How to Mitigate Self-Discharge with Emerging Additive Techniques
APR 15, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Battery Self-Discharge Mitigation Background and Objectives
Battery self-discharge represents a fundamental challenge in electrochemical energy storage systems, where stored energy gradually dissipates even when the battery is not actively used. This phenomenon occurs through various mechanisms including electrolyte decomposition, active material dissolution, and parasitic reactions at electrode interfaces. The severity of self-discharge directly impacts battery shelf life, energy efficiency, and overall system reliability across applications ranging from consumer electronics to grid-scale energy storage.
The historical development of battery technology has consistently grappled with self-discharge issues. Early lead-acid batteries exhibited significant self-discharge rates of 5-20% per month, while nickel-based chemistries improved this to 10-15% monthly. The advent of lithium-ion technology marked a substantial advancement, reducing self-discharge to 1-5% per month under optimal conditions. However, as energy density requirements increase and operating temperature ranges expand, traditional approaches to self-discharge mitigation have reached their limitations.
Contemporary battery applications demand increasingly stringent self-discharge performance. Electric vehicles require batteries that maintain charge during extended parking periods, while grid storage systems must preserve energy over seasonal cycles. Consumer devices expect multi-month standby capabilities, and aerospace applications require ultra-low self-discharge for mission-critical operations. These evolving requirements have intensified research focus on innovative mitigation strategies.
The emergence of additive-based approaches represents a paradigm shift in addressing self-discharge challenges. Unlike conventional methods that rely primarily on electrolyte formulation and electrode material optimization, additive techniques offer targeted interventions at the molecular level. These approaches can selectively inhibit specific degradation pathways while maintaining or enhancing primary battery performance characteristics.
The primary objective of investigating emerging additive techniques centers on achieving sub-1% monthly self-discharge rates across diverse operating conditions while preserving cycle life, power capability, and safety margins. Secondary objectives include developing cost-effective additive solutions that integrate seamlessly with existing manufacturing processes and demonstrate scalability across different battery chemistries and form factors.
This technological advancement pathway aims to establish a comprehensive understanding of additive mechanisms, optimize formulation strategies, and validate performance across relevant application scenarios. The ultimate goal involves translating laboratory breakthroughs into commercially viable solutions that significantly extend battery operational lifespans and enhance energy storage system economics.
The historical development of battery technology has consistently grappled with self-discharge issues. Early lead-acid batteries exhibited significant self-discharge rates of 5-20% per month, while nickel-based chemistries improved this to 10-15% monthly. The advent of lithium-ion technology marked a substantial advancement, reducing self-discharge to 1-5% per month under optimal conditions. However, as energy density requirements increase and operating temperature ranges expand, traditional approaches to self-discharge mitigation have reached their limitations.
Contemporary battery applications demand increasingly stringent self-discharge performance. Electric vehicles require batteries that maintain charge during extended parking periods, while grid storage systems must preserve energy over seasonal cycles. Consumer devices expect multi-month standby capabilities, and aerospace applications require ultra-low self-discharge for mission-critical operations. These evolving requirements have intensified research focus on innovative mitigation strategies.
The emergence of additive-based approaches represents a paradigm shift in addressing self-discharge challenges. Unlike conventional methods that rely primarily on electrolyte formulation and electrode material optimization, additive techniques offer targeted interventions at the molecular level. These approaches can selectively inhibit specific degradation pathways while maintaining or enhancing primary battery performance characteristics.
The primary objective of investigating emerging additive techniques centers on achieving sub-1% monthly self-discharge rates across diverse operating conditions while preserving cycle life, power capability, and safety margins. Secondary objectives include developing cost-effective additive solutions that integrate seamlessly with existing manufacturing processes and demonstrate scalability across different battery chemistries and form factors.
This technological advancement pathway aims to establish a comprehensive understanding of additive mechanisms, optimize formulation strategies, and validate performance across relevant application scenarios. The ultimate goal involves translating laboratory breakthroughs into commercially viable solutions that significantly extend battery operational lifespans and enhance energy storage system economics.
Market Demand for Low Self-Discharge Battery Solutions
The global battery market is experiencing unprecedented growth driven by the rapid expansion of electric vehicles, renewable energy storage systems, and portable electronic devices. Self-discharge represents a critical performance parameter that significantly impacts battery efficiency, operational costs, and user satisfaction across these applications. As energy storage requirements become more demanding, the market increasingly prioritizes solutions that minimize energy loss during storage periods.
Electric vehicle manufacturers face substantial challenges related to battery self-discharge, particularly in scenarios where vehicles remain parked for extended periods. Fleet operators and individual consumers alike demand batteries that maintain charge levels over weeks or months of inactivity. This requirement has intensified as EV adoption accelerates globally, with automotive manufacturers seeking battery technologies that can guarantee minimal capacity loss during storage and transportation phases.
