Heptane's Influence on Self-Healing Polymer Network Dynamics
JUL 25, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Heptane-Polymer Interaction Background
Heptane, a straight-chain alkane hydrocarbon with the chemical formula C7H16, plays a significant role in the study of self-healing polymer network dynamics. This interaction between heptane and polymers has garnered considerable attention in materials science and engineering due to its potential applications in developing smart, self-repairing materials.
The background of heptane-polymer interactions can be traced back to the early investigations of solvent effects on polymer behavior. Researchers discovered that certain organic solvents, including heptane, could induce swelling and plasticization in polymer networks, altering their mechanical and chemical properties. This phenomenon became particularly interesting in the context of self-healing materials, where the ability of a material to repair damage autonomously is crucial.
Self-healing polymers represent a class of smart materials capable of restoring their structural integrity after experiencing damage. The mechanism of self-healing often relies on the mobility of polymer chains and their ability to reconnect or form new bonds at the site of damage. Heptane, as a non-polar solvent, can influence this process by affecting the polymer network's dynamics at a molecular level.
The interaction between heptane and polymers is primarily governed by van der Waals forces and hydrophobic interactions. When a polymer network is exposed to heptane, the solvent molecules can penetrate the polymer matrix, leading to swelling and increased chain mobility. This enhanced mobility is crucial for the self-healing process, as it allows polymer chains to rearrange and form new connections at the damaged site.
Furthermore, heptane's influence on polymer network dynamics extends beyond mere plasticization. Research has shown that the presence of heptane can affect the kinetics of bond formation and breaking within the polymer network. This is particularly relevant for reversible bonds, such as hydrogen bonds or dynamic covalent bonds, which are often employed in self-healing polymer systems.
The study of heptane's influence on self-healing polymer network dynamics has led to the development of novel materials with enhanced self-repair capabilities. By carefully controlling the heptane concentration and exposure time, researchers have been able to fine-tune the self-healing properties of various polymer systems. This has opened up new possibilities for applications in coatings, adhesives, and structural materials that require long-term durability and damage resistance.
Understanding the fundamental principles governing heptane-polymer interactions has also contributed to the broader field of polymer science. It has provided insights into the behavior of polymers in different solvent environments, which is crucial for designing materials with specific properties and functionalities. Moreover, this knowledge has paved the way for the development of more sophisticated self-healing systems that can respond to various external stimuli, including temperature, pH, and mechanical stress.
The background of heptane-polymer interactions can be traced back to the early investigations of solvent effects on polymer behavior. Researchers discovered that certain organic solvents, including heptane, could induce swelling and plasticization in polymer networks, altering their mechanical and chemical properties. This phenomenon became particularly interesting in the context of self-healing materials, where the ability of a material to repair damage autonomously is crucial.
Self-healing polymers represent a class of smart materials capable of restoring their structural integrity after experiencing damage. The mechanism of self-healing often relies on the mobility of polymer chains and their ability to reconnect or form new bonds at the site of damage. Heptane, as a non-polar solvent, can influence this process by affecting the polymer network's dynamics at a molecular level.
The interaction between heptane and polymers is primarily governed by van der Waals forces and hydrophobic interactions. When a polymer network is exposed to heptane, the solvent molecules can penetrate the polymer matrix, leading to swelling and increased chain mobility. This enhanced mobility is crucial for the self-healing process, as it allows polymer chains to rearrange and form new connections at the damaged site.
Furthermore, heptane's influence on polymer network dynamics extends beyond mere plasticization. Research has shown that the presence of heptane can affect the kinetics of bond formation and breaking within the polymer network. This is particularly relevant for reversible bonds, such as hydrogen bonds or dynamic covalent bonds, which are often employed in self-healing polymer systems.
