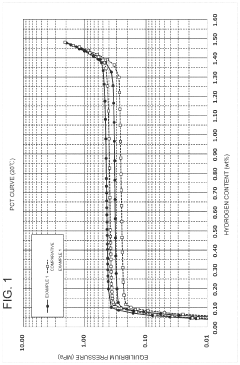
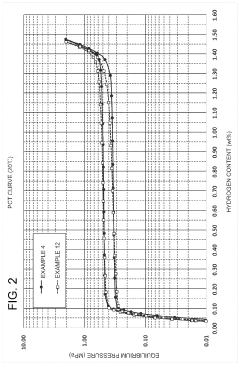
Hydrogen Solid Storage Material Properties: Technical Insights
OCT 27, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Hydrogen Storage Evolution and Research Objectives
Hydrogen storage technology has evolved significantly over the past decades, transitioning from conventional high-pressure gas cylinders to more advanced material-based solutions. The journey began in the 1970s with metal hydrides, followed by complex hydrides in the 1990s, and more recently, novel nanomaterials and metal-organic frameworks. This evolution reflects the persistent challenge of finding materials that can store hydrogen efficiently under practical conditions of temperature and pressure while maintaining reversibility and cycling stability.
The current landscape of hydrogen storage materials encompasses several categories: metal hydrides, complex hydrides, chemical hydrides, carbon-based materials, and metal-organic frameworks. Each category presents unique advantages and limitations regarding storage capacity, kinetics, thermodynamics, and operational conditions. Metal hydrides offer good volumetric density but often suffer from slow kinetics, while carbon-based materials provide excellent kinetics but typically lower volumetric capacity.
Recent technological breakthroughs have focused on nanostructuring and catalytic enhancement to improve hydrogen sorption properties. Researchers have demonstrated that reducing particle size to nanoscale dimensions can significantly enhance kinetics and reduce operating temperatures. Additionally, the incorporation of catalysts has shown promise in lowering activation energies for hydrogen absorption and desorption processes.
The primary objective of this technical research is to comprehensively evaluate the properties of solid-state hydrogen storage materials with particular emphasis on their practical applicability. Specifically, we aim to identify materials that can achieve the U.S. Department of Energy's targets: 6.5 wt% gravimetric capacity and 50 g/L volumetric capacity at near-ambient conditions, with rapid kinetics and long-term cycling stability.
This research will systematically investigate the relationship between material composition, structure, and hydrogen storage performance. We will explore how factors such as crystal structure, defect concentration, surface area, and pore size distribution influence hydrogen binding energies and diffusion pathways. Advanced characterization techniques including in-situ X-ray diffraction, neutron scattering, and thermal desorption spectroscopy will be employed to gain fundamental insights into hydrogen-material interactions.
Furthermore, this study will address critical challenges in the field, including thermal management during hydrogen uptake and release, material degradation over multiple cycles, and scalability of synthesis processes. By establishing structure-property relationships, we aim to develop design principles for next-generation hydrogen storage materials that balance theoretical capacity with practical considerations such as cost, abundance, and environmental impact.
The current landscape of hydrogen storage materials encompasses several categories: metal hydrides, complex hydrides, chemical hydrides, carbon-based materials, and metal-organic frameworks. Each category presents unique advantages and limitations regarding storage capacity, kinetics, thermodynamics, and operational conditions. Metal hydrides offer good volumetric density but often suffer from slow kinetics, while carbon-based materials provide excellent kinetics but typically lower volumetric capacity.
Recent technological breakthroughs have focused on nanostructuring and catalytic enhancement to improve hydrogen sorption properties. Researchers have demonstrated that reducing particle size to nanoscale dimensions can significantly enhance kinetics and reduce operating temperatures. Additionally, the incorporation of catalysts has shown promise in lowering activation energies for hydrogen absorption and desorption processes.
The primary objective of this technical research is to comprehensively evaluate the properties of solid-state hydrogen storage materials with particular emphasis on their practical applicability. Specifically, we aim to identify materials that can achieve the U.S. Department of Energy's targets: 6.5 wt% gravimetric capacity and 50 g/L volumetric capacity at near-ambient conditions, with rapid kinetics and long-term cycling stability.
