Nanoparticles in Biosensors: Detection Limit Testing
FEB 26, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Nanoparticle Biosensor Background and Detection Goals
Nanoparticle-based biosensors represent a revolutionary advancement in analytical detection technology, emerging from the convergence of nanotechnology and bioanalytical chemistry. This field has evolved significantly since the early 2000s, when researchers first recognized the unique optical, electrical, and magnetic properties of nanoscale materials could be harnessed for biological detection applications.
The historical development of nanoparticle biosensors can be traced through several key phases. Initial research focused on gold nanoparticles due to their exceptional stability and surface plasmon resonance properties. Subsequently, quantum dots gained prominence for their tunable fluorescence characteristics, followed by magnetic nanoparticles for their separation and concentration capabilities. More recently, carbon-based nanomaterials, including carbon nanotubes and graphene derivatives, have expanded the technological landscape.
The fundamental principle underlying nanoparticle biosensors lies in their high surface-to-volume ratio, which provides abundant binding sites for target molecules while maintaining minimal sample volume requirements. This characteristic enables enhanced sensitivity compared to conventional detection methods. Additionally, the unique physicochemical properties of nanoparticles allow for multiple signal transduction mechanisms, including optical, electrochemical, and magnetic responses.
Current technological trends indicate a shift toward multifunctional nanoparticle platforms that combine detection, amplification, and signal processing capabilities within a single system. Hybrid nanostructures incorporating multiple materials are becoming increasingly prevalent, offering synergistic effects that surpass individual component performance.
The primary detection goals in nanoparticle biosensor development center on achieving ultra-low detection limits, typically targeting femtomolar to attomolar concentrations for critical biomarkers. These objectives are driven by clinical diagnostic requirements for early disease detection, environmental monitoring needs for trace contaminant identification, and food safety applications demanding rapid pathogen detection.
Specificity enhancement represents another crucial goal, as biosensors must distinguish target analytes from complex biological matrices containing numerous interfering substances. This challenge necessitates sophisticated surface functionalization strategies and selective recognition elements.
Real-time monitoring capabilities constitute a third major objective, enabling continuous or rapid point-of-care testing scenarios. This goal requires optimization of response kinetics and signal stability while maintaining detection sensitivity.
The historical development of nanoparticle biosensors can be traced through several key phases. Initial research focused on gold nanoparticles due to their exceptional stability and surface plasmon resonance properties. Subsequently, quantum dots gained prominence for their tunable fluorescence characteristics, followed by magnetic nanoparticles for their separation and concentration capabilities. More recently, carbon-based nanomaterials, including carbon nanotubes and graphene derivatives, have expanded the technological landscape.
The fundamental principle underlying nanoparticle biosensors lies in their high surface-to-volume ratio, which provides abundant binding sites for target molecules while maintaining minimal sample volume requirements. This characteristic enables enhanced sensitivity compared to conventional detection methods. Additionally, the unique physicochemical properties of nanoparticles allow for multiple signal transduction mechanisms, including optical, electrochemical, and magnetic responses.
Current technological trends indicate a shift toward multifunctional nanoparticle platforms that combine detection, amplification, and signal processing capabilities within a single system. Hybrid nanostructures incorporating multiple materials are becoming increasingly prevalent, offering synergistic effects that surpass individual component performance.
The primary detection goals in nanoparticle biosensor development center on achieving ultra-low detection limits, typically targeting femtomolar to attomolar concentrations for critical biomarkers. These objectives are driven by clinical diagnostic requirements for early disease detection, environmental monitoring needs for trace contaminant identification, and food safety applications demanding rapid pathogen detection.
Specificity enhancement represents another crucial goal, as biosensors must distinguish target analytes from complex biological matrices containing numerous interfering substances. This challenge necessitates sophisticated surface functionalization strategies and selective recognition elements.