The renewable energy sector presents another significant market driver for low self-discharge battery solutions. Grid-scale energy storage systems require batteries capable of storing energy for extended periods without substantial losses. Solar and wind energy installations depend on storage technologies that can efficiently retain captured energy during periods of low generation or high demand variability. The economic viability of these systems directly correlates with minimizing self-discharge rates.
Consumer electronics markets continue to evolve toward devices with longer standby times and improved energy retention. Smartphones, laptops, wearable devices, and IoT sensors require batteries that maintain charge during periods of non-use. The proliferation of remote monitoring devices and emergency backup systems has created substantial demand for ultra-low self-discharge battery technologies.
Industrial applications, including medical devices, aerospace systems, and critical infrastructure backup power, represent high-value market segments where self-discharge performance directly impacts safety and reliability. These sectors demonstrate willingness to invest in premium battery solutions that incorporate advanced additive techniques to achieve superior energy retention characteristics.
The market demand extends beyond traditional lithium-ion technologies, encompassing emerging battery chemistries where additive techniques play crucial roles in controlling self-discharge mechanisms. Research institutions and battery manufacturers are actively investing in additive development programs to address market requirements for improved energy retention across diverse temperature ranges and storage conditions.
Electric vehicle manufacturers face substantial challenges related to battery self-discharge, particularly in scenarios where vehicles remain parked for extended periods. Fleet operators and individual consumers alike demand batteries that maintain charge levels over weeks or months of inactivity. This requirement has intensified as EV adoption accelerates globally, with automotive manufacturers seeking battery technologies that can guarantee minimal capacity loss during storage and transportation phases.
The renewable energy sector presents another significant market driver for low self-discharge battery solutions. Grid-scale energy storage systems require batteries capable of storing energy for extended periods without substantial losses. Solar and wind energy installations depend on storage technologies that can efficiently retain captured energy during periods of low generation or high demand variability. The economic viability of these systems directly correlates with minimizing self-discharge rates.
Consumer electronics markets continue to evolve toward devices with longer standby times and improved energy retention. Smartphones, laptops, wearable devices, and IoT sensors require batteries that maintain charge during periods of non-use. The proliferation of remote monitoring devices and emergency backup systems has created substantial demand for ultra-low self-discharge battery technologies.
Industrial applications, including medical devices, aerospace systems, and critical infrastructure backup power, represent high-value market segments where self-discharge performance directly impacts safety and reliability. These sectors demonstrate willingness to invest in premium battery solutions that incorporate advanced additive techniques to achieve superior energy retention characteristics.
The market demand extends beyond traditional lithium-ion technologies, encompassing emerging battery chemistries where additive techniques play crucial roles in controlling self-discharge mechanisms. Research institutions and battery manufacturers are actively investing in additive development programs to address market requirements for improved energy retention across diverse temperature ranges and storage conditions.
Current State and Challenges in Self-Discharge Control
Self-discharge remains one of the most persistent challenges in modern battery technology, significantly impacting energy storage systems across various applications from consumer electronics to grid-scale storage. Current lithium-ion batteries typically experience self-discharge rates ranging from 2-5% per month at room temperature, with rates increasing exponentially at elevated temperatures. This phenomenon occurs through multiple mechanisms including electrolyte decomposition, active material dissolution, and parasitic reactions at electrode interfaces.
The primary challenge lies in the complex interplay between electrolyte stability and electrode surface chemistry. Traditional carbonate-based electrolytes, while providing excellent electrochemical performance, are prone to continuous decomposition reactions that consume stored energy. These reactions are particularly pronounced at the anode-electrolyte interface, where lithium metal deposition and SEI layer instability contribute significantly to capacity loss over time.
Temperature sensitivity presents another critical challenge, as self-discharge rates can increase by factors of 2-3 for every 10°C temperature rise. This thermal dependence severely limits battery performance in automotive and stationary storage applications where temperature control is difficult or energy-intensive. Current thermal management solutions add substantial cost and complexity to battery systems.
Existing mitigation strategies have shown limited success in addressing the root causes of self-discharge. Conventional approaches focus primarily on electrolyte purification and moisture control, which only address secondary factors rather than fundamental electrochemical processes. Advanced separator technologies and coating materials have provided incremental improvements but fail to achieve the sub-1% monthly self-discharge rates required for next-generation applications.
The geographical distribution of self-discharge research reveals significant concentration in East Asian markets, particularly in China, Japan, and South Korea, where major battery manufacturers are investing heavily in additive development. However, fundamental research gaps persist in understanding the molecular-level mechanisms governing long-term electrochemical stability, creating opportunities for breakthrough innovations in additive chemistry and interface engineering.