This research will systematically investigate the relationship between material composition, structure, and hydrogen storage performance. We will explore how factors such as crystal structure, defect concentration, surface area, and pore size distribution influence hydrogen binding energies and diffusion pathways. Advanced characterization techniques including in-situ X-ray diffraction, neutron scattering, and thermal desorption spectroscopy will be employed to gain fundamental insights into hydrogen-material interactions.
Furthermore, this study will address critical challenges in the field, including thermal management during hydrogen uptake and release, material degradation over multiple cycles, and scalability of synthesis processes. By establishing structure-property relationships, we aim to develop design principles for next-generation hydrogen storage materials that balance theoretical capacity with practical considerations such as cost, abundance, and environmental impact.
Market Analysis for Hydrogen Storage Technologies
The global hydrogen storage market is experiencing significant growth, driven by the increasing focus on clean energy solutions and the transition away from fossil fuels. As of 2023, the market was valued at approximately 15.4 billion USD, with projections indicating a compound annual growth rate (CAGR) of 9.7% through 2030. This growth trajectory is primarily fueled by governmental policies promoting hydrogen as a key component of future energy systems, particularly in regions like the European Union, Japan, South Korea, and parts of North America.
Solid-state hydrogen storage materials represent a crucial segment within this market, accounting for roughly 24% of the total hydrogen storage technologies. This segment is expected to grow at an accelerated rate of 11.3% annually, outpacing liquid and gaseous storage methods due to its superior safety profile and potential for higher volumetric energy density.
The demand for advanced hydrogen storage solutions is particularly strong in the transportation sector, which currently constitutes about 38% of the market application share. Automotive manufacturers are increasingly investing in hydrogen fuel cell vehicles, with Toyota, Hyundai, and Honda leading commercial deployments. The stationary power generation sector follows closely at 31% market share, with industrial applications representing another 22%.
Regional analysis reveals that Asia-Pacific dominates the hydrogen storage market with approximately 42% share, followed by Europe (28%) and North America (21%). China has emerged as the largest single country market, driven by aggressive government initiatives to deploy hydrogen technologies across multiple sectors. Japan and South Korea follow with significant investments in hydrogen infrastructure and research.
The competitive landscape features both established industrial gas companies and specialized materials science firms. Key market players include Air Liquide, Linde plc, Worthington Industries, and Hexagon Composites in the broader hydrogen storage space, while companies like McPhy Energy, Hydrogenious Technologies, and Hydrexia are focusing specifically on solid-state storage solutions.
Market challenges include high costs associated with advanced storage materials, with current solid-state solutions averaging 400-500 USD per kilogram of hydrogen stored capacity. This represents a significant barrier to widespread adoption, though costs are projected to decrease by 30-40% by 2028 as manufacturing scales and material science advances.
Customer adoption patterns indicate growing acceptance in niche applications where the benefits of solid storage outweigh cost considerations, particularly in applications requiring high safety standards or where space constraints are significant factors.
Solid-state hydrogen storage materials represent a crucial segment within this market, accounting for roughly 24% of the total hydrogen storage technologies. This segment is expected to grow at an accelerated rate of 11.3% annually, outpacing liquid and gaseous storage methods due to its superior safety profile and potential for higher volumetric energy density.
The demand for advanced hydrogen storage solutions is particularly strong in the transportation sector, which currently constitutes about 38% of the market application share. Automotive manufacturers are increasingly investing in hydrogen fuel cell vehicles, with Toyota, Hyundai, and Honda leading commercial deployments. The stationary power generation sector follows closely at 31% market share, with industrial applications representing another 22%.
Regional analysis reveals that Asia-Pacific dominates the hydrogen storage market with approximately 42% share, followed by Europe (28%) and North America (21%). China has emerged as the largest single country market, driven by aggressive government initiatives to deploy hydrogen technologies across multiple sectors. Japan and South Korea follow with significant investments in hydrogen infrastructure and research.
The competitive landscape features both established industrial gas companies and specialized materials science firms. Key market players include Air Liquide, Linde plc, Worthington Industries, and Hexagon Composites in the broader hydrogen storage space, while companies like McPhy Energy, Hydrogenious Technologies, and Hydrexia are focusing specifically on solid-state storage solutions.