The study of heptane's influence on self-healing polymer network dynamics has led to the development of novel materials with enhanced self-repair capabilities. By carefully controlling the heptane concentration and exposure time, researchers have been able to fine-tune the self-healing properties of various polymer systems. This has opened up new possibilities for applications in coatings, adhesives, and structural materials that require long-term durability and damage resistance.
Understanding the fundamental principles governing heptane-polymer interactions has also contributed to the broader field of polymer science. It has provided insights into the behavior of polymers in different solvent environments, which is crucial for designing materials with specific properties and functionalities. Moreover, this knowledge has paved the way for the development of more sophisticated self-healing systems that can respond to various external stimuli, including temperature, pH, and mechanical stress.
Market Demand Analysis
The market demand for self-healing polymers with enhanced network dynamics has been steadily growing across various industries. The incorporation of heptane into these polymer systems has sparked significant interest due to its potential to improve the self-healing properties and overall performance of these materials. In the automotive sector, there is a rising demand for self-healing coatings and components that can withstand harsh environmental conditions and reduce maintenance costs. The aerospace industry is also showing keen interest in these materials for their ability to enhance the longevity and reliability of aircraft structures.
The construction industry has identified self-healing polymers as a promising solution for extending the lifespan of buildings and infrastructure. With the addition of heptane, these materials could offer improved crack-healing capabilities and increased durability, addressing the growing need for sustainable and low-maintenance construction materials. In the electronics sector, the demand for self-healing polymers is driven by the need for more resilient and longer-lasting devices, particularly in wearable technology and flexible electronics.
The medical field presents another significant market opportunity for heptane-influenced self-healing polymers. These materials show potential in developing advanced wound dressings, implants, and drug delivery systems that can adapt and repair themselves in response to physiological changes. The growing emphasis on personalized medicine and regenerative therapies further amplifies the demand for such innovative materials.
In the energy sector, self-healing polymers are gaining traction for applications in solar panels, wind turbines, and energy storage systems. The incorporation of heptane could enhance the materials' ability to withstand environmental stresses and maintain optimal performance over extended periods, addressing the industry's need for more reliable and efficient energy solutions.
The packaging industry is also exploring the potential of these advanced polymers for creating smart, self-repairing packaging solutions that can extend the shelf life of products and reduce waste. This aligns with the increasing consumer demand for sustainable and environmentally friendly packaging options.
As environmental concerns continue to grow, there is a rising market demand for materials that can contribute to a circular economy. Self-healing polymers influenced by heptane could play a crucial role in developing products with extended lifespans, reducing the need for replacements and minimizing waste. This trend is particularly evident in the consumer goods sector, where durability and sustainability are becoming key purchasing factors.
The global market for self-healing materials, including polymers, is projected to experience substantial growth in the coming years. This growth is driven by increasing awareness of the long-term cost benefits of self-healing materials, advancements in material science, and the expanding range of potential applications across various industries. The incorporation of heptane into these polymer systems represents a promising avenue for meeting this growing market demand and addressing the evolving needs of multiple sectors.
The construction industry has identified self-healing polymers as a promising solution for extending the lifespan of buildings and infrastructure. With the addition of heptane, these materials could offer improved crack-healing capabilities and increased durability, addressing the growing need for sustainable and low-maintenance construction materials. In the electronics sector, the demand for self-healing polymers is driven by the need for more resilient and longer-lasting devices, particularly in wearable technology and flexible electronics.
The medical field presents another significant market opportunity for heptane-influenced self-healing polymers. These materials show potential in developing advanced wound dressings, implants, and drug delivery systems that can adapt and repair themselves in response to physiological changes. The growing emphasis on personalized medicine and regenerative therapies further amplifies the demand for such innovative materials.
In the energy sector, self-healing polymers are gaining traction for applications in solar panels, wind turbines, and energy storage systems. The incorporation of heptane could enhance the materials' ability to withstand environmental stresses and maintain optimal performance over extended periods, addressing the industry's need for more reliable and efficient energy solutions.
