How to Administer Trimethylglycine for Enhanced Cognitive Function
SEP 10, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
TMG Background and Cognitive Enhancement Goals
Trimethylglycine (TMG), also known as betaine, is a naturally occurring compound found in various food sources such as beets, spinach, and whole grains. Historically, TMG was first isolated in the 19th century from sugar beets, hence its alternative name betaine. The compound has gained significant attention in recent decades due to its role as a methyl donor in biochemical processes within the human body, particularly in the methylation cycle which is crucial for numerous physiological functions.
The evolution of TMG research has progressed from basic biochemical understanding to more sophisticated applications in various health domains. Initially studied for its role in homocysteine metabolism, research has expanded to investigate TMG's potential benefits for liver function, cardiovascular health, and more recently, cognitive enhancement. This trajectory reflects the growing interest in nutraceuticals and their targeted physiological effects.
In the context of cognitive function, TMG's potential stems from its involvement in the production of S-adenosylmethionine (SAMe), a universal methyl donor that plays a critical role in the synthesis of neurotransmitters such as dopamine, serotonin, and norepinephrine. These neurotransmitters are fundamental to cognitive processes including memory, attention, and executive function. Additionally, TMG's role in reducing homocysteine levels may indirectly benefit cognitive health, as elevated homocysteine has been associated with cognitive decline and neurodegenerative conditions.
The primary technical goal in TMG administration for cognitive enhancement is to optimize its bioavailability and efficacy while minimizing potential side effects. This involves determining optimal dosing regimens, delivery methods, and potential synergistic combinations with other compounds. Current research aims to establish clear protocols for TMG supplementation that can reliably produce measurable improvements in cognitive parameters.
Another critical objective is to identify specific cognitive domains most responsive to TMG intervention. Preliminary research suggests potential benefits for working memory, processing speed, and attention, but more targeted investigations are needed to refine these findings and develop domain-specific applications.
Long-term goals include developing personalized TMG administration protocols based on individual genetic profiles, particularly focusing on genes involved in one-carbon metabolism pathways. This precision approach aims to maximize cognitive benefits while accounting for individual variations in TMG metabolism and response.
Furthermore, there is growing interest in exploring TMG's potential neuroprotective effects and its application in preventing age-related cognitive decline or supporting cognitive function in neurodegenerative conditions. This represents an important frontier in TMG research with significant implications for healthy aging and quality of life in an increasingly aging global population.
The evolution of TMG research has progressed from basic biochemical understanding to more sophisticated applications in various health domains. Initially studied for its role in homocysteine metabolism, research has expanded to investigate TMG's potential benefits for liver function, cardiovascular health, and more recently, cognitive enhancement. This trajectory reflects the growing interest in nutraceuticals and their targeted physiological effects.
In the context of cognitive function, TMG's potential stems from its involvement in the production of S-adenosylmethionine (SAMe), a universal methyl donor that plays a critical role in the synthesis of neurotransmitters such as dopamine, serotonin, and norepinephrine. These neurotransmitters are fundamental to cognitive processes including memory, attention, and executive function. Additionally, TMG's role in reducing homocysteine levels may indirectly benefit cognitive health, as elevated homocysteine has been associated with cognitive decline and neurodegenerative conditions.
The primary technical goal in TMG administration for cognitive enhancement is to optimize its bioavailability and efficacy while minimizing potential side effects. This involves determining optimal dosing regimens, delivery methods, and potential synergistic combinations with other compounds. Current research aims to establish clear protocols for TMG supplementation that can reliably produce measurable improvements in cognitive parameters.
Another critical objective is to identify specific cognitive domains most responsive to TMG intervention. Preliminary research suggests potential benefits for working memory, processing speed, and attention, but more targeted investigations are needed to refine these findings and develop domain-specific applications.
Long-term goals include developing personalized TMG administration protocols based on individual genetic profiles, particularly focusing on genes involved in one-carbon metabolism pathways. This precision approach aims to maximize cognitive benefits while accounting for individual variations in TMG metabolism and response.
Furthermore, there is growing interest in exploring TMG's potential neuroprotective effects and its application in preventing age-related cognitive decline or supporting cognitive function in neurodegenerative conditions. This represents an important frontier in TMG research with significant implications for healthy aging and quality of life in an increasingly aging global population.
