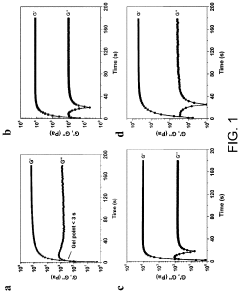
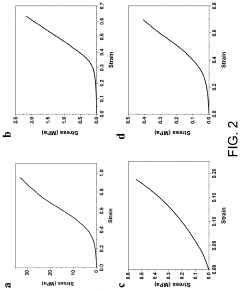
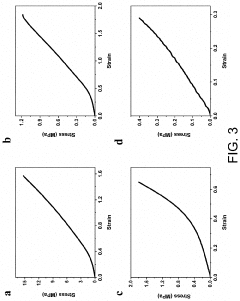
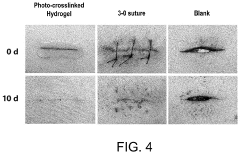
Optimize Hydrogel Crosslinking for Enhanced Mechanical Strength
MAY 12, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Hydrogel Crosslinking Background and Mechanical Goals
Hydrogels represent a class of three-dimensional polymeric networks capable of absorbing substantial amounts of water while maintaining their structural integrity. These materials have evolved from simple crosslinked polymer systems to sophisticated biomimetic structures that closely resemble natural tissues. The fundamental principle underlying hydrogel formation involves the creation of crosslinks between polymer chains, which can be achieved through various mechanisms including covalent bonding, ionic interactions, hydrogen bonding, and physical entanglements.
The historical development of hydrogels traces back to the 1960s when Wichterle and Lím first synthesized poly(2-hydroxyethyl methacrylate) for contact lens applications. Since then, the field has witnessed remarkable advancement through the introduction of natural polymers such as alginate, chitosan, and collagen, as well as synthetic alternatives like polyethylene glycol and polyacrylamide. The evolution has been driven by the need to balance water content with mechanical performance, leading to innovative crosslinking strategies.
Contemporary hydrogel applications span biomedical engineering, drug delivery systems, tissue engineering scaffolds, wound healing materials, and soft robotics. Each application domain presents unique mechanical requirements that challenge traditional crosslinking approaches. The mechanical properties of hydrogels are intrinsically linked to their crosslinking density, network topology, and polymer composition, creating a complex optimization landscape.
The primary mechanical goals for optimized hydrogel crosslinking encompass achieving enhanced tensile strength, improved elastic modulus, superior fatigue resistance, and controlled viscoelastic behavior. These objectives must be balanced against maintaining biocompatibility, preserving swelling capacity, and ensuring appropriate degradation kinetics for specific applications. The challenge lies in developing crosslinking strategies that can deliver robust mechanical performance without compromising the inherent advantages of hydrogel materials.
Current research focuses on multi-scale crosslinking approaches that combine different bonding mechanisms to create hierarchical network structures. These strategies aim to achieve mechanical properties comparable to natural tissues while maintaining the processability and functionality that make hydrogels attractive for advanced applications. The optimization process requires careful consideration of crosslinking kinetics, network homogeneity, and long-term stability under physiological conditions.
The historical development of hydrogels traces back to the 1960s when Wichterle and Lím first synthesized poly(2-hydroxyethyl methacrylate) for contact lens applications. Since then, the field has witnessed remarkable advancement through the introduction of natural polymers such as alginate, chitosan, and collagen, as well as synthetic alternatives like polyethylene glycol and polyacrylamide. The evolution has been driven by the need to balance water content with mechanical performance, leading to innovative crosslinking strategies.
Contemporary hydrogel applications span biomedical engineering, drug delivery systems, tissue engineering scaffolds, wound healing materials, and soft robotics. Each application domain presents unique mechanical requirements that challenge traditional crosslinking approaches. The mechanical properties of hydrogels are intrinsically linked to their crosslinking density, network topology, and polymer composition, creating a complex optimization landscape.
