Ionizing Rays in Nuclear Medicine: Use vs Effects
MAR 16, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Nuclear Medicine Ionizing Radiation Background and Objectives
Nuclear medicine represents a specialized branch of medical imaging and therapy that harnesses the unique properties of radioactive isotopes to diagnose and treat various diseases. This field emerged in the mid-20th century following groundbreaking discoveries in atomic physics and radiochemistry, fundamentally transforming medical practice by enabling physicians to visualize physiological processes at the molecular level rather than merely anatomical structures.
The evolution of nuclear medicine has been driven by continuous advances in radiopharmaceutical development, imaging technology, and radiation detection systems. From early applications using iodine-131 for thyroid studies in the 1940s to modern molecular imaging with positron emission tomography, the field has consistently pushed the boundaries of medical diagnostics and therapeutic interventions.
Contemporary nuclear medicine procedures utilize carefully selected radioisotopes that emit gamma rays, positrons, or beta particles, each serving specific diagnostic or therapeutic purposes. The fundamental principle involves introducing trace amounts of radioactive materials into the body, where they localize in target organs or tissues based on their biochemical properties, subsequently providing detailed functional information through external detection systems.
The primary objective of nuclear medicine ionizing radiation research focuses on optimizing the delicate balance between diagnostic efficacy and radiation safety. This involves developing radiopharmaceuticals with improved target specificity, reduced radiation burden, and enhanced imaging quality. Advanced imaging modalities such as SPECT-CT and PET-CT fusion systems exemplify this pursuit, combining functional nuclear imaging with anatomical detail while minimizing patient exposure.
Therapeutic applications of nuclear medicine, including radioimmunotherapy and targeted radionuclide therapy, aim to deliver precise radiation doses to diseased tissues while sparing healthy organs. These approaches require sophisticated dosimetry calculations and personalized treatment planning to maximize therapeutic benefit while adhering to radiation protection principles.
Current research objectives encompass the development of novel radioisotopes with optimal physical characteristics, advancement of theranostic approaches that combine diagnostic and therapeutic capabilities, and implementation of artificial intelligence algorithms for improved image reconstruction and dose optimization. The ultimate goal remains achieving superior clinical outcomes through innovative applications of ionizing radiation while maintaining the highest standards of patient safety and radiation protection.
The evolution of nuclear medicine has been driven by continuous advances in radiopharmaceutical development, imaging technology, and radiation detection systems. From early applications using iodine-131 for thyroid studies in the 1940s to modern molecular imaging with positron emission tomography, the field has consistently pushed the boundaries of medical diagnostics and therapeutic interventions.
Contemporary nuclear medicine procedures utilize carefully selected radioisotopes that emit gamma rays, positrons, or beta particles, each serving specific diagnostic or therapeutic purposes. The fundamental principle involves introducing trace amounts of radioactive materials into the body, where they localize in target organs or tissues based on their biochemical properties, subsequently providing detailed functional information through external detection systems.
The primary objective of nuclear medicine ionizing radiation research focuses on optimizing the delicate balance between diagnostic efficacy and radiation safety. This involves developing radiopharmaceuticals with improved target specificity, reduced radiation burden, and enhanced imaging quality. Advanced imaging modalities such as SPECT-CT and PET-CT fusion systems exemplify this pursuit, combining functional nuclear imaging with anatomical detail while minimizing patient exposure.
Therapeutic applications of nuclear medicine, including radioimmunotherapy and targeted radionuclide therapy, aim to deliver precise radiation doses to diseased tissues while sparing healthy organs. These approaches require sophisticated dosimetry calculations and personalized treatment planning to maximize therapeutic benefit while adhering to radiation protection principles.
Current research objectives encompass the development of novel radioisotopes with optimal physical characteristics, advancement of theranostic approaches that combine diagnostic and therapeutic capabilities, and implementation of artificial intelligence algorithms for improved image reconstruction and dose optimization. The ultimate goal remains achieving superior clinical outcomes through innovative applications of ionizing radiation while maintaining the highest standards of patient safety and radiation protection.
Market Demand for Nuclear Medicine Diagnostic and Therapeutic Applications
The global nuclear medicine market demonstrates robust growth driven by increasing prevalence of chronic diseases, aging populations, and technological advancements in imaging and therapeutic applications. Cardiovascular diseases, cancer, and neurological disorders represent the primary clinical areas fueling demand for nuclear medicine procedures. The diagnostic segment maintains dominance in market share, with single-photon emission computed tomography and positron emission tomography procedures experiencing steady adoption rates across healthcare facilities worldwide.