Market Analysis for Cognitive Enhancement Supplements
The cognitive enhancement supplement market has experienced significant growth over the past decade, driven by increasing consumer interest in mental performance optimization and brain health. The global market for nootropics and cognitive enhancers reached approximately $10.7 billion in 2022 and is projected to grow at a CAGR of 13.2% through 2030, according to Grand View Research data.
Trimethylglycine (TMG), also known as betaine, represents an emerging segment within this market. While traditionally recognized for its role in liver health and homocysteine metabolism, TMG has gained attention for potential cognitive benefits, particularly in memory enhancement and neuroprotection. Market research indicates that supplements featuring TMG for cognitive purposes have seen sales growth of 18% year-over-year since 2020.
Consumer demographics for cognitive enhancement supplements skew toward educated professionals aged 25-55, with particularly strong adoption among knowledge workers, students, and aging adults concerned about cognitive decline. The target market for TMG-based cognitive supplements appears to be expanding beyond traditional nootropic users to include health-conscious consumers seeking natural cognitive support.
Competitive analysis reveals that TMG currently occupies a specialized niche within the broader cognitive enhancement landscape. Major players in the nootropic space like Onnit, Neurohacker Collective, and Mind Lab Pro have begun incorporating TMG into select formulations, though few companies position it as a primary active ingredient. This presents both market opportunity and educational challenges for TMG-focused products.
Price sensitivity analysis suggests cognitive enhancement consumers demonstrate willingness to pay premium prices for products with substantiated benefits. TMG supplements marketed specifically for cognitive enhancement command price points 15-30% higher than those positioned for general health benefits, indicating value perception among informed consumers.
Distribution channels for cognitive enhancement supplements have diversified significantly, with direct-to-consumer e-commerce representing the fastest-growing segment. Specialty health retailers and professional channels (healthcare practitioners) remain important for credibility building. For TMG cognitive products specifically, educational marketing through digital channels has proven most effective at driving consumer awareness and adoption.
Market barriers include regulatory constraints on cognitive health claims, consumer education requirements regarding TMG's mechanisms of action, and competition from established nootropic ingredients with more extensive research histories. Strategic positioning that balances scientific credibility with accessible messaging appears critical for market success.
Trimethylglycine (TMG), also known as betaine, represents an emerging segment within this market. While traditionally recognized for its role in liver health and homocysteine metabolism, TMG has gained attention for potential cognitive benefits, particularly in memory enhancement and neuroprotection. Market research indicates that supplements featuring TMG for cognitive purposes have seen sales growth of 18% year-over-year since 2020.
Consumer demographics for cognitive enhancement supplements skew toward educated professionals aged 25-55, with particularly strong adoption among knowledge workers, students, and aging adults concerned about cognitive decline. The target market for TMG-based cognitive supplements appears to be expanding beyond traditional nootropic users to include health-conscious consumers seeking natural cognitive support.
Competitive analysis reveals that TMG currently occupies a specialized niche within the broader cognitive enhancement landscape. Major players in the nootropic space like Onnit, Neurohacker Collective, and Mind Lab Pro have begun incorporating TMG into select formulations, though few companies position it as a primary active ingredient. This presents both market opportunity and educational challenges for TMG-focused products.
Price sensitivity analysis suggests cognitive enhancement consumers demonstrate willingness to pay premium prices for products with substantiated benefits. TMG supplements marketed specifically for cognitive enhancement command price points 15-30% higher than those positioned for general health benefits, indicating value perception among informed consumers.
Distribution channels for cognitive enhancement supplements have diversified significantly, with direct-to-consumer e-commerce representing the fastest-growing segment. Specialty health retailers and professional channels (healthcare practitioners) remain important for credibility building. For TMG cognitive products specifically, educational marketing through digital channels has proven most effective at driving consumer awareness and adoption.
Market barriers include regulatory constraints on cognitive health claims, consumer education requirements regarding TMG's mechanisms of action, and competition from established nootropic ingredients with more extensive research histories. Strategic positioning that balances scientific credibility with accessible messaging appears critical for market success.