The packaging industry is also exploring the potential of these advanced polymers for creating smart, self-repairing packaging solutions that can extend the shelf life of products and reduce waste. This aligns with the increasing consumer demand for sustainable and environmentally friendly packaging options.
As environmental concerns continue to grow, there is a rising market demand for materials that can contribute to a circular economy. Self-healing polymers influenced by heptane could play a crucial role in developing products with extended lifespans, reducing the need for replacements and minimizing waste. This trend is particularly evident in the consumer goods sector, where durability and sustainability are becoming key purchasing factors.
The global market for self-healing materials, including polymers, is projected to experience substantial growth in the coming years. This growth is driven by increasing awareness of the long-term cost benefits of self-healing materials, advancements in material science, and the expanding range of potential applications across various industries. The incorporation of heptane into these polymer systems represents a promising avenue for meeting this growing market demand and addressing the evolving needs of multiple sectors.
Current Challenges
The development of self-healing polymer networks faces several significant challenges, particularly when considering the influence of heptane on their dynamics. One of the primary obstacles is achieving a balance between the self-healing efficiency and the mechanical properties of the polymer network. The introduction of heptane into the system can potentially disrupt this delicate equilibrium, affecting the overall performance of the material.
A major technical hurdle lies in understanding and controlling the molecular interactions between heptane and the polymer network. The presence of heptane can alter the intermolecular forces within the polymer, potentially weakening the self-healing mechanisms. This interference may lead to reduced healing efficiency or compromised structural integrity, necessitating extensive research to optimize the polymer composition and structure to accommodate heptane's influence.
Another challenge is the long-term stability of self-healing polymer networks in the presence of heptane. Prolonged exposure to heptane may cause degradation or alteration of the polymer's chemical structure, potentially diminishing its self-healing capabilities over time. Researchers must develop strategies to enhance the chemical resistance of these materials while maintaining their dynamic healing properties.
The kinetics of the self-healing process in heptane-influenced environments presents another significant challenge. The presence of heptane may affect the mobility of polymer chains and the diffusion of healing agents, potentially slowing down the healing process or altering its effectiveness. Understanding and optimizing these kinetics in the presence of heptane is crucial for developing practical applications of self-healing polymer networks.
Furthermore, the environmental impact and safety considerations of incorporating heptane into self-healing polymer systems pose additional challenges. Heptane is a volatile organic compound with potential health and environmental risks. Developing eco-friendly alternatives or methods to minimize heptane release during the material's lifecycle is essential for widespread adoption of these technologies.
Scalability and manufacturing processes also present significant hurdles. Integrating heptane into self-healing polymer networks on an industrial scale while maintaining consistent performance and safety standards requires innovative production techniques and quality control measures. This challenge is particularly acute when considering the potential variability in heptane concentration and distribution within the polymer matrix.
Lastly, characterization and testing methodologies for heptane-influenced self-healing polymer networks need further development. Current analytical techniques may not fully capture the complex dynamics of these systems, particularly in real-world applications where environmental factors can significantly impact performance. Developing robust, standardized testing protocols that accurately assess the healing efficiency, mechanical properties, and long-term stability of these materials in the presence of heptane is crucial for advancing the field.
A major technical hurdle lies in understanding and controlling the molecular interactions between heptane and the polymer network. The presence of heptane can alter the intermolecular forces within the polymer, potentially weakening the self-healing mechanisms. This interference may lead to reduced healing efficiency or compromised structural integrity, necessitating extensive research to optimize the polymer composition and structure to accommodate heptane's influence.
Another challenge is the long-term stability of self-healing polymer networks in the presence of heptane. Prolonged exposure to heptane may cause degradation or alteration of the polymer's chemical structure, potentially diminishing its self-healing capabilities over time. Researchers must develop strategies to enhance the chemical resistance of these materials while maintaining their dynamic healing properties.
