How to Adjust Imaging Protocols Based on Echogenicity Results
JAN 20, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Echogenicity-Based Imaging Protocol Adjustment Background and Goals
Echogenicity, defined as the ability of tissues to reflect ultrasound waves, serves as a fundamental parameter in diagnostic imaging that directly influences image quality and diagnostic accuracy. The variation in tissue echogenicity arises from differences in acoustic impedance, tissue composition, and structural characteristics, creating distinct grayscale patterns that enable clinicians to differentiate between normal and pathological conditions. However, standard imaging protocols often fail to account for these variations, resulting in suboptimal image quality, reduced diagnostic confidence, and potential misinterpretation of findings.
The evolution of ultrasound technology has progressed from basic B-mode imaging to advanced techniques incorporating tissue harmonic imaging, compound imaging, and elastography. Despite these technological advances, the challenge of adapting imaging parameters to individual patient characteristics and specific tissue echogenicity patterns remains inadequately addressed. Current clinical practice typically relies on preset protocols that may not optimize visualization across diverse patient populations and varying pathological conditions.
The primary technical goal of echogenicity-based protocol adjustment is to establish dynamic imaging frameworks that automatically or semi-automatically modify acquisition parameters in response to real-time echogenicity assessment. This includes optimizing frequency selection, gain settings, focal zone positioning, and dynamic range adjustments to enhance contrast resolution and tissue differentiation. Such adaptive approaches aim to maximize signal-to-noise ratios while minimizing artifacts that compromise diagnostic quality.
From a clinical perspective, the objective extends beyond image optimization to improving diagnostic workflow efficiency and reducing operator dependency. By implementing intelligent protocol adjustment mechanisms, healthcare facilities can standardize image quality across different operators and patient populations, ultimately enhancing diagnostic consistency and reducing examination times. This becomes particularly critical in challenging scenarios such as imaging obese patients, evaluating highly attenuating tissues, or detecting subtle pathological changes in organs with variable echogenicity.
The strategic importance of this technology lies in its potential to bridge the gap between advanced imaging hardware capabilities and practical clinical implementation, ensuring that technological innovations translate into measurable improvements in patient care and diagnostic outcomes.
The evolution of ultrasound technology has progressed from basic B-mode imaging to advanced techniques incorporating tissue harmonic imaging, compound imaging, and elastography. Despite these technological advances, the challenge of adapting imaging parameters to individual patient characteristics and specific tissue echogenicity patterns remains inadequately addressed. Current clinical practice typically relies on preset protocols that may not optimize visualization across diverse patient populations and varying pathological conditions.
The primary technical goal of echogenicity-based protocol adjustment is to establish dynamic imaging frameworks that automatically or semi-automatically modify acquisition parameters in response to real-time echogenicity assessment. This includes optimizing frequency selection, gain settings, focal zone positioning, and dynamic range adjustments to enhance contrast resolution and tissue differentiation. Such adaptive approaches aim to maximize signal-to-noise ratios while minimizing artifacts that compromise diagnostic quality.
From a clinical perspective, the objective extends beyond image optimization to improving diagnostic workflow efficiency and reducing operator dependency. By implementing intelligent protocol adjustment mechanisms, healthcare facilities can standardize image quality across different operators and patient populations, ultimately enhancing diagnostic consistency and reducing examination times. This becomes particularly critical in challenging scenarios such as imaging obese patients, evaluating highly attenuating tissues, or detecting subtle pathological changes in organs with variable echogenicity.
The strategic importance of this technology lies in its potential to bridge the gap between advanced imaging hardware capabilities and practical clinical implementation, ensuring that technological innovations translate into measurable improvements in patient care and diagnostic outcomes.
Market Demand for Adaptive Ultrasound Imaging Solutions
The global ultrasound imaging market is experiencing significant transformation driven by increasing demand for personalized diagnostic solutions and real-time adaptive imaging capabilities. Healthcare providers are seeking advanced systems that can automatically optimize imaging parameters based on tissue echogenicity characteristics, reducing operator dependency and improving diagnostic accuracy across diverse patient populations. This demand is particularly pronounced in point-of-care settings, emergency departments, and resource-limited environments where experienced sonographers may not always be available.
Clinical applications requiring adaptive ultrasound protocols span multiple medical specialties. In obstetrics and gynecology, varying tissue densities across different gestational stages and patient body habitus necessitate dynamic protocol adjustments. Abdominal imaging faces challenges with patients of different body mass indices, where echogenicity variations significantly impact image quality. Musculoskeletal and vascular imaging similarly require real-time optimization to accommodate anatomical variations and pathological changes in tissue composition.