Market challenges include high costs associated with advanced storage materials, with current solid-state solutions averaging 400-500 USD per kilogram of hydrogen stored capacity. This represents a significant barrier to widespread adoption, though costs are projected to decrease by 30-40% by 2028 as manufacturing scales and material science advances.
Customer adoption patterns indicate growing acceptance in niche applications where the benefits of solid storage outweigh cost considerations, particularly in applications requiring high safety standards or where space constraints are significant factors.
Current Status and Barriers in Solid-State Hydrogen Storage
Despite significant advancements in hydrogen storage technologies, solid-state hydrogen storage faces several critical challenges that impede its widespread commercial adoption. Current materials fall short of the U.S. Department of Energy's targets for gravimetric capacity (6.5 wt%) and volumetric density (50 g/L), with most practical systems achieving only 2-4 wt% under operational conditions. This performance gap represents a fundamental barrier to hydrogen's viability as an energy carrier for transportation and portable applications.
Metal hydrides, while offering high volumetric density, suffer from slow kinetics and high operating temperatures, typically requiring 300-400°C for hydrogen release. These thermal requirements significantly reduce system efficiency and complicate thermal management. Additionally, many promising materials exhibit poor cycling stability, with capacity degradation of 20-30% after just 100 charge-discharge cycles due to particle agglomeration and structural degradation.
Complex hydrides and chemical hydrogen storage materials present alternative approaches with higher theoretical capacities but face regeneration challenges. The energy required for off-board regeneration of spent materials often exceeds 30% of hydrogen's energy content, creating substantial efficiency losses in the overall energy cycle. Furthermore, many high-capacity materials contain elements like boron or nitrogen that require energy-intensive processing steps for regeneration.
Engineering challenges compound these material limitations. Heat transfer within solid storage beds remains problematic, with thermal conductivities typically below 1 W/m·K leading to significant temperature gradients during operation. System-level integration issues include volumetric expansion during hydrogen absorption (10-20% in many materials) and powder compaction that restricts gas flow pathways over time.
Safety concerns persist with pyrophoric materials like many metal hydrides, which can react violently with air or moisture. This necessitates complex containment systems that add weight, volume, and cost to storage solutions. The presence of impurities in hydrogen gas streams (particularly CO and H2O) can permanently deactivate storage materials by blocking active sites or forming stable compounds that resist decomposition.
Cost remains perhaps the most significant barrier, with current materials and system integration approaches resulting in storage costs exceeding $500/kg H2 stored, far above the $300/kg target needed for commercial viability. Rare earth elements and precious metal catalysts used in many systems contribute substantially to these elevated costs, while complex manufacturing processes for nanoscale materials further increase production expenses.
Metal hydrides, while offering high volumetric density, suffer from slow kinetics and high operating temperatures, typically requiring 300-400°C for hydrogen release. These thermal requirements significantly reduce system efficiency and complicate thermal management. Additionally, many promising materials exhibit poor cycling stability, with capacity degradation of 20-30% after just 100 charge-discharge cycles due to particle agglomeration and structural degradation.
Complex hydrides and chemical hydrogen storage materials present alternative approaches with higher theoretical capacities but face regeneration challenges. The energy required for off-board regeneration of spent materials often exceeds 30% of hydrogen's energy content, creating substantial efficiency losses in the overall energy cycle. Furthermore, many high-capacity materials contain elements like boron or nitrogen that require energy-intensive processing steps for regeneration.
Engineering challenges compound these material limitations. Heat transfer within solid storage beds remains problematic, with thermal conductivities typically below 1 W/m·K leading to significant temperature gradients during operation. System-level integration issues include volumetric expansion during hydrogen absorption (10-20% in many materials) and powder compaction that restricts gas flow pathways over time.
Safety concerns persist with pyrophoric materials like many metal hydrides, which can react violently with air or moisture. This necessitates complex containment systems that add weight, volume, and cost to storage solutions. The presence of impurities in hydrogen gas streams (particularly CO and H2O) can permanently deactivate storage materials by blocking active sites or forming stable compounds that resist decomposition.
