How to Design Nanoparticles for Enhanced Cellular Uptake
FEB 26, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Nanoparticle Design Background and Cellular Uptake Goals
Nanoparticle-mediated drug delivery has emerged as a transformative approach in modern medicine, addressing fundamental limitations of conventional therapeutic strategies. Traditional drug delivery systems often suffer from poor bioavailability, non-specific distribution, and limited therapeutic efficacy due to biological barriers that prevent drugs from reaching their intended cellular targets. The development of engineered nanoparticles represents a paradigm shift toward precision medicine, offering unprecedented control over drug pharmacokinetics and biodistribution.
The evolution of nanoparticle technology spans several decades, beginning with early liposomal formulations in the 1970s and progressing through successive generations of increasingly sophisticated delivery platforms. First-generation nanoparticles focused primarily on passive targeting through enhanced permeability and retention effects, while second-generation systems incorporated stealth properties to evade immune recognition. Current third-generation nanoparticles integrate active targeting mechanisms, stimuli-responsive behaviors, and multifunctional capabilities that enable precise cellular engagement.
Contemporary nanoparticle design has evolved from simple drug encapsulation vehicles to complex, intelligent systems capable of navigating biological environments with remarkable precision. This evolution reflects growing understanding of cellular biology, surface chemistry, and the intricate relationships between nanoparticle physicochemical properties and biological interactions. Modern approaches emphasize rational design principles that consider particle size, surface charge, morphology, and functionalization strategies as critical determinants of cellular uptake efficiency.
The primary objective of enhanced cellular uptake design centers on maximizing therapeutic payload delivery while minimizing off-target effects and systemic toxicity. This goal encompasses multiple interconnected targets: optimizing particle-cell interactions through surface engineering, overcoming cellular barriers via endocytic pathway manipulation, and achieving sustained intracellular drug release through controlled degradation mechanisms. Success requires balancing competing factors such as circulation stability versus cellular recognition, payload capacity versus targeting specificity, and manufacturing scalability versus design complexity.
Advanced nanoparticle systems aim to achieve selective cellular internalization through biomimetic approaches that exploit natural cellular processes. These objectives include developing particles that can distinguish between healthy and diseased cells, penetrate complex tissue architectures, and maintain therapeutic activity throughout the delivery process. The ultimate goal extends beyond simple drug delivery to encompass programmable therapeutic platforms capable of responding dynamically to cellular microenvironments and delivering personalized treatment regimens.
The evolution of nanoparticle technology spans several decades, beginning with early liposomal formulations in the 1970s and progressing through successive generations of increasingly sophisticated delivery platforms. First-generation nanoparticles focused primarily on passive targeting through enhanced permeability and retention effects, while second-generation systems incorporated stealth properties to evade immune recognition. Current third-generation nanoparticles integrate active targeting mechanisms, stimuli-responsive behaviors, and multifunctional capabilities that enable precise cellular engagement.
Contemporary nanoparticle design has evolved from simple drug encapsulation vehicles to complex, intelligent systems capable of navigating biological environments with remarkable precision. This evolution reflects growing understanding of cellular biology, surface chemistry, and the intricate relationships between nanoparticle physicochemical properties and biological interactions. Modern approaches emphasize rational design principles that consider particle size, surface charge, morphology, and functionalization strategies as critical determinants of cellular uptake efficiency.
The primary objective of enhanced cellular uptake design centers on maximizing therapeutic payload delivery while minimizing off-target effects and systemic toxicity. This goal encompasses multiple interconnected targets: optimizing particle-cell interactions through surface engineering, overcoming cellular barriers via endocytic pathway manipulation, and achieving sustained intracellular drug release through controlled degradation mechanisms. Success requires balancing competing factors such as circulation stability versus cellular recognition, payload capacity versus targeting specificity, and manufacturing scalability versus design complexity.
Advanced nanoparticle systems aim to achieve selective cellular internalization through biomimetic approaches that exploit natural cellular processes. These objectives include developing particles that can distinguish between healthy and diseased cells, penetrate complex tissue architectures, and maintain therapeutic activity throughout the delivery process. The ultimate goal extends beyond simple drug delivery to encompass programmable therapeutic platforms capable of responding dynamically to cellular microenvironments and delivering personalized treatment regimens.
