Aromatic Compounds vs Nitroaromatics: Environmental Hazards
MAR 5, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Aromatic vs Nitroaromatic Environmental Impact Background
The environmental impact of aromatic compounds and nitroaromatic compounds has emerged as a critical concern in industrial chemistry and environmental science over the past several decades. Both compound classes represent significant challenges to environmental sustainability, yet their impact mechanisms, persistence patterns, and remediation requirements differ substantially. Understanding these differences has become essential for developing effective environmental management strategies and regulatory frameworks.
Aromatic compounds, characterized by their benzene ring structures, have been widely used in industrial applications since the early 20th century. Their environmental presence stems primarily from petroleum refining, chemical manufacturing, and combustion processes. These compounds exhibit varying degrees of environmental persistence, with some demonstrating significant bioaccumulation potential and chronic toxicity effects on aquatic ecosystems.
Nitroaromatic compounds represent a more specialized subset, typically arising from explosive manufacturing, pharmaceutical production, and chemical synthesis processes. The introduction of nitro functional groups to aromatic structures fundamentally alters their environmental behavior, generally increasing their toxicity while creating unique degradation pathways. Historical contamination events, particularly at former munitions manufacturing sites, have highlighted the persistent nature of these compounds in soil and groundwater systems.
The recognition of environmental hazards associated with both compound classes has evolved significantly since the 1970s, driven by improved analytical detection methods and growing awareness of long-term ecological impacts. Early environmental studies focused primarily on acute toxicity effects, but subsequent research has revealed complex interactions with biological systems, including endocrine disruption and carcinogenic potential.
Contemporary environmental challenges involve addressing legacy contamination while preventing future releases through improved industrial practices. The development of advanced treatment technologies, including bioremediation and advanced oxidation processes, reflects the ongoing need for effective solutions to manage these persistent environmental contaminants.
Regulatory frameworks have progressively tightened controls on both aromatic and nitroaromatic compound releases, establishing increasingly stringent discharge limits and requiring comprehensive environmental monitoring programs. This regulatory evolution continues to drive innovation in pollution prevention technologies and alternative chemical processes.
Aromatic compounds, characterized by their benzene ring structures, have been widely used in industrial applications since the early 20th century. Their environmental presence stems primarily from petroleum refining, chemical manufacturing, and combustion processes. These compounds exhibit varying degrees of environmental persistence, with some demonstrating significant bioaccumulation potential and chronic toxicity effects on aquatic ecosystems.
Nitroaromatic compounds represent a more specialized subset, typically arising from explosive manufacturing, pharmaceutical production, and chemical synthesis processes. The introduction of nitro functional groups to aromatic structures fundamentally alters their environmental behavior, generally increasing their toxicity while creating unique degradation pathways. Historical contamination events, particularly at former munitions manufacturing sites, have highlighted the persistent nature of these compounds in soil and groundwater systems.
The recognition of environmental hazards associated with both compound classes has evolved significantly since the 1970s, driven by improved analytical detection methods and growing awareness of long-term ecological impacts. Early environmental studies focused primarily on acute toxicity effects, but subsequent research has revealed complex interactions with biological systems, including endocrine disruption and carcinogenic potential.
Contemporary environmental challenges involve addressing legacy contamination while preventing future releases through improved industrial practices. The development of advanced treatment technologies, including bioremediation and advanced oxidation processes, reflects the ongoing need for effective solutions to manage these persistent environmental contaminants.
Regulatory frameworks have progressively tightened controls on both aromatic and nitroaromatic compound releases, establishing increasingly stringent discharge limits and requiring comprehensive environmental monitoring programs. This regulatory evolution continues to drive innovation in pollution prevention technologies and alternative chemical processes.
Market Demand for Safer Aromatic Alternatives
The global chemical industry is experiencing unprecedented pressure to transition away from hazardous aromatic compounds, particularly nitroaromatics, driven by increasingly stringent environmental regulations and growing corporate sustainability commitments. This regulatory landscape has created substantial market opportunities for safer aromatic alternatives across multiple industrial sectors.
The pharmaceutical industry represents one of the largest demand drivers for safer aromatic alternatives, as manufacturers seek to replace traditional nitroaromatic intermediates in drug synthesis. Major pharmaceutical companies are actively reformulating existing products and designing new synthetic pathways that eliminate or minimize the use of environmentally persistent aromatic compounds. This shift is particularly pronounced in the production of antibiotics, analgesics, and cardiovascular medications where nitroaromatic compounds have historically been prevalent.