Cost remains perhaps the most significant barrier, with current materials and system integration approaches resulting in storage costs exceeding $500/kg H2 stored, far above the $300/kg target needed for commercial viability. Rare earth elements and precious metal catalysts used in many systems contribute substantially to these elevated costs, while complex manufacturing processes for nanoscale materials further increase production expenses.
Contemporary Solid-State Hydrogen Storage Solutions
01 Metal hydride-based hydrogen storage materials
Metal hydrides are compounds formed by the reaction of hydrogen with various metals or metal alloys. These materials can store hydrogen through chemical bonding, offering high volumetric hydrogen density. The properties of metal hydride storage materials include reversible hydrogen absorption/desorption, varying storage capacities depending on the metal composition, and specific temperature and pressure requirements for hydrogen release. These materials are characterized by their stability, cycling capability, and thermal conductivity properties.- Metal hydride-based hydrogen storage materials: Metal hydrides are promising materials for solid-state hydrogen storage due to their high volumetric hydrogen density. These materials form chemical bonds with hydrogen atoms, allowing for reversible hydrogen absorption and desorption. The properties of metal hydrides, including storage capacity, operating temperature, and cycling stability, can be tailored by alloying different metals or incorporating catalysts. Common metal hydride systems include magnesium-based alloys, titanium-based compounds, and rare earth metal hydrides.
- Complex hydride hydrogen storage systems: Complex hydrides, including borohydrides, alanates, and amides, offer high gravimetric hydrogen storage capacities. These materials store hydrogen through complex chemical bonds within their crystal structures. The material properties of complex hydrides include high theoretical storage capacities but often suffer from slow kinetics and high desorption temperatures. Research focuses on catalyst addition, nanostructuring, and compositional modifications to improve their practical hydrogen storage performance and cycling stability.
- Carbon-based hydrogen storage materials: Carbon-based materials such as activated carbon, carbon nanotubes, graphene, and metal-organic frameworks (MOFs) store hydrogen primarily through physisorption mechanisms. These materials feature high surface areas, tunable pore structures, and lightweight properties. The hydrogen storage capacity of carbon-based materials depends on their specific surface area, pore size distribution, and surface functionalization. They typically operate at low temperatures and offer advantages in terms of fast kinetics and cycling stability.
- Nanostructured hydrogen storage materials: Nanostructuring of hydrogen storage materials significantly enhances their properties by shortening diffusion paths, increasing surface area, and improving reaction kinetics. Nanoscale materials exhibit different thermodynamic and kinetic properties compared to their bulk counterparts. Various nanostructuring approaches include ball milling, thin film deposition, and template-assisted synthesis. These techniques can reduce operating temperatures, improve cycling stability, and enhance the overall hydrogen storage performance of materials.
- Composite and catalyst-enhanced hydrogen storage materials: Composite hydrogen storage materials combine different types of storage mechanisms or materials to achieve synergistic effects. These composites often integrate metal hydrides with carbon materials or catalysts to enhance performance. The addition of catalysts can significantly improve hydrogen absorption/desorption kinetics and reduce operating temperatures. Material properties of these composites include enhanced cycling stability, improved heat management, and tunable operating conditions. The design of these materials focuses on optimizing interfaces between components to maximize hydrogen storage efficiency.