Real-time monitoring capabilities constitute a third major objective, enabling continuous or rapid point-of-care testing scenarios. This goal requires optimization of response kinetics and signal stability while maintaining detection sensitivity.
Market Demand for Ultra-Sensitive Biosensor Detection
The global biosensor market is experiencing unprecedented growth driven by the critical need for ultra-sensitive detection capabilities across multiple sectors. Healthcare diagnostics represents the largest demand segment, where early disease detection and point-of-care testing require detection limits at femtomolar to attomolar concentrations. The COVID-19 pandemic has accelerated this demand, highlighting the necessity for rapid, accurate diagnostic tools capable of detecting minimal viral loads in asymptomatic patients.
Environmental monitoring applications constitute another significant market driver, particularly for detecting trace contaminants in water supplies, air quality assessment, and soil contamination analysis. Regulatory agencies worldwide are implementing stricter detection standards for pollutants, pesticides, and heavy metals, necessitating biosensors with enhanced sensitivity levels that traditional methods cannot achieve.
The food safety sector demands ultra-sensitive detection for pathogen identification, allergen screening, and toxin detection throughout the supply chain. Consumer awareness regarding food quality and safety has intensified market pressure for real-time monitoring systems capable of detecting single bacterial cells or trace amounts of harmful substances.
Pharmaceutical and biotechnology industries require ultra-sensitive biosensors for drug discovery, therapeutic monitoring, and biomarker identification. The growing emphasis on personalized medicine drives demand for devices capable of detecting minute concentrations of biomarkers that indicate treatment efficacy or adverse reactions.
Defense and security applications represent an emerging high-value market segment, where ultra-sensitive detection of biological warfare agents, explosives, and chemical threats is paramount. Government investments in homeland security technologies continue to fuel demand for advanced biosensor capabilities.
Market growth is further accelerated by the miniaturization trend in medical devices and the Internet of Things integration in healthcare systems. Wearable health monitors and implantable sensors require ultra-sensitive detection capabilities while maintaining compact form factors and low power consumption.
The aging global population and increasing prevalence of chronic diseases create sustained demand for continuous monitoring solutions. Ultra-sensitive biosensors enable early intervention strategies, reducing healthcare costs and improving patient outcomes through timely detection of disease progression markers.
Environmental monitoring applications constitute another significant market driver, particularly for detecting trace contaminants in water supplies, air quality assessment, and soil contamination analysis. Regulatory agencies worldwide are implementing stricter detection standards for pollutants, pesticides, and heavy metals, necessitating biosensors with enhanced sensitivity levels that traditional methods cannot achieve.
The food safety sector demands ultra-sensitive detection for pathogen identification, allergen screening, and toxin detection throughout the supply chain. Consumer awareness regarding food quality and safety has intensified market pressure for real-time monitoring systems capable of detecting single bacterial cells or trace amounts of harmful substances.
Pharmaceutical and biotechnology industries require ultra-sensitive biosensors for drug discovery, therapeutic monitoring, and biomarker identification. The growing emphasis on personalized medicine drives demand for devices capable of detecting minute concentrations of biomarkers that indicate treatment efficacy or adverse reactions.
Defense and security applications represent an emerging high-value market segment, where ultra-sensitive detection of biological warfare agents, explosives, and chemical threats is paramount. Government investments in homeland security technologies continue to fuel demand for advanced biosensor capabilities.
Market growth is further accelerated by the miniaturization trend in medical devices and the Internet of Things integration in healthcare systems. Wearable health monitors and implantable sensors require ultra-sensitive detection capabilities while maintaining compact form factors and low power consumption.
The aging global population and increasing prevalence of chronic diseases create sustained demand for continuous monitoring solutions. Ultra-sensitive biosensors enable early intervention strategies, reducing healthcare costs and improving patient outcomes through timely detection of disease progression markers.