The agrochemical sector demonstrates equally strong demand for environmentally benign aromatic alternatives. Traditional pesticide and herbicide formulations containing nitroaromatic compounds face mounting regulatory scrutiny due to their persistence in soil and groundwater systems. Agricultural chemical manufacturers are investing heavily in developing bio-based aromatic compounds and synthetic alternatives that maintain efficacy while reducing environmental impact.
Industrial applications, including dyes, pigments, and specialty chemicals, constitute another significant market segment driving demand for safer alternatives. The textile industry, in particular, is under pressure from both regulatory bodies and consumer brands to eliminate hazardous aromatic compounds from their supply chains. This has created opportunities for companies developing plant-based aromatic compounds and novel synthetic pathways that avoid nitroaromatic intermediates.
The explosives and propellants industry, traditionally reliant on nitroaromatic compounds, is exploring alternative energetic materials that provide comparable performance characteristics while reducing manufacturing and disposal risks. Military and civilian applications are both driving research into safer explosive formulations.
Market demand is further amplified by the growing emphasis on green chemistry principles and circular economy models. Companies are increasingly seeking aromatic alternatives that not only reduce environmental hazards but also offer improved biodegradability, lower toxicity profiles, and reduced carbon footprints throughout their lifecycle.
The convergence of regulatory pressure, corporate sustainability initiatives, and consumer awareness has created a robust and expanding market for safer aromatic alternatives, with demand expected to continue growing as environmental standards become more stringent globally.
The pharmaceutical industry represents one of the largest demand drivers for safer aromatic alternatives, as manufacturers seek to replace traditional nitroaromatic intermediates in drug synthesis. Major pharmaceutical companies are actively reformulating existing products and designing new synthetic pathways that eliminate or minimize the use of environmentally persistent aromatic compounds. This shift is particularly pronounced in the production of antibiotics, analgesics, and cardiovascular medications where nitroaromatic compounds have historically been prevalent.
The agrochemical sector demonstrates equally strong demand for environmentally benign aromatic alternatives. Traditional pesticide and herbicide formulations containing nitroaromatic compounds face mounting regulatory scrutiny due to their persistence in soil and groundwater systems. Agricultural chemical manufacturers are investing heavily in developing bio-based aromatic compounds and synthetic alternatives that maintain efficacy while reducing environmental impact.
Industrial applications, including dyes, pigments, and specialty chemicals, constitute another significant market segment driving demand for safer alternatives. The textile industry, in particular, is under pressure from both regulatory bodies and consumer brands to eliminate hazardous aromatic compounds from their supply chains. This has created opportunities for companies developing plant-based aromatic compounds and novel synthetic pathways that avoid nitroaromatic intermediates.
The explosives and propellants industry, traditionally reliant on nitroaromatic compounds, is exploring alternative energetic materials that provide comparable performance characteristics while reducing manufacturing and disposal risks. Military and civilian applications are both driving research into safer explosive formulations.
Market demand is further amplified by the growing emphasis on green chemistry principles and circular economy models. Companies are increasingly seeking aromatic alternatives that not only reduce environmental hazards but also offer improved biodegradability, lower toxicity profiles, and reduced carbon footprints throughout their lifecycle.
The convergence of regulatory pressure, corporate sustainability initiatives, and consumer awareness has created a robust and expanding market for safer aromatic alternatives, with demand expected to continue growing as environmental standards become more stringent globally.
Current Environmental Hazards of Nitroaromatic Compounds
Nitroaromatic compounds represent one of the most persistent and hazardous classes of environmental contaminants, posing significant threats to both ecological systems and human health. These synthetic organic chemicals, characterized by the presence of nitro functional groups (-NO2) attached to aromatic rings, have become ubiquitous pollutants due to their extensive industrial applications and inherent resistance to natural degradation processes.
The primary environmental concern stems from their exceptional persistence in various environmental matrices. Unlike their parent aromatic compounds, nitroaromatics exhibit enhanced stability due to the electron-withdrawing nature of nitro groups, which significantly reduces their susceptibility to microbial degradation and photolysis. This persistence leads to bioaccumulation in food chains, with concentrations magnifying at higher trophic levels.
Soil contamination represents a critical pathway for nitroaromatic exposure, particularly around manufacturing facilities, military installations, and former explosive production sites. Compounds such as 2,4,6-trinitrotoluene (TNT) and its metabolites can persist in soil for decades, creating long-term contamination plumes that affect groundwater resources. The low water solubility of many nitroaromatics paradoxically contributes to their environmental persistence while limiting natural attenuation processes.
