Proven Methods for Validating Echogenicity Across Platforms
JAN 20, 20268 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Echogenicity Validation Background and Objectives
Echogenicity, defined as the ability of tissue or material to reflect ultrasound waves, serves as a fundamental parameter in medical imaging diagnostics. The acoustic properties of tissues determine their appearance on ultrasound images, ranging from anechoic (black) to hyperechoic (bright white). However, the interpretation and quantification of echogenicity have historically been subjective and operator-dependent, leading to inconsistencies in clinical diagnosis and research outcomes. This challenge has become increasingly pronounced as ultrasound technology has evolved across multiple manufacturers and platforms, each employing proprietary signal processing algorithms and display optimization techniques.
The proliferation of diverse ultrasound systems in clinical and research settings has created an urgent need for standardized validation methods. Different platforms may render identical tissue structures with varying echogenic appearances due to differences in transducer frequencies, beam-forming technologies, gain settings, and post-processing algorithms. This variability compromises the reproducibility of diagnostic findings and hinders the development of quantitative ultrasound biomarkers that could be universally applied across healthcare institutions.
The primary objective of this research is to establish proven methodologies for validating echogenicity measurements across different ultrasound platforms. This involves developing standardized phantoms with known acoustic properties, defining quantitative metrics that transcend platform-specific variations, and creating validation protocols that can be implemented consistently across diverse clinical environments. The goal extends beyond mere calibration to encompass the establishment of reference standards that enable meaningful comparison of echogenicity data obtained from different systems.
Furthermore, this research aims to bridge the gap between qualitative visual assessment and quantitative acoustic analysis. By establishing validated cross-platform methodologies, the research seeks to enhance diagnostic accuracy, facilitate multi-center clinical trials, and accelerate the translation of ultrasound-based biomarkers into routine clinical practice. The ultimate objective is to ensure that echogenicity measurements become reliable, reproducible, and clinically meaningful regardless of the imaging platform employed.
The proliferation of diverse ultrasound systems in clinical and research settings has created an urgent need for standardized validation methods. Different platforms may render identical tissue structures with varying echogenic appearances due to differences in transducer frequencies, beam-forming technologies, gain settings, and post-processing algorithms. This variability compromises the reproducibility of diagnostic findings and hinders the development of quantitative ultrasound biomarkers that could be universally applied across healthcare institutions.
The primary objective of this research is to establish proven methodologies for validating echogenicity measurements across different ultrasound platforms. This involves developing standardized phantoms with known acoustic properties, defining quantitative metrics that transcend platform-specific variations, and creating validation protocols that can be implemented consistently across diverse clinical environments. The goal extends beyond mere calibration to encompass the establishment of reference standards that enable meaningful comparison of echogenicity data obtained from different systems.
Furthermore, this research aims to bridge the gap between qualitative visual assessment and quantitative acoustic analysis. By establishing validated cross-platform methodologies, the research seeks to enhance diagnostic accuracy, facilitate multi-center clinical trials, and accelerate the translation of ultrasound-based biomarkers into routine clinical practice. The ultimate objective is to ensure that echogenicity measurements become reliable, reproducible, and clinically meaningful regardless of the imaging platform employed.
Market Demand for Cross-Platform Echogenicity Standards
The medical ultrasound industry is experiencing significant transformation driven by the proliferation of imaging platforms and the increasing demand for diagnostic consistency. Healthcare providers worldwide are confronting challenges related to image quality variability across different ultrasound systems, manufacturers, and software versions. This variability directly impacts clinical decision-making, particularly in fields such as obstetrics, cardiology, and oncology where echogenicity assessment is critical for diagnosis and treatment planning.
The growing adoption of point-of-care ultrasound devices and portable imaging systems has expanded market accessibility but simultaneously intensified the need for standardized validation methods. Hospitals and imaging centers operating multiple ultrasound platforms from different vendors face operational inefficiencies and potential diagnostic discrepancies when echogenicity measurements cannot be reliably compared across systems. This challenge is particularly acute in multi-site clinical trials and telemedicine applications where images are acquired on diverse equipment and interpreted remotely.