The aging global population and rising prevalence of chronic diseases are amplifying the need for efficient diagnostic workflows. Healthcare systems are under pressure to increase throughput while maintaining diagnostic quality, creating strong market pull for intelligent imaging solutions that can reduce examination times and minimize repeat scans. Hospitals and imaging centers are increasingly prioritizing equipment investments that demonstrate measurable improvements in workflow efficiency and diagnostic confidence.
Emerging markets in Asia-Pacific and Latin America represent substantial growth opportunities, where expanding healthcare infrastructure and increasing medical imaging accessibility drive demand for user-friendly, adaptive technologies. These regions often face acute shortages of specialized imaging professionals, making automated protocol adjustment capabilities particularly valuable. The shift toward value-based healthcare models globally further incentivizes adoption of technologies that can demonstrate improved patient outcomes and operational efficiency.
Regulatory trends favoring artificial intelligence integration in medical devices and growing reimbursement support for advanced imaging techniques are creating favorable market conditions. Healthcare providers are actively seeking solutions that can seamlessly integrate echogenicity-based protocol optimization into existing clinical workflows, indicating strong commercial viability for innovations addressing this technical challenge.
Clinical applications requiring adaptive ultrasound protocols span multiple medical specialties. In obstetrics and gynecology, varying tissue densities across different gestational stages and patient body habitus necessitate dynamic protocol adjustments. Abdominal imaging faces challenges with patients of different body mass indices, where echogenicity variations significantly impact image quality. Musculoskeletal and vascular imaging similarly require real-time optimization to accommodate anatomical variations and pathological changes in tissue composition.
The aging global population and rising prevalence of chronic diseases are amplifying the need for efficient diagnostic workflows. Healthcare systems are under pressure to increase throughput while maintaining diagnostic quality, creating strong market pull for intelligent imaging solutions that can reduce examination times and minimize repeat scans. Hospitals and imaging centers are increasingly prioritizing equipment investments that demonstrate measurable improvements in workflow efficiency and diagnostic confidence.
Emerging markets in Asia-Pacific and Latin America represent substantial growth opportunities, where expanding healthcare infrastructure and increasing medical imaging accessibility drive demand for user-friendly, adaptive technologies. These regions often face acute shortages of specialized imaging professionals, making automated protocol adjustment capabilities particularly valuable. The shift toward value-based healthcare models globally further incentivizes adoption of technologies that can demonstrate improved patient outcomes and operational efficiency.
Regulatory trends favoring artificial intelligence integration in medical devices and growing reimbursement support for advanced imaging techniques are creating favorable market conditions. Healthcare providers are actively seeking solutions that can seamlessly integrate echogenicity-based protocol optimization into existing clinical workflows, indicating strong commercial viability for innovations addressing this technical challenge.