The primary challenge lies in the complex interplay between electrolyte stability and electrode surface chemistry. Traditional carbonate-based electrolytes, while providing excellent electrochemical performance, are prone to continuous decomposition reactions that consume stored energy. These reactions are particularly pronounced at the anode-electrolyte interface, where lithium metal deposition and SEI layer instability contribute significantly to capacity loss over time.
Temperature sensitivity presents another critical challenge, as self-discharge rates can increase by factors of 2-3 for every 10°C temperature rise. This thermal dependence severely limits battery performance in automotive and stationary storage applications where temperature control is difficult or energy-intensive. Current thermal management solutions add substantial cost and complexity to battery systems.
Existing mitigation strategies have shown limited success in addressing the root causes of self-discharge. Conventional approaches focus primarily on electrolyte purification and moisture control, which only address secondary factors rather than fundamental electrochemical processes. Advanced separator technologies and coating materials have provided incremental improvements but fail to achieve the sub-1% monthly self-discharge rates required for next-generation applications.
The geographical distribution of self-discharge research reveals significant concentration in East Asian markets, particularly in China, Japan, and South Korea, where major battery manufacturers are investing heavily in additive development. However, fundamental research gaps persist in understanding the molecular-level mechanisms governing long-term electrochemical stability, creating opportunities for breakthrough innovations in additive chemistry and interface engineering.
Existing Additive Solutions for Self-Discharge Reduction
01 Additive manufacturing techniques for battery electrode structures
Advanced additive manufacturing methods such as 3D printing and layer-by-layer deposition are employed to fabricate battery electrodes with controlled porosity and architecture. These techniques enable precise control over electrode geometry and material distribution, which can influence self-discharge characteristics by optimizing ion transport pathways and reducing internal short-circuit risks. The manufacturing process allows for customization of electrode structures to minimize parasitic reactions that contribute to self-discharge.- Additive manufacturing techniques for battery electrode structures: Advanced additive manufacturing methods such as 3D printing and layer-by-layer deposition can be employed to fabricate battery electrodes with optimized microstructures. These techniques enable precise control over electrode architecture, porosity, and material distribution, which can help minimize self-discharge by creating more uniform current distribution and reducing internal defects that contribute to parasitic reactions.
- Electrolyte additives for self-discharge suppression: Specialized chemical additives can be incorporated into battery electrolytes to form protective layers on electrode surfaces, preventing unwanted side reactions that cause self-discharge. These additives work by creating stable solid-electrolyte interphase layers or by scavenging reactive species that would otherwise contribute to capacity loss during storage. The selection and concentration of these additives are critical for balancing self-discharge reduction with overall battery performance.
- Conductive additives and electrode formulation optimization: The incorporation of specific conductive additives such as carbon materials, graphene, or conductive polymers into electrode formulations can improve electron transport pathways and reduce localized charge accumulation. Proper distribution and selection of these additives help minimize internal resistance and hot spots that accelerate self-discharge. The optimization of binder systems and active material particle sizes also plays a crucial role in controlling self-discharge behavior.
- Surface coating and passivation techniques: Application of protective surface coatings or passivation layers on electrode materials can effectively reduce self-discharge by preventing direct contact between active materials and electrolyte components. These coatings act as barriers to moisture, oxygen, and other contaminants while maintaining ionic conductivity. Various coating materials including metal oxides, polymers, and ceramic materials can be applied through techniques such as atomic layer deposition or chemical vapor deposition.
- Separator modification and functional additives: Enhancement of battery separators through functional additives or structural modifications can significantly reduce self-discharge rates. Modified separators with improved thermal stability, enhanced electrolyte retention, and selective ion transport properties help prevent internal short circuits and reduce shuttle effects. Incorporation of ceramic particles, functional polymers, or composite materials into separator structures creates additional barriers against self-discharge mechanisms.