Oncology applications constitute the largest market segment, with radiopharmaceuticals playing crucial roles in cancer detection, staging, and treatment monitoring. The growing incidence of cancer globally, particularly in developed nations, drives sustained demand for nuclear imaging procedures. Thyroid disorders represent another significant application area, where radioiodine therapy and diagnostic imaging maintain established clinical protocols with consistent utilization patterns.
Cardiology applications show substantial market potential, particularly in myocardial perfusion imaging and cardiac function assessment. The increasing burden of cardiovascular diseases, coupled with the superior diagnostic accuracy of nuclear medicine techniques compared to conventional imaging modalities, supports continued market expansion in this therapeutic area.
Therapeutic nuclear medicine applications exhibit accelerated growth trajectories, driven by targeted radiotherapy developments and theranostic approaches. Radiopharmaceutical therapy for neuroendocrine tumors, bone metastases, and hematological malignancies demonstrates expanding clinical adoption. The integration of diagnostic and therapeutic radiopharmaceuticals creates synergistic market opportunities, enabling personalized treatment approaches based on molecular imaging results.
Regional market dynamics reveal significant variations in adoption patterns and growth potential. Developed markets maintain steady demand supported by established healthcare infrastructure and reimbursement frameworks. Emerging markets present substantial growth opportunities driven by healthcare system modernization, increasing healthcare expenditure, and expanding access to advanced medical technologies.
Regulatory frameworks and reimbursement policies significantly influence market dynamics across different regions. The approval processes for new radiopharmaceuticals and imaging protocols directly impact market accessibility and adoption rates. Healthcare economic considerations, including cost-effectiveness analyses and comparative effectiveness research, increasingly shape clinical decision-making and market penetration strategies.
Technological convergence between nuclear medicine and other imaging modalities creates hybrid market opportunities. The integration of nuclear imaging with computed tomography and magnetic resonance imaging enhances diagnostic capabilities while expanding potential market applications and clinical utility across diverse medical specialties.
Oncology applications constitute the largest market segment, with radiopharmaceuticals playing crucial roles in cancer detection, staging, and treatment monitoring. The growing incidence of cancer globally, particularly in developed nations, drives sustained demand for nuclear imaging procedures. Thyroid disorders represent another significant application area, where radioiodine therapy and diagnostic imaging maintain established clinical protocols with consistent utilization patterns.
Cardiology applications show substantial market potential, particularly in myocardial perfusion imaging and cardiac function assessment. The increasing burden of cardiovascular diseases, coupled with the superior diagnostic accuracy of nuclear medicine techniques compared to conventional imaging modalities, supports continued market expansion in this therapeutic area.
Therapeutic nuclear medicine applications exhibit accelerated growth trajectories, driven by targeted radiotherapy developments and theranostic approaches. Radiopharmaceutical therapy for neuroendocrine tumors, bone metastases, and hematological malignancies demonstrates expanding clinical adoption. The integration of diagnostic and therapeutic radiopharmaceuticals creates synergistic market opportunities, enabling personalized treatment approaches based on molecular imaging results.
Regional market dynamics reveal significant variations in adoption patterns and growth potential. Developed markets maintain steady demand supported by established healthcare infrastructure and reimbursement frameworks. Emerging markets present substantial growth opportunities driven by healthcare system modernization, increasing healthcare expenditure, and expanding access to advanced medical technologies.
Regulatory frameworks and reimbursement policies significantly influence market dynamics across different regions. The approval processes for new radiopharmaceuticals and imaging protocols directly impact market accessibility and adoption rates. Healthcare economic considerations, including cost-effectiveness analyses and comparative effectiveness research, increasingly shape clinical decision-making and market penetration strategies.
Technological convergence between nuclear medicine and other imaging modalities creates hybrid market opportunities. The integration of nuclear imaging with computed tomography and magnetic resonance imaging enhances diagnostic capabilities while expanding potential market applications and clinical utility across diverse medical specialties.