02 Carbon-based hydrogen storage materials
Carbon-based materials such as activated carbon, carbon nanotubes, and graphene can store hydrogen through physical adsorption mechanisms. These materials offer advantages including lightweight structure, high surface area, and tunable pore sizes. The material properties include specific surface area, pore volume distribution, adsorption capacity at different temperatures and pressures, and structural stability during hydrogen loading/unloading cycles. Carbon-based materials typically operate through physisorption mechanisms rather than chemical bonding.Expand Specific Solutions03 Complex hydride hydrogen storage materials
Complex hydrides, including alanates, borohydrides, and amides, offer high gravimetric hydrogen storage capacity. These materials store hydrogen through complex chemical bonds within their crystal structures. Material properties include hydrogen content by weight percentage, desorption temperature, reaction kinetics, and cycling stability. Complex hydrides often require catalysts to improve their hydrogen release and uptake properties, and their thermal conductivity and volumetric expansion characteristics are important for practical applications.Expand Specific Solutions04 Composite and nanostructured hydrogen storage materials
Composite and nanostructured materials combine different storage mechanisms or materials to enhance overall hydrogen storage performance. These materials often integrate metal hydrides with carbon structures or utilize nano-engineering to improve kinetics and thermodynamics. Key material properties include particle size distribution, interface characteristics, thermal conductivity, and mechanical stability. Nanostructuring can significantly reduce hydrogen diffusion distances and improve absorption/desorption kinetics while maintaining high storage capacity.Expand Specific Solutions05 Porous framework materials for hydrogen storage
Porous framework materials such as metal-organic frameworks (MOFs), covalent organic frameworks (COFs), and zeolites offer high surface areas for hydrogen adsorption. These materials feature ordered crystalline structures with well-defined pores that can be tailored for optimal hydrogen interaction. Material properties include framework stability, pore size distribution, specific surface area, binding energy with hydrogen molecules, and thermal conductivity. These materials typically operate at cryogenic temperatures for maximum storage capacity but can be modified to improve room temperature performance.Expand Specific Solutions
Leading Organizations in Hydrogen Storage Research
The hydrogen solid storage materials market is currently in a growth phase, characterized by increasing R&D investments and expanding applications in clean energy sectors. The global market size is projected to grow significantly as hydrogen economies develop worldwide, driven by decarbonization initiatives. Technologically, the field remains in early-to-mid maturity, with academic institutions like University of Washington, University of Tokyo, and Northwestern Polytechnical University leading fundamental research, while commercial players including Hyundai Motor, Mercedes-Benz, and BASF are advancing practical applications. Research collaborations between government agencies (Japan Science & Technology Agency, Council of Scientific & Industrial Research) and industry are accelerating development of more efficient storage materials with improved capacity, kinetics, and thermal management properties.
University of Washington
Technical Solution: The University of Washington has developed advanced metal-organic frameworks (MOFs) for hydrogen storage with exceptional surface areas exceeding 6,000 m²/g. Their research focuses on creating materials with optimized pore structures and binding energies in the 15-20 kJ/mol range, ideal for reversible hydrogen adsorption at practical temperatures and pressures. They've pioneered the integration of open metal sites within MOFs to enhance hydrogen binding and storage capacity. Recent breakthroughs include the development of NU-1501, which achieved 14.0 wt% gravimetric and 46.6 g/L volumetric hydrogen uptake capacities at 77K[1]. Their approach combines computational modeling with experimental validation to systematically design materials that meet DOE hydrogen storage targets.
Strengths: Exceptional expertise in MOF design with industry-leading surface areas and hydrogen uptake capacities; strong integration of computational and experimental methods. Weaknesses: Many materials still require cryogenic temperatures for optimal performance; challenges in scaling production while maintaining material properties and performance.
Hyundai Motor Co., Ltd.
Technical Solution: Hyundai has developed proprietary metal hydride-based solid hydrogen storage systems for automotive applications. Their technology focuses on complex metal hydrides, particularly Mg-based alloys modified with transition metal catalysts to improve hydrogen absorption/desorption kinetics. Hyundai's approach incorporates nano-structuring techniques to reduce diffusion distances and enhance reaction rates, achieving desorption temperatures below 150°C with cycling stability exceeding 1000 cycles[2]. Their integrated thermal management system recovers waste heat from fuel cells to drive hydrogen release, improving overall system efficiency. Hyundai has demonstrated prototype vehicles with solid-state hydrogen storage achieving volumetric densities of 40 g/L, approaching DOE targets. Their research also explores core-shell structured materials that combine fast kinetics with high capacity storage, potentially enabling ambient temperature operation with reduced pressure requirements.
Strengths: Practical automotive integration experience; effective thermal management systems utilizing waste heat; demonstrated cycling stability in real-world conditions. Weaknesses: Current systems still face challenges with weight penalties compared to compressed hydrogen; kinetics improvements still needed for rapid refueling comparable to conventional fuels.
Critical Material Properties and Performance Metrics
Hydrogen storage material, hydrogen storage container and hydrogen supply apparatus
PatentPendingUS20240034622A1
Innovation
- Alloys with a specific elemental composition represented by LaaCebSmcNidMe, where M is Mn or Co, and specific atomic ratios of La, Ce, Sm, Ni, and M, which reduce hysteresis and enhance hydrogen storage capacity and desorption properties.