Market Demand for Enhanced Nanoparticle Therapeutics
The global nanomedicine market has experienced unprecedented growth driven by the urgent need for more effective therapeutic delivery systems. Enhanced cellular uptake represents a critical bottleneck in current drug delivery approaches, where conventional pharmaceuticals often suffer from poor bioavailability, non-specific distribution, and limited therapeutic efficacy. The demand for nanoparticles with superior cellular internalization capabilities stems from the fundamental challenge of overcoming biological barriers that prevent therapeutic agents from reaching their intended targets.
Cancer therapeutics constitute the largest segment driving market demand for enhanced nanoparticle systems. Traditional chemotherapy agents face significant limitations including systemic toxicity, drug resistance, and inadequate tumor penetration. The pharmaceutical industry increasingly recognizes that nanoparticles engineered for enhanced cellular uptake can address these challenges by improving drug accumulation in tumor tissues while reducing off-target effects. This has created substantial market pull for advanced nanoparticle designs that can navigate complex biological environments and achieve efficient intracellular delivery.
Neurological disorders represent another high-value market segment where enhanced cellular uptake technologies are critically needed. The blood-brain barrier poses a formidable challenge for drug delivery to the central nervous system, creating significant unmet medical needs in treating conditions such as Alzheimer's disease, Parkinson's disease, and brain tumors. Pharmaceutical companies are actively seeking nanoparticle solutions that can cross these biological barriers and achieve meaningful therapeutic concentrations within target cells.
The emergence of personalized medicine and cell-based therapies has further amplified demand for sophisticated nanoparticle delivery systems. Gene therapy, RNA interference, and CRISPR-based treatments require precise intracellular delivery mechanisms to achieve therapeutic efficacy. These advanced therapeutic modalities cannot rely on passive diffusion and instead demand engineered nanoparticles capable of facilitating active cellular internalization processes.
Regulatory agencies have begun establishing clearer pathways for nanoparticle-based therapeutics, reducing market uncertainty and encouraging investment in enhanced delivery technologies. The success of approved nanomedicines has demonstrated commercial viability and created confidence among pharmaceutical companies to pursue more sophisticated nanoparticle designs. This regulatory clarity has translated into increased research funding and accelerated development timelines for next-generation nanoparticle therapeutics with enhanced cellular uptake capabilities.
Cancer therapeutics constitute the largest segment driving market demand for enhanced nanoparticle systems. Traditional chemotherapy agents face significant limitations including systemic toxicity, drug resistance, and inadequate tumor penetration. The pharmaceutical industry increasingly recognizes that nanoparticles engineered for enhanced cellular uptake can address these challenges by improving drug accumulation in tumor tissues while reducing off-target effects. This has created substantial market pull for advanced nanoparticle designs that can navigate complex biological environments and achieve efficient intracellular delivery.
Neurological disorders represent another high-value market segment where enhanced cellular uptake technologies are critically needed. The blood-brain barrier poses a formidable challenge for drug delivery to the central nervous system, creating significant unmet medical needs in treating conditions such as Alzheimer's disease, Parkinson's disease, and brain tumors. Pharmaceutical companies are actively seeking nanoparticle solutions that can cross these biological barriers and achieve meaningful therapeutic concentrations within target cells.
The emergence of personalized medicine and cell-based therapies has further amplified demand for sophisticated nanoparticle delivery systems. Gene therapy, RNA interference, and CRISPR-based treatments require precise intracellular delivery mechanisms to achieve therapeutic efficacy. These advanced therapeutic modalities cannot rely on passive diffusion and instead demand engineered nanoparticles capable of facilitating active cellular internalization processes.
Regulatory agencies have begun establishing clearer pathways for nanoparticle-based therapeutics, reducing market uncertainty and encouraging investment in enhanced delivery technologies. The success of approved nanomedicines has demonstrated commercial viability and created confidence among pharmaceutical companies to pursue more sophisticated nanoparticle designs. This regulatory clarity has translated into increased research funding and accelerated development timelines for next-generation nanoparticle therapeutics with enhanced cellular uptake capabilities.