Current State of Echogenicity Assessment and Protocol Limitations
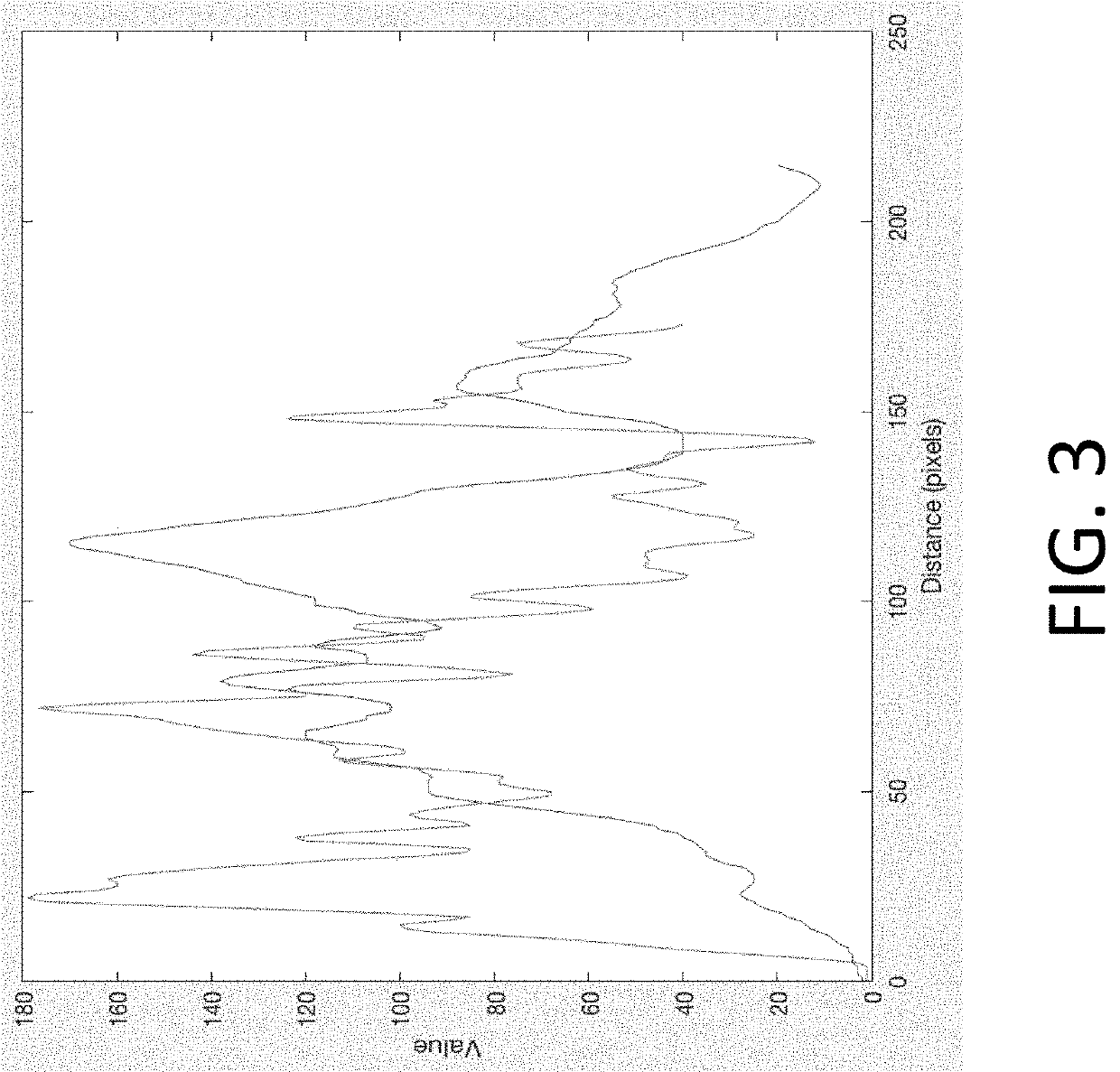
Echogenicity assessment currently relies on qualitative visual interpretation by sonographers and radiologists, who categorize tissues based on their acoustic reflectivity relative to surrounding structures. Standard classifications include hyperechoic, isoechoic, hypoechoic, and anechoic patterns. However, this subjective approach introduces significant inter-observer variability, with studies reporting concordance rates ranging from 60% to 85% depending on tissue type and operator experience. The lack of standardized quantitative metrics makes it challenging to establish consistent protocols for imaging parameter adjustments.
Most ultrasound systems operate with preset imaging protocols optimized for average tissue characteristics, failing to account for individual variations in echogenicity. These fixed parameters include frequency selection, gain settings, time-gain compensation curves, and dynamic range configurations. When encountering tissues with unexpected echogenic properties, operators manually adjust settings based on personal experience rather than systematic guidelines. This reactive approach often results in suboptimal image quality, particularly in challenging scenarios such as highly attenuating tissues or complex heterogeneous structures.
Current protocol limitations become particularly evident in specific clinical contexts. Obese patients present increased acoustic attenuation requiring lower frequencies and higher gain, yet standardized adjustment algorithms remain absent. Similarly, fibrotic tissues exhibiting altered echogenicity patterns demand customized imaging parameters that are not systematically defined in existing protocols. The situation is further complicated by equipment variations across manufacturers, as different platforms employ proprietary signal processing algorithms that respond differently to echogenicity changes.
Recent technological advances have introduced computer-aided quantitative ultrasound techniques, including texture analysis and acoustic parameter mapping. However, these tools remain largely confined to research settings and have not been integrated into routine clinical workflow for real-time protocol adjustment. The gap between echogenicity assessment capabilities and actionable protocol modification strategies represents a critical limitation in contemporary ultrasound practice, directly impacting diagnostic accuracy and examination efficiency across diverse patient populations and pathological conditions.
Most ultrasound systems operate with preset imaging protocols optimized for average tissue characteristics, failing to account for individual variations in echogenicity. These fixed parameters include frequency selection, gain settings, time-gain compensation curves, and dynamic range configurations. When encountering tissues with unexpected echogenic properties, operators manually adjust settings based on personal experience rather than systematic guidelines. This reactive approach often results in suboptimal image quality, particularly in challenging scenarios such as highly attenuating tissues or complex heterogeneous structures.