Regulatory bodies and accreditation organizations are increasingly emphasizing quality assurance protocols that ensure measurement reproducibility across platforms. The demand for cross-platform echogenicity standards is being driven by several key stakeholders including hospital networks seeking to optimize equipment procurement, clinical researchers requiring consistent imaging protocols, and medical device manufacturers aiming to demonstrate system interoperability. Insurance providers and healthcare payers are also showing interest in standardized imaging metrics as part of value-based care initiatives.
The market opportunity extends beyond traditional clinical settings into emerging applications such as artificial intelligence-based diagnostic tools, which require standardized input data for algorithm training and validation. Telehealth expansion and remote diagnostic services further amplify the need for platform-independent echogenicity validation methods that can ensure diagnostic accuracy regardless of the acquisition device. Additionally, the growing emphasis on personalized medicine and longitudinal patient monitoring creates demand for imaging standards that enable reliable comparison of echogenicity measurements over time and across different healthcare facilities.
Current market dynamics indicate strong institutional interest in solutions that can bridge the gap between proprietary imaging technologies while maintaining diagnostic fidelity, positioning cross-platform validation standards as a critical enabler for next-generation ultrasound applications.
The growing adoption of point-of-care ultrasound devices and portable imaging systems has expanded market accessibility but simultaneously intensified the need for standardized validation methods. Hospitals and imaging centers operating multiple ultrasound platforms from different vendors face operational inefficiencies and potential diagnostic discrepancies when echogenicity measurements cannot be reliably compared across systems. This challenge is particularly acute in multi-site clinical trials and telemedicine applications where images are acquired on diverse equipment and interpreted remotely.
Regulatory bodies and accreditation organizations are increasingly emphasizing quality assurance protocols that ensure measurement reproducibility across platforms. The demand for cross-platform echogenicity standards is being driven by several key stakeholders including hospital networks seeking to optimize equipment procurement, clinical researchers requiring consistent imaging protocols, and medical device manufacturers aiming to demonstrate system interoperability. Insurance providers and healthcare payers are also showing interest in standardized imaging metrics as part of value-based care initiatives.
The market opportunity extends beyond traditional clinical settings into emerging applications such as artificial intelligence-based diagnostic tools, which require standardized input data for algorithm training and validation. Telehealth expansion and remote diagnostic services further amplify the need for platform-independent echogenicity validation methods that can ensure diagnostic accuracy regardless of the acquisition device. Additionally, the growing emphasis on personalized medicine and longitudinal patient monitoring creates demand for imaging standards that enable reliable comparison of echogenicity measurements over time and across different healthcare facilities.
Current market dynamics indicate strong institutional interest in solutions that can bridge the gap between proprietary imaging technologies while maintaining diagnostic fidelity, positioning cross-platform validation standards as a critical enabler for next-generation ultrasound applications.
Current Challenges in Multi-Platform Echogenicity Validation
Multi-platform echogenicity validation faces significant technical obstacles stemming from fundamental differences in ultrasound hardware architectures and signal processing methodologies. Variations in transducer frequencies, beam-forming algorithms, and acoustic impedance characteristics across different manufacturers create substantial inconsistencies in echogenicity measurements. These hardware-level disparities result in the same tissue structure producing markedly different grayscale intensity values when imaged by different ultrasound systems, undermining the reliability of cross-platform comparisons.
The absence of universally accepted standardization protocols represents a critical impediment to validation efforts. Current industry practices lack consensus on reference phantoms, calibration procedures, and measurement metrics for echogenicity assessment. Different institutions employ disparate methodologies, making it nearly impossible to establish reproducible validation frameworks. This fragmentation is exacerbated by proprietary image processing algorithms that manufacturers implement as competitive advantages, creating black-box systems that resist standardized evaluation.