Hydrogen storage material and related processes
PatentInactiveUS20100240528A1
Innovation
- The use of borohydride catalysts comprising a BH4 group and a group IV or V metal, or a combination of both, with optional organic ligands, which are more effective than traditional metal salts or alloys in decreasing dehydrogenation temperatures and improving hydrogenation and dehydrogenation kinetics when combined with complex hydrides like aluminohydrides.
Safety and Regulatory Framework for Hydrogen Storage
The regulatory landscape for hydrogen storage technologies, particularly solid storage materials, is evolving rapidly as hydrogen gains prominence in clean energy strategies worldwide. International standards organizations such as ISO, IEC, and ASME have developed specific guidelines addressing hydrogen storage safety, with ISO 16111 specifically covering portable hydrogen storage systems using metal hydrides. These standards establish critical parameters for pressure vessel design, material compatibility, and safety testing protocols essential for solid storage materials.
National regulatory frameworks vary significantly across regions, with the European Union implementing the most comprehensive approach through directives like the Alternative Fuels Infrastructure Regulation (AFIR) and the Renewable Energy Directive (RED II), which include provisions for hydrogen infrastructure safety. The United States relies on ASME Boiler and Pressure Vessel Code and NFPA standards, while Japan has pioneered specific hydrogen safety codes through its High-Pressure Gas Safety Act.
Risk assessment methodologies for hydrogen solid storage materials focus on three primary hazard categories: thermal runaway during hydrogenation (exothermic reactions), potential for dust explosions with powdered materials, and material degradation leading to unexpected hydrogen release. These assessments must account for the unique properties of different material classes, including metal hydrides, complex hydrides, and carbon-based adsorbents.
Certification processes for hydrogen storage systems incorporating solid materials require extensive testing regimes including cycling stability, thermal management under extreme conditions, and resistance to mechanical shock. Materials must demonstrate compliance with toxicity regulations, particularly for systems containing elements like boron or aluminum compounds that may present environmental hazards if improperly handled.
Future regulatory developments are trending toward performance-based standards rather than prescriptive requirements, allowing for innovation while maintaining safety. Harmonization efforts between major markets are accelerating to prevent regulatory fragmentation that could impede technology deployment. The International Partnership for Hydrogen and Fuel Cells in the Economy (IPHE) is coordinating global regulatory approaches to establish consistent safety frameworks.
Insurance considerations represent a significant challenge for hydrogen technologies, with underwriters requiring extensive safety data before providing coverage for novel storage systems. This has prompted industry consortia to develop standardized risk assessment protocols specifically for solid storage materials to facilitate insurance provision and commercial deployment.
National regulatory frameworks vary significantly across regions, with the European Union implementing the most comprehensive approach through directives like the Alternative Fuels Infrastructure Regulation (AFIR) and the Renewable Energy Directive (RED II), which include provisions for hydrogen infrastructure safety. The United States relies on ASME Boiler and Pressure Vessel Code and NFPA standards, while Japan has pioneered specific hydrogen safety codes through its High-Pressure Gas Safety Act.
Risk assessment methodologies for hydrogen solid storage materials focus on three primary hazard categories: thermal runaway during hydrogenation (exothermic reactions), potential for dust explosions with powdered materials, and material degradation leading to unexpected hydrogen release. These assessments must account for the unique properties of different material classes, including metal hydrides, complex hydrides, and carbon-based adsorbents.
Certification processes for hydrogen storage systems incorporating solid materials require extensive testing regimes including cycling stability, thermal management under extreme conditions, and resistance to mechanical shock. Materials must demonstrate compliance with toxicity regulations, particularly for systems containing elements like boron or aluminum compounds that may present environmental hazards if improperly handled.
Future regulatory developments are trending toward performance-based standards rather than prescriptive requirements, allowing for innovation while maintaining safety. Harmonization efforts between major markets are accelerating to prevent regulatory fragmentation that could impede technology deployment. The International Partnership for Hydrogen and Fuel Cells in the Economy (IPHE) is coordinating global regulatory approaches to establish consistent safety frameworks.