The primary mechanical goals for optimized hydrogel crosslinking encompass achieving enhanced tensile strength, improved elastic modulus, superior fatigue resistance, and controlled viscoelastic behavior. These objectives must be balanced against maintaining biocompatibility, preserving swelling capacity, and ensuring appropriate degradation kinetics for specific applications. The challenge lies in developing crosslinking strategies that can deliver robust mechanical performance without compromising the inherent advantages of hydrogel materials.
Current research focuses on multi-scale crosslinking approaches that combine different bonding mechanisms to create hierarchical network structures. These strategies aim to achieve mechanical properties comparable to natural tissues while maintaining the processability and functionality that make hydrogels attractive for advanced applications. The optimization process requires careful consideration of crosslinking kinetics, network homogeneity, and long-term stability under physiological conditions.
Market Demand for High-Strength Hydrogel Applications
The global hydrogel market is experiencing unprecedented growth driven by diverse applications requiring enhanced mechanical properties. Biomedical applications represent the largest segment, where high-strength hydrogels are essential for load-bearing tissue engineering scaffolds, particularly in cartilage and bone regeneration. The aging population worldwide has intensified demand for durable implantable devices and prosthetics that can withstand physiological stresses while maintaining biocompatibility.
Wound care management constitutes another significant market driver, with advanced wound dressings requiring superior tensile strength to prevent tearing during application and removal. Chronic wound treatment, affecting millions globally, demands hydrogels that maintain structural integrity under dynamic conditions while providing optimal healing environments.
The pharmaceutical industry increasingly relies on mechanically robust hydrogels for controlled drug delivery systems. Injectable hydrogels must possess sufficient strength to maintain their structure post-injection while enabling sustained therapeutic release. This application has gained particular traction in oncology and diabetes management, where precise drug delivery timing is critical.
Industrial applications are emerging as a substantial growth area, with high-strength hydrogels finding use in water treatment, agriculture, and smart materials. Water purification systems require hydrogels capable of withstanding high pressure and flow rates while maintaining filtration efficiency. Agricultural applications demand hydrogels that can endure soil mechanical stress while providing sustained water and nutrient release.
The cosmetics and personal care sector represents a rapidly expanding market segment, where mechanically enhanced hydrogels enable longer-lasting skincare products and more durable contact lenses. Consumer expectations for product longevity and performance have driven manufacturers to seek hydrogels with superior mechanical properties.
Emerging applications in soft robotics and flexible electronics are creating new market opportunities. These sectors require hydrogels with exceptional mechanical strength while maintaining flexibility and conductivity. The growing Internet of Things ecosystem and wearable technology market are particularly driving demand for mechanically robust, biocompatible hydrogels that can function reliably under repeated deformation cycles.
Wound care management constitutes another significant market driver, with advanced wound dressings requiring superior tensile strength to prevent tearing during application and removal. Chronic wound treatment, affecting millions globally, demands hydrogels that maintain structural integrity under dynamic conditions while providing optimal healing environments.
The pharmaceutical industry increasingly relies on mechanically robust hydrogels for controlled drug delivery systems. Injectable hydrogels must possess sufficient strength to maintain their structure post-injection while enabling sustained therapeutic release. This application has gained particular traction in oncology and diabetes management, where precise drug delivery timing is critical.
Industrial applications are emerging as a substantial growth area, with high-strength hydrogels finding use in water treatment, agriculture, and smart materials. Water purification systems require hydrogels capable of withstanding high pressure and flow rates while maintaining filtration efficiency. Agricultural applications demand hydrogels that can endure soil mechanical stress while providing sustained water and nutrient release.
The cosmetics and personal care sector represents a rapidly expanding market segment, where mechanically enhanced hydrogels enable longer-lasting skincare products and more durable contact lenses. Consumer expectations for product longevity and performance have driven manufacturers to seek hydrogels with superior mechanical properties.