Image acquisition parameters introduce another layer of complexity to validation challenges. Variables such as gain settings, time-gain compensation, dynamic range adjustments, and focal zone positioning significantly influence echogenicity measurements. Even minor variations in these parameters can produce substantial differences in quantitative assessments, yet standardizing these settings across platforms proves problematic due to platform-specific optimization requirements and clinical workflow considerations.
Environmental and operational factors further complicate validation efforts. Temperature fluctuations affect acoustic properties of validation phantoms, while operator-dependent variables including probe positioning, contact pressure, and scanning techniques introduce human-factor inconsistencies. These elements create reproducibility challenges that extend beyond pure technical considerations into practical implementation domains.
Data interpretation and quantification methodologies present additional obstacles. The conversion from raw radiofrequency data to displayable B-mode images involves multiple transformation steps that vary across platforms. Establishing equivalent regions of interest, accounting for speckle noise patterns, and normalizing intensity scales require sophisticated analytical approaches that remain underdeveloped. The lack of robust statistical frameworks for comparing echogenicity measurements across heterogeneous datasets limits the scientific rigor of validation studies.
The absence of universally accepted standardization protocols represents a critical impediment to validation efforts. Current industry practices lack consensus on reference phantoms, calibration procedures, and measurement metrics for echogenicity assessment. Different institutions employ disparate methodologies, making it nearly impossible to establish reproducible validation frameworks. This fragmentation is exacerbated by proprietary image processing algorithms that manufacturers implement as competitive advantages, creating black-box systems that resist standardized evaluation.
Image acquisition parameters introduce another layer of complexity to validation challenges. Variables such as gain settings, time-gain compensation, dynamic range adjustments, and focal zone positioning significantly influence echogenicity measurements. Even minor variations in these parameters can produce substantial differences in quantitative assessments, yet standardizing these settings across platforms proves problematic due to platform-specific optimization requirements and clinical workflow considerations.
Environmental and operational factors further complicate validation efforts. Temperature fluctuations affect acoustic properties of validation phantoms, while operator-dependent variables including probe positioning, contact pressure, and scanning techniques introduce human-factor inconsistencies. These elements create reproducibility challenges that extend beyond pure technical considerations into practical implementation domains.
Data interpretation and quantification methodologies present additional obstacles. The conversion from raw radiofrequency data to displayable B-mode images involves multiple transformation steps that vary across platforms. Establishing equivalent regions of interest, accounting for speckle noise patterns, and normalizing intensity scales require sophisticated analytical approaches that remain underdeveloped. The lack of robust statistical frameworks for comparing echogenicity measurements across heterogeneous datasets limits the scientific rigor of validation studies.
Existing Cross-Platform Validation Methodologies
01 Ultrasound contrast agents for enhanced echogenicity
Contrast agents containing microbubbles or nanoparticles are used to enhance echogenicity in ultrasound imaging. These agents improve visualization of blood flow, tissue perfusion, and organ structures by increasing the acoustic impedance difference between tissues. The contrast agents can be formulated with various shell materials and gas cores to optimize their acoustic properties and stability.- Ultrasound contrast agents for enhanced echogenicity: Contrast agents containing microbubbles or nanoparticles are used to enhance echogenicity in ultrasound imaging. These agents improve visualization of blood flow, tissue perfusion, and anatomical structures by increasing the acoustic impedance difference between tissues. The contrast agents can be formulated with various shell materials and gas cores to optimize their acoustic properties and stability.
- Echogenic medical devices and implants: Medical devices such as catheters, needles, and implants are designed with echogenic properties to improve their visibility during ultrasound-guided procedures. These devices incorporate materials or surface modifications that enhance ultrasound reflection, allowing clinicians to accurately track device placement and positioning in real-time imaging. Various coating techniques and material compositions are employed to achieve optimal echogenicity.
- Tissue characterization using echogenicity analysis: Methods and systems for analyzing tissue echogenicity patterns to characterize tissue properties and detect abnormalities. These techniques involve processing ultrasound signals to quantify echogenicity levels, texture patterns, and spatial distributions. The analysis can be used for diagnostic purposes, including differentiation between normal and pathological tissues, assessment of tissue composition, and monitoring of disease progression.