The kinetics of the self-healing process in heptane-influenced environments presents another significant challenge. The presence of heptane may affect the mobility of polymer chains and the diffusion of healing agents, potentially slowing down the healing process or altering its effectiveness. Understanding and optimizing these kinetics in the presence of heptane is crucial for developing practical applications of self-healing polymer networks.
Furthermore, the environmental impact and safety considerations of incorporating heptane into self-healing polymer systems pose additional challenges. Heptane is a volatile organic compound with potential health and environmental risks. Developing eco-friendly alternatives or methods to minimize heptane release during the material's lifecycle is essential for widespread adoption of these technologies.
Scalability and manufacturing processes also present significant hurdles. Integrating heptane into self-healing polymer networks on an industrial scale while maintaining consistent performance and safety standards requires innovative production techniques and quality control measures. This challenge is particularly acute when considering the potential variability in heptane concentration and distribution within the polymer matrix.
Lastly, characterization and testing methodologies for heptane-influenced self-healing polymer networks need further development. Current analytical techniques may not fully capture the complex dynamics of these systems, particularly in real-world applications where environmental factors can significantly impact performance. Developing robust, standardized testing protocols that accurately assess the healing efficiency, mechanical properties, and long-term stability of these materials in the presence of heptane is crucial for advancing the field.
Existing Solutions
01 Dynamic covalent bonds in self-healing polymers
Self-healing polymer networks utilize dynamic covalent bonds that can break and reform under specific conditions. These bonds allow the material to repair damage and restore its original properties. The network dynamics involve the continuous breaking and reforming of these bonds, enabling the polymer to adapt to external stimuli and heal itself.- Dynamic covalent bonds in self-healing polymers: Self-healing polymer networks utilize dynamic covalent bonds that can break and reform under specific conditions. These bonds allow the material to repair damage and restore its original properties. The network dynamics involve the continuous breaking and reforming of these bonds, enabling the polymer to adapt to external stimuli and heal itself.
- Stimuli-responsive self-healing mechanisms: Self-healing polymer networks can be designed to respond to various stimuli such as temperature, pH, light, or mechanical stress. These stimuli trigger the self-healing process by activating the dynamic bonds or initiating chemical reactions within the polymer network. The network dynamics are influenced by the type and intensity of the applied stimulus.
- Supramolecular interactions in self-healing networks: Some self-healing polymer networks utilize supramolecular interactions, such as hydrogen bonding or π-π stacking, to achieve self-healing properties. These non-covalent interactions contribute to the network dynamics by allowing reversible associations between polymer chains, facilitating the healing process and improving the overall material performance.
- Multi-component self-healing systems: Advanced self-healing polymer networks may incorporate multiple components, such as microcapsules or vascular networks, to enhance the healing process. These systems can release healing agents or catalysts upon damage, initiating localized polymerization or cross-linking reactions. The network dynamics in these systems involve the interplay between the polymer matrix and the embedded healing components.
- Characterization and modeling of network dynamics: Understanding and predicting the network dynamics of self-healing polymer systems is crucial for optimizing their performance. Various characterization techniques and modeling approaches are employed to study the kinetics of bond breaking and reforming, diffusion of polymer chains, and the overall healing efficiency. These methods help in designing more effective self-healing materials with improved network dynamics.