Emerging applications in soft robotics and flexible electronics are creating new market opportunities. These sectors require hydrogels with exceptional mechanical strength while maintaining flexibility and conductivity. The growing Internet of Things ecosystem and wearable technology market are particularly driving demand for mechanically robust, biocompatible hydrogels that can function reliably under repeated deformation cycles.
Current Crosslinking Methods and Mechanical Limitations
Hydrogel crosslinking represents a fundamental approach to enhance mechanical properties through the formation of three-dimensional network structures. Current methodologies primarily encompass chemical crosslinking, physical crosslinking, and hybrid approaches, each presenting distinct advantages and inherent limitations that constrain optimal mechanical performance.
Chemical crosslinking methods utilize covalent bond formation to create permanent network structures. Free radical polymerization remains the most prevalent technique, employing initiators such as ammonium persulfate or photoinitiators under UV exposure. This approach typically achieves crosslinking densities ranging from 0.1 to 10 mol%, resulting in compressive strengths between 0.1 to 2 MPa for most hydrogel systems. However, the irreversible nature of covalent bonds often leads to brittleness and limited self-healing capabilities.
Physical crosslinking mechanisms rely on non-covalent interactions including hydrogen bonding, electrostatic interactions, and hydrophobic associations. Ionic crosslinking using divalent cations like calcium or magnesium ions demonstrates reversibility and biocompatibility but typically yields lower mechanical strength, with elastic moduli rarely exceeding 100 kPa. Temperature-responsive crosslinking through thermosensitive polymers offers controllable gelation but suffers from thermal instability under physiological conditions.
The primary mechanical limitations stem from the inherent trade-off between water content and mechanical strength. High water content, essential for many applications, directly correlates with reduced crosslinking density and compromised mechanical integrity. Most conventional hydrogels exhibit tensile strengths below 1 MPa and elongation at break limited to 100-300%, significantly restricting their application scope.
Network heterogeneity represents another critical limitation, where uneven crosslink distribution creates stress concentration points leading to premature failure. Traditional crosslinking methods often result in random network formation, producing mechanical anisotropy and unpredictable failure modes. Additionally, swelling-induced stress can cause network degradation over time, particularly in ionically crosslinked systems.
Current double-network strategies attempt to address these limitations by combining sacrificial networks with elastic networks. While achieving improved toughness values up to 1000 J/m², these systems require complex synthesis procedures and often compromise other properties such as transparency or biocompatibility. The challenge remains in developing crosslinking methodologies that simultaneously optimize strength, toughness, and functionality while maintaining the advantageous properties that make hydrogels attractive for diverse applications.
Chemical crosslinking methods utilize covalent bond formation to create permanent network structures. Free radical polymerization remains the most prevalent technique, employing initiators such as ammonium persulfate or photoinitiators under UV exposure. This approach typically achieves crosslinking densities ranging from 0.1 to 10 mol%, resulting in compressive strengths between 0.1 to 2 MPa for most hydrogel systems. However, the irreversible nature of covalent bonds often leads to brittleness and limited self-healing capabilities.
Physical crosslinking mechanisms rely on non-covalent interactions including hydrogen bonding, electrostatic interactions, and hydrophobic associations. Ionic crosslinking using divalent cations like calcium or magnesium ions demonstrates reversibility and biocompatibility but typically yields lower mechanical strength, with elastic moduli rarely exceeding 100 kPa. Temperature-responsive crosslinking through thermosensitive polymers offers controllable gelation but suffers from thermal instability under physiological conditions.
The primary mechanical limitations stem from the inherent trade-off between water content and mechanical strength. High water content, essential for many applications, directly correlates with reduced crosslinking density and compromised mechanical integrity. Most conventional hydrogels exhibit tensile strengths below 1 MPa and elongation at break limited to 100-300%, significantly restricting their application scope.