02 Surface coating additives to reduce self-discharge
Specialized coating materials and surface treatment additives are applied to electrode surfaces to create protective barriers that minimize unwanted electrochemical reactions. These coatings act as passivation layers that prevent direct contact between active materials and electrolytes, thereby reducing self-discharge rates. The additives can include polymeric materials, ceramic coatings, or carbon-based layers that maintain ionic conductivity while blocking electronic pathways responsible for self-discharge.Expand Specific Solutions03 Electrolyte additives for self-discharge mitigation
Chemical additives incorporated into battery electrolytes help stabilize the electrode-electrolyte interface and suppress side reactions that lead to self-discharge. These additives can form stable solid electrolyte interphase layers, scavenge reactive species, or modify the electrochemical window of the electrolyte system. The formulation of electrolyte additives is critical for extending shelf life and maintaining charge retention during storage periods.Expand Specific Solutions04 Conductive additives and their impact on self-discharge
The selection and distribution of conductive additives within electrode formulations significantly affects self-discharge behavior. Carbon-based conductive additives such as carbon black, graphene, or carbon nanotubes are optimized in terms of type, particle size, and loading amount to balance electrical conductivity with minimal contribution to self-discharge pathways. Proper dispersion techniques ensure uniform distribution while preventing the formation of conductive networks that could facilitate unwanted charge transfer.Expand Specific Solutions05 Binder additives and separator modifications for self-discharge control
Polymeric binder additives and separator material modifications play important roles in controlling self-discharge mechanisms. Advanced binder systems provide mechanical stability while minimizing ionic resistance and preventing micro-short circuits. Separator additives or functional coatings enhance the barrier properties against dendrite penetration and shuttle effects, which are common causes of self-discharge in various battery chemistries. These materials are engineered to maintain structural integrity while providing selective ion transport.Expand Specific Solutions
Key Players in Battery Additive and Materials Industry
The self-discharge mitigation technology landscape is in a mature growth phase, driven by increasing demand for high-performance energy storage solutions across automotive, consumer electronics, and industrial applications. The market demonstrates significant scale with established players like Panasonic Intellectual Property Management and Canon leading consumer battery innovations, while industrial giants such as China Petroleum & Chemical Corp. and Air Liquide SA drive chemical additive development. Technology maturity varies considerably across segments, with companies like Semiconductor Energy Laboratory and Hitachi High-Tech America advancing cutting-edge materials science, while traditional manufacturers like Kuraray and W.R. Grace focus on proven additive formulations. Research institutions including South China Normal University and Centre National de la Recherche Scientifique contribute fundamental breakthroughs in electrochemical mechanisms. The competitive landscape shows convergence between chemical processing expertise and advanced materials engineering, indicating technology consolidation phases ahead.
Panasonic Intellectual Property Management Co. Ltd.
Technical Solution: Panasonic has developed advanced electrolyte additive formulations specifically designed to minimize self-discharge in lithium-ion batteries. Their approach focuses on surface film-forming additives that create stable solid electrolyte interphase (SEI) layers on electrode surfaces, effectively reducing parasitic reactions that contribute to self-discharge. The company has implemented proprietary organic carbonate-based additives combined with fluorinated compounds that enhance the passivation of electrode materials. These additives work by forming protective layers that prevent continuous electrolyte decomposition and minimize lithium inventory loss during storage periods.
Strengths: Extensive patent portfolio and proven commercial implementation in consumer electronics. Weaknesses: Limited focus on next-generation battery chemistries beyond traditional lithium-ion systems.
Centre National de la Recherche Scientifique
Technical Solution: CNRS has conducted extensive research on molecular-level additive design for self-discharge mitigation through computational modeling and experimental validation. Their approach involves the development of redox-active organic additives that can reversibly capture and release electrons, effectively buffering against self-discharge reactions. The research focuses on designing molecules with specific HOMO-LUMO energy levels that can intercept unwanted electron transfer processes while remaining electrochemically stable. They have also investigated ionic liquid-based additives that provide enhanced electrochemical windows and reduced volatility, contributing to long-term battery stability and reduced self-discharge rates in various battery chemistries.
Strengths: Cutting-edge fundamental research capabilities and strong theoretical understanding of electrochemical processes. Weaknesses: Limited commercial manufacturing experience and technology transfer capabilities for industrial implementation.
Core Innovations in Emerging Battery Additive Techniques
Lithium-ion battery with improved self-discharge characteristics
PatentWO2008094206A9
Innovation
- Incorporating a Lewis basic species such as dimethyl acetamide or N-methylpyrrolidone into the non-aqueous electrolyte of lithium-ion batteries in small amounts (0.01-1.0 volume percent) to inhibit self-discharge and reduce sensitivity to water impurities.
