How to Implement Lithium Nitride Solutions in Emerging Technology
AUG 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Lithium Nitride Technology Background and Objectives
Lithium nitride (Li₃N) represents a significant frontier in materials science, having evolved from a laboratory curiosity to a promising component in multiple emerging technologies. The compound was first synthesized in the late 19th century but remained largely unexplored until recent decades when its unique properties began attracting substantial research interest. Historically, lithium nitride's high ionic conductivity and ability to store hydrogen were recognized, but practical applications remained limited due to production challenges and stability issues.
The technological evolution of lithium nitride has accelerated dramatically in the past decade, driven by the global push for sustainable energy solutions and advanced materials. Research has expanded from basic property studies to application-focused development, particularly in energy storage, semiconductor technology, and advanced ceramics. This progression reflects broader trends in materials science toward multi-functional compounds that can address complex technological challenges.
Current objectives in lithium nitride research and implementation center on several key areas. Primary among these is enhancing lithium-ion battery performance, where lithium nitride shows promise as a solid-state electrolyte material that could significantly improve safety and energy density compared to conventional liquid electrolytes. Additionally, researchers aim to develop lithium nitride-based hydrogen storage systems that could revolutionize clean energy infrastructure by providing efficient, high-capacity storage solutions.
Another critical objective involves improving manufacturing scalability and cost-effectiveness. Despite its promising properties, widespread implementation of lithium nitride technologies has been hindered by production challenges, including sensitivity to moisture and oxygen during synthesis and processing. Developing robust, economical manufacturing methods represents a crucial step toward commercial viability.
The semiconductor industry presents another frontier, with objectives focused on utilizing lithium nitride's unique electronic properties in next-generation devices. Research aims to integrate lithium nitride into semiconductor fabrication processes to enhance performance characteristics such as electron mobility and thermal management.
Looking forward, the technological trajectory suggests increasing convergence between lithium nitride research and other advanced materials, particularly in hybrid systems that combine lithium nitride with complementary compounds to achieve enhanced functionality. This trend aligns with broader movements toward materials by design, where compounds are engineered at the molecular level for specific applications.
The ultimate goal driving lithium nitride technology development is to transition from laboratory demonstrations to commercially viable products that address pressing technological challenges, particularly in energy storage, electronics, and sustainable manufacturing. Success in this domain could contribute significantly to addressing global challenges in energy efficiency and technological advancement.
The technological evolution of lithium nitride has accelerated dramatically in the past decade, driven by the global push for sustainable energy solutions and advanced materials. Research has expanded from basic property studies to application-focused development, particularly in energy storage, semiconductor technology, and advanced ceramics. This progression reflects broader trends in materials science toward multi-functional compounds that can address complex technological challenges.
Current objectives in lithium nitride research and implementation center on several key areas. Primary among these is enhancing lithium-ion battery performance, where lithium nitride shows promise as a solid-state electrolyte material that could significantly improve safety and energy density compared to conventional liquid electrolytes. Additionally, researchers aim to develop lithium nitride-based hydrogen storage systems that could revolutionize clean energy infrastructure by providing efficient, high-capacity storage solutions.
Another critical objective involves improving manufacturing scalability and cost-effectiveness. Despite its promising properties, widespread implementation of lithium nitride technologies has been hindered by production challenges, including sensitivity to moisture and oxygen during synthesis and processing. Developing robust, economical manufacturing methods represents a crucial step toward commercial viability.
The semiconductor industry presents another frontier, with objectives focused on utilizing lithium nitride's unique electronic properties in next-generation devices. Research aims to integrate lithium nitride into semiconductor fabrication processes to enhance performance characteristics such as electron mobility and thermal management.
Looking forward, the technological trajectory suggests increasing convergence between lithium nitride research and other advanced materials, particularly in hybrid systems that combine lithium nitride with complementary compounds to achieve enhanced functionality. This trend aligns with broader movements toward materials by design, where compounds are engineered at the molecular level for specific applications.
The ultimate goal driving lithium nitride technology development is to transition from laboratory demonstrations to commercially viable products that address pressing technological challenges, particularly in energy storage, electronics, and sustainable manufacturing. Success in this domain could contribute significantly to addressing global challenges in energy efficiency and technological advancement.