Aquatic ecosystems face severe impacts from nitroaromatic contamination, with these compounds demonstrating acute and chronic toxicity to aquatic organisms. Studies have documented significant effects on fish reproduction, development, and immune system function at environmentally relevant concentrations. The compounds' lipophilic nature facilitates bioaccumulation in fatty tissues, leading to biomagnification through aquatic food webs.
Human health risks associated with nitroaromatic exposure are multifaceted and severe. Many nitroaromatic compounds are classified as probable or confirmed carcinogens, with occupational exposure studies linking them to increased rates of bladder cancer, liver dysfunction, and hematological disorders. The compounds' ability to form reactive metabolites upon biotransformation contributes to their mutagenic and carcinogenic potential.
Atmospheric contamination occurs through industrial emissions and volatilization from contaminated sites. Airborne nitroaromatics can undergo photochemical reactions, forming secondary pollutants that contribute to urban air quality degradation. Workers in industries handling these compounds face elevated exposure risks through inhalation and dermal contact pathways.
The remediation challenges posed by nitroaromatic contamination are substantial due to their chemical stability and resistance to conventional treatment technologies. Traditional biological treatment systems often prove ineffective, necessitating advanced oxidation processes or specialized bioremediation approaches using adapted microbial consortia.
The primary environmental concern stems from their exceptional persistence in various environmental matrices. Unlike their parent aromatic compounds, nitroaromatics exhibit enhanced stability due to the electron-withdrawing nature of nitro groups, which significantly reduces their susceptibility to microbial degradation and photolysis. This persistence leads to bioaccumulation in food chains, with concentrations magnifying at higher trophic levels.
Soil contamination represents a critical pathway for nitroaromatic exposure, particularly around manufacturing facilities, military installations, and former explosive production sites. Compounds such as 2,4,6-trinitrotoluene (TNT) and its metabolites can persist in soil for decades, creating long-term contamination plumes that affect groundwater resources. The low water solubility of many nitroaromatics paradoxically contributes to their environmental persistence while limiting natural attenuation processes.
Aquatic ecosystems face severe impacts from nitroaromatic contamination, with these compounds demonstrating acute and chronic toxicity to aquatic organisms. Studies have documented significant effects on fish reproduction, development, and immune system function at environmentally relevant concentrations. The compounds' lipophilic nature facilitates bioaccumulation in fatty tissues, leading to biomagnification through aquatic food webs.
Human health risks associated with nitroaromatic exposure are multifaceted and severe. Many nitroaromatic compounds are classified as probable or confirmed carcinogens, with occupational exposure studies linking them to increased rates of bladder cancer, liver dysfunction, and hematological disorders. The compounds' ability to form reactive metabolites upon biotransformation contributes to their mutagenic and carcinogenic potential.
Atmospheric contamination occurs through industrial emissions and volatilization from contaminated sites. Airborne nitroaromatics can undergo photochemical reactions, forming secondary pollutants that contribute to urban air quality degradation. Workers in industries handling these compounds face elevated exposure risks through inhalation and dermal contact pathways.
The remediation challenges posed by nitroaromatic contamination are substantial due to their chemical stability and resistance to conventional treatment technologies. Traditional biological treatment systems often prove ineffective, necessitating advanced oxidation processes or specialized bioremediation approaches using adapted microbial consortia.
Existing Hazard Mitigation Solutions
01 Biodegradation and bioremediation of nitroaromatic compounds
Methods and compositions for the biological degradation of nitroaromatic compounds in contaminated environments. These approaches utilize microorganisms, enzymes, or biological systems capable of breaking down toxic nitroaromatic pollutants into less harmful substances. The biodegradation processes can involve aerobic or anaerobic pathways, and may employ naturally occurring or genetically modified organisms to enhance degradation efficiency in soil, water, or industrial waste streams.- Biodegradation and bioremediation of nitroaromatic compounds: Methods and compositions for the biological degradation of nitroaromatic compounds in contaminated environments. These approaches utilize microorganisms, enzymes, or biological systems capable of breaking down toxic nitroaromatic pollutants into less harmful substances. The biodegradation processes can involve aerobic or anaerobic pathways, and may employ naturally occurring or genetically modified organisms to enhance degradation efficiency in soil, water, or industrial waste streams.