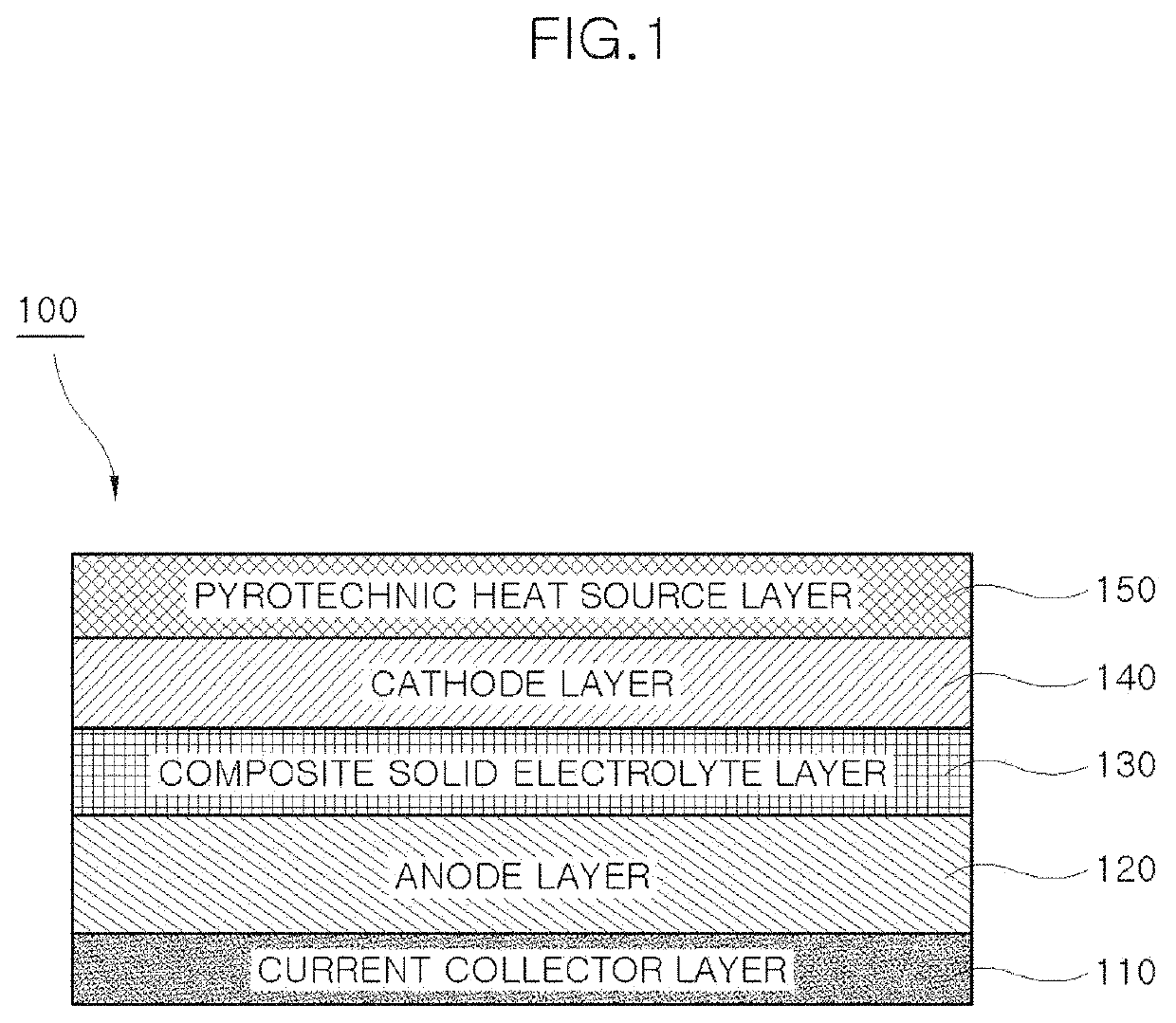
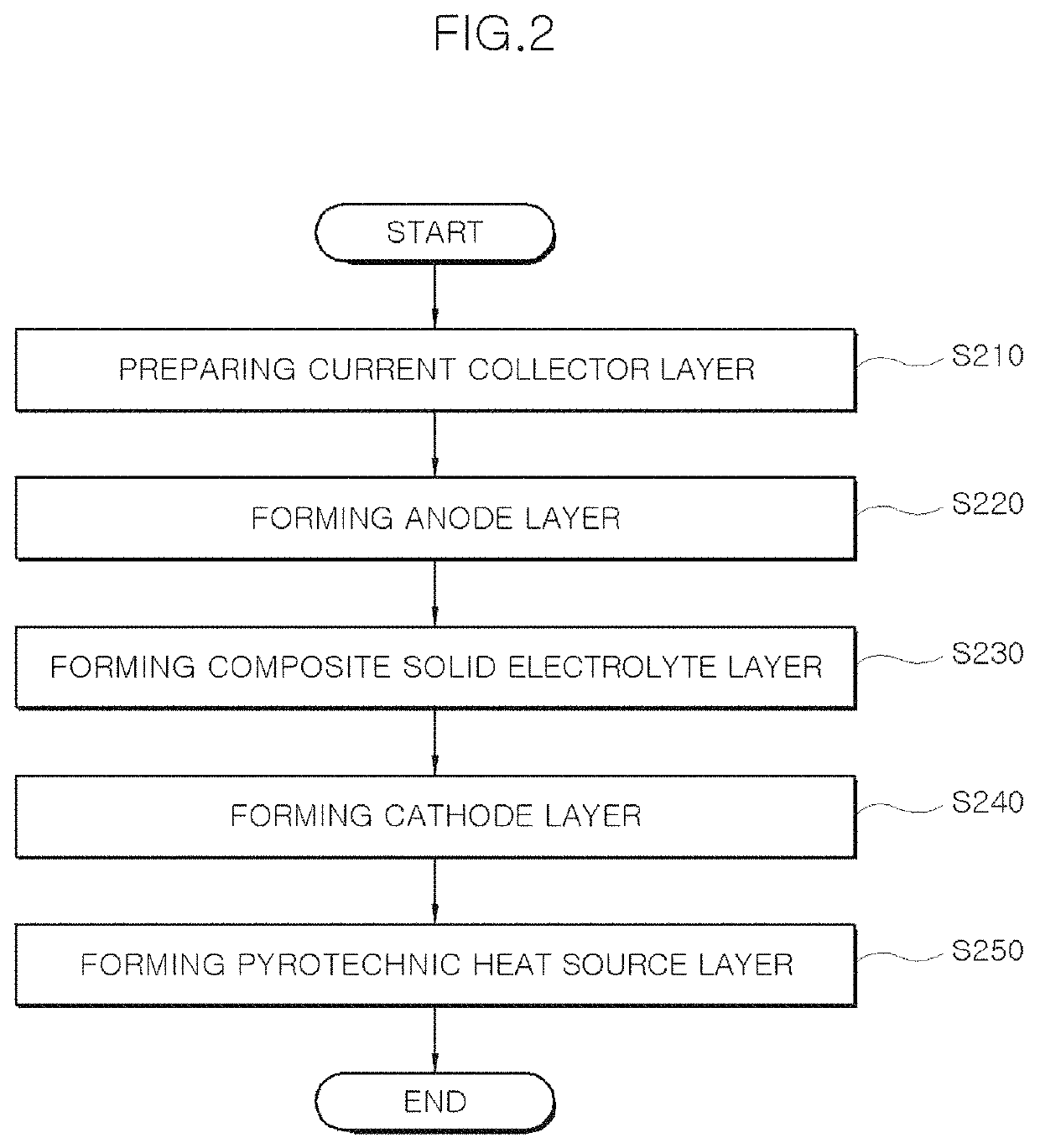
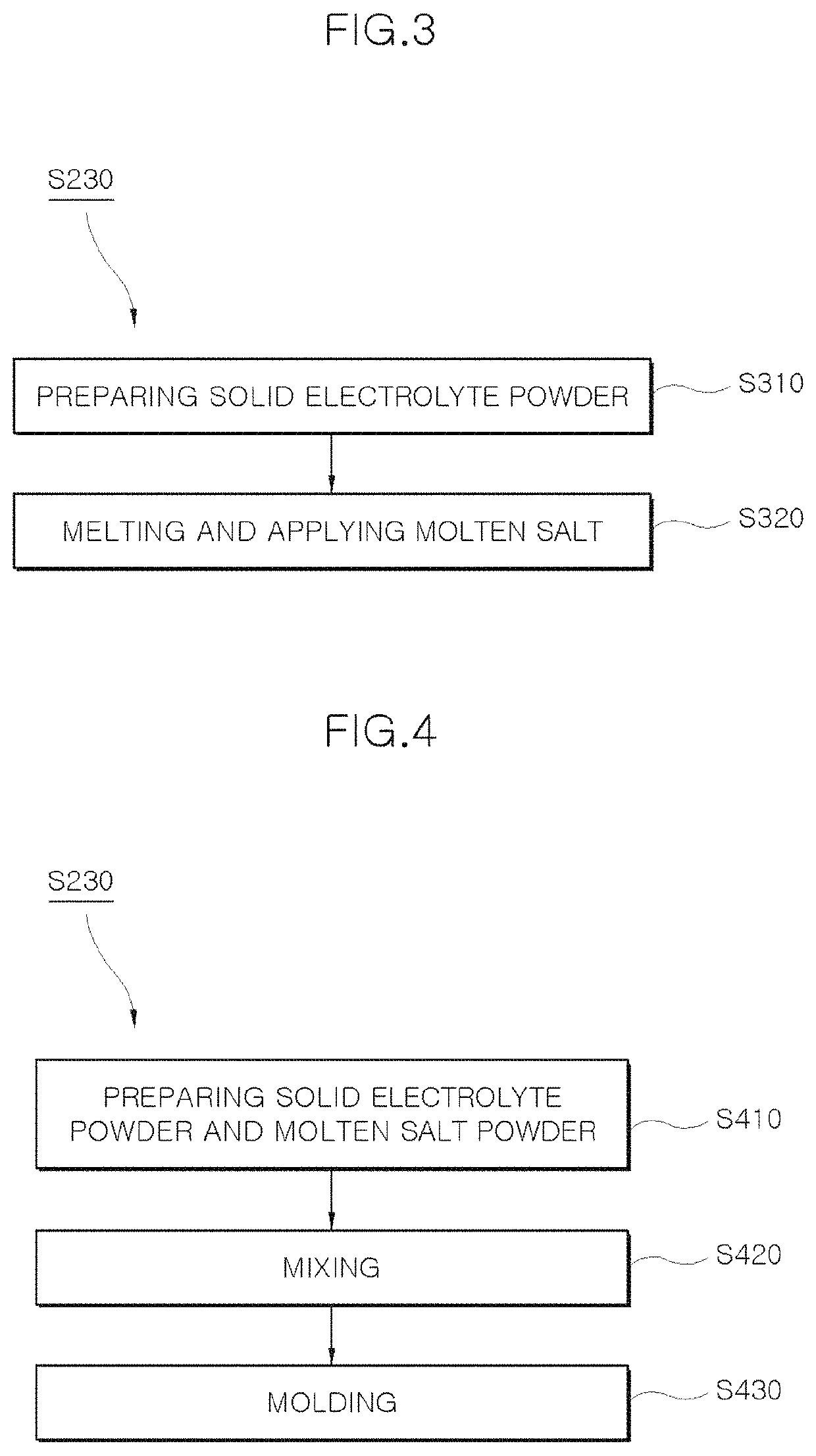
Composite solid electrolyte without self-discharge, battery unit cell having same, and method of manufacturing composite solid electrolyte
PatentActiveUS10998583B1
Innovation
- A composite solid electrolyte is developed by incorporating molten salt powder or a molten salt passivation layer, which acts as an electrical insulator at room temperature, combined with solid electrolyte powder, to prevent self-discharge and enhance ion conductivity, stability, and operational efficiency.
Environmental Impact Assessment of Battery Additives
The environmental implications of battery additives designed to mitigate self-discharge present a complex landscape of considerations spanning their entire lifecycle. As the battery industry increasingly adopts novel additive formulations to enhance performance and longevity, understanding their ecological footprint becomes paramount for sustainable technology development.
Manufacturing processes for advanced battery additives often involve sophisticated chemical synthesis routes that can generate significant environmental burdens. Many emerging additives, particularly those containing rare earth elements or complex organic compounds, require energy-intensive production methods and specialized purification steps. The carbon footprint associated with additive production can be substantial, especially for materials requiring high-temperature processing or multiple synthesis stages.