Current State and Detection Limit Challenges in Nanoparticle Biosensors
Nanoparticle-based biosensors have emerged as a transformative technology in analytical chemistry and biomedical diagnostics, leveraging the unique optical, electrical, and magnetic properties of nanoscale materials. The current landscape encompasses diverse nanoparticle platforms including gold nanoparticles, quantum dots, magnetic nanoparticles, and carbon-based nanomaterials, each offering distinct advantages for specific detection applications. These systems have demonstrated remarkable sensitivity improvements over conventional biosensing methods, with some achieving detection limits in the femtomolar to attomolar range.
The integration of nanoparticles into biosensor architectures has enabled signal amplification through various mechanisms, including plasmonic enhancement, fluorescence resonance energy transfer, and electrochemical catalysis. Surface plasmon resonance-based sensors utilizing gold nanoparticles have shown particular promise in label-free detection, while quantum dot-based systems excel in multiplexed analysis capabilities. Recent developments in hybrid nanocomposite materials have further expanded the detection capabilities, combining multiple functionalities within single sensing platforms.
Despite significant technological advances, several critical challenges continue to limit the practical implementation of nanoparticle biosensors. The primary obstacle remains achieving consistent and reproducible detection limits across different operational conditions and sample matrices. Variability in nanoparticle synthesis, surface functionalization, and bioconjugation processes directly impacts sensor performance and reliability. Additionally, the complex interplay between nanoparticle properties and biological environments often leads to unpredictable signal responses and baseline drift.
Matrix interference effects pose another substantial challenge, particularly in complex biological samples where proteins, salts, and other biomolecules can significantly affect nanoparticle behavior and sensor response. The phenomenon of protein corona formation on nanoparticle surfaces can alter their binding kinetics and specificity, leading to compromised detection limits and increased false positive rates. Furthermore, the long-term stability of nanoparticle-based sensors remains problematic, with degradation of optical properties and biorecognition elements affecting sustained performance.
Current detection limit achievements vary significantly across different nanoparticle platforms and target analytes. While laboratory demonstrations have reported impressive sensitivity levels, translating these results to real-world applications remains challenging due to environmental factors, sample preparation requirements, and instrument standardization issues.
The integration of nanoparticles into biosensor architectures has enabled signal amplification through various mechanisms, including plasmonic enhancement, fluorescence resonance energy transfer, and electrochemical catalysis. Surface plasmon resonance-based sensors utilizing gold nanoparticles have shown particular promise in label-free detection, while quantum dot-based systems excel in multiplexed analysis capabilities. Recent developments in hybrid nanocomposite materials have further expanded the detection capabilities, combining multiple functionalities within single sensing platforms.
Despite significant technological advances, several critical challenges continue to limit the practical implementation of nanoparticle biosensors. The primary obstacle remains achieving consistent and reproducible detection limits across different operational conditions and sample matrices. Variability in nanoparticle synthesis, surface functionalization, and bioconjugation processes directly impacts sensor performance and reliability. Additionally, the complex interplay between nanoparticle properties and biological environments often leads to unpredictable signal responses and baseline drift.
Matrix interference effects pose another substantial challenge, particularly in complex biological samples where proteins, salts, and other biomolecules can significantly affect nanoparticle behavior and sensor response. The phenomenon of protein corona formation on nanoparticle surfaces can alter their binding kinetics and specificity, leading to compromised detection limits and increased false positive rates. Furthermore, the long-term stability of nanoparticle-based sensors remains problematic, with degradation of optical properties and biorecognition elements affecting sustained performance.
Current detection limit achievements vary significantly across different nanoparticle platforms and target analytes. While laboratory demonstrations have reported impressive sensitivity levels, translating these results to real-world applications remains challenging due to environmental factors, sample preparation requirements, and instrument standardization issues.