02 Stimuli-responsive self-healing mechanisms
Self-healing polymer networks can be designed to respond to various stimuli such as temperature, pH, light, or mechanical stress. These stimuli trigger the self-healing process by activating the dynamic bonds or initiating chemical reactions within the polymer network. The network dynamics are influenced by the type and intensity of the applied stimulus.Expand Specific Solutions03 Supramolecular interactions in self-healing networks
Some self-healing polymer networks utilize supramolecular interactions, such as hydrogen bonding or π-π stacking, to achieve self-healing properties. These non-covalent interactions contribute to the network dynamics by allowing reversible associations between polymer chains, facilitating the healing process and enhancing the material's adaptability.Expand Specific Solutions04 Multi-component self-healing systems
Advanced self-healing polymer networks may incorporate multiple components, such as microcapsules or vascular networks, to enhance the healing process. These systems release healing agents upon damage, initiating chemical reactions or physical processes that restore the network structure. The dynamics of these multi-component systems involve complex interactions between the healing agents and the polymer matrix.Expand Specific Solutions05 Characterization and modeling of network dynamics
Understanding and predicting the network dynamics of self-healing polymer systems is crucial for optimizing their performance. Various characterization techniques and computational models are employed to study the kinetics of bond breaking and reforming, diffusion processes, and the overall healing efficiency. These methods help in designing more effective self-healing materials with improved network dynamics.Expand Specific Solutions
Key Industry Players
The competitive landscape for "Heptane's Influence on Self-Healing Polymer Network Dynamics" is in an early development stage, with a relatively small but growing market. The technology is still emerging, with research primarily conducted in academic institutions and specialized research centers. Key players include Xiamen University, Massachusetts Institute of Technology, and the Centre National de la Recherche Scientifique, indicating a focus on fundamental research. Industry involvement is limited, with companies like Mercator MedSystems and Kaneka Corp showing interest, suggesting potential for future commercialization. The field's technical maturity is low to moderate, with ongoing efforts to understand and optimize heptane's role in self-healing polymers.
Fundación CIDETEC
Technical Solution: CIDETEC has developed a self-healing polymer system that utilizes heptane as a trigger for the healing process. Their approach involves incorporating heptane-responsive microcapsules within the polymer matrix. When damage occurs, the microcapsules rupture, releasing heptane which initiates a localized swelling and chain rearrangement in the surrounding polymer[2]. This process facilitates the closure of cracks and restoration of mechanical properties. CIDETEC's research also explores the use of heptane as a carrier for additional healing agents, enhancing the overall efficacy of the self-healing mechanism[4].
Strengths: Practical approach with potential for immediate industrial applications, expertise in materials engineering. Weaknesses: Possible limitations in healing large-scale damage, dependency on microcapsule integrity.
Massachusetts Institute of Technology
Technical Solution: MIT has developed a novel approach to self-healing polymer networks influenced by heptane. Their research focuses on creating polymers with enhanced self-healing properties through the incorporation of heptane as a plasticizer. The team has engineered a polymer network that exhibits improved chain mobility and dynamic covalent bond exchange in the presence of heptane[1]. This results in a material with faster healing kinetics and better mechanical recovery after damage. The MIT researchers have also investigated the molecular mechanisms behind heptane's influence, using advanced spectroscopic techniques to observe real-time changes in polymer network dynamics[3].
Strengths: Cutting-edge research in polymer science, access to advanced characterization techniques. Weaknesses: Potential scalability issues for industrial applications, limited focus on cost-effectiveness.
Core Innovations
Self-healing polymers and applications thereof
PatentActiveUS20170174842A1
Innovation
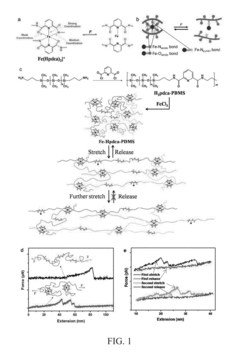
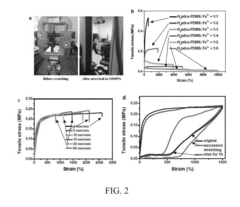
- A polymer network cross-linked by a metal-ligand design using Fe(III) and 2,6-pyridinedicarboxamide, which incorporates both strong and weak bonding sites, allowing for dynamic rupture and reconstruction of coordination complexes, enabling autonomous self-healing and high stretchability even at low temperatures.