Market Analysis for Lithium Nitride Applications
The global market for lithium nitride applications is experiencing significant growth, driven by the increasing demand for advanced materials in emerging technologies. The market size for lithium nitride was valued at approximately $127 million in 2022 and is projected to reach $312 million by 2030, representing a compound annual growth rate (CAGR) of 11.8% during the forecast period.
Energy storage represents the largest application segment, accounting for nearly 42% of the market share. Lithium nitride's exceptional ionic conductivity properties make it an ideal candidate for solid-state batteries, which are gaining traction due to their enhanced safety profiles and higher energy densities compared to conventional lithium-ion batteries. Major automotive manufacturers and battery producers are investing heavily in this technology, with companies like Toyota, Samsung, and Solid Power leading research initiatives.
The electronics sector constitutes the second-largest market segment at 28%, where lithium nitride is utilized in semiconductor manufacturing and as a component in advanced electronic devices. The material's unique properties enable the development of smaller, more efficient electronic components with improved thermal management capabilities.
Geographically, Asia-Pacific dominates the market with a 45% share, primarily due to the strong presence of electronics manufacturing and battery production facilities in countries like China, Japan, and South Korea. North America follows with 30% market share, driven by substantial investments in clean energy technologies and advanced materials research.
Market penetration challenges include high production costs, with lithium nitride synthesis requiring specialized equipment and precise processing conditions. Current production costs average $850-1,200 per kilogram, significantly higher than alternative materials. However, economies of scale and improved manufacturing processes are expected to reduce costs by 30-40% over the next five years.
Customer adoption barriers include limited awareness of lithium nitride's benefits among potential end-users and concerns regarding long-term stability in various applications. Industry surveys indicate that only 22% of potential industrial users are familiar with lithium nitride's full range of applications.
Market growth opportunities are particularly strong in emerging applications such as quantum computing, where lithium nitride's unique electronic properties show promise for developing next-generation quantum bits. Additionally, the material's potential in hydrogen storage systems represents a significant untapped market, especially as hydrogen economies develop globally.
Energy storage represents the largest application segment, accounting for nearly 42% of the market share. Lithium nitride's exceptional ionic conductivity properties make it an ideal candidate for solid-state batteries, which are gaining traction due to their enhanced safety profiles and higher energy densities compared to conventional lithium-ion batteries. Major automotive manufacturers and battery producers are investing heavily in this technology, with companies like Toyota, Samsung, and Solid Power leading research initiatives.
The electronics sector constitutes the second-largest market segment at 28%, where lithium nitride is utilized in semiconductor manufacturing and as a component in advanced electronic devices. The material's unique properties enable the development of smaller, more efficient electronic components with improved thermal management capabilities.
Geographically, Asia-Pacific dominates the market with a 45% share, primarily due to the strong presence of electronics manufacturing and battery production facilities in countries like China, Japan, and South Korea. North America follows with 30% market share, driven by substantial investments in clean energy technologies and advanced materials research.
Market penetration challenges include high production costs, with lithium nitride synthesis requiring specialized equipment and precise processing conditions. Current production costs average $850-1,200 per kilogram, significantly higher than alternative materials. However, economies of scale and improved manufacturing processes are expected to reduce costs by 30-40% over the next five years.
Customer adoption barriers include limited awareness of lithium nitride's benefits among potential end-users and concerns regarding long-term stability in various applications. Industry surveys indicate that only 22% of potential industrial users are familiar with lithium nitride's full range of applications.
Market growth opportunities are particularly strong in emerging applications such as quantum computing, where lithium nitride's unique electronic properties show promise for developing next-generation quantum bits. Additionally, the material's potential in hydrogen storage systems represents a significant untapped market, especially as hydrogen economies develop globally.
Current State and Technical Challenges of Lithium Nitride
Lithium nitride (Li₃N) has emerged as a promising material in various technological applications, yet its current state of development presents both opportunities and significant challenges. Globally, research on lithium nitride has intensified over the past decade, with major advancements occurring primarily in North America, Europe, and East Asia. The United States, Japan, China, and Germany lead in patent filings related to lithium nitride technologies, indicating concentrated innovation hubs.