Existing Detection Limit Testing Methods and Solutions
01 Optical detection methods for nanoparticles
Optical detection techniques utilize light-based methods to identify and quantify nanoparticles at low concentrations. These methods include fluorescence spectroscopy, surface plasmon resonance, and light scattering techniques. The detection limit can be improved by enhancing signal intensity through various optical configurations and using high-sensitivity detectors. These approaches enable the detection of individual nanoparticles or very low concentrations in solution.- Optical detection methods for nanoparticles: Optical detection techniques utilize light-based methods to identify and quantify nanoparticles at low concentrations. These methods include fluorescence spectroscopy, surface plasmon resonance, and light scattering techniques. The detection limit can be improved by enhancing signal intensity through various optical configurations and using high-sensitivity detectors. These approaches enable the detection of individual nanoparticles or extremely low concentrations in various sample matrices.
- Electrochemical sensing for nanoparticle detection: Electrochemical methods provide sensitive detection of nanoparticles by measuring electrical signals generated through redox reactions or changes in conductivity. These techniques can achieve low detection limits by utilizing modified electrodes, nanomaterial-based sensors, and signal amplification strategies. The methods are particularly effective for metallic nanoparticles and can be adapted for various environmental and biological samples.
- Mass spectrometry-based nanoparticle analysis: Mass spectrometry techniques enable precise detection and characterization of nanoparticles by measuring their mass-to-charge ratio. Single particle inductively coupled plasma mass spectrometry is particularly effective for achieving ultra-low detection limits. These methods can determine particle size distribution, composition, and concentration simultaneously, making them valuable for comprehensive nanoparticle analysis in complex matrices.
- Biosensor platforms for nanoparticle detection: Biosensor-based approaches integrate biological recognition elements with transduction mechanisms to detect nanoparticles with high specificity and sensitivity. These platforms may employ antibodies, aptamers, or other biomolecules for selective nanoparticle capture and detection. The integration of nanomaterials in biosensor design can further enhance detection limits through signal amplification and improved binding efficiency.
- Microfluidic and lab-on-chip systems for nanoparticle quantification: Microfluidic devices and lab-on-chip technologies provide miniaturized platforms for nanoparticle detection with reduced sample volumes and enhanced sensitivity. These systems integrate sample preparation, separation, and detection functions in compact formats. The controlled flow dynamics and small detection volumes enable improved detection limits compared to conventional methods, making them suitable for point-of-care and field applications.
02 Electrochemical sensing for nanoparticle detection
Electrochemical methods provide sensitive detection of nanoparticles by measuring electrical signals generated through redox reactions or changes in conductivity. These techniques can achieve low detection limits by utilizing modified electrodes, nanomaterial-based sensors, and signal amplification strategies. The methods are particularly effective for metallic nanoparticles and can be adapted for various sample matrices.Expand Specific Solutions03 Mass spectrometry-based nanoparticle analysis
Mass spectrometry techniques enable precise detection and characterization of nanoparticles with excellent sensitivity. Single particle inductively coupled plasma mass spectrometry and related methods can detect individual nanoparticles and determine their size distribution and composition. These approaches achieve detection limits in the parts per trillion range and provide detailed information about nanoparticle properties.Expand Specific Solutions04 Biosensor-based nanoparticle detection systems
Biosensors integrate biological recognition elements with transduction mechanisms to detect nanoparticles with high specificity and sensitivity. These systems employ antibodies, aptamers, or other biomolecules to selectively bind target nanoparticles, generating measurable signals. The detection limit can be enhanced through signal amplification techniques and optimized sensor design, enabling detection at very low concentrations in complex biological samples.Expand Specific Solutions05 Microfluidic and lab-on-chip detection platforms
Microfluidic devices and lab-on-chip systems provide miniaturized platforms for nanoparticle detection with reduced sample volumes and improved detection limits. These platforms integrate sample preparation, separation, and detection functions in compact devices. The small dimensions enhance sensitivity through increased surface-to-volume ratios and enable rapid analysis with minimal reagent consumption.Expand Specific Solutions
Key Players in Nanoparticle Biosensor Industry
The nanoparticles in biosensors market for detection limit testing represents a rapidly evolving sector in the early-to-mid growth stage, driven by increasing demand for ultra-sensitive diagnostic tools. The market demonstrates significant expansion potential, particularly in healthcare and environmental monitoring applications. Technology maturity varies considerably across players, with established corporations like Koninklijke Philips NV and Sony Group Corp. leveraging advanced manufacturing capabilities, while research institutions including University of Rochester, Technical University of Denmark, and Peking University focus on fundamental breakthroughs in nanoparticle synthesis and sensor integration. Leading academic centers such as Institute of Science Tokyo and Politecnico di Milano are advancing detection methodologies, while specialized companies like Sumitomo Electric Industries contribute materials expertise. The competitive landscape shows a hybrid ecosystem where industrial giants collaborate with research institutions to overcome current detection limit barriers and commercialize next-generation biosensing platforms.