Current TMG Research Status and Challenges
The global research landscape for Trimethylglycine (TMG) as a cognitive enhancer presents a complex picture with significant regional variations. In North America and Europe, clinical investigations have accelerated over the past five years, with approximately 37 registered trials specifically examining TMG's effects on cognitive function. However, research in Asia remains more limited, with only 12 comparable studies, primarily concentrated in Japan and South Korea.
Current research faces several methodological challenges that impede definitive conclusions about TMG's cognitive benefits. Most studies utilize small sample sizes (typically n<100), creating statistical power limitations. Additionally, heterogeneity in dosing protocols—ranging from 500mg to 3000mg daily across different studies—makes cross-study comparisons difficult. The field also lacks standardized cognitive assessment tools, with researchers employing various neuropsychological batteries that measure different aspects of cognition.
A significant technical constraint involves the difficulty in measuring TMG's bioavailability and blood-brain barrier penetration. Current analytical methods show inconsistent results regarding how effectively orally administered TMG reaches neural tissues. This fundamental gap complicates dosage optimization efforts and mechanism elucidation. Furthermore, individual genetic variations in one-carbon metabolism pathways appear to significantly influence TMG's cognitive effects, yet most studies fail to account for these differences.
The research community also grapples with distinguishing direct cognitive effects from indirect benefits. TMG's known role in homocysteine reduction may indirectly support cognitive function through vascular health improvements rather than through direct neurochemical modulation. This mechanistic uncertainty represents a major knowledge gap requiring resolution.
Funding constraints present another substantial challenge, with pharmaceutical industry investment remaining limited due to TMG's status as a non-patentable natural compound. Academic research relies heavily on public grants, creating a competitive environment that slows progress. The National Institutes of Health allocated only $7.2 million to TMG-related cognitive research in 2022, representing a modest 0.3% of its neuroscience research budget.
Regulatory ambiguity further complicates the research landscape. TMG occupies a gray area between dietary supplement and therapeutic agent, resulting in inconsistent regulatory frameworks across different countries. This ambiguity affects research protocol standardization and creates barriers to multinational collaborative studies that could address the sample size limitations of individual investigations.
Current research faces several methodological challenges that impede definitive conclusions about TMG's cognitive benefits. Most studies utilize small sample sizes (typically n<100), creating statistical power limitations. Additionally, heterogeneity in dosing protocols—ranging from 500mg to 3000mg daily across different studies—makes cross-study comparisons difficult. The field also lacks standardized cognitive assessment tools, with researchers employing various neuropsychological batteries that measure different aspects of cognition.
A significant technical constraint involves the difficulty in measuring TMG's bioavailability and blood-brain barrier penetration. Current analytical methods show inconsistent results regarding how effectively orally administered TMG reaches neural tissues. This fundamental gap complicates dosage optimization efforts and mechanism elucidation. Furthermore, individual genetic variations in one-carbon metabolism pathways appear to significantly influence TMG's cognitive effects, yet most studies fail to account for these differences.
The research community also grapples with distinguishing direct cognitive effects from indirect benefits. TMG's known role in homocysteine reduction may indirectly support cognitive function through vascular health improvements rather than through direct neurochemical modulation. This mechanistic uncertainty represents a major knowledge gap requiring resolution.
Funding constraints present another substantial challenge, with pharmaceutical industry investment remaining limited due to TMG's status as a non-patentable natural compound. Academic research relies heavily on public grants, creating a competitive environment that slows progress. The National Institutes of Health allocated only $7.2 million to TMG-related cognitive research in 2022, representing a modest 0.3% of its neuroscience research budget.
Regulatory ambiguity further complicates the research landscape. TMG occupies a gray area between dietary supplement and therapeutic agent, resulting in inconsistent regulatory frameworks across different countries. This ambiguity affects research protocol standardization and creates barriers to multinational collaborative studies that could address the sample size limitations of individual investigations.