- Chemical reduction and transformation of nitroaromatic compounds: Chemical methods for reducing or transforming nitroaromatic compounds to minimize their environmental hazards. These techniques involve catalytic reduction, chemical oxidation, or other transformation processes that convert toxic nitroaromatic substances into less hazardous products. The methods may employ various catalysts, reducing agents, or reaction conditions to achieve efficient conversion while minimizing secondary pollution.
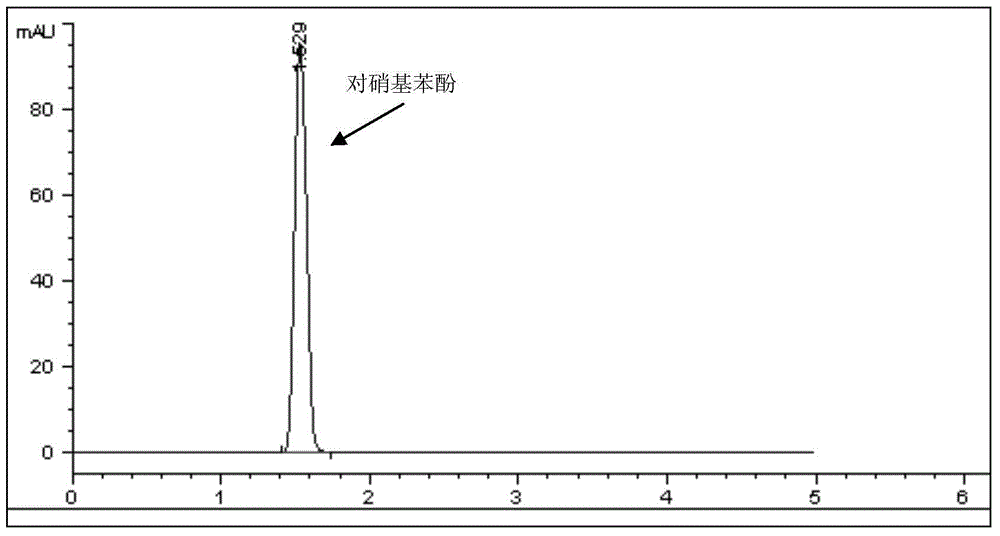
- Detection and monitoring of aromatic and nitroaromatic pollutants: Technologies and methods for detecting, identifying, and monitoring aromatic and nitroaromatic compounds in environmental samples. These approaches include analytical techniques, sensor systems, and monitoring devices that can identify the presence and concentration of hazardous aromatic pollutants in air, water, soil, or industrial emissions. The detection methods enable environmental assessment and compliance monitoring for pollution control.
- Adsorption and removal of aromatic pollutants from contaminated media: Techniques for removing aromatic and nitroaromatic compounds from contaminated environments through adsorption processes. These methods utilize adsorbent materials such as activated carbon, modified clays, polymeric materials, or nanomaterials to capture and remove toxic aromatic pollutants from water, air, or soil. The adsorption approaches provide effective means for environmental remediation and pollution control in various contaminated sites.
- Safe handling and disposal of nitroaromatic waste materials: Methods and systems for the safe handling, treatment, and disposal of waste materials containing aromatic and nitroaromatic compounds. These approaches address the environmental hazards associated with industrial waste, manufacturing byproducts, or contaminated materials containing toxic aromatic substances. The methods may include stabilization, encapsulation, thermal treatment, or other disposal techniques that prevent environmental release and minimize ecological risks.
