Amide: Best Practices for Long-Term Cryogenic Storage
FEB 28, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Amide Cryogenic Storage Background and Objectives
Amide compounds represent a fundamental class of organic molecules characterized by the presence of a carbonyl group (C=O) linked to a nitrogen atom, forming the distinctive -CONH- functional group. These versatile chemical entities serve as critical building blocks in pharmaceutical synthesis, polymer chemistry, and advanced materials science. The significance of amides extends beyond their structural simplicity, as they exhibit unique physicochemical properties including hydrogen bonding capabilities, thermal stability, and diverse reactivity patterns that make them indispensable in modern chemical applications.
The evolution of cryogenic storage technology has emerged as a pivotal factor in preserving the integrity and functionality of temperature-sensitive amide compounds. Traditional storage methods often prove inadequate for maintaining the structural stability of complex amide derivatives, particularly those containing labile functional groups or stereochemical centers. The development of ultra-low temperature preservation techniques has revolutionized the field, enabling researchers and manufacturers to maintain compound libraries and pharmaceutical intermediates for extended periods without degradation.
Historical challenges in amide preservation have driven significant technological advancement in cryogenic storage methodologies. Early approaches suffered from issues including freeze-thaw damage, crystallization-induced structural changes, and moisture-related degradation. The recognition that different amide structures require tailored storage protocols has led to the development of compound-specific preservation strategies, incorporating factors such as molecular weight, polarity, and thermal sensitivity into storage design considerations.
The primary objective of establishing best practices for long-term cryogenic storage of amides centers on maximizing compound stability while maintaining chemical and biological activity over extended timeframes. This encompasses developing standardized protocols that minimize degradation pathways, prevent unwanted chemical transformations, and preserve the original properties of stored materials. The goal extends to creating reproducible storage conditions that ensure consistent results across different facilities and applications.
Contemporary research focuses on optimizing storage parameters including temperature selection, cooling rates, container materials, and atmospheric conditions to achieve maximum preservation efficiency. The integration of advanced monitoring systems and quality control measures represents a crucial component in validating storage effectiveness and detecting potential degradation before significant losses occur. These technological objectives aim to establish industry-wide standards that enhance the reliability and cost-effectiveness of amide compound preservation across pharmaceutical, research, and industrial applications.
The evolution of cryogenic storage technology has emerged as a pivotal factor in preserving the integrity and functionality of temperature-sensitive amide compounds. Traditional storage methods often prove inadequate for maintaining the structural stability of complex amide derivatives, particularly those containing labile functional groups or stereochemical centers. The development of ultra-low temperature preservation techniques has revolutionized the field, enabling researchers and manufacturers to maintain compound libraries and pharmaceutical intermediates for extended periods without degradation.
Historical challenges in amide preservation have driven significant technological advancement in cryogenic storage methodologies. Early approaches suffered from issues including freeze-thaw damage, crystallization-induced structural changes, and moisture-related degradation. The recognition that different amide structures require tailored storage protocols has led to the development of compound-specific preservation strategies, incorporating factors such as molecular weight, polarity, and thermal sensitivity into storage design considerations.
The primary objective of establishing best practices for long-term cryogenic storage of amides centers on maximizing compound stability while maintaining chemical and biological activity over extended timeframes. This encompasses developing standardized protocols that minimize degradation pathways, prevent unwanted chemical transformations, and preserve the original properties of stored materials. The goal extends to creating reproducible storage conditions that ensure consistent results across different facilities and applications.
Contemporary research focuses on optimizing storage parameters including temperature selection, cooling rates, container materials, and atmospheric conditions to achieve maximum preservation efficiency. The integration of advanced monitoring systems and quality control measures represents a crucial component in validating storage effectiveness and detecting potential degradation before significant losses occur. These technological objectives aim to establish industry-wide standards that enhance the reliability and cost-effectiveness of amide compound preservation across pharmaceutical, research, and industrial applications.
Market Demand for Long-Term Amide Preservation
The pharmaceutical and biotechnology industries represent the primary drivers of market demand for long-term amide preservation technologies. Pharmaceutical companies require stable storage solutions for peptide-based drugs, protein therapeutics, and complex organic compounds containing amide bonds. The growing pipeline of peptide therapeutics, particularly in oncology and metabolic disorders, has created substantial demand for reliable cryogenic storage methodologies that maintain molecular integrity over extended periods.