Current protocol limitations become particularly evident in specific clinical contexts. Obese patients present increased acoustic attenuation requiring lower frequencies and higher gain, yet standardized adjustment algorithms remain absent. Similarly, fibrotic tissues exhibiting altered echogenicity patterns demand customized imaging parameters that are not systematically defined in existing protocols. The situation is further complicated by equipment variations across manufacturers, as different platforms employ proprietary signal processing algorithms that respond differently to echogenicity changes.
Recent technological advances have introduced computer-aided quantitative ultrasound techniques, including texture analysis and acoustic parameter mapping. However, these tools remain largely confined to research settings and have not been integrated into routine clinical workflow for real-time protocol adjustment. The gap between echogenicity assessment capabilities and actionable protocol modification strategies represents a critical limitation in contemporary ultrasound practice, directly impacting diagnostic accuracy and examination efficiency across diverse patient populations and pathological conditions.
Existing Solutions for Echogenicity-Driven Protocol Adjustment
01 Ultrasound imaging protocols for tissue characterization
Methods and systems for optimizing ultrasound imaging protocols to assess tissue echogenicity involve adjusting imaging parameters such as frequency, gain, and focus depth. These protocols enable standardized evaluation of tissue characteristics by analyzing echo patterns and intensity distributions. Advanced signal processing techniques are employed to enhance image quality and improve the accuracy of echogenicity measurements across different tissue types.- Ultrasound imaging protocols for tissue characterization: Methods and systems for optimizing ultrasound imaging protocols to assess tissue echogenicity involve adjusting imaging parameters such as frequency, gain, and focus depth. These protocols enable standardized evaluation of tissue characteristics by analyzing echo patterns and intensity distributions. Advanced signal processing techniques are employed to enhance image quality and improve the accuracy of echogenicity measurements across different tissue types.
- Contrast-enhanced ultrasound imaging techniques: Imaging protocols utilizing contrast agents to enhance echogenicity involve the administration of microbubble-based contrast media that improve visualization of vascular structures and tissue perfusion. These techniques include specific timing sequences and imaging modes designed to capture contrast enhancement phases. The protocols optimize detection sensitivity and enable better differentiation between normal and pathological tissues based on their echogenic properties.
- Automated echogenicity analysis and classification systems: Computer-implemented methods for automated analysis of echogenicity patterns utilize machine learning algorithms and image processing techniques to classify tissue types. These systems extract quantitative features from ultrasound images and apply pattern recognition to standardize echogenicity assessment. The protocols include calibration procedures and reference standards to ensure consistent measurements across different imaging devices and operators.
- Multi-modal imaging integration for echogenicity assessment: Protocols combining ultrasound echogenicity data with other imaging modalities enable comprehensive tissue characterization. These approaches involve registration and fusion of ultrasound images with computed tomography, magnetic resonance imaging, or other diagnostic imaging data. The integrated protocols provide enhanced spatial resolution and improved diagnostic accuracy by correlating echogenic features with anatomical and functional information from complementary imaging techniques.
- Real-time echogenicity monitoring during interventional procedures: Dynamic imaging protocols for monitoring echogenicity changes during therapeutic interventions include real-time feedback mechanisms and adaptive parameter adjustment. These methods track temporal variations in tissue echogenicity to guide needle placement, monitor ablation procedures, or assess treatment response. The protocols incorporate motion compensation and artifact reduction techniques to maintain image quality during active procedures.