- Echogenic pharmaceutical formulations: Pharmaceutical compositions formulated to exhibit echogenic properties for ultrasound-guided drug delivery and monitoring. These formulations include therapeutic agents combined with echogenic components that enable visualization during administration and tracking of distribution within the body. The echogenic properties facilitate precise targeting and real-time monitoring of drug delivery to specific anatomical sites.
- Image processing methods for echogenicity enhancement: Advanced signal and image processing techniques designed to enhance echogenicity visualization in ultrasound imaging. These methods include algorithms for noise reduction, contrast enhancement, and feature extraction that improve the detection and interpretation of echogenic structures. The processing techniques can be implemented in real-time imaging systems to provide improved diagnostic capabilities and measurement accuracy.
02 Echogenic medical devices and implants
Medical devices such as catheters, needles, and implants are designed with echogenic properties to improve their visibility during ultrasound-guided procedures. These devices incorporate materials or surface modifications that enhance ultrasound reflection, allowing clinicians to accurately track device placement and positioning in real-time imaging. Various coating techniques and material compositions are employed to achieve optimal echogenicity.Expand Specific Solutions03 Tissue characterization based on echogenicity patterns
Diagnostic methods utilize echogenicity patterns to characterize and differentiate tissue types, identify pathological conditions, and assess disease progression. Analysis of echo texture, intensity, and distribution patterns enables detection of abnormalities such as tumors, cysts, and inflammatory changes. Advanced image processing algorithms and machine learning techniques are applied to quantify echogenicity features for improved diagnostic accuracy.Expand Specific Solutions04 Echogenic drug delivery systems
Drug delivery systems are engineered with echogenic properties to enable ultrasound-guided targeting and monitoring of therapeutic agents. These systems combine pharmaceutical compounds with echogenic carriers that can be tracked during administration and allow for controlled release at specific anatomical sites. The echogenic properties facilitate real-time visualization of drug distribution and accumulation in target tissues.Expand Specific Solutions05 Methods for measuring and quantifying echogenicity
Standardized techniques and systems are developed for objective measurement and quantification of echogenicity in ultrasound imaging. These methods involve calibration protocols, reference standards, and computational algorithms to assess echo intensity levels and tissue acoustic properties. Quantitative echogenicity measurements support reproducible diagnostic assessments and enable comparison of results across different imaging systems and clinical settings.Expand Specific Solutions
Major Players in Ultrasound Platform Development
The research on validating echogenicity across platforms represents an emerging technical domain at the intersection of medical imaging and standardization methodologies. The competitive landscape reflects early-to-mid stage development, with market participation spanning technology giants, specialized medical device manufacturers, and academic institutions. Key players include Huawei Technologies and Agilent Technologies, who bring advanced signal processing and analytical instrumentation capabilities, alongside research-driven entities like Sichuan University contributing foundational validation methodologies. The technology maturity varies significantly—while established companies like Novartis and Agilent possess robust quality control frameworks, newer entrants are still developing cross-platform standardization protocols. Market scale remains relatively niche, primarily serving ultrasound equipment manufacturers and clinical research facilities requiring reproducible echogenicity measurements across different imaging systems and vendor platforms.
Agilent Technologies, Inc.
Technical Solution: Agilent has developed comprehensive validation methodologies for ultrasound echogenicity standardization across multiple platforms. Their approach utilizes tissue-mimicking phantoms with calibrated acoustic properties and reference materials to ensure consistent echogenicity measurements. The validation framework incorporates quantitative ultrasound (QUS) parameters including backscatter coefficient analysis, attenuation measurements, and grayscale standardization protocols. Their system employs automated quality assurance tools that perform periodic calibration checks using standardized test objects with known acoustic impedance values, enabling cross-platform comparison and longitudinal study reliability in clinical and research settings.