The synthesis of high-purity lithium nitride remains one of the foremost technical challenges. Traditional methods involve direct reaction of lithium metal with nitrogen gas at elevated temperatures (approximately 400-800°C), which presents safety concerns due to lithium's high reactivity. Alternative synthesis routes, including plasma-assisted deposition and mechanochemical methods, have been developed but face scalability limitations for industrial production.
Stability issues constitute another significant barrier to widespread implementation. Lithium nitride is highly reactive with moisture and oxygen, forming lithium hydroxide and ammonia upon exposure to air. This necessitates stringent handling protocols and specialized packaging solutions, substantially increasing manufacturing complexity and costs. Current encapsulation technologies provide only limited protection against environmental degradation over extended periods.
Ion conductivity optimization represents a critical technical challenge for lithium nitride's application in solid-state batteries. While lithium nitride exhibits one of the highest lithium-ion conductivities among solid electrolytes (approximately 10⁻³ S/cm at room temperature), interfacial resistance issues at electrode-electrolyte boundaries significantly reduce overall battery performance. Research efforts are focused on developing composite materials and interface engineering solutions to address these limitations.
Manufacturing scalability presents perhaps the most significant obstacle to commercial viability. Current production methods are predominantly laboratory-scale, with limited throughput and high production costs. The transition to industrial-scale manufacturing requires substantial process engineering innovations to maintain material quality while achieving economic viability.
Material compatibility challenges also persist when integrating lithium nitride with existing technologies. Its high reactivity with common battery components, including cathode materials and current collectors, necessitates the development of specialized interface layers and compatible materials systems.
Regulatory and safety considerations further complicate implementation efforts. The reactive nature of lithium nitride raises concerns regarding transportation, storage, and disposal protocols. Comprehensive safety standards specific to lithium nitride applications are still evolving, creating uncertainty for commercial deployment.
The synthesis of high-purity lithium nitride remains one of the foremost technical challenges. Traditional methods involve direct reaction of lithium metal with nitrogen gas at elevated temperatures (approximately 400-800°C), which presents safety concerns due to lithium's high reactivity. Alternative synthesis routes, including plasma-assisted deposition and mechanochemical methods, have been developed but face scalability limitations for industrial production.
Stability issues constitute another significant barrier to widespread implementation. Lithium nitride is highly reactive with moisture and oxygen, forming lithium hydroxide and ammonia upon exposure to air. This necessitates stringent handling protocols and specialized packaging solutions, substantially increasing manufacturing complexity and costs. Current encapsulation technologies provide only limited protection against environmental degradation over extended periods.
Ion conductivity optimization represents a critical technical challenge for lithium nitride's application in solid-state batteries. While lithium nitride exhibits one of the highest lithium-ion conductivities among solid electrolytes (approximately 10⁻³ S/cm at room temperature), interfacial resistance issues at electrode-electrolyte boundaries significantly reduce overall battery performance. Research efforts are focused on developing composite materials and interface engineering solutions to address these limitations.
Manufacturing scalability presents perhaps the most significant obstacle to commercial viability. Current production methods are predominantly laboratory-scale, with limited throughput and high production costs. The transition to industrial-scale manufacturing requires substantial process engineering innovations to maintain material quality while achieving economic viability.
Material compatibility challenges also persist when integrating lithium nitride with existing technologies. Its high reactivity with common battery components, including cathode materials and current collectors, necessitates the development of specialized interface layers and compatible materials systems.
Regulatory and safety considerations further complicate implementation efforts. The reactive nature of lithium nitride raises concerns regarding transportation, storage, and disposal protocols. Comprehensive safety standards specific to lithium nitride applications are still evolving, creating uncertainty for commercial deployment.