02 Chemical reduction and transformation of nitroaromatic compounds
Chemical methods for reducing or transforming nitroaromatic compounds to minimize their environmental hazards. These techniques involve catalytic reduction, chemical oxidation, or other transformation processes that convert toxic nitroaromatic substances into less hazardous products. The methods may utilize various catalysts, reducing agents, or reaction conditions to achieve efficient conversion while minimizing secondary pollution.Expand Specific Solutions03 Detection and monitoring of aromatic and nitroaromatic pollutants
Technologies and methods for detecting, identifying, and monitoring aromatic and nitroaromatic compounds in environmental samples. These approaches include analytical techniques, sensor systems, and monitoring devices that can identify the presence and concentration of hazardous aromatic pollutants in air, water, soil, or industrial emissions. The detection methods enable environmental assessment and compliance monitoring for pollution control.Expand Specific Solutions04 Adsorption and removal of aromatic pollutants from contaminated media
Techniques for removing aromatic and nitroaromatic compounds from contaminated environments through adsorption processes. These methods employ various adsorbent materials such as activated carbon, modified clays, polymeric materials, or nanomaterials to capture and remove toxic aromatic pollutants from water, air, or soil. The adsorption approaches provide effective means for environmental remediation and pollution control in industrial and municipal applications.Expand Specific Solutions05 Safe handling and disposal of nitroaromatic waste materials
Methods and systems for the safe handling, treatment, and disposal of waste materials containing aromatic and nitroaromatic compounds. These approaches address the environmental hazards associated with industrial waste, manufacturing byproducts, or contaminated materials containing toxic aromatic substances. The methods may include stabilization, encapsulation, thermal treatment, or other disposal techniques that prevent environmental release and minimize ecological risks.Expand Specific Solutions
Key Players in Aromatic Chemical Industry
The aromatic compounds versus nitroaromatics environmental hazards field represents a mature but evolving market driven by increasing environmental regulations and sustainability demands. The industry is experiencing significant growth, with market expansion fueled by stricter safety standards and green chemistry initiatives. Technology maturity varies considerably across players, with established chemical giants like BASF Corp., DuPont de Nemours, and 3M Innovative Properties leading in advanced remediation and detection technologies. Specialty companies such as Anellotech and P2 Science are pioneering innovative bio-based alternatives to traditional aromatic compounds, while fragrance leaders Givaudan SA, Firmenich SA, and Symrise are developing safer aromatic formulations. Academic institutions including Oregon State University, Shanghai University, and Nanjing University contribute cutting-edge research in environmental impact assessment and mitigation strategies. The competitive landscape shows a clear division between traditional chemical manufacturers focusing on compliance solutions and emerging companies developing sustainable alternatives to address environmental concerns.
BASF Corp.
Technical Solution: BASF has developed comprehensive environmental monitoring and remediation technologies for aromatic compounds and nitroaromatics. Their approach includes advanced catalytic oxidation processes for degrading nitroaromatic pollutants in industrial wastewater, utilizing proprietary catalyst formulations that achieve over 95% removal efficiency. The company has implemented bioaugmentation strategies using specialized microbial consortia capable of metabolizing both simple aromatic compounds and complex nitroaromatics like TNT and DNT. BASF's integrated treatment systems combine physical-chemical pretreatment with biological degradation, followed by advanced oxidation processes to ensure complete mineralization of hazardous compounds.
Strengths: Extensive industrial experience, proven large-scale implementation, comprehensive treatment approach. Weaknesses: High capital investment requirements, complex system maintenance needs.
DuPont de Nemours, Inc.
Technical Solution: DuPont has pioneered innovative membrane separation technologies specifically designed for removing aromatic and nitroaromatic contaminants from environmental matrices. Their proprietary polymer membranes demonstrate selective permeability characteristics, effectively concentrating hazardous compounds for subsequent treatment while allowing clean water passage. The company's approach integrates electrochemical reduction processes that convert nitroaromatic compounds to less toxic aromatic amines, followed by advanced oxidation to achieve complete degradation. DuPont's systems incorporate real-time monitoring capabilities using spectroscopic sensors to track contaminant levels and treatment efficiency, ensuring regulatory compliance and optimal performance.
Strengths: Advanced membrane technology, selective separation capabilities, integrated monitoring systems. Weaknesses: Membrane fouling issues, limited applicability to highly concentrated waste streams.
Core Innovations in Nitroaromatic Risk Assessment
Arthrobacter and application thereof
PatentInactiveCN104946556A
Innovation
- A strain of Arthrobacter bacteria named YC-RL1 was isolated and identified. This strain can 100% degrade 100 mg/L of naphthalene, fluorene, biphenyl, 1,2,3,4-tetrachlorobenzene and parabens within 4 days. The nitrophenol mixture maintains degradability at higher salt concentrations and is used in environmental remediation and industrial wastewater treatment by preparing bacterial agents and biological cleaners.