Koninklijke Philips NV
Technical Solution: Philips has developed advanced nanoparticle-based biosensor platforms focusing on point-of-care diagnostics with enhanced detection limits. Their technology integrates gold nanoparticles and magnetic nanoparticles in immunoassay formats, achieving detection limits in the picomolar range for cardiac biomarkers and infectious disease markers. The company's biosensor systems utilize surface plasmon resonance enhancement and magnetic separation techniques to improve signal-to-noise ratios, enabling detection of target analytes at concentrations as low as 0.1 ng/mL. Their proprietary nanoparticle functionalization methods ensure stable conjugation with antibodies while maintaining high sensitivity and specificity in complex biological matrices.
Strengths: Strong commercial presence in healthcare diagnostics, robust manufacturing capabilities, extensive clinical validation experience. Weaknesses: Higher cost compared to traditional methods, limited customization for research applications, dependency on proprietary reagents.
The Regents of the University of California
Technical Solution: UC researchers have pioneered quantum dot-based biosensors with exceptional detection limits for single-molecule detection applications. Their nanoparticle biosensor platforms utilize semiconductor quantum dots and plasmonic nanostructures to achieve detection limits down to attomolar concentrations. The technology employs FRET-based mechanisms and surface-enhanced fluorescence to amplify signals from target biomolecules. Recent developments include multiplexed detection systems capable of simultaneously detecting multiple biomarkers with detection limits of 10^-18 M for DNA sequences and 10^-15 M for protein targets. Their innovative approach combines microfluidics with nanoparticle arrays to create highly sensitive diagnostic platforms suitable for early disease detection and environmental monitoring applications.
Strengths: Cutting-edge research capabilities, strong intellectual property portfolio, excellent academic-industry collaborations. Weaknesses: Technology transfer challenges, limited manufacturing scalability, longer development timelines for commercialization.
Core Innovations in Nanoparticle Detection Enhancement
Method for the topographically-selective passivation of micro- and nanoscale devices
PatentInactiveUS20190369092A1
Innovation
- A topographically selective passivation method using self-assembling hydrogel nanoparticles to mask non-sensing regions, allowing specific binding of capture molecules only to the active sensing areas on substrates with distinct topography, such as photonic crystals.
Plasmon-enhanced fluorescence biochemical sensors
PatentWO2023154567A1
Innovation
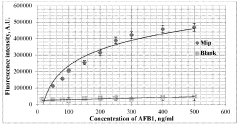
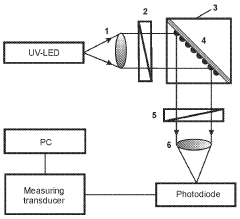
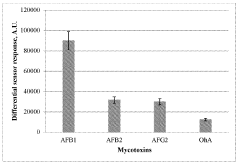
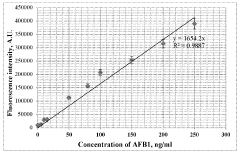
- A plasmon-enhanced fluorescence sensor platform utilizing molecularly imprinted polymer membranes with embedded silver nanoparticles that selectively bind aflatoxins, enhancing fluorescence detection through localized surface plasmon resonance, thereby achieving ultralow detection limits and improved selectivity.