Current Status and Challenges of Ionizing Radiation in Nuclear Medicine
Nuclear medicine currently stands at a critical juncture where the therapeutic benefits of ionizing radiation must be carefully balanced against potential health risks. The field has achieved remarkable diagnostic and therapeutic capabilities through radiopharmaceuticals, yet faces mounting challenges related to radiation safety, dose optimization, and long-term patient outcomes.
The diagnostic applications of ionizing radiation in nuclear medicine have reached unprecedented sophistication. Single Photon Emission Computed Tomography (SPECT) and Positron Emission Tomography (PET) imaging provide exceptional molecular-level insights into physiological processes. These modalities utilize radioisotopes such as Technetium-99m, Fluorine-18, and Gallium-68 to visualize organ function, detect malignancies, and monitor treatment responses with remarkable precision.
Therapeutic nuclear medicine has expanded significantly with the development of targeted radiopharmaceuticals. Radioimmunotherapy using agents like Yttrium-90 and Lutetium-177 has demonstrated efficacy in treating neuroendocrine tumors and certain lymphomas. Additionally, radioisotope therapies for thyroid disorders using Iodine-131 remain cornerstone treatments, while emerging alpha-particle therapies show promise for treating metastatic cancers.
However, significant challenges persist in managing radiation exposure risks. Patient dose optimization remains complex due to individual variations in radiopharmaceutical uptake and clearance. Healthcare workers face occupational exposure concerns, particularly in high-volume nuclear medicine departments. The challenge extends to developing standardized protocols that minimize radiation dose while maintaining diagnostic accuracy and therapeutic efficacy.
Regulatory frameworks struggle to keep pace with rapidly evolving radiopharmaceutical technologies. Current guidelines often lack specificity for novel radioisotopes and delivery mechanisms, creating uncertainty in clinical implementation. The shortage of critical medical radioisotopes, exemplified by the global Molybdenum-99 supply crisis, highlights infrastructure vulnerabilities that threaten service continuity.
Technical limitations in radiation detection and measurement systems pose additional challenges. Accurate dosimetry for internal radiation exposure remains difficult, particularly for organs with complex geometries or heterogeneous uptake patterns. This uncertainty complicates risk-benefit assessments and personalized treatment planning.
The integration of artificial intelligence and machine learning technologies offers potential solutions for dose optimization and risk prediction, yet implementation faces validation and regulatory hurdles that must be systematically addressed.
The diagnostic applications of ionizing radiation in nuclear medicine have reached unprecedented sophistication. Single Photon Emission Computed Tomography (SPECT) and Positron Emission Tomography (PET) imaging provide exceptional molecular-level insights into physiological processes. These modalities utilize radioisotopes such as Technetium-99m, Fluorine-18, and Gallium-68 to visualize organ function, detect malignancies, and monitor treatment responses with remarkable precision.
Therapeutic nuclear medicine has expanded significantly with the development of targeted radiopharmaceuticals. Radioimmunotherapy using agents like Yttrium-90 and Lutetium-177 has demonstrated efficacy in treating neuroendocrine tumors and certain lymphomas. Additionally, radioisotope therapies for thyroid disorders using Iodine-131 remain cornerstone treatments, while emerging alpha-particle therapies show promise for treating metastatic cancers.
However, significant challenges persist in managing radiation exposure risks. Patient dose optimization remains complex due to individual variations in radiopharmaceutical uptake and clearance. Healthcare workers face occupational exposure concerns, particularly in high-volume nuclear medicine departments. The challenge extends to developing standardized protocols that minimize radiation dose while maintaining diagnostic accuracy and therapeutic efficacy.
Regulatory frameworks struggle to keep pace with rapidly evolving radiopharmaceutical technologies. Current guidelines often lack specificity for novel radioisotopes and delivery mechanisms, creating uncertainty in clinical implementation. The shortage of critical medical radioisotopes, exemplified by the global Molybdenum-99 supply crisis, highlights infrastructure vulnerabilities that threaten service continuity.
Technical limitations in radiation detection and measurement systems pose additional challenges. Accurate dosimetry for internal radiation exposure remains difficult, particularly for organs with complex geometries or heterogeneous uptake patterns. This uncertainty complicates risk-benefit assessments and personalized treatment planning.
The integration of artificial intelligence and machine learning technologies offers potential solutions for dose optimization and risk prediction, yet implementation faces validation and regulatory hurdles that must be systematically addressed.