Insurance considerations represent a significant challenge for hydrogen technologies, with underwriters requiring extensive safety data before providing coverage for novel storage systems. This has prompted industry consortia to develop standardized risk assessment protocols specifically for solid storage materials to facilitate insurance provision and commercial deployment.
Environmental Impact Assessment of Storage Materials
The environmental impact of hydrogen solid storage materials must be comprehensively evaluated across their entire lifecycle. Current metal hydride storage systems demonstrate significant advantages over conventional fossil fuel technologies, with potential greenhouse gas emission reductions of 40-60% when hydrogen is produced from renewable sources. However, the production of these materials, particularly complex hydrides containing rare earth elements, raises sustainability concerns due to energy-intensive mining and refining processes.
Manufacturing processes for advanced storage materials like metal-organic frameworks (MOFs) and carbon-based nanostructures require substantial energy inputs and potentially hazardous chemicals. Life cycle assessments indicate that the environmental footprint of these materials is heavily front-loaded, with 60-75% of lifetime emissions occurring during production phases. This underscores the importance of developing cleaner manufacturing techniques and utilizing renewable energy in production facilities.
The operational phase of hydrogen storage materials presents minimal direct environmental impacts, as the hydrogen storage and release processes are generally non-polluting. The primary environmental benefit comes from displacing fossil fuel consumption, with each kilogram of hydrogen potentially offsetting 9-12 kg of CO2 emissions when used in fuel cell applications. Material degradation over multiple charge-discharge cycles remains a concern, as performance loss may lead to premature replacement and additional resource consumption.
End-of-life considerations reveal both challenges and opportunities. Many metal hydride materials contain valuable elements that can be recovered and recycled, potentially reducing the need for virgin material extraction by 30-40%. However, current recycling infrastructure for these specialized materials remains underdeveloped. Certain complex hydrides may also pose environmental risks if improperly disposed of, as they can release reactive compounds or leach metals into soil and groundwater.
Water consumption represents another critical environmental factor, particularly for hydrolysis-based hydrogen storage systems. These systems can require 9-15 liters of water per kilogram of hydrogen produced, raising concerns about deployment in water-scarce regions. Advanced closed-loop systems that recapture and reuse water are under development but remain technologically immature.
Land use impacts vary significantly between storage material types. While the physical footprint of solid storage systems is generally smaller than compressed gas alternatives, the upstream mining operations for certain metal components can cause substantial habitat disruption and biodiversity loss. Responsible sourcing practices and rehabilitation requirements are essential to mitigate these impacts.
Manufacturing processes for advanced storage materials like metal-organic frameworks (MOFs) and carbon-based nanostructures require substantial energy inputs and potentially hazardous chemicals. Life cycle assessments indicate that the environmental footprint of these materials is heavily front-loaded, with 60-75% of lifetime emissions occurring during production phases. This underscores the importance of developing cleaner manufacturing techniques and utilizing renewable energy in production facilities.
The operational phase of hydrogen storage materials presents minimal direct environmental impacts, as the hydrogen storage and release processes are generally non-polluting. The primary environmental benefit comes from displacing fossil fuel consumption, with each kilogram of hydrogen potentially offsetting 9-12 kg of CO2 emissions when used in fuel cell applications. Material degradation over multiple charge-discharge cycles remains a concern, as performance loss may lead to premature replacement and additional resource consumption.
End-of-life considerations reveal both challenges and opportunities. Many metal hydride materials contain valuable elements that can be recovered and recycled, potentially reducing the need for virgin material extraction by 30-40%. However, current recycling infrastructure for these specialized materials remains underdeveloped. Certain complex hydrides may also pose environmental risks if improperly disposed of, as they can release reactive compounds or leach metals into soil and groundwater.
Water consumption represents another critical environmental factor, particularly for hydrolysis-based hydrogen storage systems. These systems can require 9-15 liters of water per kilogram of hydrogen produced, raising concerns about deployment in water-scarce regions. Advanced closed-loop systems that recapture and reuse water are under development but remain technologically immature.
Land use impacts vary significantly between storage material types. While the physical footprint of solid storage systems is generally smaller than compressed gas alternatives, the upstream mining operations for certain metal components can cause substantial habitat disruption and biodiversity loss. Responsible sourcing practices and rehabilitation requirements are essential to mitigate these impacts.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!