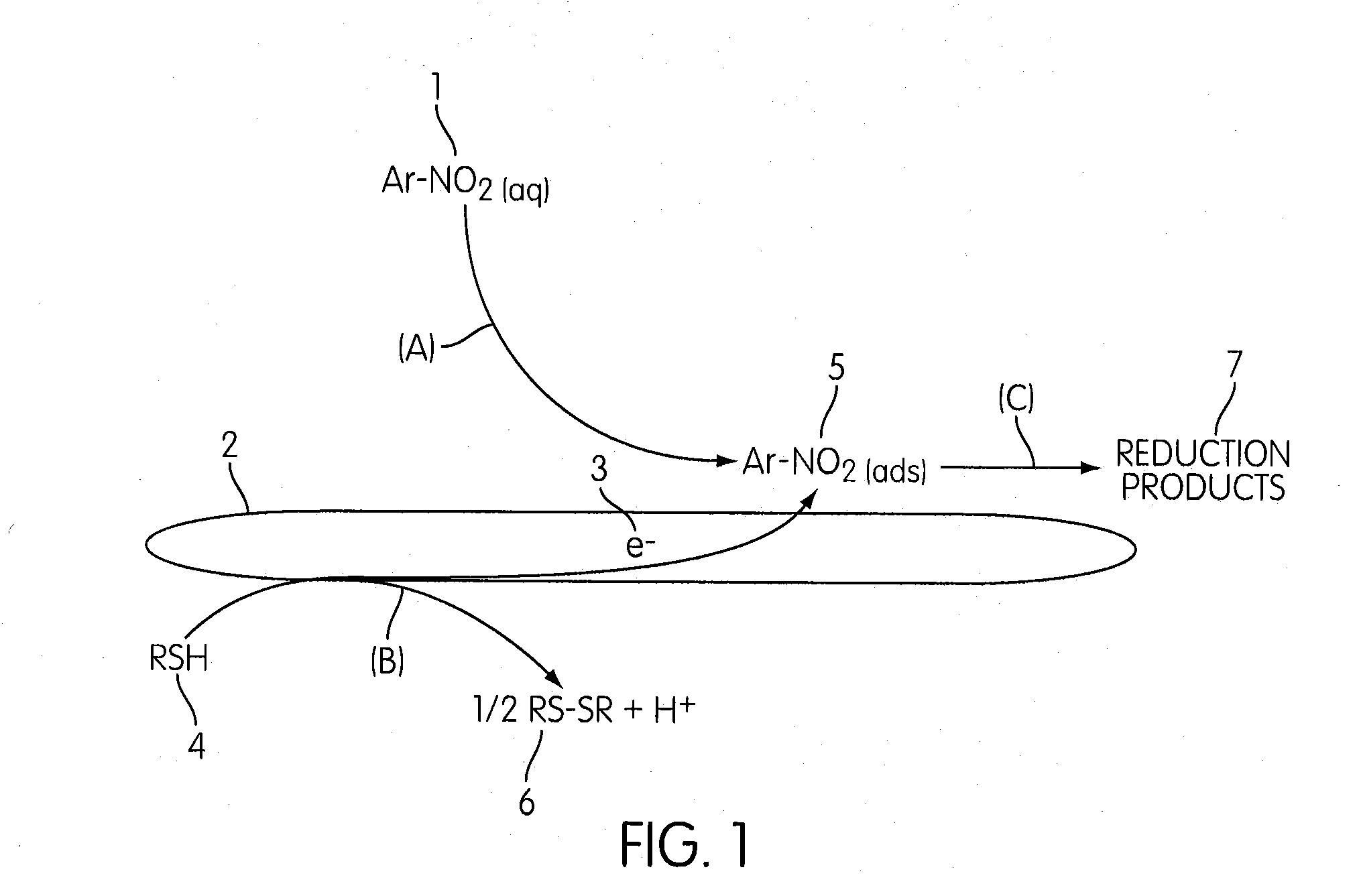
Method for remediating nitro and/or halogenated compound-contaminated soil, sediment or water using graphitic carbon as a catalytic sorbent
PatentInactiveUS20100158612A1
Innovation
- The use of graphitic carbon as both an adsorbent and catalyst, in conjunction with reducing agents like elemental iron or sulfur-containing compounds, to facilitate reductive degradation of adsorbed organic contaminants, converting them into more water-soluble and biodegradable compounds.
Environmental Regulations for Aromatic Compounds
The regulatory landscape for aromatic compounds has evolved significantly over the past decades, driven by mounting scientific evidence of their environmental and health impacts. International frameworks such as the Stockholm Convention on Persistent Organic Pollutants and the Rotterdam Convention have established foundational principles for controlling hazardous aromatic substances across national boundaries.
In the United States, the Environmental Protection Agency regulates aromatic compounds under multiple statutory authorities. The Clean Air Act addresses volatile aromatic compounds as hazardous air pollutants, with specific emission standards for benzene, toluene, and xylene isomers. The Clean Water Act establishes discharge limits for aromatic compounds in industrial wastewater, while the Resource Conservation and Recovery Act governs the disposal of aromatic waste materials.