Academic and research institutions constitute another significant market segment, driven by the need to preserve valuable research samples, synthetic intermediates, and reference standards. Universities, government research facilities, and private research organizations require cost-effective solutions for maintaining extensive compound libraries and biological samples. The increasing emphasis on reproducible research and long-term studies has amplified the importance of proper amide preservation protocols.
The chemical manufacturing sector demonstrates growing interest in long-term amide storage solutions, particularly for specialty chemicals and advanced materials. Companies producing high-value synthetic intermediates, catalysts, and performance materials need reliable preservation methods to maintain product quality during extended storage periods. This demand is particularly pronounced in industries where batch-to-batch consistency is critical for downstream applications.
Biobanking and clinical research organizations represent an emerging market segment with specific requirements for amide-containing biological samples. The expansion of personalized medicine initiatives and large-scale epidemiological studies has created demand for standardized preservation protocols that ensure sample viability across decades of storage.
Market growth is further stimulated by regulatory requirements in pharmaceutical development, where stability data and long-term storage validation are mandatory for drug approval processes. The increasing complexity of modern therapeutics, including antibody-drug conjugates and peptide-protein hybrids, has elevated the importance of specialized preservation techniques.
Geographic demand patterns show concentration in regions with established pharmaceutical and biotechnology clusters, including North America, Europe, and Asia-Pacific. The market exhibits seasonal variations related to research funding cycles and pharmaceutical development timelines, with sustained growth projected due to expanding therapeutic pipelines and increasing research activities globally.
Academic and research institutions constitute another significant market segment, driven by the need to preserve valuable research samples, synthetic intermediates, and reference standards. Universities, government research facilities, and private research organizations require cost-effective solutions for maintaining extensive compound libraries and biological samples. The increasing emphasis on reproducible research and long-term studies has amplified the importance of proper amide preservation protocols.
The chemical manufacturing sector demonstrates growing interest in long-term amide storage solutions, particularly for specialty chemicals and advanced materials. Companies producing high-value synthetic intermediates, catalysts, and performance materials need reliable preservation methods to maintain product quality during extended storage periods. This demand is particularly pronounced in industries where batch-to-batch consistency is critical for downstream applications.
Biobanking and clinical research organizations represent an emerging market segment with specific requirements for amide-containing biological samples. The expansion of personalized medicine initiatives and large-scale epidemiological studies has created demand for standardized preservation protocols that ensure sample viability across decades of storage.
Market growth is further stimulated by regulatory requirements in pharmaceutical development, where stability data and long-term storage validation are mandatory for drug approval processes. The increasing complexity of modern therapeutics, including antibody-drug conjugates and peptide-protein hybrids, has elevated the importance of specialized preservation techniques.
Geographic demand patterns show concentration in regions with established pharmaceutical and biotechnology clusters, including North America, Europe, and Asia-Pacific. The market exhibits seasonal variations related to research funding cycles and pharmaceutical development timelines, with sustained growth projected due to expanding therapeutic pipelines and increasing research activities globally.
Current Challenges in Amide Cryogenic Storage
Long-term cryogenic storage of amides presents several critical challenges that significantly impact storage efficiency, material integrity, and operational costs. These challenges stem from the unique physicochemical properties of amides and the extreme conditions required for cryogenic preservation.
Thermal stability represents a primary concern during extended cryogenic storage periods. Amides undergo structural changes when subjected to repeated freeze-thaw cycles, leading to molecular degradation and reduced functional efficacy. The formation of ice crystals within amide solutions can cause mechanical stress on molecular structures, resulting in protein denaturation or compound fragmentation. This thermal instability becomes particularly pronounced in storage systems experiencing temperature fluctuations.
Container compatibility issues pose significant technical barriers to effective amide preservation. Many conventional cryogenic storage materials exhibit brittleness at ultra-low temperatures, leading to container failure and sample contamination. The interaction between amide compounds and storage vessel materials can result in adsorption losses, chemical reactions, or leaching of container components into stored samples. Selecting appropriate container materials that maintain structural integrity while preventing sample interaction remains a complex engineering challenge.