02 Contrast-enhanced ultrasound imaging techniques
Imaging protocols utilizing contrast agents to enhance echogenicity involve the administration of microbubble-based agents that improve visualization of vascular structures and tissue perfusion. These techniques include specific timing sequences and imaging modes designed to capture contrast enhancement phases. The protocols optimize detection sensitivity and enable differentiation between normal and pathological tissues based on their contrast uptake patterns.Expand Specific Solutions03 Automated echogenicity analysis and classification systems
Computer-implemented methods for automated assessment of tissue echogenicity utilize machine learning algorithms and image processing techniques to classify tissues based on their acoustic properties. These systems analyze grayscale distributions, texture patterns, and spatial relationships to generate quantitative echogenicity scores. The protocols incorporate standardized reference databases and calibration procedures to ensure consistent and reproducible measurements across different imaging platforms.Expand Specific Solutions04 Multi-modal imaging integration for echogenicity assessment
Protocols combining ultrasound echogenicity data with other imaging modalities enable comprehensive tissue characterization. These approaches integrate information from multiple sources to create enhanced diagnostic frameworks. The methods include registration techniques to align images from different modalities and fusion algorithms to combine complementary information, improving the overall accuracy of tissue assessment and pathology detection.Expand Specific Solutions05 Real-time echogenicity monitoring during interventional procedures
Dynamic imaging protocols for monitoring echogenicity changes during therapeutic interventions provide real-time feedback for procedure guidance. These methods track temporal variations in tissue acoustic properties to assess treatment efficacy and detect complications. The protocols incorporate adaptive imaging parameters that adjust automatically based on tissue response, enabling continuous visualization and quantification of echogenicity changes throughout the procedure.Expand Specific Solutions
Key Players in Ultrasound Imaging and Protocol Optimization
The ultrasound echogenicity-based imaging protocol adjustment field represents a mature yet evolving market segment within medical imaging, currently in a growth phase driven by AI integration and automation demands. Major established players including Siemens Healthineers AG, Koninklijke Philips NV, and Shenzhen Mindray Bio-Medical Electronics dominate with comprehensive ultrasound platforms featuring advanced echogenicity analysis capabilities. Mid-tier innovators like Sonoscape Medical Corp. and SuperSonic Imagine SA contribute specialized elastography and tissue characterization technologies. The technology maturity varies across the competitive landscape: while core echogenicity detection is well-established among leaders like Siemens Healthineers and Philips, emerging companies such as AmCad BioMed Corp. and Optellum Ltd. are advancing AI-powered automated protocol optimization, representing next-generation solutions. The market shows strong consolidation with established medical device manufacturers leveraging extensive clinical networks, while specialized firms focus on niche applications in oncology and vascular imaging, indicating a bifurcated competitive structure between comprehensive system providers and targeted innovation specialists.