Current Implementation Solutions for Lithium Nitride
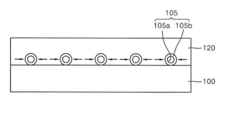
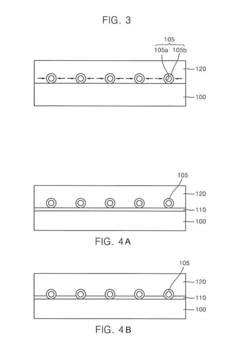
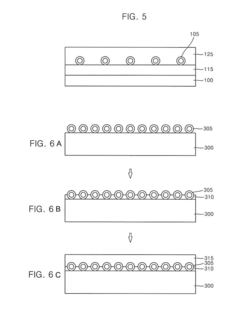
01 Synthesis and preparation methods of lithium nitride
Various methods for synthesizing lithium nitride have been developed, including direct reaction of lithium with nitrogen gas, plasma-assisted processes, and chemical vapor deposition techniques. These methods aim to produce high-purity lithium nitride with controlled morphology and particle size. The synthesis conditions, such as temperature, pressure, and reaction time, significantly influence the properties of the resulting lithium nitride material.- Synthesis and preparation methods of lithium nitride: Various methods for synthesizing lithium nitride have been developed, including direct reaction of lithium with nitrogen gas, plasma-assisted processes, and chemical vapor deposition techniques. These methods aim to control the purity, particle size, and crystallinity of the resulting lithium nitride, which is important for its applications in energy storage and other fields. The synthesis conditions, such as temperature, pressure, and reaction time, significantly affect the properties of the final product.
- Lithium nitride as solid electrolyte material: Lithium nitride serves as an effective solid electrolyte material in lithium batteries due to its high ionic conductivity. When used in solid-state batteries, it facilitates lithium ion transport while preventing dendrite formation. The material can be modified or combined with other compounds to enhance its stability and conductivity properties, making it valuable for next-generation energy storage technologies that require safer and more efficient electrolytes.
- Lithium nitride in electrode materials: Lithium nitride is utilized in the development of advanced electrode materials for lithium-ion batteries. It can be incorporated into anode compositions to improve capacity, cycling stability, and rate capability. The nitrogen-rich environment created by lithium nitride helps to stabilize the electrode structure during charging and discharging cycles, reducing capacity fade and extending battery life. Various composite materials combining lithium nitride with carbon or metal oxides have shown promising electrochemical performance.
- Protective coatings and surface treatments using lithium nitride: Lithium nitride can be applied as protective coatings on various materials, including metals and semiconductors. These coatings provide protection against oxidation, corrosion, and other forms of degradation. The deposition of lithium nitride films can be achieved through techniques such as sputtering, chemical vapor deposition, or atomic layer deposition. The resulting coatings exhibit good adhesion, uniformity, and protective properties, making them valuable for extending the lifetime of sensitive components.
- Lithium nitride in hydrogen storage applications: Lithium nitride demonstrates potential as a hydrogen storage material due to its ability to form lithium imide and lithium amide upon reaction with hydrogen. This reversible reaction allows for the storage and release of hydrogen under moderate conditions, making it attractive for clean energy applications. The hydrogen storage capacity, kinetics, and cycling stability of lithium nitride-based materials can be enhanced through various strategies, including nanostructuring, catalyst addition, and compositional modifications.
02 Applications in battery technology
Lithium nitride serves as an important material in advanced battery technologies, particularly as a solid electrolyte or electrode component in lithium-ion batteries. It offers high ionic conductivity, which facilitates lithium ion transport within battery systems. Additionally, lithium nitride can be used as a precursor for creating protective layers on electrode surfaces, enhancing battery performance, cycle life, and safety characteristics.Expand Specific Solutions03 Lithium nitride in semiconductor manufacturing
Lithium nitride finds applications in semiconductor manufacturing processes, particularly as a material for thin film deposition. It can be used in the fabrication of electronic devices, serving as a dielectric layer or as a component in compound semiconductor structures. The unique properties of lithium nitride, including its electronic band structure and thermal stability, make it suitable for specific semiconductor applications.Expand Specific Solutions04 Lithium nitride composites and hybrid materials
Research has focused on developing lithium nitride-based composite and hybrid materials to enhance specific properties. These composites often combine lithium nitride with other materials such as carbon, metal oxides, or polymers to create materials with improved mechanical, thermal, or electrochemical characteristics. Such hybrid materials find applications in energy storage, catalysis, and advanced materials science.Expand Specific Solutions05 Hydrogen storage and catalytic applications
Lithium nitride demonstrates potential in hydrogen storage applications due to its ability to absorb and release hydrogen under specific conditions. Additionally, lithium nitride and its derivatives show catalytic properties that can be utilized in various chemical reactions, including ammonia synthesis and decomposition. The material's surface properties and reactivity make it valuable for these energy-related and catalytic applications.Expand Specific Solutions
Key Industry Players in Lithium Nitride Development
The lithium nitride technology market is currently in an early growth phase, characterized by increasing research activities and emerging commercial applications. The market size is expanding, driven by growing demand in semiconductor manufacturing, energy storage, and advanced materials sectors. From a technical maturity perspective, the field is transitioning from research to commercialization, with key players demonstrating varying levels of expertise. Leading semiconductor companies like Applied Materials, Tokyo Electron, and TSMC are developing integration processes, while specialized materials firms such as BNNT Materials and Adem Technologies focus on novel synthesis methods. Academic institutions including Northwestern University and Central South University contribute fundamental research, while energy companies like Svolt Energy and Tianqi Lithium are exploring applications in battery technology. The competitive landscape reflects a diverse ecosystem of established corporations and specialized startups working to overcome implementation challenges.