Standardization and Validation Protocols for Detection Testing
The establishment of robust standardization and validation protocols for detection limit testing in nanoparticle-based biosensors represents a critical foundation for ensuring reproducible and reliable analytical performance across different platforms and applications. Current standardization efforts focus on harmonizing measurement methodologies, calibration procedures, and performance metrics to enable meaningful comparison of detection capabilities between various biosensor systems.
International standardization organizations, including ISO and ASTM, are actively developing comprehensive guidelines that address the unique challenges posed by nanoparticle-enhanced detection systems. These protocols encompass sample preparation procedures, environmental control parameters, and statistical analysis methods specifically tailored for nanoscale detection phenomena. The standardization framework emphasizes the importance of establishing reference materials and certified standards that account for the complex interactions between nanoparticles and biological analytes.
Validation protocols for detection limit determination require multi-laboratory collaborative studies to establish method precision, accuracy, and robustness across different operational conditions. These validation frameworks incorporate systematic approaches for evaluating inter-laboratory reproducibility, operator variability, and instrument-to-instrument consistency. The protocols mandate rigorous documentation of experimental conditions, including buffer compositions, temperature controls, and measurement timing sequences that significantly impact nanoparticle-based detection performance.
Quality assurance measures within validation protocols include the implementation of control charts, proficiency testing programs, and continuous monitoring systems that track long-term analytical performance. These measures ensure that detection limit claims remain valid throughout the operational lifetime of biosensor systems and provide mechanisms for identifying systematic drift or degradation in analytical capabilities.
The validation process also addresses the challenge of matrix effects and interference studies, requiring comprehensive evaluation of biosensor performance in complex biological samples. This includes establishing acceptance criteria for method selectivity, specificity, and the ability to maintain stated detection limits in the presence of potential interferents commonly encountered in real-world applications.
International standardization organizations, including ISO and ASTM, are actively developing comprehensive guidelines that address the unique challenges posed by nanoparticle-enhanced detection systems. These protocols encompass sample preparation procedures, environmental control parameters, and statistical analysis methods specifically tailored for nanoscale detection phenomena. The standardization framework emphasizes the importance of establishing reference materials and certified standards that account for the complex interactions between nanoparticles and biological analytes.
Validation protocols for detection limit determination require multi-laboratory collaborative studies to establish method precision, accuracy, and robustness across different operational conditions. These validation frameworks incorporate systematic approaches for evaluating inter-laboratory reproducibility, operator variability, and instrument-to-instrument consistency. The protocols mandate rigorous documentation of experimental conditions, including buffer compositions, temperature controls, and measurement timing sequences that significantly impact nanoparticle-based detection performance.
Quality assurance measures within validation protocols include the implementation of control charts, proficiency testing programs, and continuous monitoring systems that track long-term analytical performance. These measures ensure that detection limit claims remain valid throughout the operational lifetime of biosensor systems and provide mechanisms for identifying systematic drift or degradation in analytical capabilities.
The validation process also addresses the challenge of matrix effects and interference studies, requiring comprehensive evaluation of biosensor performance in complex biological samples. This includes establishing acceptance criteria for method selectivity, specificity, and the ability to maintain stated detection limits in the presence of potential interferents commonly encountered in real-world applications.
Safety Assessment of Nanoparticles in Biosensor Applications
The integration of nanoparticles in biosensor applications has raised significant safety concerns that require comprehensive assessment frameworks. While these nanoscale materials offer unprecedented sensitivity and specificity in biological detection systems, their potential toxicological effects on human health and environmental systems demand rigorous evaluation protocols. The unique physicochemical properties that make nanoparticles effective in biosensing applications, such as high surface-to-volume ratios and enhanced reactivity, simultaneously contribute to their potential biological hazards.