Network heterogeneity represents another critical limitation, where uneven crosslink distribution creates stress concentration points leading to premature failure. Traditional crosslinking methods often result in random network formation, producing mechanical anisotropy and unpredictable failure modes. Additionally, swelling-induced stress can cause network degradation over time, particularly in ionically crosslinked systems.
Current double-network strategies attempt to address these limitations by combining sacrificial networks with elastic networks. While achieving improved toughness values up to 1000 J/m², these systems require complex synthesis procedures and often compromise other properties such as transparency or biocompatibility. The challenge remains in developing crosslinking methodologies that simultaneously optimize strength, toughness, and functionality while maintaining the advantageous properties that make hydrogels attractive for diverse applications.
Existing Crosslinking Optimization Solutions
01 Cross-linking enhancement for improved mechanical properties
Cross-linking strategies are employed to enhance the mechanical strength of hydrogels by forming covalent or non-covalent bonds between polymer chains. This approach increases the structural integrity and load-bearing capacity of the hydrogel network, resulting in improved tensile strength, compressive strength, and elastic modulus. Various cross-linking agents and methods can be utilized to achieve optimal mechanical performance.- Cross-linking enhancement for improved mechanical properties: Various cross-linking methods and agents are employed to enhance the mechanical strength of hydrogels. Chemical cross-linking through covalent bonds and physical cross-linking through non-covalent interactions can significantly improve the tensile strength, compressive strength, and elastic modulus of hydrogel materials. The degree of cross-linking directly correlates with the mechanical performance of the resulting hydrogel structure.
- Composite reinforcement with nanoparticles and fibers: Incorporation of reinforcing materials such as nanoparticles, nanofibers, or microfibers into hydrogel matrices can dramatically improve mechanical strength. These reinforcing agents create a composite structure that distributes stress more effectively and increases the overall load-bearing capacity. The reinforcement can be achieved through various nanomaterials including carbon nanotubes, graphene oxide, and ceramic particles.
- Double network and interpenetrating polymer networks: The formation of double network structures or interpenetrating polymer networks creates hydrogels with superior mechanical properties. This approach involves combining two or more polymer networks with different characteristics, where one network provides toughness while another contributes to strength. The synergistic effect of multiple networks results in hydrogels with enhanced fracture resistance and mechanical durability.
- Polymer concentration and molecular weight optimization: The mechanical strength of hydrogels can be controlled by optimizing polymer concentration and molecular weight parameters. Higher polymer concentrations generally lead to increased mechanical properties due to greater chain entanglement and cross-link density. Molecular weight distribution and polymer architecture also play crucial roles in determining the final mechanical characteristics of the hydrogel system.
- pH and ionic strength modification for strength enhancement: Adjustment of environmental conditions such as pH and ionic strength can significantly influence hydrogel mechanical properties. Ionic interactions and hydrogen bonding can be manipulated to create stronger intermolecular forces within the hydrogel network. These modifications can lead to reversible or irreversible changes in mechanical strength depending on the specific polymer system and environmental conditions applied.