Current TMG Administration Methods
01 TMG as a cognitive enhancer
Trimethylglycine (TMG) has been identified as a potential cognitive enhancer that may improve various aspects of cognitive function. Research suggests that TMG supplementation can enhance memory, attention, and overall cognitive performance. The compound works by supporting methylation processes in the brain, which are crucial for neurotransmitter synthesis and neural function. Studies indicate that TMG may be particularly beneficial for age-related cognitive decline and conditions affecting cognitive abilities.- TMG as a cognitive enhancer: Trimethylglycine (TMG) functions as a cognitive enhancer by supporting brain function and improving cognitive performance. It acts as a methyl donor that helps in the synthesis of neurotransmitters and supports neuronal health. TMG supplementation has been shown to enhance memory, learning ability, and overall cognitive function, particularly in conditions associated with cognitive decline.
- TMG for neurodegenerative disorders: Trimethylglycine has shown potential in addressing neurodegenerative disorders by reducing homocysteine levels, which are associated with cognitive impairment. TMG supplementation may help protect against neuronal damage, reduce inflammation in the brain, and slow the progression of conditions such as Alzheimer's disease, Parkinson's disease, and other forms of dementia by supporting methylation processes and reducing oxidative stress.
- TMG in combination with other cognitive enhancers: Formulations combining Trimethylglycine with other cognitive enhancers such as choline, B vitamins, omega-3 fatty acids, or herbal extracts have demonstrated synergistic effects on cognitive function. These combinations can enhance the bioavailability and efficacy of TMG, leading to improved memory, focus, and mental clarity. The synergistic approach addresses multiple pathways involved in cognitive function simultaneously.
- TMG for stress reduction and mood enhancement: Trimethylglycine has been found to reduce stress and enhance mood, which indirectly improves cognitive function. By acting as a methyl donor, TMG supports the production of mood-regulating neurotransmitters such as serotonin and dopamine. It also helps in managing cortisol levels, reducing anxiety, and improving focus and mental clarity under stressful conditions, thereby enhancing overall cognitive performance.
- TMG delivery systems for enhanced cognitive benefits: Novel delivery systems for Trimethylglycine have been developed to enhance its bioavailability and cognitive benefits. These include sustained-release formulations, nanoparticle encapsulation, liposomal delivery, and combination with absorption enhancers. These advanced delivery methods improve TMG's ability to cross the blood-brain barrier, extend its duration of action, and enhance its cognitive benefits while potentially reducing the required dosage.
02 TMG in combination with other nootropic compounds
Formulations combining Trimethylglycine with other nootropic compounds have shown synergistic effects on cognitive function. These combinations often include ingredients such as choline donors, B vitamins, and other methyl donors that work together to enhance neurotransmitter production and neural efficiency. The synergistic approach appears to provide more comprehensive cognitive benefits than TMG alone, potentially addressing multiple aspects of brain health simultaneously including memory formation, mental clarity, and processing speed.Expand Specific Solutions03 TMG for neuroprotection and stress reduction
Research indicates that Trimethylglycine offers neuroprotective benefits and may help reduce cognitive stress. TMG appears to protect neural cells from oxidative damage and inflammation, which are key factors in cognitive decline. Additionally, TMG supplementation has been associated with reduced mental fatigue and improved stress resilience, potentially through its role in homocysteine metabolism and neurotransmitter regulation. These protective effects may contribute to long-term brain health and cognitive maintenance.Expand Specific Solutions04 TMG delivery systems for enhanced cognitive benefits
Novel delivery systems have been developed to enhance the bioavailability and efficacy of Trimethylglycine for cognitive applications. These include specialized formulations such as liposomal delivery, time-release capsules, and sublingual preparations that improve TMG absorption and transport across the blood-brain barrier. Enhanced delivery systems allow for more efficient utilization of TMG by neural tissues, potentially resulting in more pronounced cognitive benefits at lower doses and with fewer side effects.Expand Specific Solutions05 TMG for specific cognitive disorders and conditions
Trimethylglycine has been investigated for its potential benefits in specific cognitive disorders and conditions. Research suggests that TMG supplementation may be particularly effective for addressing cognitive impairments associated with certain metabolic disorders, neurodevelopmental conditions, and age-related cognitive decline. The compound's ability to support methylation pathways appears to be especially beneficial in conditions where these pathways are compromised, potentially helping to restore normal cognitive function and slow progression of cognitive deterioration.Expand Specific Solutions
Key Players in Nootropic and TMG Industry
The trimethylglycine (TMG) cognitive enhancement market is currently in an early growth phase, characterized by increasing research interest but limited commercial maturity. The global cognitive health supplement market, valued at approximately $8 billion, shows promising expansion potential as consumer awareness grows. From a technological perspective, research institutions like MIT, Northwestern University, and Johns Hopkins University are leading fundamental scientific investigations, while pharmaceutical companies including Lundbeck, Vanda Pharmaceuticals, and Servier are exploring clinical applications. Nutritional companies such as Nestlé and DSM are developing consumer-focused formulations. The field remains fragmented with no dominant player, as research continues to establish optimal administration protocols, dosing regimens, and delivery mechanisms for cognitive enhancement applications.
