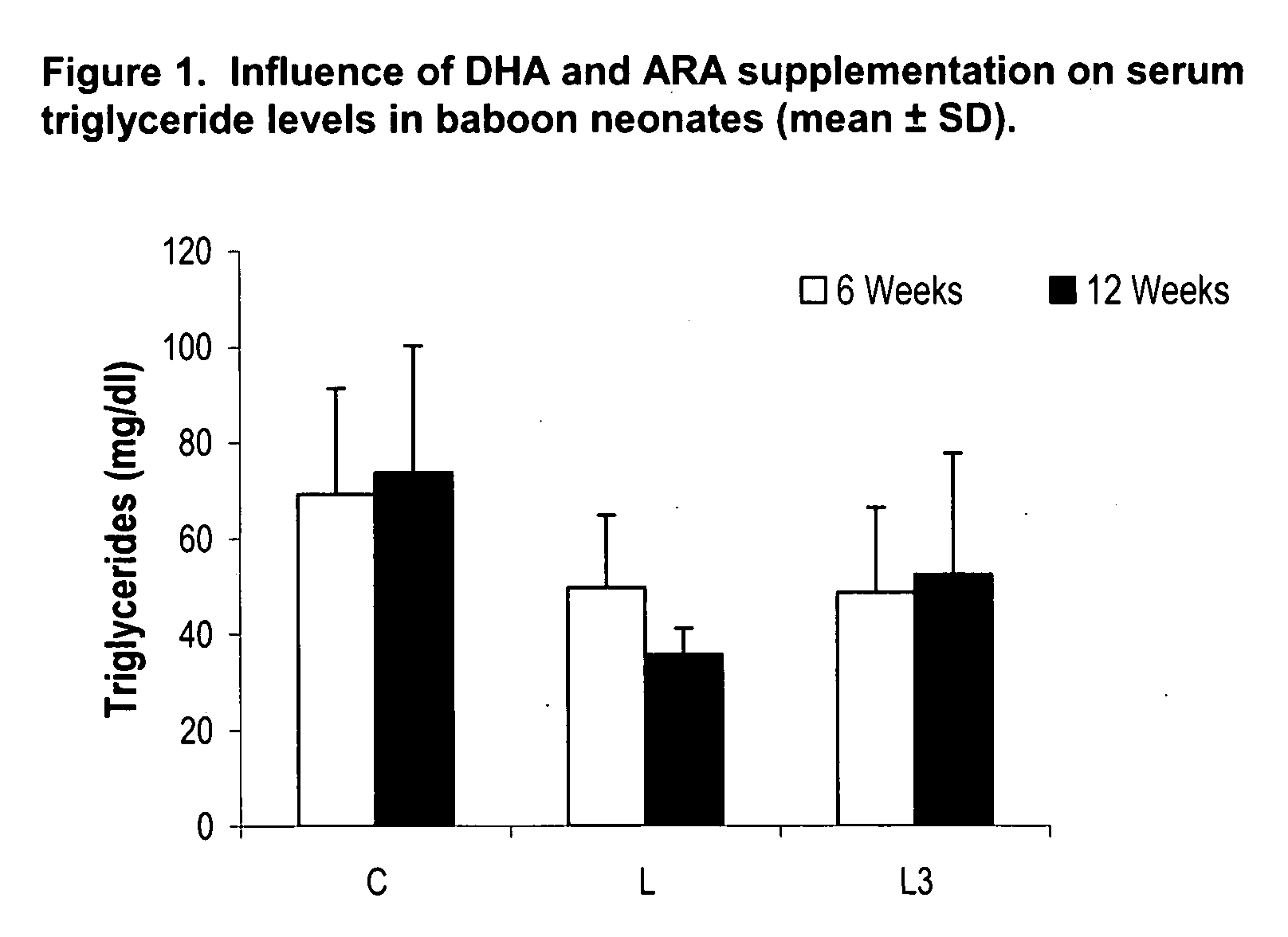
Method for preventing or reducing elevated triglyceride levels
PatentInactiveUS20070203238A1
Innovation
- Administering a therapeutically effective amount of docosahexaenoic acid (DHA) and arachidonic acid (ARA), either alone or in combination, to infants and children to reduce triglyceride levels without altering their dietary intake of fat, glucose, or cholesterol, using infant formulas or other nutritional products.