Current Ionizing Radiation Solutions in Nuclear Medicine Practice
01 Protection against ionizing radiation damage
Compositions and methods for protecting biological systems, materials, or surfaces from the harmful effects of ionizing radiation. These formulations may include antioxidants, radical scavengers, and protective agents that mitigate radiation-induced damage at the cellular or molecular level. Applications include medical treatments, industrial processes, and protective coatings.- Protection against ionizing radiation damage: Compositions and methods for protecting biological systems, cells, and tissues from damage caused by ionizing radiation. These formulations may include antioxidants, radical scavengers, and protective agents that mitigate the harmful effects of ionizing rays on living organisms. The protective mechanisms involve neutralizing free radicals and preventing cellular damage at the molecular level.
- Detection and measurement of ionizing radiation: Devices, systems, and methods for detecting, measuring, and monitoring ionizing radiation exposure. These technologies include radiation detectors, dosimeters, and sensing apparatus that can accurately quantify radiation levels in various environments. The detection mechanisms may involve scintillation materials, semiconductor devices, or other radiation-sensitive components.
- Sterilization and disinfection using ionizing radiation: Methods and apparatus for sterilizing medical devices, pharmaceutical products, food items, and other materials using ionizing radiation. These processes utilize controlled doses of radiation to eliminate microorganisms, bacteria, and pathogens without significantly affecting the physical or chemical properties of the treated materials. Applications include medical equipment sterilization and food preservation.
- Shielding materials and barriers against ionizing radiation: Compositions, materials, and structural designs for shielding against ionizing radiation exposure. These include specialized materials with high atomic numbers, composite structures, and protective barriers that absorb or deflect ionizing rays. Applications range from medical imaging facilities to nuclear power plants and space exploration equipment.
- Medical and therapeutic applications of ionizing radiation: Methods and systems for using ionizing radiation in medical diagnosis and treatment, particularly in cancer therapy and imaging procedures. These applications involve controlled delivery of radiation to target specific tissues or tumors while minimizing exposure to healthy surrounding tissues. Techniques include radiotherapy, computed tomography, and other diagnostic imaging modalities.
02 Ionizing radiation sterilization processes
Methods and systems for sterilizing products, materials, or medical devices using ionizing radiation such as gamma rays, electron beams, or X-rays. These processes ensure microbial inactivation while maintaining product integrity. The technology is widely applied in pharmaceutical, medical device, and food industries for achieving sterility assurance levels.Expand Specific Solutions03 Detection and measurement of ionizing radiation
Devices, sensors, and methods for detecting, measuring, and monitoring ionizing radiation exposure. These systems may include dosimeters, radiation detectors, and imaging equipment that provide accurate quantification of radiation levels for safety, diagnostic, or research purposes. Applications span medical imaging, nuclear safety, and environmental monitoring.Expand Specific Solutions04 Radiation shielding materials and structures
Materials, composites, and structural designs specifically developed to attenuate or block ionizing radiation. These solutions incorporate high-density materials, specialized polymers, or layered structures to provide effective shielding in medical facilities, nuclear installations, and aerospace applications. The formulations balance protection efficiency with practical considerations such as weight and cost.Expand Specific Solutions05 Therapeutic applications of ionizing radiation
Methods and compositions for using ionizing radiation in therapeutic treatments, particularly in oncology for tumor ablation and cancer therapy. These approaches involve controlled delivery of radiation doses to target tissues while minimizing damage to surrounding healthy cells. The technology includes radiation sensitizers, dose optimization protocols, and combination therapies.Expand Specific Solutions
Key Players in Nuclear Medicine and Radiopharmaceutical Industry
The ionizing rays in nuclear medicine field represents a mature yet rapidly evolving sector, currently in an advanced development stage with significant market expansion driven by increasing cancer incidence and technological innovations. The market demonstrates substantial growth potential, particularly in targeted radiotherapy and precision medicine applications. Technology maturity varies across segments, with established players like Elekta AB and Varian Medical Systems leading conventional radiation therapy systems, while emerging companies such as Alpha Tau Medical Ltd. and Nanobiotix SA pioneer novel approaches like Alpha DaRT and nanoparticle-enhanced radiotherapy. The competitive landscape includes major medical device manufacturers (Koninklijke Philips NV), specialized biotechnology firms (G1 Therapeutics, Genentech), and government entities (Rosatom State Atomic Energy Corp.), alongside prominent research institutions. This diverse ecosystem reflects the field's transition from traditional radiotherapy toward personalized, targeted treatments with improved efficacy and reduced side effects.