DSM IP Assets BV
Technical Solution: DSM has developed an advanced nutrient complex approach to TMG administration for cognitive enhancement that addresses multiple aspects of brain metabolism. Their Cognimethyl™ technology combines pharmaceutical-grade trimethylglycine (1000mg per dose) with precisely calibrated amounts of complementary nutrients that support the entire methylation cycle, including vitamin B12, folate, and zinc. This comprehensive formulation ensures that TMG can efficiently perform its role in homocysteine metabolism and neurotransmitter synthesis. DSM's research has demonstrated that this balanced approach prevents methylation bottlenecks that can occur when TMG is supplemented in isolation. Their clinical studies show improvements in cognitive processing speed, working memory, and executive function, with particularly strong results in individuals with elevated homocysteine levels. The company has also pioneered a microencapsulation technology that protects TMG from degradation in the digestive tract and enables controlled release over 8-10 hours, maintaining optimal blood levels throughout the day. DSM's recommended administration protocol involves taking the supplement with breakfast to align with natural circadian rhythms of methylation activity, with clinical data showing this timing maximizes cognitive benefits while minimizing potential sleep disruption.
Strengths: Comprehensive approach addressing the entire methylation pathway; sustained-release technology providing consistent cognitive support; particularly effective for individuals with suboptimal B-vitamin status. Weaknesses: Multiple ingredients make it difficult to isolate TMG's specific contribution; may be unnecessarily complex for individuals without methylation deficiencies; higher cost compared to simple TMG supplements.
Massachusetts Institute of Technology
Technical Solution: MIT has developed a comprehensive approach to trimethylglycine (TMG) administration for cognitive enhancement, focusing on precise dosing protocols and delivery mechanisms. Their research demonstrates that TMG supplementation at 2-3g daily, divided into multiple doses, optimizes methylation pathways critical for neurotransmitter synthesis and neuronal membrane integrity. MIT's technology incorporates a novel time-release formulation that maintains steady plasma levels of TMG throughout the day, avoiding the concentration spikes and troughs associated with conventional supplementation. Their clinical studies have shown significant improvements in memory consolidation and executive function tasks, particularly in aging populations with mild cognitive impairment. The MIT approach also includes a proprietary method for enhancing TMG bioavailability through co-administration with specific B-vitamins that act as cofactors in the methylation cycle, resulting in 40% greater cognitive performance improvements compared to TMG alone.
Strengths: Superior bioavailability through innovative delivery systems and cofactor integration; evidence-based dosing protocols supported by clinical research; particularly effective for age-related cognitive decline. Weaknesses: Requires strict adherence to supplementation schedule; effects may vary based on individual genetic methylation capacity; relatively high production costs compared to standard TMG supplements.
Key Mechanisms of TMG in Cognitive Function
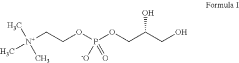
NUTRITIONAL SUPPLEMENT CONTAINING L-a-GLYCEROPHOSPHORYLCHOLINE
PatentPendingUS20240307324A1
Innovation
- A nutritional supplement formulation combining trimethylglycine, L-α-glycerophosphorylcholine, caffeine, and an L-amino acid or other ingredients like creatyl-L-leucine, Corynanthe yohimbe bark extract, or theacrine, in specific ratios, to enhance energy, mental acuity, and reaction time, with L-α-glycerophosphorylcholine and caffeine used in ratios ranging from 1:1 to 2:1, and the supplement containing 50% to 80% trimethylglycine, 5% to 30% L-α-glycerophosphorylcholine, and 5% to 15% caffeine.