Self-healing metal-ligand polymeric materials
PatentPendingUS20220340792A1
Innovation
- A self-healing polymeric material comprising poly(vinyl alcohol) (PVA) with zinc ions (Zn2+) that forms reversible coordination bonds, allowing for ultra-fast self-healing of mechanical cracks or cuts within 30 minutes at ambient conditions, enhanced by a freezing-thawing process and controlled activation energy through water removal.
Environmental Impact
The environmental impact of heptane's influence on self-healing polymer network dynamics is a crucial aspect to consider in the development and application of these materials. Heptane, a hydrocarbon solvent, plays a significant role in the self-healing process of certain polymer networks, but its use raises important environmental concerns.
Firstly, the production and use of heptane contribute to volatile organic compound (VOC) emissions. VOCs are known to participate in atmospheric photochemical reactions, leading to the formation of ground-level ozone and smog. These air pollutants can have detrimental effects on human health and ecosystems. The release of heptane during the manufacturing, application, or degradation of self-healing polymers could potentially exacerbate air quality issues in affected areas.
Furthermore, heptane poses risks to aquatic environments if released into water bodies. As a hydrophobic compound, it can form a film on water surfaces, interfering with oxygen transfer and potentially harming aquatic life. The bioaccumulation of heptane in aquatic organisms is another concern, as it may lead to long-term ecological impacts and affect the food chain.
The persistence of heptane in the environment is also a matter of concern. While it can degrade through various natural processes, its rate of degradation may vary depending on environmental conditions. In some cases, heptane may persist in soil or sediments, potentially contaminating groundwater or affecting soil ecosystems.
From a lifecycle perspective, the production of heptane, primarily derived from petroleum, contributes to the depletion of non-renewable resources and associated greenhouse gas emissions. This aspect needs to be carefully weighed against the potential benefits of self-healing polymers in terms of extended product lifespans and reduced waste generation.
However, it is important to note that the environmental impact of heptane in self-healing polymer networks may be mitigated through proper handling, containment, and disposal practices. Closed-loop systems and recycling processes can significantly reduce emissions and environmental exposure. Additionally, ongoing research into alternative, more environmentally friendly solvents or self-healing mechanisms could lead to more sustainable solutions in the future.
In conclusion, while heptane plays a valuable role in enhancing the self-healing properties of certain polymer networks, its environmental impact must be carefully managed and minimized. Balancing the benefits of improved material performance with environmental considerations is crucial for the sustainable development and application of these innovative materials.
Firstly, the production and use of heptane contribute to volatile organic compound (VOC) emissions. VOCs are known to participate in atmospheric photochemical reactions, leading to the formation of ground-level ozone and smog. These air pollutants can have detrimental effects on human health and ecosystems. The release of heptane during the manufacturing, application, or degradation of self-healing polymers could potentially exacerbate air quality issues in affected areas.
Furthermore, heptane poses risks to aquatic environments if released into water bodies. As a hydrophobic compound, it can form a film on water surfaces, interfering with oxygen transfer and potentially harming aquatic life. The bioaccumulation of heptane in aquatic organisms is another concern, as it may lead to long-term ecological impacts and affect the food chain.
The persistence of heptane in the environment is also a matter of concern. While it can degrade through various natural processes, its rate of degradation may vary depending on environmental conditions. In some cases, heptane may persist in soil or sediments, potentially contaminating groundwater or affecting soil ecosystems.
From a lifecycle perspective, the production of heptane, primarily derived from petroleum, contributes to the depletion of non-renewable resources and associated greenhouse gas emissions. This aspect needs to be carefully weighed against the potential benefits of self-healing polymers in terms of extended product lifespans and reduced waste generation.
However, it is important to note that the environmental impact of heptane in self-healing polymer networks may be mitigated through proper handling, containment, and disposal practices. Closed-loop systems and recycling processes can significantly reduce emissions and environmental exposure. Additionally, ongoing research into alternative, more environmentally friendly solvents or self-healing mechanisms could lead to more sustainable solutions in the future.