Strengths: Industry-leading precision instrumentation expertise, established quality control protocols, comprehensive phantom development capabilities. Weaknesses: High implementation costs, requires specialized training for proper calibration procedures.
Sichuan University
Technical Solution: Sichuan University has conducted research on ultrasound image standardization with focus on developing cost-effective validation methods suitable for resource-limited settings. Their approach includes simplified phantom designs using locally available materials, protocol standardization for common clinical applications, and development of software-based correction algorithms for echogenicity normalization. The research emphasizes practical implementation strategies for ensuring measurement consistency across different equipment generations and manufacturers commonly found in developing healthcare systems, with particular attention to portable and point-of-care ultrasound devices requiring field validation capabilities.
Strengths: Cost-effective solutions, practical implementation focus, adaptable to resource-constrained environments. Weaknesses: May lack precision of commercial systems, limited validation in high-end research applications.
Key Patents in Echogenicity Standardization
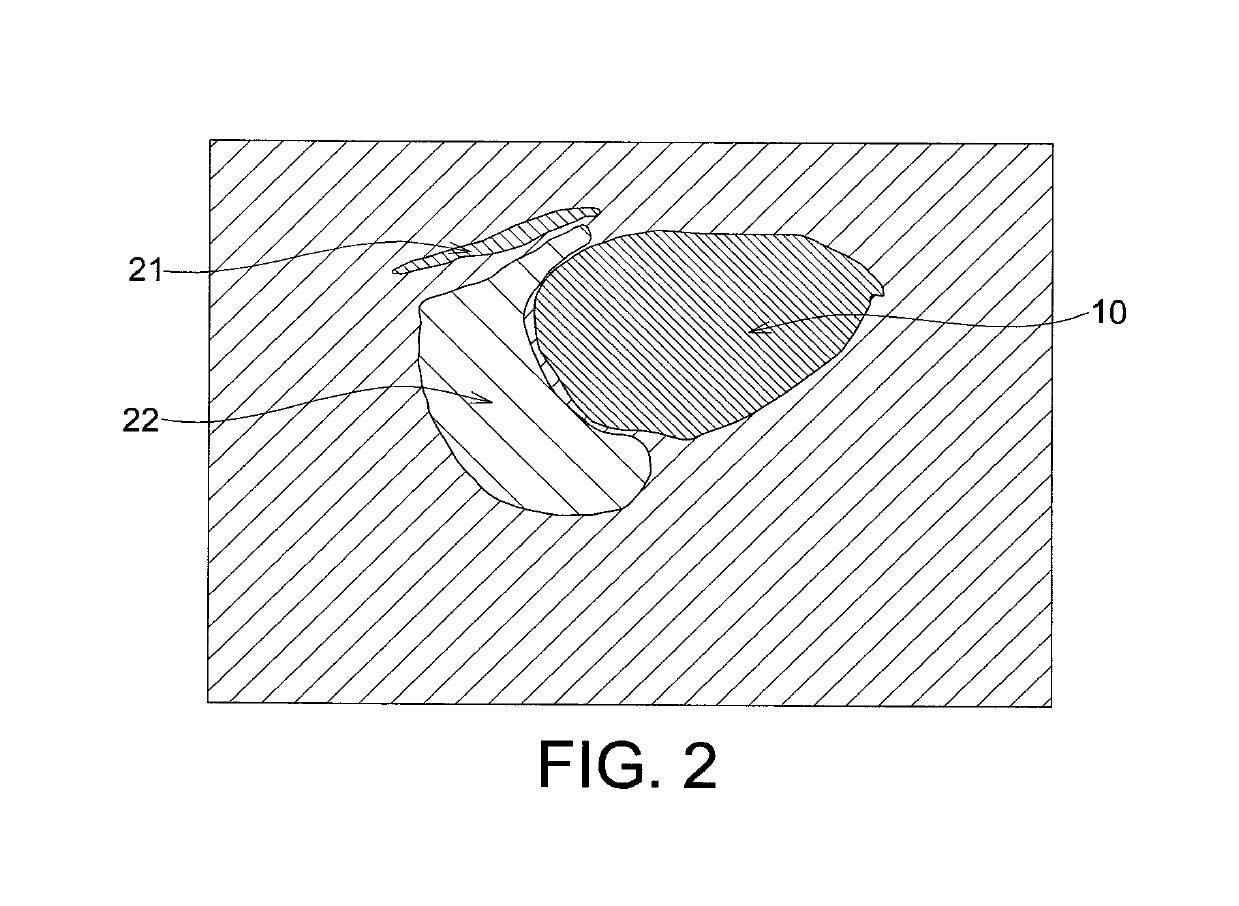
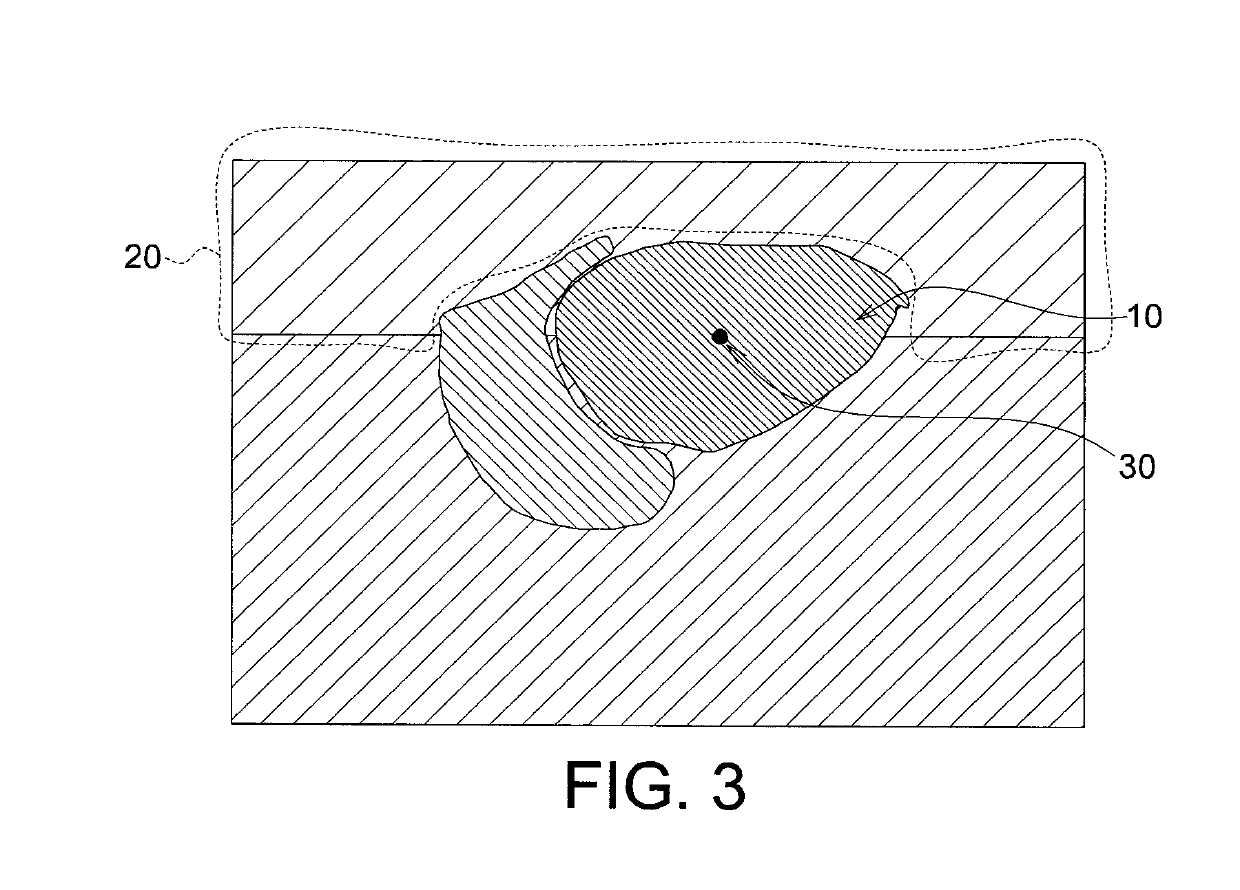
Echogenicity quantification method and calibration method for ultrasonic device using echogenicity index
PatentActiveUS10249037B2
Innovation
- An echogenicity quantification method that calculates an echogenicity index by averaging and normalizing grayscale values within a Region Of Interest (ROI) and a reference region, excluding outliers, to provide an objective and consistent measure across different ultrasonic devices and operators.