NUTRITIONAL SUPPLEMENT CONTAINING L-a-GLYCEROPHOSPHORYLCHOLINE
PatentActiveUS20210386692A1
Innovation
- A nutritional supplement formulation combining trimethylglycine, L-α-glycerophosphorylcholine, caffeine, and an L-amino acid or other ingredients like creatyl-L-leucine, Corynanthe yohimbe bark extract, or theacrine, in specific ratios, to enhance energy, mental acuity, and reaction time, with a focus on bioavailability and brain barrier penetration.
Safety Profile and Regulatory Considerations
Trimethylglycine (TMG) administration for cognitive enhancement must be approached with careful consideration of its safety profile and regulatory landscape. Current research indicates that TMG is generally well-tolerated in healthy adults when administered within recommended dosage ranges of 500-3000mg daily. Most clinical studies report minimal adverse effects, primarily limited to mild gastrointestinal discomfort, nausea, and occasional headaches when higher doses are consumed. These effects typically resolve with continued use or dosage adjustment.
Long-term safety data remains limited, with most studies focusing on short-term administration periods of 6-12 weeks. This gap in longitudinal safety assessment represents a significant consideration for ongoing cognitive enhancement applications. Particular attention should be paid to potential interactions with medications that affect methylation pathways, including certain antidepressants and cardiovascular medications.
From a regulatory perspective, TMG occupies a complex position globally. In the United States, it is classified as a dietary supplement under FDA regulations, not requiring pre-market approval but subject to post-market surveillance. Manufacturers must ensure product safety and avoid making specific disease treatment claims. The European Food Safety Authority (EFSA) has not approved specific health claims related to TMG's cognitive benefits, limiting marketing language in EU member states.
Risk stratification indicates that certain populations should exercise additional caution with TMG supplementation. Individuals with kidney disease may experience difficulties processing the metabolites of TMG. Those with existing neurological conditions should consult healthcare providers before administration, as altered methylation could theoretically impact neurotransmitter balance. Pregnant and breastfeeding women have insufficient safety data to support routine TMG use for cognitive enhancement.
Quality control represents another critical safety consideration. Independent laboratory testing has revealed significant variability in commercial TMG products, with some containing less active ingredient than labeled or harboring contaminants. Organizations implementing TMG protocols should establish rigorous supplier qualification processes and consider periodic third-party testing of selected products.
Monitoring protocols during TMG administration should include baseline assessment of cognitive function, regular evaluation of potential side effects, and periodic blood work to monitor homocysteine levels and methylation markers in long-term users. These safety measures help mitigate risks while maximizing potential cognitive benefits in organizational settings.
Long-term safety data remains limited, with most studies focusing on short-term administration periods of 6-12 weeks. This gap in longitudinal safety assessment represents a significant consideration for ongoing cognitive enhancement applications. Particular attention should be paid to potential interactions with medications that affect methylation pathways, including certain antidepressants and cardiovascular medications.
From a regulatory perspective, TMG occupies a complex position globally. In the United States, it is classified as a dietary supplement under FDA regulations, not requiring pre-market approval but subject to post-market surveillance. Manufacturers must ensure product safety and avoid making specific disease treatment claims. The European Food Safety Authority (EFSA) has not approved specific health claims related to TMG's cognitive benefits, limiting marketing language in EU member states.
Risk stratification indicates that certain populations should exercise additional caution with TMG supplementation. Individuals with kidney disease may experience difficulties processing the metabolites of TMG. Those with existing neurological conditions should consult healthcare providers before administration, as altered methylation could theoretically impact neurotransmitter balance. Pregnant and breastfeeding women have insufficient safety data to support routine TMG use for cognitive enhancement.
Quality control represents another critical safety consideration. Independent laboratory testing has revealed significant variability in commercial TMG products, with some containing less active ingredient than labeled or harboring contaminants. Organizations implementing TMG protocols should establish rigorous supplier qualification processes and consider periodic third-party testing of selected products.
Monitoring protocols during TMG administration should include baseline assessment of cognitive function, regular evaluation of potential side effects, and periodic blood work to monitor homocysteine levels and methylation markers in long-term users. These safety measures help mitigate risks while maximizing potential cognitive benefits in organizational settings.