Alpha Tau Medical Ltd.
Technical Solution: Alpha Tau Medical develops Alpha DaRT (Diffusing alpha-emitters Radiation Therapy), a novel cancer treatment technology that uses alpha-emitting radioisotopes delivered through intratumoral seeds. The system utilizes Radium-224 atoms that decay to release alpha particles with high linear energy transfer, providing potent localized radiation effects while minimizing systemic exposure. The alpha particles have a short range in tissue, concentrating radiation damage within the tumor microenvironment. This approach aims to achieve superior tumor control compared to conventional external beam radiation while reducing side effects through highly localized dose delivery and immune system activation.
Strengths: Highly localized radiation delivery, potential for enhanced immune response activation. Weaknesses: Limited to accessible tumors, early-stage clinical development with limited long-term data.
Elekta AB
Technical Solution: Elekta develops advanced linear accelerators and stereotactic radiosurgery systems that utilize high-energy ionizing radiation for precise cancer treatment. Their Unity MR-Linac combines MRI guidance with radiation therapy, enabling real-time tumor tracking and adaptive treatment planning. The company's solutions incorporate sophisticated dose calculation algorithms and beam shaping technologies to maximize therapeutic effects while minimizing radiation exposure to healthy tissues. Their systems feature advanced imaging capabilities for treatment verification and patient positioning accuracy.
Strengths: Market-leading precision in radiation delivery, excellent integration of imaging and treatment systems. Weaknesses: High equipment costs, requires specialized training and infrastructure.
Core Innovations in Radiation Safety and Dose Optimization
Monitoring radiation exposure and radiosensitivity using ambient ionization mass spectrometry
PatentWO2018009700A1
Innovation
- The use of surface desorption ionization mass spectrometry methods and apparatus for real-time analysis of molecular profiles to identify and quantify radiation exposure effects, allowing for personalized treatments and reducing the risk of secondary cancers by determining maximum radiation exposure and tailoring therapies based on individual sensitivity.