European Union regulations present a comprehensive approach through the REACH Regulation, which requires extensive registration, evaluation, and authorization of aromatic chemical substances. The Industrial Emissions Directive sets strict emission limit values for aromatic compounds from industrial installations. Additionally, the Water Framework Directive identifies priority aromatic substances requiring enhanced monitoring and control measures.
Nitroaromatic compounds face particularly stringent regulatory oversight due to their enhanced toxicity profiles. The European Chemicals Agency has classified numerous nitroaromatic substances as substances of very high concern, triggering authorization requirements and substitution obligations. Similar classifications exist under various national chemical management systems worldwide.
Occupational exposure limits for aromatic compounds vary significantly across jurisdictions but generally follow similar risk assessment methodologies. The American Conference of Governmental Industrial Hygienists and equivalent international bodies regularly update threshold limit values based on emerging toxicological data.
Recent regulatory trends indicate increasing emphasis on mixture toxicity assessment, recognizing that aromatic compounds rarely exist in isolation within environmental matrices. This shift toward cumulative risk assessment approaches represents a fundamental evolution in regulatory thinking, potentially leading to more restrictive future standards for aromatic compound emissions and exposures.
In the United States, the Environmental Protection Agency regulates aromatic compounds under multiple statutory authorities. The Clean Air Act addresses volatile aromatic compounds as hazardous air pollutants, with specific emission standards for benzene, toluene, and xylene isomers. The Clean Water Act establishes discharge limits for aromatic compounds in industrial wastewater, while the Resource Conservation and Recovery Act governs the disposal of aromatic waste materials.
European Union regulations present a comprehensive approach through the REACH Regulation, which requires extensive registration, evaluation, and authorization of aromatic chemical substances. The Industrial Emissions Directive sets strict emission limit values for aromatic compounds from industrial installations. Additionally, the Water Framework Directive identifies priority aromatic substances requiring enhanced monitoring and control measures.
Nitroaromatic compounds face particularly stringent regulatory oversight due to their enhanced toxicity profiles. The European Chemicals Agency has classified numerous nitroaromatic substances as substances of very high concern, triggering authorization requirements and substitution obligations. Similar classifications exist under various national chemical management systems worldwide.
Occupational exposure limits for aromatic compounds vary significantly across jurisdictions but generally follow similar risk assessment methodologies. The American Conference of Governmental Industrial Hygienists and equivalent international bodies regularly update threshold limit values based on emerging toxicological data.