Current State and Challenges in Nanoparticle Cellular Uptake
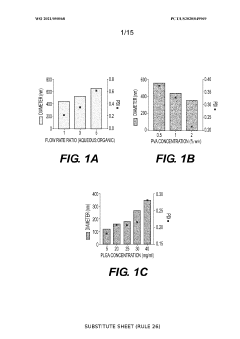
The field of nanoparticle-mediated drug delivery has witnessed remarkable progress over the past two decades, yet significant challenges persist in achieving optimal cellular uptake efficiency. Current nanoparticle systems demonstrate highly variable uptake rates across different cell types, with efficiency ranging from less than 1% to approximately 15% in most therapeutic applications. This substantial variation stems from the complex interplay between nanoparticle physicochemical properties and cellular membrane dynamics.
Size-dependent uptake mechanisms represent one of the most critical challenges in current nanoparticle design. Particles smaller than 50 nanometers typically undergo clathrin-mediated endocytosis, while larger particles may trigger macropinocytosis or phagocytosis. However, the optimal size window varies significantly among different cell types, creating difficulties in developing universally effective delivery systems. Additionally, particles often become trapped in endosomal compartments, limiting their therapeutic efficacy.
Surface functionalization remains a double-edged sword in nanoparticle development. While positive surface charges generally enhance cellular interaction through electrostatic attraction to negatively charged cell membranes, they also increase cytotoxicity and trigger rapid clearance by the immune system. Conversely, neutral or negatively charged particles show improved biocompatibility but often exhibit reduced cellular uptake rates. The challenge lies in achieving the delicate balance between uptake efficiency and biocompatibility.
Protein corona formation presents another significant obstacle that current technologies struggle to address effectively. Upon exposure to biological fluids, nanoparticles rapidly acquire a protein corona that fundamentally alters their surface properties and cellular recognition patterns. This phenomenon often negates carefully engineered surface modifications, leading to unpredictable in vivo behavior that differs substantially from in vitro observations.
Current targeting strategies, including passive and active targeting approaches, face substantial limitations in clinical translation. Passive targeting through the enhanced permeability and retention effect shows inconsistent results across different tumor types and patient populations. Active targeting using ligand-receptor interactions, while promising in laboratory settings, often fails to achieve expected specificity in complex biological environments due to receptor heterogeneity and competitive binding.
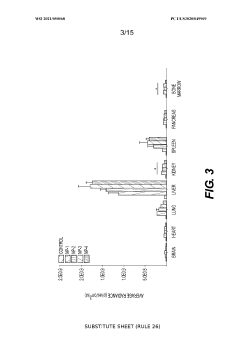
The heterogeneity of cellular uptake mechanisms across different tissues and disease states poses additional challenges for developing standardized nanoparticle formulations. Cancer cells, for instance, may exhibit altered endocytic pathways compared to healthy cells, requiring disease-specific optimization strategies that complicate universal therapeutic applications.
Size-dependent uptake mechanisms represent one of the most critical challenges in current nanoparticle design. Particles smaller than 50 nanometers typically undergo clathrin-mediated endocytosis, while larger particles may trigger macropinocytosis or phagocytosis. However, the optimal size window varies significantly among different cell types, creating difficulties in developing universally effective delivery systems. Additionally, particles often become trapped in endosomal compartments, limiting their therapeutic efficacy.
Surface functionalization remains a double-edged sword in nanoparticle development. While positive surface charges generally enhance cellular interaction through electrostatic attraction to negatively charged cell membranes, they also increase cytotoxicity and trigger rapid clearance by the immune system. Conversely, neutral or negatively charged particles show improved biocompatibility but often exhibit reduced cellular uptake rates. The challenge lies in achieving the delicate balance between uptake efficiency and biocompatibility.
Protein corona formation presents another significant obstacle that current technologies struggle to address effectively. Upon exposure to biological fluids, nanoparticles rapidly acquire a protein corona that fundamentally alters their surface properties and cellular recognition patterns. This phenomenon often negates carefully engineered surface modifications, leading to unpredictable in vivo behavior that differs substantially from in vitro observations.
Current targeting strategies, including passive and active targeting approaches, face substantial limitations in clinical translation. Passive targeting through the enhanced permeability and retention effect shows inconsistent results across different tumor types and patient populations. Active targeting using ligand-receptor interactions, while promising in laboratory settings, often fails to achieve expected specificity in complex biological environments due to receptor heterogeneity and competitive binding.