Cytotoxicity represents the primary safety concern in nanoparticle-based biosensors, particularly for applications involving direct contact with biological tissues or fluids. Metal oxide nanoparticles commonly used in biosensors, including titanium dioxide, zinc oxide, and silver nanoparticles, have demonstrated varying degrees of cellular toxicity depending on size, surface chemistry, and concentration. In vitro studies have revealed that nanoparticles can induce oxidative stress, membrane damage, and DNA fragmentation in various cell lines, with smaller particles generally exhibiting higher toxicity due to increased cellular uptake and intracellular accumulation.
Biocompatibility assessment protocols for nanoparticle-based biosensors must encompass multiple evaluation criteria, including hemolysis testing, inflammatory response analysis, and long-term bioaccumulation studies. The ISO 10993 series provides standardized guidelines for biological evaluation of medical devices, which can be adapted for nanoparticle biosensor applications. However, conventional toxicity testing methods may not adequately capture the unique behavior of nanomaterials, necessitating the development of nano-specific assessment protocols that consider particle dissolution, protein corona formation, and cellular internalization mechanisms.
Environmental safety considerations extend beyond immediate human health impacts to encompass broader ecological effects. Nanoparticles released from biosensor devices during manufacturing, use, or disposal can potentially accumulate in soil and water systems, affecting microbial communities and aquatic organisms. Life cycle assessment approaches are increasingly being employed to evaluate the environmental footprint of nanoparticle biosensors from production to end-of-life disposal.
Regulatory frameworks for nanoparticle safety in biosensor applications remain fragmented across different jurisdictions. The European Union's REACH regulation and the FDA's nanotechnology guidance documents provide some oversight, but specific standards for biosensor applications are still evolving. Risk assessment methodologies must balance the beneficial applications of nanoparticle biosensors against potential adverse effects, requiring interdisciplinary collaboration between toxicologists, materials scientists, and regulatory experts to establish appropriate safety thresholds and monitoring protocols.
Cytotoxicity represents the primary safety concern in nanoparticle-based biosensors, particularly for applications involving direct contact with biological tissues or fluids. Metal oxide nanoparticles commonly used in biosensors, including titanium dioxide, zinc oxide, and silver nanoparticles, have demonstrated varying degrees of cellular toxicity depending on size, surface chemistry, and concentration. In vitro studies have revealed that nanoparticles can induce oxidative stress, membrane damage, and DNA fragmentation in various cell lines, with smaller particles generally exhibiting higher toxicity due to increased cellular uptake and intracellular accumulation.
Biocompatibility assessment protocols for nanoparticle-based biosensors must encompass multiple evaluation criteria, including hemolysis testing, inflammatory response analysis, and long-term bioaccumulation studies. The ISO 10993 series provides standardized guidelines for biological evaluation of medical devices, which can be adapted for nanoparticle biosensor applications. However, conventional toxicity testing methods may not adequately capture the unique behavior of nanomaterials, necessitating the development of nano-specific assessment protocols that consider particle dissolution, protein corona formation, and cellular internalization mechanisms.
Environmental safety considerations extend beyond immediate human health impacts to encompass broader ecological effects. Nanoparticles released from biosensor devices during manufacturing, use, or disposal can potentially accumulate in soil and water systems, affecting microbial communities and aquatic organisms. Life cycle assessment approaches are increasingly being employed to evaluate the environmental footprint of nanoparticle biosensors from production to end-of-life disposal.
Regulatory frameworks for nanoparticle safety in biosensor applications remain fragmented across different jurisdictions. The European Union's REACH regulation and the FDA's nanotechnology guidance documents provide some oversight, but specific standards for biosensor applications are still evolving. Risk assessment methodologies must balance the beneficial applications of nanoparticle biosensors against potential adverse effects, requiring interdisciplinary collaboration between toxicologists, materials scientists, and regulatory experts to establish appropriate safety thresholds and monitoring protocols.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!