02 Composite reinforcement with nanoparticles and fibers
Incorporation of reinforcing materials such as nanoparticles, nanofibers, or microfibers into hydrogel matrices significantly enhances mechanical strength. These reinforcing agents act as load-bearing elements and stress distributors within the hydrogel network, improving resistance to deformation and failure. The composite approach allows for tailored mechanical properties while maintaining the hydrogel's inherent characteristics.Expand Specific Solutions03 Double network and interpenetrating polymer networks
Double network structures and interpenetrating polymer networks create synergistic effects that dramatically improve hydrogel mechanical strength. These systems combine multiple polymer networks with different properties, where one network provides toughness while another contributes to elasticity. The resulting hydrogels exhibit superior mechanical performance compared to single-network systems.Expand Specific Solutions04 Polymer concentration and molecular weight optimization
Adjusting polymer concentration and molecular weight parameters directly influences hydrogel mechanical properties. Higher polymer concentrations generally lead to increased cross-link density and improved mechanical strength, while optimal molecular weight selection ensures proper chain entanglement and network formation. These parameters must be carefully balanced to achieve desired mechanical performance without compromising other properties.Expand Specific Solutions05 Surface modification and gradient structures
Surface modification techniques and gradient structural designs enhance hydrogel mechanical properties through localized strengthening and stress distribution optimization. These approaches create heterogeneous structures with varying mechanical properties across different regions, allowing for improved load transfer and resistance to mechanical failure. The modifications can be achieved through various chemical and physical treatment methods.Expand Specific Solutions
Key Players in Hydrogel and Crosslinking Industry
The hydrogel crosslinking optimization field represents a mature technology sector experiencing significant growth driven by expanding biomedical applications. The market demonstrates substantial scale with diverse applications spanning tissue engineering, drug delivery, and medical devices. The competitive landscape features a multi-tiered structure including established chemical giants like BASF Corp., Evonik Operations GmbH, and Beiersdorf AG providing foundational materials and manufacturing capabilities. Specialized biotechnology companies such as Regentis Biomaterials Ltd., SupraPolix BV, and MediFab Co., Ltd. focus on advanced hydrogel formulations and medical applications. Leading research institutions including MIT, University of Tokyo, Sichuan University, and South China University of Technology drive fundamental innovation in crosslinking mechanisms and material properties. The technology maturity varies across applications, with basic hydrogel systems being commercially established while advanced smart and responsive crosslinking systems remain in development phases, indicating continued innovation potential.
BASF Corp.
Technical Solution: BASF has developed advanced crosslinking technologies for hydrogels using multi-functional crosslinkers and controlled polymerization techniques. Their approach focuses on optimizing the crosslink density through precise control of crosslinker concentration and reaction conditions. They utilize both chemical crosslinking with glutaraldehyde and physical crosslinking methods to achieve enhanced mechanical properties. The company has implemented dual-crosslinking systems that combine covalent and ionic crosslinks, resulting in hydrogels with improved tensile strength and elasticity. Their proprietary crosslinking agents allow for tunable mechanical properties while maintaining biocompatibility for various applications.
Strengths: Extensive chemical expertise and industrial-scale production capabilities. Weaknesses: Limited focus on biomedical applications compared to specialized biomaterial companies.
Massachusetts Institute of Technology
Technical Solution: MIT has pioneered innovative crosslinking strategies including double-network hydrogels and dynamic covalent crosslinking systems. Their research focuses on creating tough hydrogels through sacrificial bond mechanisms and interpenetrating polymer networks. They have developed reversible crosslinking methods using boronate ester bonds and disulfide linkages that provide self-healing properties while maintaining high mechanical strength. The institute's approach includes using nanoparticle reinforcement combined with optimized crosslinking to achieve exceptional toughness and fatigue resistance. Their work on hybrid crosslinking systems demonstrates significant improvements in both strength and flexibility.
Strengths: Cutting-edge research capabilities and innovative crosslinking mechanisms. Weaknesses: Academic focus may limit immediate commercial scalability and industrial implementation.
Core Innovations in Advanced Crosslinking Methods
Double-crosslinked hydrogel composite material, preparation method therefor, and use thereof
PatentWO2025190125A1
Innovation
- By using acrylic monomers and zwitterionic monomers to react under light conditions in the presence of nanoparticles to form a physical-chemical double-crosslinked copolymer, a double-crosslinked hydrogel composite material with high mechanical strength, high adhesion strength and good anti-swelling properties is constructed.
Highly strong and tough photo-crosslinked hydrogel material and its preparation and application
PatentActiveUS20230130864A1
Innovation
- A highly strong and tough photo-crosslinked hydrogel material is developed by cross-linking photosensitive groups between polymer derivatives, specifically using o-nitrobenzyl phototrigger modified polymer derivatives, double bond groups modified polymer derivatives, and photoinitiators to enhance mechanical properties through improved load transfer and polymerization processes.