Siemens Healthineers AG
Technical Solution: Siemens Healthineers has developed advanced ultrasound imaging systems with adaptive imaging protocols based on tissue echogenicity assessment. Their technology employs real-time tissue characterization algorithms that automatically adjust frequency, gain, and dynamic range parameters according to detected echogenicity patterns[2][5]. The system utilizes machine learning-based tissue classification to identify hypoechoic, isoechoic, and hyperechoic regions, then optimizes imaging parameters including transmit power, time-gain compensation curves, and focal zone positioning. Their ACUSON series incorporates intelligent tissue-specific presets that modify imaging protocols based on echogenic properties, enabling enhanced visualization of structures with varying acoustic impedance[7][9].
Strengths: Comprehensive automated adjustment capabilities with AI-driven optimization; extensive clinical validation across multiple applications. Weaknesses: High system cost; requires significant training for optimal utilization of advanced features.
Koninklijke Philips NV
Technical Solution: Philips has implemented adaptive imaging technology in their EPIQ and Affiniti ultrasound platforms that dynamically adjusts scanning protocols based on echogenicity analysis. Their solution features real-time tissue characterization using multi-parametric analysis of echo amplitude, texture patterns, and attenuation coefficients[3][8]. The system automatically modulates frequency selection, harmonic imaging modes, and compound imaging angles based on tissue echogenic properties. Philips' proprietary algorithms assess echogenicity heterogeneity and adjust focal zones, beam density, and processing parameters to optimize image quality for both highly echogenic structures like calcifications and hypoechoic lesions. The technology includes preset optimization libraries for organ-specific imaging protocols[6][11].
Strengths: Excellent integration with clinical workflows; robust performance across diverse patient populations and body habitus. Weaknesses: Limited customization options for advanced users; moderate learning curve for protocol optimization.
Core Innovations in Automated Protocol Adaptation Algorithms
Systems and methods for automated image recognition of implants and compositions with long-lasting echogenicity
PatentInactiveUS20220015742A1
Innovation
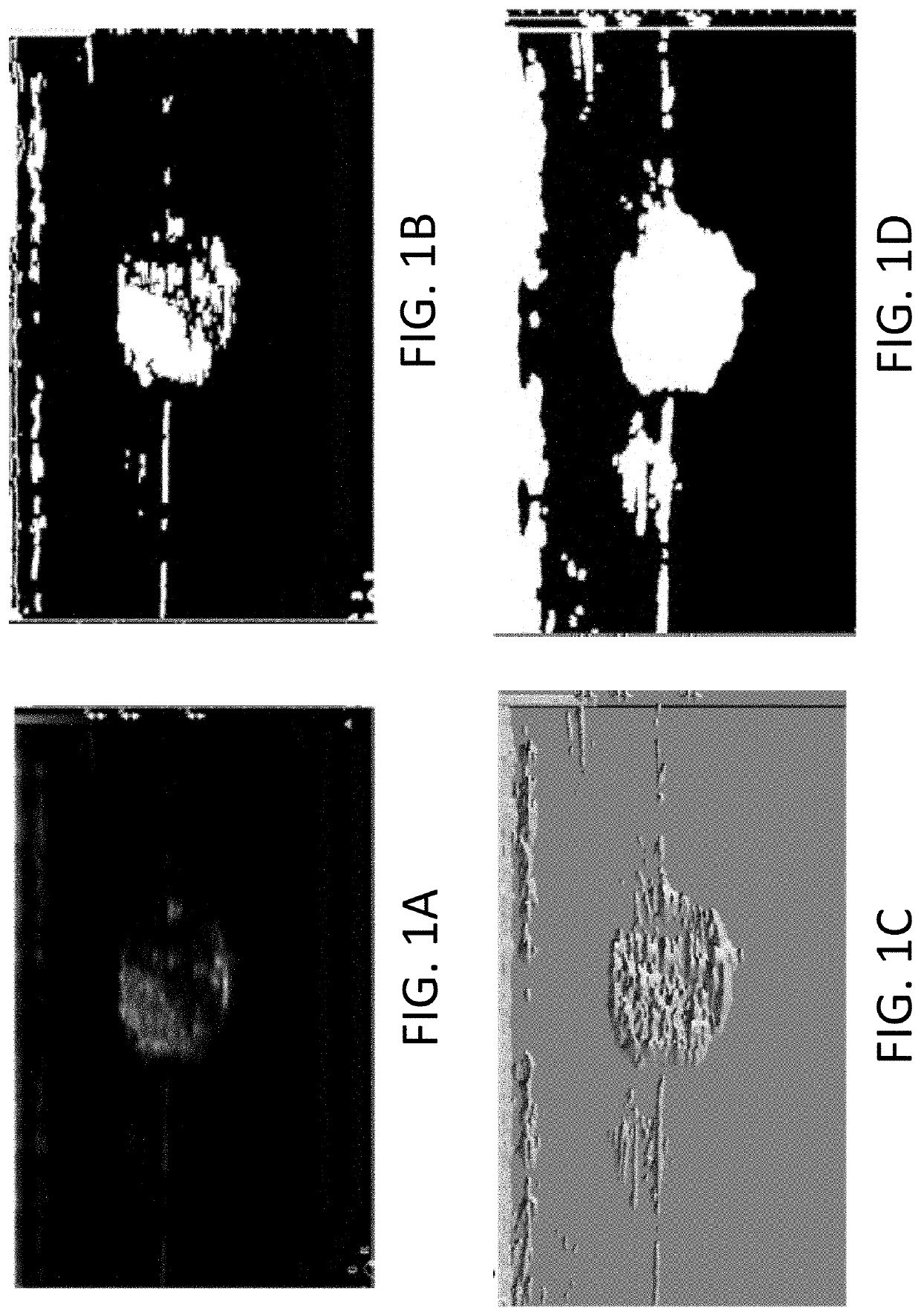
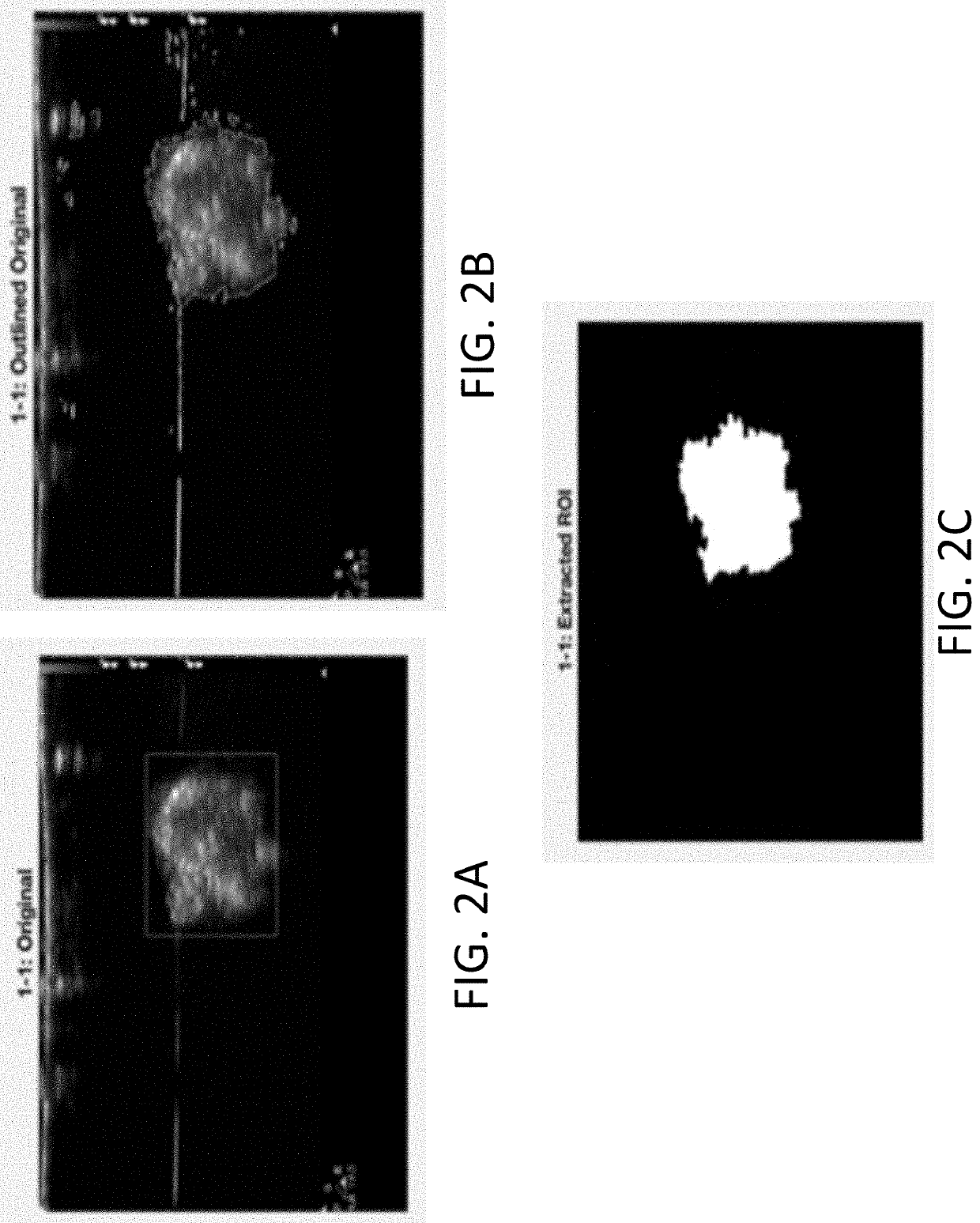
- The development of systems and methods for automatically detecting and analyzing bodily implants using algorithms that extract Regions of Interest (ROIs) from medical images, combined with the use of non-gas ultrasound contrast agents that maintain echogenicity for extended periods, allowing for long-term visualization and tracking of implants.