Method and system for generating collective output for validation
PatentWO2007103424A3
Innovation
- A logic engine is configured to guide users through the entire validation workflow, from project initiation to individual study design, providing systematic guidance for identifying, arranging, conducting and recording validation studies for chemistry kits and laboratory operations.
- Automatic generation of sample plate layouts for individual assays combined with unified data output that records studies, assays, results and outcomes across various forensic criteria in an integrated manner.
- An integrated validation platform that manages the complete validation process for verifying precision, sensitivity, accuracy, and reproducibility characteristics of biological tests such as DNA identification from start to finish.
Regulatory Standards for Ultrasound Quality Assurance
Ultrasound quality assurance operates within a comprehensive regulatory framework designed to ensure diagnostic accuracy, patient safety, and cross-platform consistency. The U.S. Food and Drug Administration (FDA) establishes baseline requirements through 21 CFR Part 1020.33, which mandates performance standards for diagnostic ultrasound equipment, including specifications for acoustic output measurements and safety limits. These regulations require manufacturers to provide detailed technical documentation demonstrating compliance with acoustic output parameters, particularly the mechanical index and thermal index values that directly influence echogenicity measurements.
The International Electrotechnical Commission (IEC) provides globally recognized standards, notably IEC 60601-2-37, which specifies essential performance requirements and test methods for ultrasound equipment. This standard addresses image quality parameters including spatial resolution, contrast resolution, and penetration depth—all critical factors affecting echogenicity validation. The American Institute of Ultrasound in Medicine (AIUM) complements these technical standards with practice guidelines that emphasize routine quality control testing protocols, recommending specific phantoms and test procedures for verifying system performance consistency.
Accreditation bodies such as the American College of Radiology (ACR) and the Intersocietal Accreditation Commission (IAC) enforce quality assurance programs requiring periodic equipment testing and documentation. These programs mandate quarterly phantom testing using standardized tissue-mimicking materials with known acoustic properties, enabling objective assessment of echogenicity representation across different platforms. The ACR's ultrasound accreditation program specifically requires facilities to maintain quality control records demonstrating consistent system performance over time.
European regulations under the Medical Device Regulation (MDR 2017/745) impose stringent post-market surveillance requirements, compelling manufacturers to continuously monitor device performance and report deviations. This regulatory environment necessitates robust validation methodologies that can demonstrate echogenicity consistency not only during initial certification but throughout the product lifecycle. Compliance with these multifaceted regulatory standards ensures that echogenicity validation methods meet internationally accepted benchmarks for reliability and reproducibility, forming the foundation for meaningful cross-platform comparisons in clinical practice.
The International Electrotechnical Commission (IEC) provides globally recognized standards, notably IEC 60601-2-37, which specifies essential performance requirements and test methods for ultrasound equipment. This standard addresses image quality parameters including spatial resolution, contrast resolution, and penetration depth—all critical factors affecting echogenicity validation. The American Institute of Ultrasound in Medicine (AIUM) complements these technical standards with practice guidelines that emphasize routine quality control testing protocols, recommending specific phantoms and test procedures for verifying system performance consistency.