Svolt Energy Technology Co., Ltd.
Technical Solution: Svolt has developed advanced lithium nitride-based solid-state electrolytes for next-generation batteries. Their proprietary technology utilizes lithium nitride (Li3N) as a key component in solid-state battery architecture, creating a stable interface between lithium metal anodes and solid electrolytes. The company's approach involves synthesizing lithium nitride through controlled reaction of lithium with nitrogen at specific temperatures and pressures, resulting in highly pure Li3N with optimized crystallinity. This material serves as an excellent lithium-ion conductor with ionic conductivity exceeding 10^-3 S/cm at room temperature[1]. Svolt's implementation includes a gradient design where lithium nitride forms a protective layer between the lithium metal anode and the bulk electrolyte, effectively suppressing dendrite formation while maintaining high ionic conductivity. Their manufacturing process incorporates specialized deposition techniques that ensure uniform lithium nitride layer formation, critical for battery performance and safety[3].
Strengths: Superior ionic conductivity compared to conventional electrolytes, excellent chemical stability with lithium metal, and effective dendrite suppression capabilities. Weaknesses: Manufacturing complexity requiring precise control of nitrogen atmosphere, higher production costs compared to liquid electrolytes, and challenges in scaling production to commercial volumes.
Tianqi Lithium Corp.
Technical Solution: Tianqi Lithium has pioneered an innovative approach to lithium nitride implementation in energy storage systems. Their technology centers on using lithium nitride as a critical component in advanced battery systems, particularly as an interface modifier between electrodes and electrolytes. The company has developed a proprietary process for synthesizing high-purity lithium nitride powder with controlled particle size distribution (typically 1-5 μm), which is then integrated into battery components through specialized coating techniques. Their implementation involves creating a thin (50-200 nm) lithium nitride layer on electrode surfaces that serves as an artificial solid electrolyte interphase (SEI), significantly enhancing electrode stability and cycle life[2]. Tianqi's research has demonstrated that these lithium nitride interfaces can reduce interfacial resistance by up to 60% compared to conventional systems while providing protection against unwanted side reactions with electrolytes[4]. The company has also explored lithium nitride's application in lithium-sulfur batteries, where it helps contain polysulfide shuttling effects.
Strengths: Excellent interfacial stability with various electrode materials, significant improvement in battery cycle life, and compatibility with existing manufacturing processes. Weaknesses: Sensitivity to moisture requiring stringent handling protocols, higher material costs compared to traditional battery components, and limited long-term stability data in real-world applications.
Critical Patents and Research in Lithium Nitride Technology
Process for the preparation of lithium nitride in a pulverised state
PatentInactiveGB199027A
Innovation
- Heating a lithium amalgam in a nitrogen atmosphere to distill off mercury, resulting in lithium nitride in a finely pulverised state, which can be used as a catalyst for ammonia synthesis without pressure or at low pressures and temperatures.