The heterogeneity of cellular uptake mechanisms across different tissues and disease states poses additional challenges for developing standardized nanoparticle formulations. Cancer cells, for instance, may exhibit altered endocytic pathways compared to healthy cells, requiring disease-specific optimization strategies that complicate universal therapeutic applications.
Existing Solutions for Improving Cellular Uptake Efficiency
01 Surface modification of nanoparticles to enhance cellular uptake
Nanoparticles can be surface-modified with various functional groups, ligands, or coatings to improve their interaction with cell membranes and enhance cellular uptake. Surface modifications such as PEGylation, targeting ligands, or charge modifications can increase biocompatibility and facilitate receptor-mediated endocytosis. These modifications help nanoparticles overcome biological barriers and improve their internalization efficiency into target cells.- Surface modification of nanoparticles to enhance cellular uptake: Nanoparticles can be surface-modified with various functional groups, ligands, or coatings to improve their interaction with cell membranes and enhance cellular uptake. Surface modifications such as PEGylation, targeting ligands, or charge modifications can increase biocompatibility and facilitate receptor-mediated endocytosis. These modifications help nanoparticles overcome biological barriers and improve their internalization efficiency into target cells.
- Size and shape optimization of nanoparticles for cellular internalization: The physical properties of nanoparticles, particularly their size and shape, significantly influence cellular uptake mechanisms. Smaller nanoparticles generally exhibit higher cellular uptake rates compared to larger particles. Spherical, rod-shaped, or other geometric configurations can affect the interaction with cell membranes and the pathway of internalization. Optimizing these parameters can enhance the efficiency of nanoparticle delivery into cells.
- Targeting ligand conjugation for receptor-mediated uptake: Conjugating specific targeting ligands to nanoparticles enables receptor-mediated endocytosis, which is a highly efficient cellular uptake mechanism. Ligands such as antibodies, peptides, or small molecules can bind to specific receptors overexpressed on target cells, facilitating selective internalization. This approach enhances the specificity and efficiency of nanoparticle delivery to particular cell types or tissues.
- Use of cell-penetrating peptides to facilitate nanoparticle entry: Cell-penetrating peptides can be incorporated into nanoparticle formulations to enhance their ability to cross cell membranes. These peptides interact with membrane components and facilitate translocation of nanoparticles into the cytoplasm through various mechanisms including direct penetration and endocytosis. This strategy improves the intracellular delivery of nanoparticles and their therapeutic or diagnostic cargo.
- Endosomal escape mechanisms for nanoparticle delivery: After cellular uptake through endocytosis, nanoparticles often become trapped in endosomes or lysosomes, limiting their therapeutic efficacy. Strategies to promote endosomal escape include pH-responsive materials, fusogenic peptides, or membrane-disrupting agents that facilitate the release of nanoparticles into the cytoplasm. These mechanisms ensure that the nanoparticle cargo reaches its intended intracellular target and maintains its biological activity.
02 Size and shape optimization of nanoparticles for cellular internalization
The physical characteristics of nanoparticles, particularly their size and shape, play a critical role in determining cellular uptake efficiency. Smaller nanoparticles generally exhibit better cellular penetration, while specific shapes can influence the mechanism of internalization. Optimization of these parameters can significantly enhance the rate and extent of nanoparticle uptake by cells through various endocytic pathways.Expand Specific Solutions03 Targeting ligand conjugation for receptor-mediated uptake
Conjugating specific targeting ligands to nanoparticles enables receptor-mediated endocytosis, which significantly enhances cellular uptake specificity and efficiency. These ligands can include antibodies, peptides, or small molecules that recognize and bind to receptors overexpressed on target cells. This approach allows for selective delivery of nanoparticles to specific cell types while minimizing off-target effects.Expand Specific Solutions04 Charge-based strategies for enhanced membrane interaction
The surface charge of nanoparticles significantly influences their interaction with negatively charged cell membranes. Positively charged nanoparticles typically exhibit enhanced cellular uptake through electrostatic interactions with the cell membrane. Charge optimization strategies, including the use of cationic polymers or lipids, can be employed to improve nanoparticle-cell membrane interactions and facilitate cellular internalization.Expand Specific Solutions05 Stimuli-responsive nanoparticles for controlled cellular uptake
Nanoparticles can be designed to respond to specific stimuli such as pH, temperature, or enzymatic activity to trigger or enhance cellular uptake. These stimuli-responsive systems can undergo conformational changes or release targeting moieties in response to the cellular microenvironment, thereby improving uptake efficiency. Such smart nanoparticle systems enable controlled and site-specific delivery with enhanced cellular internalization at target locations.Expand Specific Solutions
Key Players in Nanoparticle and Drug Delivery Industry
The nanoparticle design field for enhanced cellular uptake represents a rapidly evolving sector characterized by significant academic-industry collaboration and advancing technological maturity. The market demonstrates substantial growth potential driven by pharmaceutical and biomedical applications, with major research contributions from leading institutions including MIT, Yale University, Northwestern University, and Zhejiang University establishing foundational technologies. Commercial players like Nanosys Inc., NanoPharmaceuticals LLC, ModernaTX Inc., and Nanovis LLC are translating academic breakthroughs into viable products, while organizations such as Fraunhofer-Gesellschaft and CNRS provide crucial applied research infrastructure. The technology has progressed beyond proof-of-concept stages, with several companies achieving market deployment in specific applications, indicating a maturing competitive landscape where both established pharmaceutical companies and specialized nanotechnology firms compete for market share in this high-potential therapeutic delivery sector.