Raw material extraction for additive components raises additional environmental concerns. Lithium-based additives contribute to the growing demand for lithium resources, intensifying mining pressures on salt flats and hard rock deposits. Similarly, additives incorporating cobalt, nickel, or other transition metals perpetuate the environmental challenges associated with mining these materials, including habitat disruption and water contamination risks.
The biodegradability and toxicity profiles of battery additives vary significantly across different chemical classes. Organic additives such as vinylene carbonate and fluoroethylene carbonate generally exhibit better environmental compatibility compared to heavy metal-based alternatives. However, fluorinated compounds present persistence concerns due to their strong carbon-fluorine bonds, potentially leading to bioaccumulation in environmental systems.
End-of-life management represents a critical environmental consideration for additive-enhanced batteries. While some additives can be recovered through existing recycling processes, others may complicate material separation and purification steps. The presence of certain additives can alter the chemistry of recycling streams, potentially requiring modified processing techniques or generating new waste streams.
Regulatory frameworks are evolving to address the environmental impact of battery additives, with increasing emphasis on lifecycle assessments and sustainable chemistry principles. The European Union's Battery Regulation and similar initiatives worldwide are driving manufacturers toward more environmentally conscious additive selection and development practices, promoting the adoption of green chemistry approaches in additive design.
Manufacturing processes for advanced battery additives often involve sophisticated chemical synthesis routes that can generate significant environmental burdens. Many emerging additives, particularly those containing rare earth elements or complex organic compounds, require energy-intensive production methods and specialized purification steps. The carbon footprint associated with additive production can be substantial, especially for materials requiring high-temperature processing or multiple synthesis stages.
Raw material extraction for additive components raises additional environmental concerns. Lithium-based additives contribute to the growing demand for lithium resources, intensifying mining pressures on salt flats and hard rock deposits. Similarly, additives incorporating cobalt, nickel, or other transition metals perpetuate the environmental challenges associated with mining these materials, including habitat disruption and water contamination risks.