Contamination prevention during long-term storage requires sophisticated control mechanisms. Microbial growth, although significantly reduced at cryogenic temperatures, can still occur during handling procedures or temperature excursions. Cross-contamination between samples stored in proximity presents additional risks, particularly in high-density storage systems. The challenge intensifies when considering the need for periodic sample access without compromising the integrity of remaining stored materials.
Sample retrieval and handling procedures introduce operational complexities that can compromise amide stability. The transition from cryogenic to ambient temperatures must be carefully controlled to prevent thermal shock and condensation formation. Automated retrieval systems, while reducing human error, require sophisticated engineering solutions to maintain temperature control throughout the access process.
Storage infrastructure limitations constrain the scalability and reliability of long-term amide preservation. Power supply interruptions, equipment failures, and maintenance requirements can result in temperature excursions that compromise sample integrity. The high energy consumption associated with maintaining cryogenic temperatures presents both economic and environmental sustainability challenges.
Quality monitoring and validation throughout extended storage periods remain technically challenging. Traditional analytical methods may not be suitable for cryogenic conditions, requiring specialized instrumentation and protocols. Establishing reliable indicators of sample degradation without compromising storage conditions requires innovative monitoring approaches that can operate continuously under extreme temperature conditions.
Thermal stability represents a primary concern during extended cryogenic storage periods. Amides undergo structural changes when subjected to repeated freeze-thaw cycles, leading to molecular degradation and reduced functional efficacy. The formation of ice crystals within amide solutions can cause mechanical stress on molecular structures, resulting in protein denaturation or compound fragmentation. This thermal instability becomes particularly pronounced in storage systems experiencing temperature fluctuations.
Container compatibility issues pose significant technical barriers to effective amide preservation. Many conventional cryogenic storage materials exhibit brittleness at ultra-low temperatures, leading to container failure and sample contamination. The interaction between amide compounds and storage vessel materials can result in adsorption losses, chemical reactions, or leaching of container components into stored samples. Selecting appropriate container materials that maintain structural integrity while preventing sample interaction remains a complex engineering challenge.
Contamination prevention during long-term storage requires sophisticated control mechanisms. Microbial growth, although significantly reduced at cryogenic temperatures, can still occur during handling procedures or temperature excursions. Cross-contamination between samples stored in proximity presents additional risks, particularly in high-density storage systems. The challenge intensifies when considering the need for periodic sample access without compromising the integrity of remaining stored materials.
Sample retrieval and handling procedures introduce operational complexities that can compromise amide stability. The transition from cryogenic to ambient temperatures must be carefully controlled to prevent thermal shock and condensation formation. Automated retrieval systems, while reducing human error, require sophisticated engineering solutions to maintain temperature control throughout the access process.
Storage infrastructure limitations constrain the scalability and reliability of long-term amide preservation. Power supply interruptions, equipment failures, and maintenance requirements can result in temperature excursions that compromise sample integrity. The high energy consumption associated with maintaining cryogenic temperatures presents both economic and environmental sustainability challenges.
Quality monitoring and validation throughout extended storage periods remain technically challenging. Traditional analytical methods may not be suitable for cryogenic conditions, requiring specialized instrumentation and protocols. Establishing reliable indicators of sample degradation without compromising storage conditions requires innovative monitoring approaches that can operate continuously under extreme temperature conditions.
Existing Amide Cryopreservation Solutions
01 Use of stabilizers and antioxidants to prevent amide degradation
Stabilizers and antioxidants can be incorporated into amide-containing formulations to prevent oxidative degradation and improve storage stability. These additives help protect amide compounds from chemical breakdown during storage by scavenging free radicals and preventing oxidation reactions. The use of appropriate stabilizing agents can significantly extend the shelf life of amide-based products and maintain their chemical integrity over time.- Use of stabilizers and antioxidants to prevent amide degradation: Stabilizers and antioxidants can be incorporated into amide-containing formulations to prevent oxidative degradation and improve storage stability. These additives help protect amide compounds from chemical breakdown during storage by scavenging free radicals and preventing oxidation reactions. The use of appropriate stabilizing agents can significantly extend the shelf life of amide-based products and maintain their chemical integrity over time.
- pH adjustment and buffering systems for amide stability: Controlling the pH of amide formulations through buffering systems is critical for maintaining storage stability. Amides can be susceptible to hydrolysis under acidic or alkaline conditions, and maintaining an optimal pH range can prevent degradation. Buffer systems help maintain consistent pH levels during storage, protecting amide compounds from pH-induced decomposition and ensuring product stability over extended periods.