Method and Apparatus for Providing Depth-of-Interaction Detection Using Positron Emission Tomography (PET)
PatentActiveUS20090008562A1
Innovation
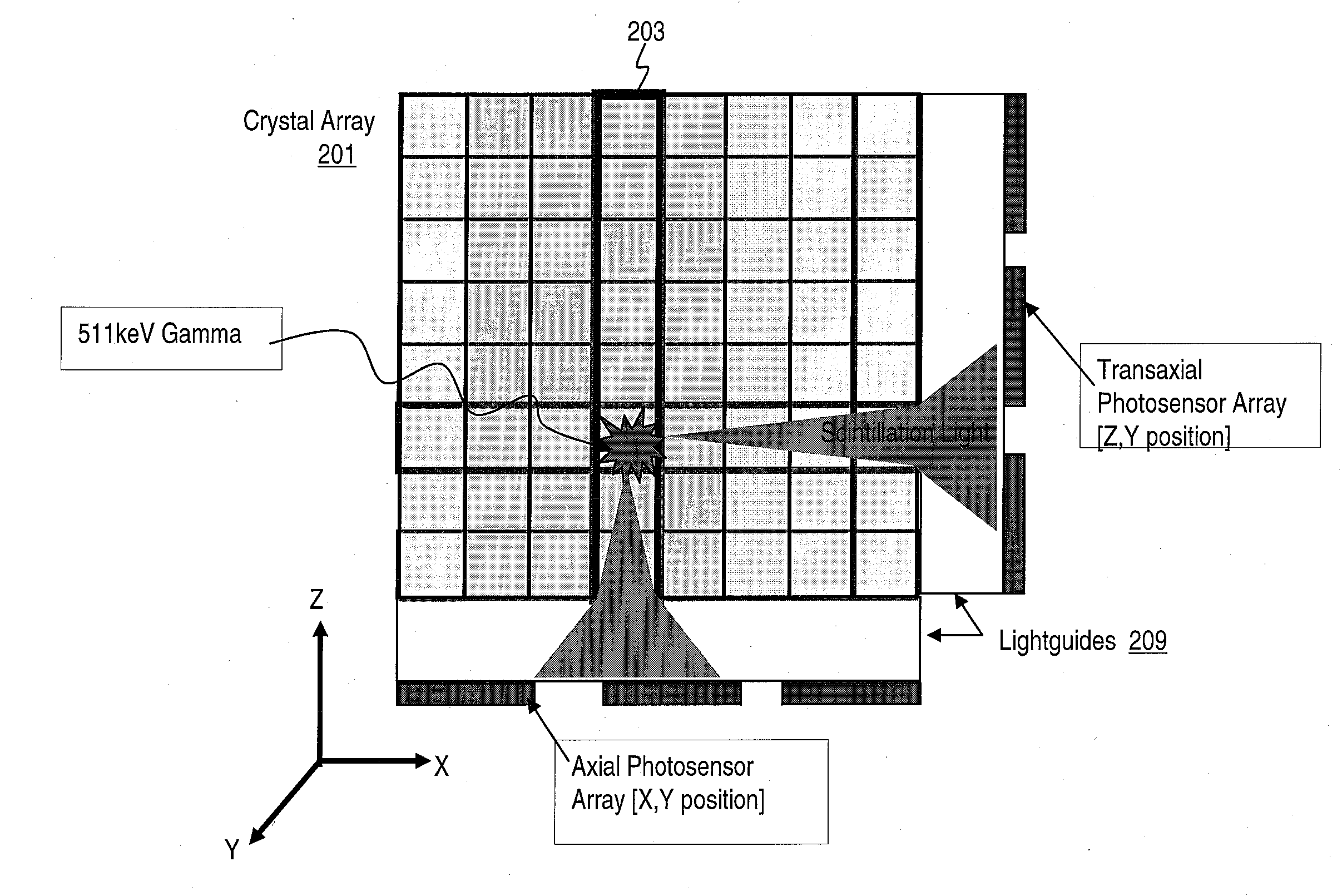
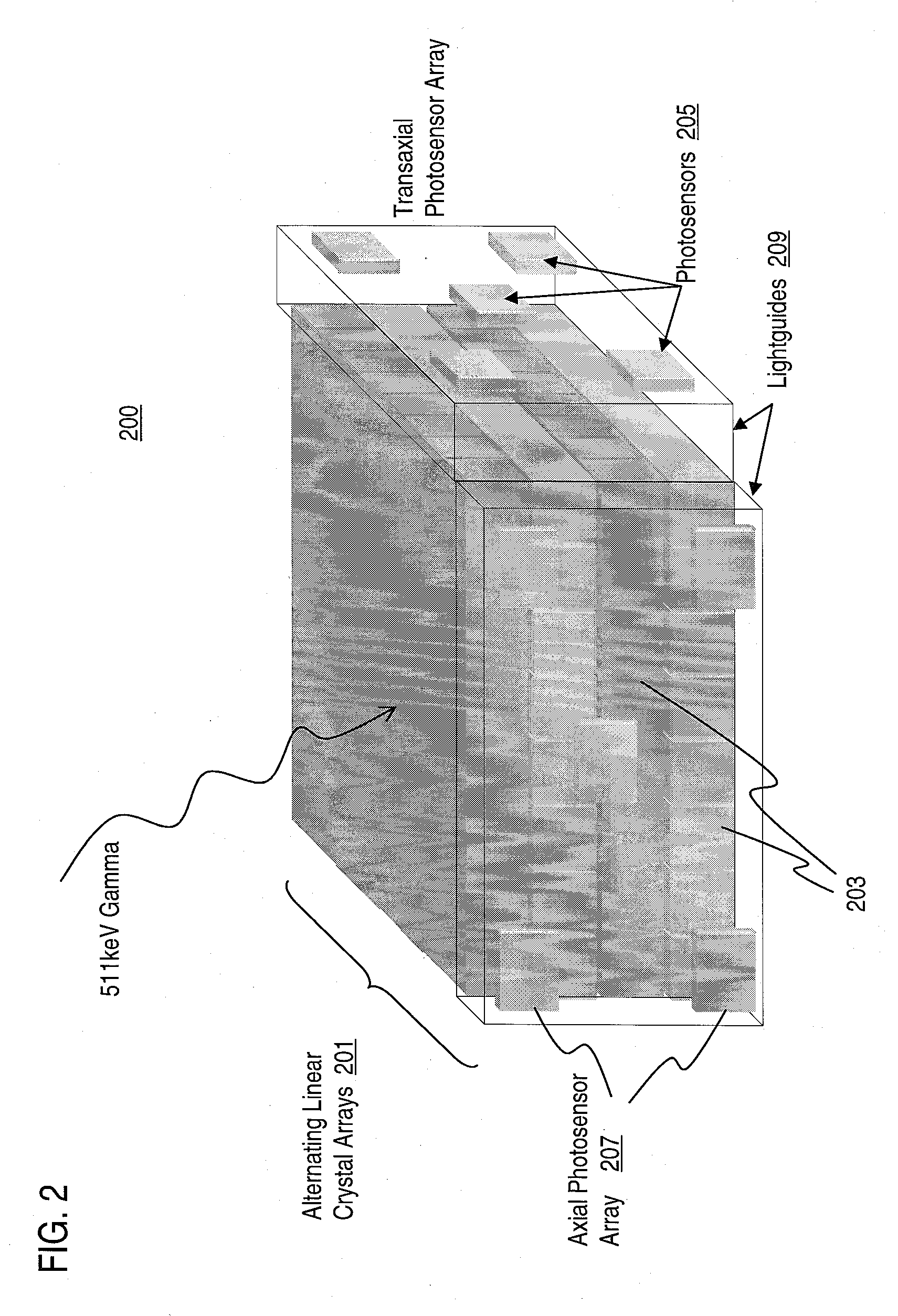
- A PET detector system using a scintillation block with alternating transaxial and axial linear arrays of pixellated scintillators, where light is shared between pixels to provide depth-of-interaction information, allowing for dual event localization and minimizing edge effects by reading out data from multiple sides of the scintillation array.
Regulatory Framework for Nuclear Medicine Radiation Safety
The regulatory framework for nuclear medicine radiation safety represents a comprehensive multi-layered system designed to protect patients, healthcare workers, and the general public from the potential harmful effects of ionizing radiation while enabling beneficial medical applications. This framework operates through international guidelines, national regulations, and institutional protocols that collectively establish standards for radiation protection in nuclear medicine practices.
At the international level, organizations such as the International Atomic Energy Agency (IAEA) and the International Commission on Radiological Protection (ICRP) provide fundamental safety standards and recommendations that serve as the foundation for national regulatory systems. These guidelines establish the basic principles of radiation protection including justification, optimization, and dose limitation, which form the cornerstone of all nuclear medicine safety protocols.
National regulatory authorities, such as the Nuclear Regulatory Commission in the United States and equivalent bodies in other countries, translate international standards into legally binding requirements. These regulations cover licensing requirements for nuclear medicine facilities, qualification standards for personnel, protocols for handling radioactive materials, and mandatory reporting procedures for radiation incidents. The regulatory scope encompasses everything from facility design and equipment specifications to waste management and emergency response procedures.