Accreditation bodies such as the American College of Radiology (ACR) and the Intersocietal Accreditation Commission (IAC) enforce quality assurance programs requiring periodic equipment testing and documentation. These programs mandate quarterly phantom testing using standardized tissue-mimicking materials with known acoustic properties, enabling objective assessment of echogenicity representation across different platforms. The ACR's ultrasound accreditation program specifically requires facilities to maintain quality control records demonstrating consistent system performance over time.
European regulations under the Medical Device Regulation (MDR 2017/745) impose stringent post-market surveillance requirements, compelling manufacturers to continuously monitor device performance and report deviations. This regulatory environment necessitates robust validation methodologies that can demonstrate echogenicity consistency not only during initial certification but throughout the product lifecycle. Compliance with these multifaceted regulatory standards ensures that echogenicity validation methods meet internationally accepted benchmarks for reliability and reproducibility, forming the foundation for meaningful cross-platform comparisons in clinical practice.
Phantom-Based Validation Protocol Development
Phantom-based validation protocols represent a systematic approach to ensuring consistent echogenicity measurements across different ultrasound platforms and imaging conditions. These protocols utilize tissue-mimicking phantoms with known acoustic properties as reference standards, enabling objective assessment of system performance and cross-platform comparability. The development of robust validation protocols requires careful consideration of phantom material composition, geometric design, and testing procedures that can accommodate variations in transducer frequencies, imaging modes, and manufacturer-specific processing algorithms.
The foundation of effective phantom-based validation lies in selecting or fabricating phantoms with stable, reproducible acoustic characteristics that closely simulate biological tissue properties. Standard phantoms typically incorporate materials such as agar, gelatin, or polyvinyl alcohol cryogel matrices embedded with scattering particles to achieve controlled echogenicity levels. These phantoms must demonstrate long-term stability, minimal temperature dependence, and uniform acoustic properties throughout their volume to serve as reliable reference standards.
Protocol development necessitates establishing standardized imaging parameters and acquisition procedures that minimize operator-dependent variability while accommodating platform-specific capabilities. This includes defining optimal transducer positioning, gain settings, focal zone placement, and image depth configurations. Documentation of environmental conditions such as room temperature and phantom equilibration time is essential for reproducibility. The protocol should incorporate multiple regions of interest within the phantom to assess spatial uniformity and enable statistical analysis of measurement precision.
Quantitative analysis methods form a critical component of validation protocols, typically involving grayscale histogram analysis, mean pixel intensity measurements, and signal-to-noise ratio calculations. These metrics provide objective benchmarks for comparing echogenicity measurements across different systems and time points. Establishing acceptance criteria based on coefficient of variation thresholds and inter-platform correlation coefficients enables systematic quality assurance and facilitates identification of platform-specific biases or calibration drift that may compromise clinical measurement reliability.
The foundation of effective phantom-based validation lies in selecting or fabricating phantoms with stable, reproducible acoustic characteristics that closely simulate biological tissue properties. Standard phantoms typically incorporate materials such as agar, gelatin, or polyvinyl alcohol cryogel matrices embedded with scattering particles to achieve controlled echogenicity levels. These phantoms must demonstrate long-term stability, minimal temperature dependence, and uniform acoustic properties throughout their volume to serve as reliable reference standards.
Protocol development necessitates establishing standardized imaging parameters and acquisition procedures that minimize operator-dependent variability while accommodating platform-specific capabilities. This includes defining optimal transducer positioning, gain settings, focal zone placement, and image depth configurations. Documentation of environmental conditions such as room temperature and phantom equilibration time is essential for reproducibility. The protocol should incorporate multiple regions of interest within the phantom to assess spatial uniformity and enable statistical analysis of measurement precision.
Quantitative analysis methods form a critical component of validation protocols, typically involving grayscale histogram analysis, mean pixel intensity measurements, and signal-to-noise ratio calculations. These metrics provide objective benchmarks for comparing echogenicity measurements across different systems and time points. Establishing acceptance criteria based on coefficient of variation thresholds and inter-platform correlation coefficients enables systematic quality assurance and facilitates identification of platform-specific biases or calibration drift that may compromise clinical measurement reliability.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!