- Packaging and container selection for amide preservation: The selection of appropriate packaging materials and containers plays a crucial role in maintaining amide storage stability. Barrier properties of packaging materials can protect amide compounds from moisture, oxygen, and light exposure, which are common factors causing degradation. Specialized container designs and materials that minimize permeability and provide protection from environmental factors can significantly enhance the storage stability of amide-containing products.
- Temperature control and storage conditions optimization: Optimizing storage temperature and environmental conditions is essential for maintaining amide stability. Amide compounds may undergo thermal degradation or physical changes at elevated temperatures. Establishing appropriate storage temperature ranges and controlling environmental factors such as humidity can prevent degradation reactions and maintain product quality. Specific storage protocols and temperature-controlled systems can be implemented to ensure long-term stability of amide formulations.
- Formulation design with co-solvents and excipients for enhanced stability: The incorporation of specific co-solvents and excipients in amide formulations can improve storage stability by creating a more stable chemical environment. These additives can modify the solubility characteristics, reduce chemical reactivity, and provide physical stability to amide compounds. Careful selection of compatible excipients and solvent systems can minimize degradation pathways and enhance the overall stability profile of amide-based products during storage.
02 pH adjustment and buffering systems for amide stability
Controlling the pH of amide-containing formulations through buffering systems is critical for maintaining storage stability. Amides can be susceptible to hydrolysis under acidic or alkaline conditions, and maintaining an optimal pH range can prevent degradation. Buffer systems help maintain consistent pH levels during storage, protecting amide compounds from pH-induced decomposition and ensuring product stability over extended periods.Expand Specific Solutions03 Packaging and container selection for amide preservation
The selection of appropriate packaging materials and containers plays a crucial role in maintaining amide storage stability. Barrier properties of packaging materials can protect amide compounds from moisture, oxygen, and light exposure, which are common causes of degradation. Specialized container designs and materials that minimize permeability and provide protection from environmental factors can significantly enhance the storage stability of amide-containing products.Expand Specific Solutions04 Temperature control and storage conditions optimization
Optimizing storage temperature and environmental conditions is essential for maintaining amide stability. Amide compounds may undergo thermal degradation or physical changes at elevated temperatures, and controlling storage conditions can prevent such deterioration. Specific temperature ranges and humidity levels can be established to maximize stability, and formulations can be designed to remain stable under various storage conditions encountered during distribution and use.Expand Specific Solutions05 Formulation design with co-solvents and excipients for enhanced stability
The incorporation of specific co-solvents and excipients in amide formulations can enhance storage stability by preventing crystallization, aggregation, or chemical degradation. Careful selection of compatible excipients and solvent systems can create a stable matrix that protects amide compounds from degradation pathways. These formulation strategies may include the use of specific polymers, surfactants, or other additives that interact favorably with amide compounds to maintain their stability throughout the product shelf life.Expand Specific Solutions
Key Players in Cryogenic Storage Industry
The amide cryogenic storage technology sector represents an emerging niche within the broader biopreservation and industrial gas markets, currently in early-to-mid development stages with significant growth potential driven by expanding biotechnology and pharmaceutical applications. The global cryogenic storage market, valued at approximately $7 billion, is experiencing robust growth as demand increases for biological sample preservation and industrial applications. Technology maturity varies considerably across market participants, with established industrial gas leaders like Air Liquide SA and Praxair Technology demonstrating advanced cryogenic infrastructure capabilities, while specialized biopreservation companies such as BioLife Solutions and TMRW Life Sciences focus on innovative storage solutions for biological materials. Chemical manufacturers including Kyowa Hakko Bio, Mitsubishi Gas Chemical, and AlzChem contribute essential amide compounds and preservation media. The competitive landscape also features automotive players like General Motors and specialized medical technology firms like Cook Medical Technologies, indicating diverse application potential across industries seeking optimized long-term cryogenic storage protocols.
BioLife Solutions, Inc.
Technical Solution: BioLife Solutions specializes in developing advanced biopreservation media and automated thaw systems for long-term cryogenic storage of biological materials. Their CryoStor series provides optimized formulations that minimize cellular damage during freeze-thaw cycles, incorporating proprietary cryoprotectants and stabilizing agents. The company's solutions are specifically designed to maintain cell viability and functionality during extended storage periods at ultra-low temperatures, with comprehensive protocols for controlled rate freezing and rapid thawing processes.