Biocompatibility and Safety Standards for Hydrogels
Biocompatibility assessment represents a fundamental prerequisite for hydrogel applications in biomedical fields, particularly when crosslinking optimization aims to enhance mechanical strength. The evaluation framework encompasses cytotoxicity testing, hemocompatibility analysis, and inflammatory response assessment. Standard protocols such as ISO 10993 series provide comprehensive guidelines for biological evaluation of medical devices, establishing systematic approaches for hydrogel safety validation.
Cytotoxicity evaluation forms the cornerstone of biocompatibility testing, employing both direct and indirect contact methods to assess cellular viability. Enhanced crosslinking densities may introduce potential concerns regarding leachable compounds or altered surface properties that could affect cell proliferation and differentiation. In vitro studies utilizing standardized cell lines, including L929 fibroblasts and human dermal fibroblasts, provide initial screening data for cytotoxic effects.
Hemocompatibility testing becomes critical for hydrogels intended for blood-contacting applications or systemic delivery. Hemolysis assays, platelet adhesion studies, and coagulation pathway analysis evaluate blood-material interactions. Optimized crosslinking strategies must maintain hemolysis rates below 5% according to ASTM F756 standards, while minimizing thrombogenic potential through careful surface chemistry control.
Inflammatory response assessment involves comprehensive evaluation of immune system activation following hydrogel implantation. Standardized animal models, typically following ISO 10993-6 guidelines, monitor inflammatory markers including interleukin levels, macrophage activation, and tissue histopathology. Enhanced mechanical properties achieved through crosslinking optimization should not compromise the hydrogel's ability to integrate with surrounding tissues without triggering chronic inflammatory responses.
Regulatory compliance requires adherence to region-specific standards including FDA guidance documents, European Medical Device Regulation, and pharmacopeial requirements. Documentation of crosslinking agent selection, residual monomer quantification, and degradation product characterization supports regulatory submissions. Long-term biocompatibility studies extending 26 weeks demonstrate sustained safety profiles essential for permanent or semi-permanent implant applications.
Sterilization compatibility represents an additional safety consideration, as enhanced crosslinking may alter hydrogel response to gamma irradiation, ethylene oxide, or steam sterilization methods. Validation studies must confirm maintained biocompatibility following sterilization processes while preserving the optimized mechanical properties achieved through crosslinking enhancement.
Cytotoxicity evaluation forms the cornerstone of biocompatibility testing, employing both direct and indirect contact methods to assess cellular viability. Enhanced crosslinking densities may introduce potential concerns regarding leachable compounds or altered surface properties that could affect cell proliferation and differentiation. In vitro studies utilizing standardized cell lines, including L929 fibroblasts and human dermal fibroblasts, provide initial screening data for cytotoxic effects.
Hemocompatibility testing becomes critical for hydrogels intended for blood-contacting applications or systemic delivery. Hemolysis assays, platelet adhesion studies, and coagulation pathway analysis evaluate blood-material interactions. Optimized crosslinking strategies must maintain hemolysis rates below 5% according to ASTM F756 standards, while minimizing thrombogenic potential through careful surface chemistry control.
Inflammatory response assessment involves comprehensive evaluation of immune system activation following hydrogel implantation. Standardized animal models, typically following ISO 10993-6 guidelines, monitor inflammatory markers including interleukin levels, macrophage activation, and tissue histopathology. Enhanced mechanical properties achieved through crosslinking optimization should not compromise the hydrogel's ability to integrate with surrounding tissues without triggering chronic inflammatory responses.
Regulatory compliance requires adherence to region-specific standards including FDA guidance documents, European Medical Device Regulation, and pharmacopeial requirements. Documentation of crosslinking agent selection, residual monomer quantification, and degradation product characterization supports regulatory submissions. Long-term biocompatibility studies extending 26 weeks demonstrate sustained safety profiles essential for permanent or semi-permanent implant applications.