The biodegradability and toxicity profiles of battery additives vary significantly across different chemical classes. Organic additives such as vinylene carbonate and fluoroethylene carbonate generally exhibit better environmental compatibility compared to heavy metal-based alternatives. However, fluorinated compounds present persistence concerns due to their strong carbon-fluorine bonds, potentially leading to bioaccumulation in environmental systems.
End-of-life management represents a critical environmental consideration for additive-enhanced batteries. While some additives can be recovered through existing recycling processes, others may complicate material separation and purification steps. The presence of certain additives can alter the chemistry of recycling streams, potentially requiring modified processing techniques or generating new waste streams.
Regulatory frameworks are evolving to address the environmental impact of battery additives, with increasing emphasis on lifecycle assessments and sustainable chemistry principles. The European Union's Battery Regulation and similar initiatives worldwide are driving manufacturers toward more environmentally conscious additive selection and development practices, promoting the adoption of green chemistry approaches in additive design.
Safety Standards for Advanced Battery Additive Systems
The development of safety standards for advanced battery additive systems represents a critical regulatory frontier as emerging additive techniques gain prominence in mitigating self-discharge phenomena. Current safety frameworks primarily address conventional battery chemistries and fail to adequately encompass the unique risks and behaviors introduced by novel additive materials designed to reduce parasitic reactions and improve charge retention.
Existing international standards such as IEC 62133, UN 38.3, and UL 1642 provide foundational safety requirements for lithium-ion batteries but lack specific provisions for evaluating additive-enhanced systems. These standards focus on thermal runaway, mechanical abuse, and electrical safety testing without considering the long-term stability interactions between host electrolytes and specialized additives like solid electrolyte interphase modifiers, redox shuttles, or surface passivation agents.
The regulatory gap becomes particularly pronounced when addressing additive migration, chemical compatibility, and degradation byproduct formation over extended operational periods. Traditional safety testing protocols may not capture the delayed onset of safety risks that could emerge from additive decomposition or unexpected electrochemical interactions under stress conditions. This necessitates the development of accelerated aging tests specifically designed for additive-containing systems.
Emerging safety standard initiatives are beginning to address these challenges through collaborative efforts between battery manufacturers, additive suppliers, and regulatory bodies. Proposed frameworks emphasize comprehensive material characterization, including thermal stability analysis of additive compounds, gas evolution monitoring during cycling, and long-term compatibility assessments between additives and cell components.
Key areas requiring standardization include additive purity specifications, maximum allowable concentrations, thermal decomposition temperature thresholds, and mandatory disclosure requirements for additive chemical compositions. Additionally, new testing methodologies must evaluate the cumulative safety impact of multiple additive systems within single battery cells, as many manufacturers employ cocktail approaches combining various functional additives.
The development of these standards must balance innovation encouragement with safety assurance, ensuring that emerging additive technologies can be rapidly deployed while maintaining rigorous safety benchmarks. International harmonization of these standards will be essential for global market acceptance and regulatory compliance across different jurisdictions.
Existing international standards such as IEC 62133, UN 38.3, and UL 1642 provide foundational safety requirements for lithium-ion batteries but lack specific provisions for evaluating additive-enhanced systems. These standards focus on thermal runaway, mechanical abuse, and electrical safety testing without considering the long-term stability interactions between host electrolytes and specialized additives like solid electrolyte interphase modifiers, redox shuttles, or surface passivation agents.
The regulatory gap becomes particularly pronounced when addressing additive migration, chemical compatibility, and degradation byproduct formation over extended operational periods. Traditional safety testing protocols may not capture the delayed onset of safety risks that could emerge from additive decomposition or unexpected electrochemical interactions under stress conditions. This necessitates the development of accelerated aging tests specifically designed for additive-containing systems.
Emerging safety standard initiatives are beginning to address these challenges through collaborative efforts between battery manufacturers, additive suppliers, and regulatory bodies. Proposed frameworks emphasize comprehensive material characterization, including thermal stability analysis of additive compounds, gas evolution monitoring during cycling, and long-term compatibility assessments between additives and cell components.
Key areas requiring standardization include additive purity specifications, maximum allowable concentrations, thermal decomposition temperature thresholds, and mandatory disclosure requirements for additive chemical compositions. Additionally, new testing methodologies must evaluate the cumulative safety impact of multiple additive systems within single battery cells, as many manufacturers employ cocktail approaches combining various functional additives.
The development of these standards must balance innovation encouragement with safety assurance, ensuring that emerging additive technologies can be rapidly deployed while maintaining rigorous safety benchmarks. International harmonization of these standards will be essential for global market acceptance and regulatory compliance across different jurisdictions.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!