Nanosys, Inc.
Technical Solution: Nanosys has developed quantum dot-based nanoparticles with engineered surface properties for enhanced cellular uptake in imaging and diagnostic applications. Their technology utilizes semiconductor nanocrystals with precisely controlled surface functionalization to optimize cellular internalization. The company's approach involves coating quantum dots with biocompatible polymers and targeting ligands that facilitate receptor-mediated endocytosis. Their nanoparticles are designed with tunable optical properties and surface chemistry modifications that enable both enhanced cellular uptake and real-time tracking of internalization processes. The technology demonstrates superior cellular uptake efficiency compared to conventional fluorescent markers, with applications in cellular imaging, diagnostics, and theranostics.
Strengths: Established commercial presence in nanotechnology, expertise in quantum dot manufacturing, strong intellectual property portfolio. Weaknesses: Primarily focused on imaging applications, potential toxicity concerns with quantum dot materials limiting therapeutic applications.
The Regents of the University of California
Technical Solution: UC system has developed innovative nanoparticle designs incorporating biomimetic approaches for enhanced cellular uptake. Their technology focuses on cell membrane-coated nanoparticles that leverage natural cellular recognition mechanisms to improve internalization. The research includes development of hybrid nanoparticles combining synthetic cores with natural cell membrane coatings, achieving enhanced biocompatibility and reduced immunogenicity. Their approach utilizes red blood cell membranes, cancer cell membranes, and platelet membranes as coating materials, resulting in nanoparticles that can evade immune recognition while maintaining high cellular uptake efficiency. The technology demonstrates significant improvements in targeting specificity and cellular internalization rates, with applications spanning drug delivery, imaging, and therapeutic interventions.
Strengths: Innovative biomimetic approach, strong research foundation, diverse application potential across multiple therapeutic areas. Weaknesses: Early-stage technology requiring further development, complex manufacturing processes for membrane-coated particles.
Core Innovations in Nanoparticle Surface Engineering
Nanoparticles for selective tissue or cellular uptake
PatentPendingUS20220339294A1
Innovation
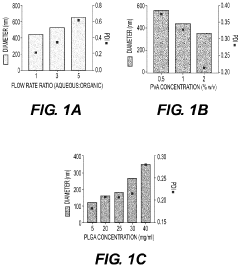
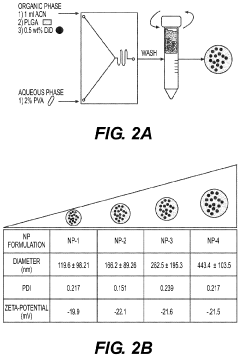
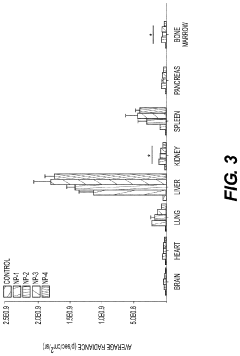
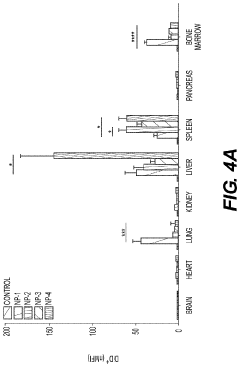
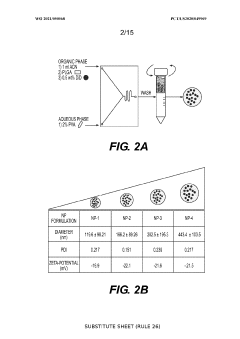
- Development of biodegradable polymeric nanoparticles with controlled sizes between 70 nm and 220 nm, manufactured using a microfluidic system, which are selectively taken up by lung cells and bone marrow cells without the need for targeting agents, allowing for the delivery of therapeutic, diagnostic, and prophylactic agents.