Nitride thin film stucture and method of forming the same
PatentInactiveUS20150087137A1
Innovation
- A thin film structure is formed by coating a substrate with hollow structures, which reduces stress and dislocations in the nitride thin film, allowing for high-quality film growth with improved light extraction efficiency and reduced substrate warpage, achieved through the use of hollow structures that control refractive index and provide an epitaxial lateral overgrowth effect.
Environmental Impact and Sustainability Considerations
The implementation of lithium nitride solutions in emerging technologies necessitates careful consideration of environmental impacts throughout the entire lifecycle. Lithium extraction processes, particularly from brine pools and hard rock mining, pose significant environmental challenges including water depletion, habitat disruption, and chemical contamination. Traditional lithium mining operations can consume between 500,000 to 2 million gallons of water per ton of lithium extracted, creating substantial ecological pressure in water-scarce regions.
Manufacturing processes for lithium nitride compounds generate considerable carbon emissions, estimated at 15-20 tons of CO2 equivalent per ton of processed material. Additionally, the production phase involves energy-intensive high-temperature synthesis methods that typically operate at 600-800°C, contributing further to the environmental footprint. Chemical waste streams containing nitrogen compounds present potential risks for water systems if not properly managed.
Sustainability improvements are emerging across the lithium nitride value chain. Advanced extraction technologies such as direct lithium extraction (DLE) can reduce water consumption by up to 70% compared to traditional evaporation methods. Closed-loop manufacturing systems are being developed to recapture and reuse process chemicals, minimizing waste discharge by approximately 85%. Energy efficiency innovations in synthesis processes have demonstrated potential to reduce energy requirements by 30-40% through catalytic approaches and optimized reaction pathways.
End-of-life considerations represent a critical sustainability challenge. Current recycling rates for lithium compounds remain below 5% globally, creating significant material loss. Emerging technologies for lithium nitride recovery from spent products show promise, with laboratory-scale processes achieving recovery rates of 80-90%. Implementation of circular economy principles could potentially reduce primary lithium demand by 25-30% by 2030, according to industry projections.
Regulatory frameworks are evolving to address these environmental concerns. The European Union's Battery Directive revision proposes mandatory recycled content requirements and carbon footprint declarations for battery materials. Several countries are implementing extended producer responsibility (EPR) programs that hold manufacturers accountable for the entire lifecycle environmental impact of their products containing lithium compounds.
Life cycle assessment (LCA) studies indicate that despite production-phase impacts, lithium nitride applications in energy storage and conversion technologies can deliver net environmental benefits through operational efficiencies. For example, lithium nitride-enabled solid-state batteries could reduce lifetime carbon emissions by 25-35% compared to conventional lithium-ion technologies when considering full cradle-to-grave impacts.
Manufacturing processes for lithium nitride compounds generate considerable carbon emissions, estimated at 15-20 tons of CO2 equivalent per ton of processed material. Additionally, the production phase involves energy-intensive high-temperature synthesis methods that typically operate at 600-800°C, contributing further to the environmental footprint. Chemical waste streams containing nitrogen compounds present potential risks for water systems if not properly managed.
Sustainability improvements are emerging across the lithium nitride value chain. Advanced extraction technologies such as direct lithium extraction (DLE) can reduce water consumption by up to 70% compared to traditional evaporation methods. Closed-loop manufacturing systems are being developed to recapture and reuse process chemicals, minimizing waste discharge by approximately 85%. Energy efficiency innovations in synthesis processes have demonstrated potential to reduce energy requirements by 30-40% through catalytic approaches and optimized reaction pathways.
End-of-life considerations represent a critical sustainability challenge. Current recycling rates for lithium compounds remain below 5% globally, creating significant material loss. Emerging technologies for lithium nitride recovery from spent products show promise, with laboratory-scale processes achieving recovery rates of 80-90%. Implementation of circular economy principles could potentially reduce primary lithium demand by 25-30% by 2030, according to industry projections.
Regulatory frameworks are evolving to address these environmental concerns. The European Union's Battery Directive revision proposes mandatory recycled content requirements and carbon footprint declarations for battery materials. Several countries are implementing extended producer responsibility (EPR) programs that hold manufacturers accountable for the entire lifecycle environmental impact of their products containing lithium compounds.