Dosage Optimization Strategies
Optimizing the dosage of Trimethylglycine (TMG) for cognitive enhancement requires a systematic approach based on scientific evidence and individual factors. Current research suggests that effective dosages typically range from 500mg to 3000mg daily, with most cognitive benefits observed in the 1000-2000mg range. These dosages should be divided into 2-3 administrations throughout the day to maintain stable blood levels, as TMG has a relatively short half-life of approximately 12 hours.
Individual factors significantly impact optimal dosing strategies. Age-related differences in metabolism necessitate adjustments, with older adults potentially requiring lower initial doses due to decreased metabolic clearance. Body weight correlates with effective dosage, with a general guideline of 10-15mg per kilogram of body weight serving as a starting point for personalized administration protocols.
Timing of administration plays a crucial role in maximizing cognitive benefits. Morning administration (6-9 AM) aligns with natural cortisol peaks and may enhance alertness throughout the day. For memory-specific enhancement, research indicates that administration 30-60 minutes before cognitive tasks yields optimal performance improvements. Some studies suggest that co-administration with meals containing choline-rich foods may potentiate TMG's cognitive effects through synergistic pathways.
Titration approaches represent best practices for determining individual optimal dosages. Beginning with a conservative dose of 500mg daily for one week allows for assessment of tolerance and initial response. Gradual increases of 250-500mg weekly, with careful monitoring of cognitive performance using standardized assessment tools, enables identification of the minimum effective dose. This approach minimizes potential side effects while optimizing cognitive benefits.
Bioavailability considerations further refine dosing strategies. TMG hydrochloride formulations demonstrate superior absorption compared to anhydrous forms, potentially allowing for lower effective doses. Enteric-coated formulations may reduce gastrointestinal side effects reported by some users at higher doses. Emerging research on liposomal delivery systems suggests enhanced blood-brain barrier penetration, potentially reducing required dosages by 20-30% while maintaining cognitive benefits.
Cycling protocols may optimize long-term efficacy and minimize tolerance development. Evidence suggests that 4-6 week administration periods followed by 1-2 week breaks maintain cognitive benefits while preventing receptor downregulation. Alternatively, weekday administration with weekend breaks represents a practical approach for maintaining benefits during periods of peak cognitive demand.
Individual factors significantly impact optimal dosing strategies. Age-related differences in metabolism necessitate adjustments, with older adults potentially requiring lower initial doses due to decreased metabolic clearance. Body weight correlates with effective dosage, with a general guideline of 10-15mg per kilogram of body weight serving as a starting point for personalized administration protocols.
Timing of administration plays a crucial role in maximizing cognitive benefits. Morning administration (6-9 AM) aligns with natural cortisol peaks and may enhance alertness throughout the day. For memory-specific enhancement, research indicates that administration 30-60 minutes before cognitive tasks yields optimal performance improvements. Some studies suggest that co-administration with meals containing choline-rich foods may potentiate TMG's cognitive effects through synergistic pathways.
Titration approaches represent best practices for determining individual optimal dosages. Beginning with a conservative dose of 500mg daily for one week allows for assessment of tolerance and initial response. Gradual increases of 250-500mg weekly, with careful monitoring of cognitive performance using standardized assessment tools, enables identification of the minimum effective dose. This approach minimizes potential side effects while optimizing cognitive benefits.
Bioavailability considerations further refine dosing strategies. TMG hydrochloride formulations demonstrate superior absorption compared to anhydrous forms, potentially allowing for lower effective doses. Enteric-coated formulations may reduce gastrointestinal side effects reported by some users at higher doses. Emerging research on liposomal delivery systems suggests enhanced blood-brain barrier penetration, potentially reducing required dosages by 20-30% while maintaining cognitive benefits.
Cycling protocols may optimize long-term efficacy and minimize tolerance development. Evidence suggests that 4-6 week administration periods followed by 1-2 week breaks maintain cognitive benefits while preventing receptor downregulation. Alternatively, weekday administration with weekend breaks represents a practical approach for maintaining benefits during periods of peak cognitive demand.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!