Sterilization compatibility represents an additional safety consideration, as enhanced crosslinking may alter hydrogel response to gamma irradiation, ethylene oxide, or steam sterilization methods. Validation studies must confirm maintained biocompatibility following sterilization processes while preserving the optimized mechanical properties achieved through crosslinking enhancement.
Environmental Impact of Crosslinking Agents
The environmental implications of crosslinking agents used in hydrogel synthesis have become increasingly significant as the demand for mechanically robust hydrogels expands across biomedical, agricultural, and industrial applications. Traditional chemical crosslinking agents, particularly those based on formaldehyde derivatives such as glutaraldehyde and glyoxal, pose substantial environmental concerns due to their toxicity and persistence in ecosystems. These compounds can leach into soil and water systems, potentially causing long-term contamination and adverse effects on aquatic life.
The manufacturing processes of conventional crosslinking agents often involve energy-intensive chemical synthesis routes that generate significant carbon emissions and hazardous waste byproducts. Petrochemical-derived crosslinkers, including various acrylate and methacrylate compounds, contribute to the depletion of fossil fuel resources and release volatile organic compounds during production and application phases. The disposal of hydrogels containing these agents presents additional challenges, as many crosslinked networks resist biodegradation and accumulate in landfills.
Emerging bio-based crosslinking alternatives demonstrate promising environmental profiles while maintaining mechanical enhancement capabilities. Natural crosslinking agents derived from plant extracts, such as genipin from gardenia fruits and tannic acid from various botanical sources, offer biodegradable pathways with reduced ecological footprint. These compounds typically exhibit lower toxicity profiles and can be sourced through sustainable agricultural practices, creating circular economy opportunities.
Physical crosslinking methods, including freeze-thaw cycling and ionic crosslinking using naturally occurring minerals, present environmentally benign alternatives that eliminate the need for synthetic chemical agents entirely. These approaches leverage natural processes to achieve mechanical strengthening while maintaining complete biodegradability of the resulting hydrogel networks.
The lifecycle assessment of crosslinking agents reveals that environmental impact extends beyond immediate toxicity concerns to encompass resource consumption, energy requirements, and end-of-life disposal considerations. Regulatory frameworks increasingly emphasize the need for green chemistry approaches in hydrogel development, driving innovation toward sustainable crosslinking solutions that balance mechanical performance with environmental responsibility.
The manufacturing processes of conventional crosslinking agents often involve energy-intensive chemical synthesis routes that generate significant carbon emissions and hazardous waste byproducts. Petrochemical-derived crosslinkers, including various acrylate and methacrylate compounds, contribute to the depletion of fossil fuel resources and release volatile organic compounds during production and application phases. The disposal of hydrogels containing these agents presents additional challenges, as many crosslinked networks resist biodegradation and accumulate in landfills.
Emerging bio-based crosslinking alternatives demonstrate promising environmental profiles while maintaining mechanical enhancement capabilities. Natural crosslinking agents derived from plant extracts, such as genipin from gardenia fruits and tannic acid from various botanical sources, offer biodegradable pathways with reduced ecological footprint. These compounds typically exhibit lower toxicity profiles and can be sourced through sustainable agricultural practices, creating circular economy opportunities.
Physical crosslinking methods, including freeze-thaw cycling and ionic crosslinking using naturally occurring minerals, present environmentally benign alternatives that eliminate the need for synthetic chemical agents entirely. These approaches leverage natural processes to achieve mechanical strengthening while maintaining complete biodegradability of the resulting hydrogel networks.
The lifecycle assessment of crosslinking agents reveals that environmental impact extends beyond immediate toxicity concerns to encompass resource consumption, energy requirements, and end-of-life disposal considerations. Regulatory frameworks increasingly emphasize the need for green chemistry approaches in hydrogel development, driving innovation toward sustainable crosslinking solutions that balance mechanical performance with environmental responsibility.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!