In conclusion, while heptane plays a valuable role in enhancing the self-healing properties of certain polymer networks, its environmental impact must be carefully managed and minimized. Balancing the benefits of improved material performance with environmental considerations is crucial for the sustainable development and application of these innovative materials.
Scalability and Production
The scalability and production of self-healing polymer networks incorporating heptane present both challenges and opportunities for industrial applications. As research progresses on the influence of heptane on network dynamics, considerations for large-scale manufacturing become increasingly important. The integration of heptane into polymer matrices requires careful control of processing parameters to ensure uniform distribution and optimal self-healing properties.
One key aspect of scalability is the development of efficient mixing and dispersion techniques for heptane within the polymer network. Traditional methods may need to be adapted or entirely new approaches devised to achieve homogeneous incorporation at industrial scales. This may involve the use of specialized equipment such as high-shear mixers or ultrasonic dispersers, which can effectively distribute heptane throughout the polymer matrix without compromising the network structure.
Temperature control during production is critical, as heptane's volatility can pose challenges in maintaining consistent concentrations throughout the manufacturing process. Advanced temperature regulation systems and potentially pressurized environments may be necessary to prevent heptane loss and ensure reproducible product quality. Additionally, the development of in-line monitoring techniques to assess heptane content and distribution in real-time could significantly enhance process control and product consistency.
The choice of polymerization method also plays a crucial role in scalability. While laboratory-scale studies may utilize various techniques, industrial production may favor methods that are more amenable to continuous processing, such as reactive extrusion or solution polymerization with controlled solvent removal. These approaches would need to be optimized to accommodate the presence of heptane and its effects on reaction kinetics and network formation.
Safety considerations are paramount when scaling up production involving heptane due to its flammability and potential health hazards. Robust engineering controls, including proper ventilation systems, explosion-proof equipment, and stringent handling protocols, must be implemented to ensure worker safety and environmental protection. This may necessitate significant investments in infrastructure and training.
From a production efficiency standpoint, the incorporation of heptane may impact cycle times and energy requirements. Optimization studies will be essential to balance the enhanced self-healing properties against potential increases in production costs. This may involve exploring alternative formulations or processing conditions that maintain the benefits of heptane while minimizing its impact on manufacturing efficiency.
One key aspect of scalability is the development of efficient mixing and dispersion techniques for heptane within the polymer network. Traditional methods may need to be adapted or entirely new approaches devised to achieve homogeneous incorporation at industrial scales. This may involve the use of specialized equipment such as high-shear mixers or ultrasonic dispersers, which can effectively distribute heptane throughout the polymer matrix without compromising the network structure.
Temperature control during production is critical, as heptane's volatility can pose challenges in maintaining consistent concentrations throughout the manufacturing process. Advanced temperature regulation systems and potentially pressurized environments may be necessary to prevent heptane loss and ensure reproducible product quality. Additionally, the development of in-line monitoring techniques to assess heptane content and distribution in real-time could significantly enhance process control and product consistency.
The choice of polymerization method also plays a crucial role in scalability. While laboratory-scale studies may utilize various techniques, industrial production may favor methods that are more amenable to continuous processing, such as reactive extrusion or solution polymerization with controlled solvent removal. These approaches would need to be optimized to accommodate the presence of heptane and its effects on reaction kinetics and network formation.
Safety considerations are paramount when scaling up production involving heptane due to its flammability and potential health hazards. Robust engineering controls, including proper ventilation systems, explosion-proof equipment, and stringent handling protocols, must be implemented to ensure worker safety and environmental protection. This may necessitate significant investments in infrastructure and training.
From a production efficiency standpoint, the incorporation of heptane may impact cycle times and energy requirements. Optimization studies will be essential to balance the enhanced self-healing properties against potential increases in production costs. This may involve exploring alternative formulations or processing conditions that maintain the benefits of heptane while minimizing its impact on manufacturing efficiency.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!