Strengths: Industry-leading expertise in biopreservation with proven track record in maintaining cell viability. Weaknesses: Limited to biological applications, may not address broader industrial cryogenic storage needs.
Praxair Technology, Inc.
Technical Solution: Praxair Technology develops advanced cryogenic storage systems utilizing high-performance insulation materials and precision temperature control mechanisms. Their solutions incorporate multi-layer vacuum insulation, automated liquid nitrogen delivery systems, and sophisticated monitoring technologies to ensure stable long-term storage conditions. The company's approach emphasizes energy efficiency through optimized heat transfer reduction and automated replenishment systems that minimize temperature fluctuations during extended storage periods, particularly for industrial and laboratory applications.
Strengths: Strong engineering capabilities in cryogenic systems with focus on energy efficiency and automation. Weaknesses: Primarily focused on industrial applications, may lack specialized solutions for sensitive biological materials.
Core Innovations in Amide Stability Enhancement
Method for storing n-alkenyl carboxylic acid tertiary amide
PatentWO2006118319A1
Innovation
- Wetting the inner wall surface of storage containers with N-alkenyl carboxylic acid tertiary amides and controlling the temperature difference between the inner wall surface and the liquid within a specified range (not lower than -10°C) to maintain stability.
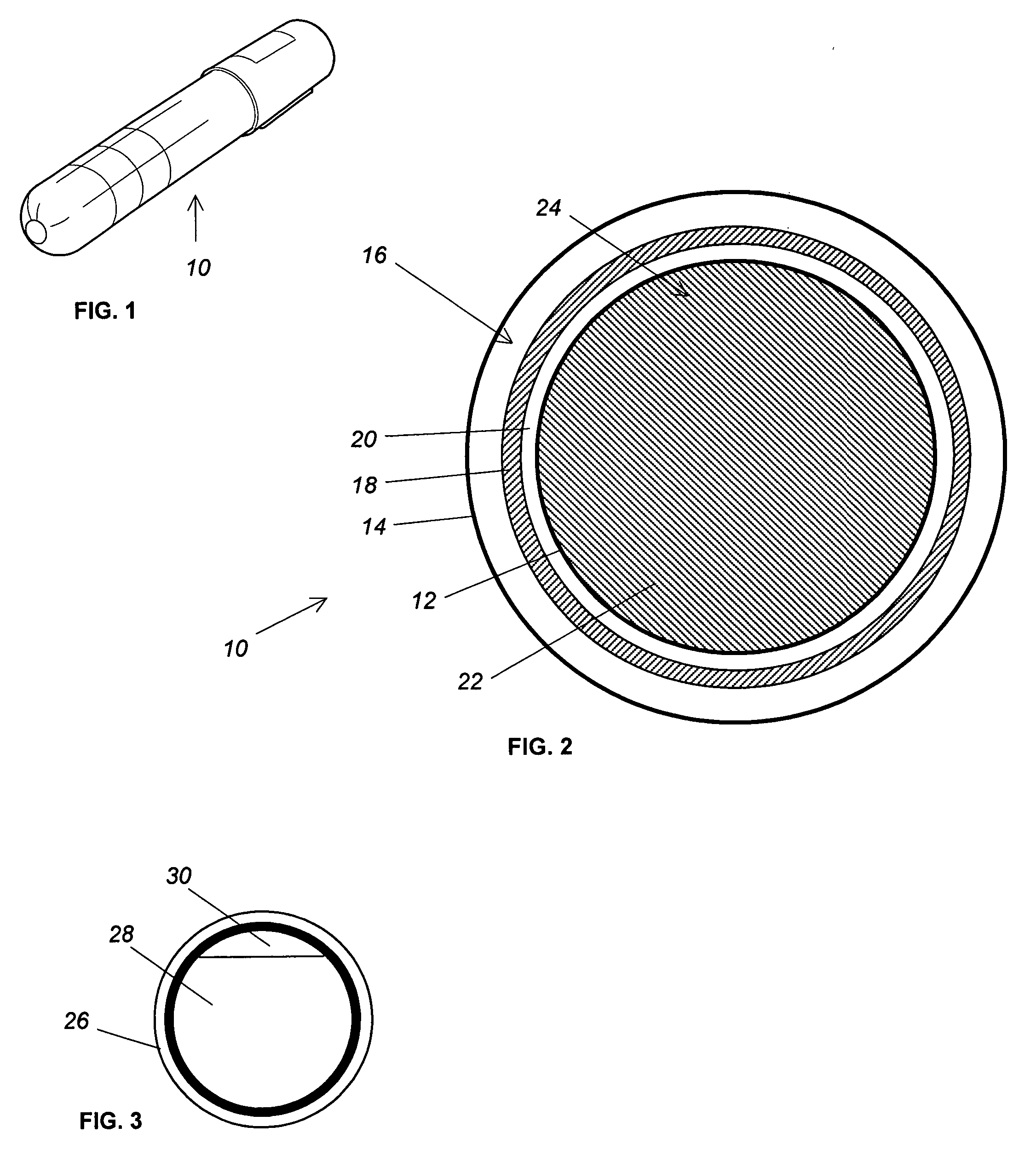
Reduced boiloff cryogen storage
PatentInactiveUS20060218940A1
Innovation
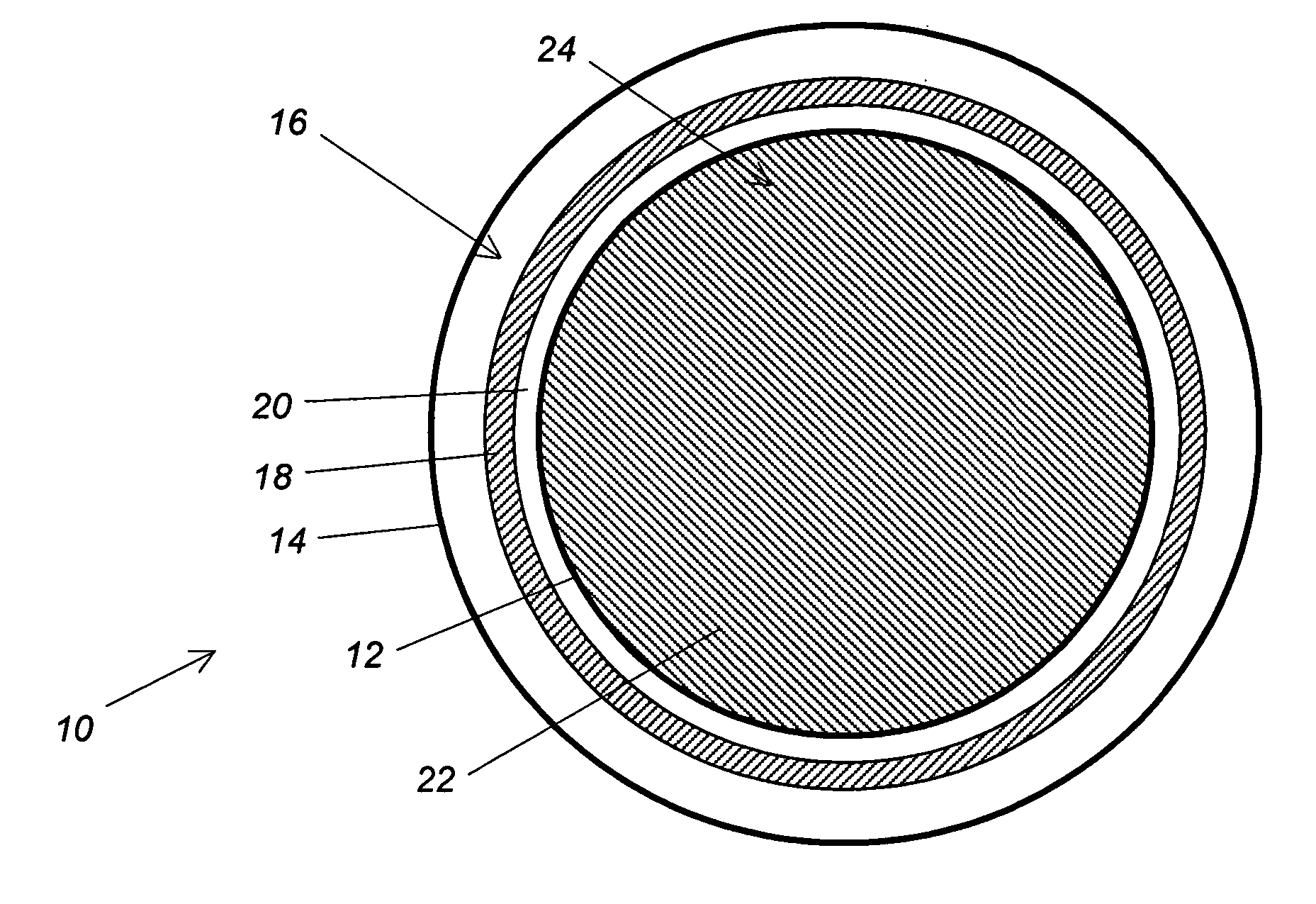
- A storage system with a nanoporous foam structure inside a tank shell, comprising a protective outer layer, an impermeable inner layer, and intermediate insulation layers, which reduces vapor pressure and heat leakage, thereby lowering boiloff rates without significant weight or complexity addition.