Institutional compliance mechanisms ensure that individual healthcare facilities adhere to regulatory requirements through comprehensive radiation safety programs. These programs typically include appointment of qualified radiation safety officers, establishment of radiation safety committees, implementation of ALARA (As Low As Reasonably Achievable) principles, and regular monitoring of radiation exposure levels for both patients and staff.
The regulatory framework also addresses specific aspects of nuclear medicine practice including patient dose optimization protocols, quality assurance requirements for imaging equipment, and standardized procedures for radiopharmaceutical administration. Regular inspections, audits, and compliance assessments ensure ongoing adherence to safety standards while accommodating technological advances and evolving best practices in the field.
At the international level, organizations such as the International Atomic Energy Agency (IAEA) and the International Commission on Radiological Protection (ICRP) provide fundamental safety standards and recommendations that serve as the foundation for national regulatory systems. These guidelines establish the basic principles of radiation protection including justification, optimization, and dose limitation, which form the cornerstone of all nuclear medicine safety protocols.
National regulatory authorities, such as the Nuclear Regulatory Commission in the United States and equivalent bodies in other countries, translate international standards into legally binding requirements. These regulations cover licensing requirements for nuclear medicine facilities, qualification standards for personnel, protocols for handling radioactive materials, and mandatory reporting procedures for radiation incidents. The regulatory scope encompasses everything from facility design and equipment specifications to waste management and emergency response procedures.