Nanoparticles for selective tissue or cellular uptake
PatentWO2021050568A1
Innovation
- Development of biodegradable polymeric nanoparticles with controlled sizes between 70 nm and 220 nm, manufactured using a microfluidic system, which allows for selective uptake by lung cells and bone marrow cells without the need for targeting agents, enabling the delivery of therapeutic, diagnostic, and prophylactic agents.
Regulatory Framework for Nanoparticle-Based Therapeutics
The regulatory landscape for nanoparticle-based therapeutics represents one of the most complex and evolving areas in pharmaceutical oversight. Unlike conventional drug formulations, nanoparticles present unique challenges due to their size-dependent properties, potential for altered biodistribution, and novel mechanisms of cellular interaction. Regulatory agencies worldwide have recognized that traditional assessment frameworks may be insufficient for evaluating these advanced therapeutic systems.
The United States Food and Drug Administration has established specific guidance documents addressing nanotechnology applications in drug products. The FDA's approach emphasizes the importance of characterizing physicochemical properties that may influence safety and efficacy, including particle size distribution, surface charge, morphology, and stability profiles. The agency requires comprehensive data on how nanoparticle design parameters affect cellular uptake mechanisms, as these directly impact therapeutic outcomes and potential adverse effects.
European regulatory authorities through the European Medicines Agency have developed parallel frameworks that focus heavily on quality-by-design principles for nanomedicines. The EMA guidelines emphasize the critical relationship between nanoparticle design features and their biological behavior, requiring detailed documentation of how surface modifications, targeting ligands, and size optimization strategies influence cellular internalization pathways.
International harmonization efforts have emerged through organizations such as the International Council for Harmonisation, which has begun developing unified standards for nanoparticle characterization and safety assessment. These initiatives recognize that enhanced cellular uptake, while therapeutically beneficial, may also present unique safety considerations that require specialized evaluation protocols.
Manufacturing quality control represents another crucial regulatory dimension, as nanoparticle production must demonstrate consistent control over parameters that directly influence cellular uptake efficiency. Regulatory frameworks now require real-time monitoring of critical quality attributes throughout the manufacturing process, ensuring that batch-to-batch variations do not compromise the designed cellular interaction profiles.
The regulatory pathway for nanoparticle therapeutics continues to evolve as scientific understanding advances, with agencies increasingly requiring mechanistic data linking design features to cellular uptake performance and clinical outcomes.
The United States Food and Drug Administration has established specific guidance documents addressing nanotechnology applications in drug products. The FDA's approach emphasizes the importance of characterizing physicochemical properties that may influence safety and efficacy, including particle size distribution, surface charge, morphology, and stability profiles. The agency requires comprehensive data on how nanoparticle design parameters affect cellular uptake mechanisms, as these directly impact therapeutic outcomes and potential adverse effects.
European regulatory authorities through the European Medicines Agency have developed parallel frameworks that focus heavily on quality-by-design principles for nanomedicines. The EMA guidelines emphasize the critical relationship between nanoparticle design features and their biological behavior, requiring detailed documentation of how surface modifications, targeting ligands, and size optimization strategies influence cellular internalization pathways.
International harmonization efforts have emerged through organizations such as the International Council for Harmonisation, which has begun developing unified standards for nanoparticle characterization and safety assessment. These initiatives recognize that enhanced cellular uptake, while therapeutically beneficial, may also present unique safety considerations that require specialized evaluation protocols.
Manufacturing quality control represents another crucial regulatory dimension, as nanoparticle production must demonstrate consistent control over parameters that directly influence cellular uptake efficiency. Regulatory frameworks now require real-time monitoring of critical quality attributes throughout the manufacturing process, ensuring that batch-to-batch variations do not compromise the designed cellular interaction profiles.