Adjusting acquisition protocols for dynamic medical imaging using dynamic models
PatentActiveUS20100183206A1
Innovation
- A method and apparatus for adjusting dynamic medical imaging acquisition protocols using anatomic and dynamic models to compensate for temporal variations in patient motion and contrast agent flow, allowing for more standardized, reproducible, and efficient image acquisition by fitting anatomic models to patient data and adjusting protocol parameters accordingly.
Clinical Validation and Regulatory Requirements for Adaptive Protocols
The implementation of adaptive imaging protocols based on echogenicity results necessitates rigorous clinical validation to ensure patient safety and diagnostic accuracy. Clinical trials must demonstrate that protocol adjustments maintain or improve image quality while optimizing examination efficiency across diverse patient populations. Validation studies typically require multi-center participation to establish statistical significance and account for variations in equipment, operator expertise, and patient demographics. These studies must document the correlation between echogenicity measurements and optimal imaging parameters, establishing evidence-based thresholds for protocol modifications.
Regulatory frameworks governing adaptive protocols vary significantly across jurisdictions, with agencies such as the FDA, EMA, and NMPA requiring comprehensive documentation of algorithm performance and clinical outcomes. Manufacturers must provide substantial evidence that automated adjustments do not compromise diagnostic capability or introduce systematic errors. The regulatory submission process demands detailed technical documentation including algorithm validation data, failure mode analysis, and risk mitigation strategies. Software as a Medical Device (SaMD) classifications often apply to adaptive imaging systems, requiring ongoing post-market surveillance and periodic safety updates.
Quality management systems must incorporate specific controls for adaptive protocols, including verification procedures for echogenicity measurement accuracy and protocol adjustment logic. Clinical sites implementing these technologies require standardized training programs and competency assessments to ensure consistent application. Documentation requirements extend beyond initial approval to include continuous monitoring of protocol performance, adverse event reporting, and periodic recalibration procedures.
Ethical considerations surrounding adaptive protocols include informed consent processes that clearly communicate the automated nature of adjustments and potential limitations. Regulatory bodies increasingly emphasize the need for transparency in algorithmic decision-making and the maintenance of clinician oversight capabilities. Compliance with data privacy regulations becomes critical when adaptive systems utilize patient-specific information for protocol optimization, requiring robust data governance frameworks and security measures to protect sensitive medical information throughout the imaging workflow.