Institutional compliance mechanisms ensure that individual healthcare facilities adhere to regulatory requirements through comprehensive radiation safety programs. These programs typically include appointment of qualified radiation safety officers, establishment of radiation safety committees, implementation of ALARA (As Low As Reasonably Achievable) principles, and regular monitoring of radiation exposure levels for both patients and staff.
The regulatory framework also addresses specific aspects of nuclear medicine practice including patient dose optimization protocols, quality assurance requirements for imaging equipment, and standardized procedures for radiopharmaceutical administration. Regular inspections, audits, and compliance assessments ensure ongoing adherence to safety standards while accommodating technological advances and evolving best practices in the field.
Risk-Benefit Assessment in Nuclear Medicine Applications
The risk-benefit assessment in nuclear medicine applications represents a critical evaluation framework that balances therapeutic advantages against potential radiation-induced adverse effects. This assessment methodology has evolved significantly as nuclear medicine has expanded from simple diagnostic imaging to complex therapeutic interventions, necessitating sophisticated analytical approaches to quantify both immediate clinical benefits and long-term radiation risks.
Contemporary risk assessment models in nuclear medicine employ standardized metrics such as effective dose calculations, organ-specific absorbed doses, and lifetime attributable risk estimates. These quantitative frameworks enable clinicians to make informed decisions by comparing the probability of diagnostic accuracy or therapeutic success against the statistical likelihood of radiation-induced complications, including secondary malignancies and deterministic effects.
The benefit component of this assessment encompasses multiple dimensions, including diagnostic accuracy improvements, therapeutic efficacy rates, and quality-adjusted life years gained. For diagnostic procedures, benefits are measured through enhanced disease detection sensitivity, specificity improvements, and reduced need for invasive procedures. Therapeutic applications demonstrate benefits through tumor response rates, progression-free survival, and overall survival improvements.
Risk quantification involves sophisticated dosimetry calculations that account for patient-specific factors such as age, gender, organ sensitivity, and pre-existing conditions. Pediatric populations require specialized risk models due to increased radiosensitivity and longer life expectancy, while elderly patients may have different risk-benefit profiles based on competing mortality risks and reduced radiation sensitivity.
Modern assessment protocols integrate real-time monitoring systems and adaptive dosing strategies to optimize the risk-benefit ratio throughout treatment courses. These dynamic approaches utilize biomarkers, imaging responses, and toxicity monitoring to adjust radiation exposure levels, ensuring maximum therapeutic benefit while minimizing unnecessary radiation burden.
Regulatory frameworks worldwide have established standardized guidelines for conducting risk-benefit assessments, requiring comprehensive documentation of decision-making processes and patient-specific justifications for nuclear medicine procedures. These protocols ensure consistent application of assessment principles across different clinical settings and geographic regions.
Contemporary risk assessment models in nuclear medicine employ standardized metrics such as effective dose calculations, organ-specific absorbed doses, and lifetime attributable risk estimates. These quantitative frameworks enable clinicians to make informed decisions by comparing the probability of diagnostic accuracy or therapeutic success against the statistical likelihood of radiation-induced complications, including secondary malignancies and deterministic effects.
The benefit component of this assessment encompasses multiple dimensions, including diagnostic accuracy improvements, therapeutic efficacy rates, and quality-adjusted life years gained. For diagnostic procedures, benefits are measured through enhanced disease detection sensitivity, specificity improvements, and reduced need for invasive procedures. Therapeutic applications demonstrate benefits through tumor response rates, progression-free survival, and overall survival improvements.
Risk quantification involves sophisticated dosimetry calculations that account for patient-specific factors such as age, gender, organ sensitivity, and pre-existing conditions. Pediatric populations require specialized risk models due to increased radiosensitivity and longer life expectancy, while elderly patients may have different risk-benefit profiles based on competing mortality risks and reduced radiation sensitivity.
Modern assessment protocols integrate real-time monitoring systems and adaptive dosing strategies to optimize the risk-benefit ratio throughout treatment courses. These dynamic approaches utilize biomarkers, imaging responses, and toxicity monitoring to adjust radiation exposure levels, ensuring maximum therapeutic benefit while minimizing unnecessary radiation burden.
Regulatory frameworks worldwide have established standardized guidelines for conducting risk-benefit assessments, requiring comprehensive documentation of decision-making processes and patient-specific justifications for nuclear medicine procedures. These protocols ensure consistent application of assessment principles across different clinical settings and geographic regions.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!