Safety Regulations for Cryogenic Chemical Storage
Cryogenic storage of amides requires strict adherence to comprehensive safety regulations established by multiple regulatory bodies. The Occupational Safety and Health Administration (OSHA) mandates specific protocols for handling materials at extremely low temperatures, including requirements for personal protective equipment, ventilation systems, and emergency response procedures. These regulations are complemented by guidelines from the Compressed Gas Association (CGA) and the National Institute for Occupational Safety and Health (NIOSH), which provide detailed specifications for cryogenic storage facility design and operational safety measures.
International standards such as ISO 21009 series and NFPA 55 establish critical requirements for cryogenic fluid storage systems. These standards mandate proper insulation specifications, pressure relief systems, and containment protocols specifically relevant to amide storage applications. The regulations require double-wall vacuum-insulated storage vessels with appropriate pressure monitoring systems and automatic venting mechanisms to prevent dangerous pressure buildup during long-term storage periods.
Personnel safety regulations emphasize mandatory training programs covering cryogenic hazard recognition, proper handling techniques, and emergency response procedures. Workers must be certified in the use of specialized protective equipment including cryogenic gloves, face shields, and insulated aprons. Regulations also require regular medical monitoring for personnel exposed to cryogenic environments, with specific attention to potential respiratory and skin exposure risks associated with amide vapors at low temperatures.
Facility design regulations mandate specific architectural requirements including adequate ventilation rates, oxygen monitoring systems, and emergency evacuation procedures. Storage areas must maintain minimum clearance distances from ignition sources and populated areas, with specialized flooring materials capable of withstanding extreme temperature variations. Fire suppression systems must be specifically designed for cryogenic environments, often requiring inert gas flooding systems rather than traditional water-based suppression methods.
Environmental compliance regulations address waste disposal, emission monitoring, and groundwater protection measures. The Environmental Protection Agency (EPA) requires specific documentation for amide storage operations, including regular environmental impact assessments and contamination prevention protocols. These regulations mandate secondary containment systems and groundwater monitoring wells in facilities storing significant quantities of cryogenic amides for extended periods.
International standards such as ISO 21009 series and NFPA 55 establish critical requirements for cryogenic fluid storage systems. These standards mandate proper insulation specifications, pressure relief systems, and containment protocols specifically relevant to amide storage applications. The regulations require double-wall vacuum-insulated storage vessels with appropriate pressure monitoring systems and automatic venting mechanisms to prevent dangerous pressure buildup during long-term storage periods.
Personnel safety regulations emphasize mandatory training programs covering cryogenic hazard recognition, proper handling techniques, and emergency response procedures. Workers must be certified in the use of specialized protective equipment including cryogenic gloves, face shields, and insulated aprons. Regulations also require regular medical monitoring for personnel exposed to cryogenic environments, with specific attention to potential respiratory and skin exposure risks associated with amide vapors at low temperatures.
Facility design regulations mandate specific architectural requirements including adequate ventilation rates, oxygen monitoring systems, and emergency evacuation procedures. Storage areas must maintain minimum clearance distances from ignition sources and populated areas, with specialized flooring materials capable of withstanding extreme temperature variations. Fire suppression systems must be specifically designed for cryogenic environments, often requiring inert gas flooding systems rather than traditional water-based suppression methods.