Regulatory frameworks governing adaptive protocols vary significantly across jurisdictions, with agencies such as the FDA, EMA, and NMPA requiring comprehensive documentation of algorithm performance and clinical outcomes. Manufacturers must provide substantial evidence that automated adjustments do not compromise diagnostic capability or introduce systematic errors. The regulatory submission process demands detailed technical documentation including algorithm validation data, failure mode analysis, and risk mitigation strategies. Software as a Medical Device (SaMD) classifications often apply to adaptive imaging systems, requiring ongoing post-market surveillance and periodic safety updates.
Quality management systems must incorporate specific controls for adaptive protocols, including verification procedures for echogenicity measurement accuracy and protocol adjustment logic. Clinical sites implementing these technologies require standardized training programs and competency assessments to ensure consistent application. Documentation requirements extend beyond initial approval to include continuous monitoring of protocol performance, adverse event reporting, and periodic recalibration procedures.
Ethical considerations surrounding adaptive protocols include informed consent processes that clearly communicate the automated nature of adjustments and potential limitations. Regulatory bodies increasingly emphasize the need for transparency in algorithmic decision-making and the maintenance of clinician oversight capabilities. Compliance with data privacy regulations becomes critical when adaptive systems utilize patient-specific information for protocol optimization, requiring robust data governance frameworks and security measures to protect sensitive medical information throughout the imaging workflow.
Standardization Challenges in Echogenicity-Based Imaging Workflows
Standardization challenges in echogenicity-based imaging workflows represent a critical barrier to the widespread adoption and optimization of ultrasound protocols. The fundamental issue stems from the subjective nature of echogenicity assessment, where tissue characterization depends heavily on operator interpretation and equipment variability. Different ultrasound systems employ varying acoustic output parameters, frequency ranges, and signal processing algorithms, leading to inconsistent echogenicity measurements across platforms. This lack of uniformity complicates the establishment of universal protocols for adjusting imaging parameters based on echogenicity findings.
The absence of standardized terminology and classification systems further exacerbates workflow challenges. While terms such as hyperechoic, hypoechoic, and isoechoic are widely used, their precise definitions and threshold values remain institution-dependent. This variability creates difficulties in developing automated protocol adjustment systems and hinders effective communication among healthcare providers. Additionally, the lack of quantitative metrics for echogenicity assessment limits the reproducibility of imaging protocols across different clinical settings.
Equipment calibration and quality assurance protocols vary significantly across healthcare facilities, introducing another layer of complexity. Without standardized calibration procedures, the same tissue may exhibit different echogenicity characteristics on different machines, making protocol adjustments unreliable. The integration of artificial intelligence and machine learning tools for echogenicity analysis has introduced new standardization requirements, as these systems require consistent training datasets and validation methodologies.
Regulatory frameworks and clinical guidelines have not kept pace with technological advances in echogenicity-based imaging. The lack of consensus on best practices for protocol adjustment creates uncertainty for practitioners and institutions seeking to implement systematic approaches. International collaboration efforts are needed to establish unified standards for echogenicity measurement, reporting, and protocol optimization. Addressing these standardization challenges requires coordinated efforts among equipment manufacturers, professional societies, regulatory bodies, and clinical practitioners to develop comprehensive frameworks that ensure consistency while maintaining flexibility for specific clinical applications.
The absence of standardized terminology and classification systems further exacerbates workflow challenges. While terms such as hyperechoic, hypoechoic, and isoechoic are widely used, their precise definitions and threshold values remain institution-dependent. This variability creates difficulties in developing automated protocol adjustment systems and hinders effective communication among healthcare providers. Additionally, the lack of quantitative metrics for echogenicity assessment limits the reproducibility of imaging protocols across different clinical settings.
Equipment calibration and quality assurance protocols vary significantly across healthcare facilities, introducing another layer of complexity. Without standardized calibration procedures, the same tissue may exhibit different echogenicity characteristics on different machines, making protocol adjustments unreliable. The integration of artificial intelligence and machine learning tools for echogenicity analysis has introduced new standardization requirements, as these systems require consistent training datasets and validation methodologies.
Regulatory frameworks and clinical guidelines have not kept pace with technological advances in echogenicity-based imaging. The lack of consensus on best practices for protocol adjustment creates uncertainty for practitioners and institutions seeking to implement systematic approaches. International collaboration efforts are needed to establish unified standards for echogenicity measurement, reporting, and protocol optimization. Addressing these standardization challenges requires coordinated efforts among equipment manufacturers, professional societies, regulatory bodies, and clinical practitioners to develop comprehensive frameworks that ensure consistency while maintaining flexibility for specific clinical applications.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!