The regulatory pathway for nanoparticle therapeutics continues to evolve as scientific understanding advances, with agencies increasingly requiring mechanistic data linking design features to cellular uptake performance and clinical outcomes.
Safety Assessment and Biocompatibility of Engineered Nanoparticles
The safety assessment and biocompatibility evaluation of engineered nanoparticles designed for enhanced cellular uptake represents a critical regulatory and scientific imperative that directly impacts their clinical translation potential. As nanoparticles are engineered with specific surface modifications, targeting ligands, and physicochemical properties to improve cellular internalization, these design modifications can significantly alter their biological interactions and toxicological profiles compared to conventional nanomaterials.
Comprehensive cytotoxicity evaluation forms the foundation of safety assessment, requiring systematic testing across multiple cell lines representative of target tissues and potential off-target organs. Standard viability assays including MTT, WST-1, and LDH release tests must be complemented by more sophisticated analyses such as flow cytometry-based apoptosis detection and real-time cellular impedance monitoring to capture both acute and chronic toxicity responses. The enhanced uptake characteristics of these engineered nanoparticles necessitate particular attention to intracellular accumulation patterns and potential organelle-specific damage.
Hemocompatibility assessment becomes increasingly complex when nanoparticles are designed with active targeting mechanisms or surface charge modifications that enhance cellular uptake. Hemolysis testing, platelet aggregation studies, and complement activation assays must account for the dynamic nature of protein corona formation on engineered surfaces. The interaction between targeting ligands and blood components can lead to unexpected immunological responses or altered pharmacokinetic profiles that differ substantially from non-targeted formulations.
Genotoxicity evaluation requires specialized consideration for uptake-enhanced nanoparticles due to their increased intracellular bioavailability and potential nuclear translocation. Standard Ames testing must be supplemented with mammalian cell-based assays including micronucleus formation, chromosomal aberration analysis, and DNA damage response pathway activation studies. The enhanced cellular penetration capabilities may result in increased genetic material exposure, necessitating more stringent safety thresholds.
Long-term biocompatibility studies must address the fate of accumulated nanoparticles within cells and tissues, particularly focusing on biodegradation pathways, clearance mechanisms, and potential for chronic inflammatory responses. The engineered surface modifications that enhance uptake may also influence biodistribution patterns and elimination kinetics, requiring extended observation periods and comprehensive histopathological evaluation to ensure acceptable safety profiles for clinical applications.
Comprehensive cytotoxicity evaluation forms the foundation of safety assessment, requiring systematic testing across multiple cell lines representative of target tissues and potential off-target organs. Standard viability assays including MTT, WST-1, and LDH release tests must be complemented by more sophisticated analyses such as flow cytometry-based apoptosis detection and real-time cellular impedance monitoring to capture both acute and chronic toxicity responses. The enhanced uptake characteristics of these engineered nanoparticles necessitate particular attention to intracellular accumulation patterns and potential organelle-specific damage.
Hemocompatibility assessment becomes increasingly complex when nanoparticles are designed with active targeting mechanisms or surface charge modifications that enhance cellular uptake. Hemolysis testing, platelet aggregation studies, and complement activation assays must account for the dynamic nature of protein corona formation on engineered surfaces. The interaction between targeting ligands and blood components can lead to unexpected immunological responses or altered pharmacokinetic profiles that differ substantially from non-targeted formulations.
Genotoxicity evaluation requires specialized consideration for uptake-enhanced nanoparticles due to their increased intracellular bioavailability and potential nuclear translocation. Standard Ames testing must be supplemented with mammalian cell-based assays including micronucleus formation, chromosomal aberration analysis, and DNA damage response pathway activation studies. The enhanced cellular penetration capabilities may result in increased genetic material exposure, necessitating more stringent safety thresholds.
Long-term biocompatibility studies must address the fate of accumulated nanoparticles within cells and tissues, particularly focusing on biodegradation pathways, clearance mechanisms, and potential for chronic inflammatory responses. The engineered surface modifications that enhance uptake may also influence biodistribution patterns and elimination kinetics, requiring extended observation periods and comprehensive histopathological evaluation to ensure acceptable safety profiles for clinical applications.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!