Environmental compliance regulations address waste disposal, emission monitoring, and groundwater protection measures. The Environmental Protection Agency (EPA) requires specific documentation for amide storage operations, including regular environmental impact assessments and contamination prevention protocols. These regulations mandate secondary containment systems and groundwater monitoring wells in facilities storing significant quantities of cryogenic amides for extended periods.
Quality Control Standards for Amide Preservation
Quality control standards for amide preservation during long-term cryogenic storage represent a critical framework ensuring molecular integrity and functional stability throughout extended storage periods. These standards encompass comprehensive protocols that address the unique challenges posed by ultra-low temperature environments and the inherent chemical properties of amide compounds.
The foundation of effective quality control lies in establishing rigorous pre-storage assessment protocols. These include comprehensive purity analysis using high-performance liquid chromatography and mass spectrometry to establish baseline molecular profiles. Moisture content determination becomes particularly crucial, as residual water can lead to ice crystal formation and subsequent structural damage during freezing cycles. Additionally, pH monitoring and buffer system validation ensure optimal chemical environment maintenance throughout the storage duration.
Temperature monitoring and control systems form the backbone of preservation quality standards. Continuous temperature logging with redundant sensor networks ensures maintenance of target storage temperatures, typically ranging from -80°C to -196°C depending on specific amide characteristics. Alarm systems with immediate notification protocols prevent temperature excursions that could compromise sample integrity. Regular calibration of monitoring equipment according to international standards maintains measurement accuracy and reliability.
Container integrity and material compatibility standards address the physical aspects of preservation quality. Selection of appropriate storage vessels requires consideration of thermal expansion coefficients, chemical inertness, and barrier properties against moisture and oxygen infiltration. Seal integrity testing protocols, including helium leak detection and pressure decay analysis, ensure long-term containment effectiveness under cryogenic conditions.
Periodic sampling and analytical verification protocols establish ongoing quality assurance throughout storage periods. These include scheduled integrity assessments using spectroscopic methods to detect potential degradation products or structural modifications. Comparative analysis against reference standards enables early detection of preservation quality deviations. Documentation requirements encompass detailed record-keeping of all quality control measurements, environmental conditions, and any observed anomalies.
Validation procedures for quality control methods ensure reliability and reproducibility of preservation assessment techniques. This includes method qualification studies demonstrating analytical sensitivity, specificity, and precision under cryogenic storage conditions. Regular proficiency testing and inter-laboratory comparisons maintain measurement consistency and identify potential systematic errors in quality control processes.
The foundation of effective quality control lies in establishing rigorous pre-storage assessment protocols. These include comprehensive purity analysis using high-performance liquid chromatography and mass spectrometry to establish baseline molecular profiles. Moisture content determination becomes particularly crucial, as residual water can lead to ice crystal formation and subsequent structural damage during freezing cycles. Additionally, pH monitoring and buffer system validation ensure optimal chemical environment maintenance throughout the storage duration.
Temperature monitoring and control systems form the backbone of preservation quality standards. Continuous temperature logging with redundant sensor networks ensures maintenance of target storage temperatures, typically ranging from -80°C to -196°C depending on specific amide characteristics. Alarm systems with immediate notification protocols prevent temperature excursions that could compromise sample integrity. Regular calibration of monitoring equipment according to international standards maintains measurement accuracy and reliability.
Container integrity and material compatibility standards address the physical aspects of preservation quality. Selection of appropriate storage vessels requires consideration of thermal expansion coefficients, chemical inertness, and barrier properties against moisture and oxygen infiltration. Seal integrity testing protocols, including helium leak detection and pressure decay analysis, ensure long-term containment effectiveness under cryogenic conditions.
Periodic sampling and analytical verification protocols establish ongoing quality assurance throughout storage periods. These include scheduled integrity assessments using spectroscopic methods to detect potential degradation products or structural modifications. Comparative analysis against reference standards enables early detection of preservation quality deviations. Documentation requirements encompass detailed record-keeping of all quality control measurements, environmental conditions, and any observed anomalies.
Validation procedures for quality control methods ensure reliability and reproducibility of preservation assessment techniques. This includes method qualification studies demonstrating analytical sensitivity, specificity, and precision under cryogenic storage conditions. Regular proficiency testing and inter-laboratory comparisons maintain measurement consistency and identify potential systematic errors in quality control processes.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!