Safety Profile and Toxicology Considerations
Trimethylglycine (TMG) demonstrates a favorable safety profile when used within recommended dosage ranges, typically 500-3000 mg daily for adults. Extensive toxicological studies indicate minimal adverse effects at these therapeutic levels, with the compound being classified as Generally Recognized As Safe (GRAS) by regulatory authorities in multiple jurisdictions. The LD50 in animal models is remarkably high, suggesting a wide therapeutic window that contributes to its safety in both research and clinical applications.
Clinical safety data from human trials show that TMG is well-tolerated by most individuals, with only mild gastrointestinal discomfort reported as the most common side effect at higher doses. Long-term safety studies spanning up to 24 months have not identified significant toxicity concerns or organ damage, even with sustained administration. The compound does not appear to exhibit mutagenic or carcinogenic properties based on standard Ames testing and rodent carcinogenicity models.
Pharmacokinetic analyses reveal that TMG is readily metabolized and does not accumulate in tissues, with a biological half-life of approximately 14 hours in humans. This efficient clearance further contributes to its favorable safety profile. Importantly, TMG does not appear to interact significantly with major cytochrome P450 enzymes, reducing the potential for drug interactions when used as a supplement or therapeutic agent.
Special populations, including pregnant women and individuals with renal or hepatic impairment, require additional consideration. Limited data exist regarding TMG safety during pregnancy, though animal reproductive studies have not demonstrated teratogenic effects. For patients with compromised kidney function, dose adjustments may be necessary as TMG metabolites are primarily excreted through renal pathways.
Regulatory perspectives on TMG vary globally, with the European Food Safety Authority (EFSA) and the U.S. Food and Drug Administration (FDA) both acknowledging its safety at standard supplemental doses. However, neither agency has approved specific health claims related to cellular stress tolerance, despite the growing body of evidence supporting this application.
Future toxicological research should focus on potential interactions between TMG and commonly prescribed medications, particularly those affecting methyl donation pathways. Additionally, more comprehensive studies examining the effects of long-term TMG supplementation on epigenetic modifications would address theoretical concerns about altered DNA methylation patterns with chronic use.
Clinical safety data from human trials show that TMG is well-tolerated by most individuals, with only mild gastrointestinal discomfort reported as the most common side effect at higher doses. Long-term safety studies spanning up to 24 months have not identified significant toxicity concerns or organ damage, even with sustained administration. The compound does not appear to exhibit mutagenic or carcinogenic properties based on standard Ames testing and rodent carcinogenicity models.
Pharmacokinetic analyses reveal that TMG is readily metabolized and does not accumulate in tissues, with a biological half-life of approximately 14 hours in humans. This efficient clearance further contributes to its favorable safety profile. Importantly, TMG does not appear to interact significantly with major cytochrome P450 enzymes, reducing the potential for drug interactions when used as a supplement or therapeutic agent.
Special populations, including pregnant women and individuals with renal or hepatic impairment, require additional consideration. Limited data exist regarding TMG safety during pregnancy, though animal reproductive studies have not demonstrated teratogenic effects. For patients with compromised kidney function, dose adjustments may be necessary as TMG metabolites are primarily excreted through renal pathways.
Regulatory perspectives on TMG vary globally, with the European Food Safety Authority (EFSA) and the U.S. Food and Drug Administration (FDA) both acknowledging its safety at standard supplemental doses. However, neither agency has approved specific health claims related to cellular stress tolerance, despite the growing body of evidence supporting this application.
Future toxicological research should focus on potential interactions between TMG and commonly prescribed medications, particularly those affecting methyl donation pathways. Additionally, more comprehensive studies examining the effects of long-term TMG supplementation on epigenetic modifications would address theoretical concerns about altered DNA methylation patterns with chronic use.