Life cycle assessment (LCA) studies indicate that despite production-phase impacts, lithium nitride applications in energy storage and conversion technologies can deliver net environmental benefits through operational efficiencies. For example, lithium nitride-enabled solid-state batteries could reduce lifetime carbon emissions by 25-35% compared to conventional lithium-ion technologies when considering full cradle-to-grave impacts.
Supply Chain Analysis and Material Sourcing Strategies
The lithium nitride supply chain presents unique challenges due to the specialized nature of this compound and its growing importance in emerging technologies. Currently, the global supply chain for lithium nitride remains underdeveloped compared to more established materials, with limited dedicated production facilities concentrated primarily in North America, Europe, and East Asia. This geographical concentration creates potential vulnerabilities in supply security, particularly as demand increases across multiple technology sectors.
Raw material sourcing represents the first critical challenge in the lithium nitride supply chain. High-purity lithium metal, the primary precursor, faces its own supply constraints due to competing demands from the battery industry. Strategic partnerships with lithium producers who can guarantee consistent quality and volume are becoming essential for companies implementing lithium nitride solutions. Forward-thinking organizations are increasingly securing long-term supply agreements to mitigate price volatility and availability risks.
Production scaling presents another significant hurdle. The synthesis of high-quality lithium nitride requires specialized equipment and controlled environments to prevent contamination and ensure consistent properties. Current manufacturing processes remain relatively small-scale and often batch-oriented, limiting production volumes and increasing unit costs. Investment in continuous flow production methods and automated quality control systems represents a promising direction for improving manufacturing efficiency.
Quality control throughout the supply chain is paramount, as impurities can significantly impact the performance of lithium nitride in advanced applications. Developing standardized testing protocols and certification processes will be crucial as the market matures. Leading companies are implementing comprehensive traceability systems to monitor material quality from raw material sourcing through final product integration.
Alternative sourcing strategies are emerging to address supply constraints. Research into synthetic routes using more abundant precursors is advancing, though these approaches typically face challenges in achieving the same performance characteristics as traditionally produced lithium nitride. Recycling pathways are also being explored, particularly for recovering lithium nitride from end-of-life products, though these processes remain in early development stages.
Regulatory considerations further complicate the supply chain landscape. The transport and handling of reactive materials like lithium nitride are subject to stringent safety regulations that vary by region, creating compliance challenges for global supply chains. Companies must develop expertise in navigating these regulatory frameworks to ensure uninterrupted material flow across international boundaries.
Raw material sourcing represents the first critical challenge in the lithium nitride supply chain. High-purity lithium metal, the primary precursor, faces its own supply constraints due to competing demands from the battery industry. Strategic partnerships with lithium producers who can guarantee consistent quality and volume are becoming essential for companies implementing lithium nitride solutions. Forward-thinking organizations are increasingly securing long-term supply agreements to mitigate price volatility and availability risks.
Production scaling presents another significant hurdle. The synthesis of high-quality lithium nitride requires specialized equipment and controlled environments to prevent contamination and ensure consistent properties. Current manufacturing processes remain relatively small-scale and often batch-oriented, limiting production volumes and increasing unit costs. Investment in continuous flow production methods and automated quality control systems represents a promising direction for improving manufacturing efficiency.
Quality control throughout the supply chain is paramount, as impurities can significantly impact the performance of lithium nitride in advanced applications. Developing standardized testing protocols and certification processes will be crucial as the market matures. Leading companies are implementing comprehensive traceability systems to monitor material quality from raw material sourcing through final product integration.
Alternative sourcing strategies are emerging to address supply constraints. Research into synthetic routes using more abundant precursors is advancing, though these approaches typically face challenges in achieving the same performance characteristics as traditionally produced lithium nitride. Recycling pathways are also being explored, particularly for recovering lithium nitride from end-of-life products, though these processes remain in early development stages.
Regulatory considerations further complicate the supply chain landscape. The transport and handling of reactive materials like lithium nitride are subject to stringent safety regulations that vary by region, creating compliance challenges for global supply chains. Companies must develop expertise in navigating these regulatory frameworks to ensure uninterrupted material flow across international boundaries.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!