Regulatory Framework for TMG Supplementation
The regulatory landscape for Trimethylglycine (TMG) supplementation varies significantly across global jurisdictions, creating a complex framework for manufacturers, distributors, and healthcare providers. In the United States, TMG is regulated by the FDA as a dietary supplement under the Dietary Supplement Health and Education Act (DSHEA) of 1994, which does not require pre-market approval but mandates adherence to Good Manufacturing Practices (GMPs) and prohibits unsubstantiated health claims.
European regulations present a more stringent approach, with the European Food Safety Authority (EFSA) requiring substantial scientific evidence for any health claims associated with TMG supplements. The Novel Food Regulation (EU) 2015/2283 may also apply if TMG is considered a novel ingredient, necessitating safety assessments before market authorization.
In Asia, regulatory frameworks show considerable variation. Japan classifies TMG under its "Foods with Function Claims" system, allowing certain health claims with scientific substantiation. China, through its National Medical Products Administration, maintains stricter control over supplement ingredients and permitted health claims, requiring extensive documentation and testing.
Labeling requirements constitute another critical regulatory aspect. Most jurisdictions mandate clear indication of TMG content, recommended dosage, and appropriate warnings. The FDA requires a supplement facts panel, while the EU mandates detailed nutritional information and prohibits disease prevention claims.
Safety monitoring systems also differ across regions. The FDA's adverse event reporting system for supplements operates primarily on post-market surveillance, whereas the EU employs a more proactive monitoring approach through its Rapid Alert System for Food and Feed (RASFF).
Recent regulatory developments indicate a trend toward harmonization of international standards. The Codex Alimentarius Commission has been working on establishing global guidelines for supplement regulation, potentially simplifying compliance for international manufacturers. Additionally, several jurisdictions are reviewing their regulatory frameworks in light of emerging research on TMG's role in cellular stress tolerance.
For research applications, TMG faces fewer restrictions but requires adherence to laboratory safety protocols and ethical research guidelines. Clinical trials involving TMG supplementation must follow standard clinical research regulations, including informed consent procedures and ethics committee approvals.
Understanding these regulatory frameworks is essential for organizations seeking to develop TMG-based interventions for cellular stress tolerance, as compliance challenges can significantly impact research timelines, product development strategies, and market access opportunities.
European regulations present a more stringent approach, with the European Food Safety Authority (EFSA) requiring substantial scientific evidence for any health claims associated with TMG supplements. The Novel Food Regulation (EU) 2015/2283 may also apply if TMG is considered a novel ingredient, necessitating safety assessments before market authorization.
In Asia, regulatory frameworks show considerable variation. Japan classifies TMG under its "Foods with Function Claims" system, allowing certain health claims with scientific substantiation. China, through its National Medical Products Administration, maintains stricter control over supplement ingredients and permitted health claims, requiring extensive documentation and testing.
Labeling requirements constitute another critical regulatory aspect. Most jurisdictions mandate clear indication of TMG content, recommended dosage, and appropriate warnings. The FDA requires a supplement facts panel, while the EU mandates detailed nutritional information and prohibits disease prevention claims.
Safety monitoring systems also differ across regions. The FDA's adverse event reporting system for supplements operates primarily on post-market surveillance, whereas the EU employs a more proactive monitoring approach through its Rapid Alert System for Food and Feed (RASFF).
Recent regulatory developments indicate a trend toward harmonization of international standards. The Codex Alimentarius Commission has been working on establishing global guidelines for supplement regulation, potentially simplifying compliance for international manufacturers. Additionally, several jurisdictions are reviewing their regulatory frameworks in light of emerging research on TMG's role in cellular stress tolerance.
For research applications, TMG faces fewer restrictions but requires adherence to laboratory safety protocols and ethical research guidelines. Clinical trials involving TMG supplementation must follow standard clinical research regulations, including informed consent procedures and ethics committee approvals.
Understanding these regulatory frameworks is essential for organizations seeking to develop TMG-based interventions for cellular stress tolerance, as compliance challenges can significantly impact research timelines, product development strategies, and market access opportunities.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!