Recent regulatory trends indicate increasing emphasis on mixture toxicity assessment, recognizing that aromatic compounds rarely exist in isolation within environmental matrices. This shift toward cumulative risk assessment approaches represents a fundamental evolution in regulatory thinking, potentially leading to more restrictive future standards for aromatic compound emissions and exposures.
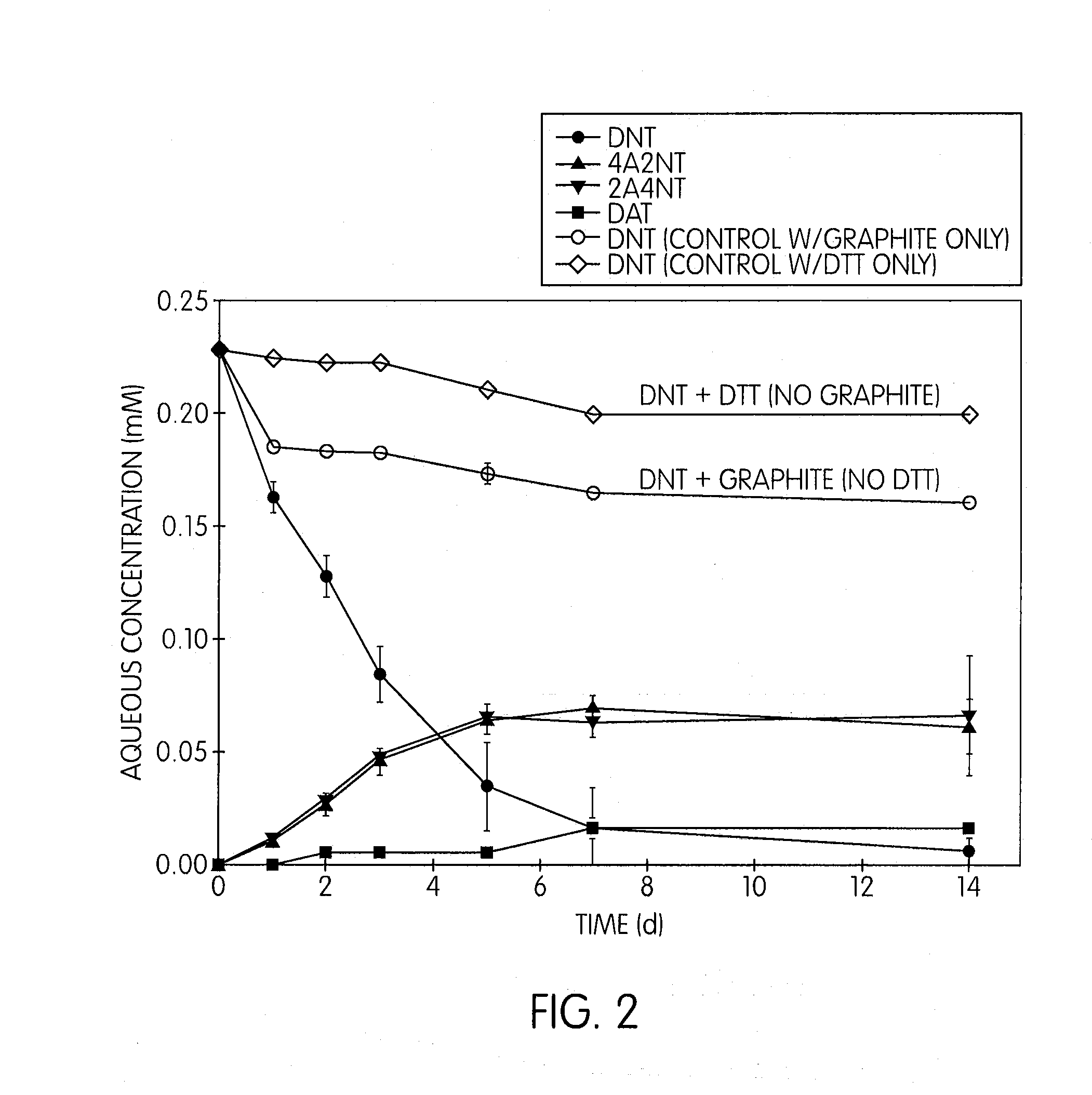
Biodegradation Pathways for Nitroaromatic Remediation
Biodegradation represents the most sustainable and environmentally friendly approach for nitroaromatic compound remediation, leveraging natural microbial processes to transform these persistent pollutants into less harmful substances. The biological degradation of nitroaromatic compounds occurs through several distinct metabolic pathways, each characterized by specific enzymatic mechanisms and environmental conditions.
The aerobic degradation pathway primarily involves the initial reduction of nitro groups to amino groups through nitroreductase enzymes. This process typically occurs in oxygen-rich environments where microorganisms such as Pseudomonas, Rhodococcus, and Bacillus species utilize nitroaromatic compounds as carbon or nitrogen sources. The pathway proceeds through sequential reduction steps, converting nitro groups to nitroso, hydroxylamine, and finally amino derivatives, which can then undergo ring cleavage through conventional aromatic degradation mechanisms.
Anaerobic biodegradation pathways offer alternative routes particularly effective for highly substituted nitroaromatic compounds that resist aerobic treatment. Under oxygen-limited conditions, specialized anaerobic bacteria employ reductive dehalogenation and nitro group reduction mechanisms. These pathways often involve complex electron transport chains and specialized enzymes capable of handling recalcitrant compounds that remain stable under aerobic conditions.
Co-metabolic degradation represents another significant pathway where nitroaromatic compounds are transformed by enzymes produced for the metabolism of other substrates. This process is particularly relevant for compounds that cannot serve as sole carbon sources but can be degraded when suitable co-substrates are present. Methanotrophic and ammonia-oxidizing bacteria frequently demonstrate co-metabolic capabilities for nitroaromatic transformation.
The effectiveness of biodegradation pathways depends critically on environmental factors including pH, temperature, nutrient availability, and the presence of inhibitory substances. Bioaugmentation strategies involving the introduction of specialized microbial consortia and biostimulation through nutrient supplementation have shown promising results in enhancing natural biodegradation rates. Recent advances in genetic engineering have also enabled the development of enhanced microbial strains with improved degradation capabilities for specific nitroaromatic compounds.
The aerobic degradation pathway primarily involves the initial reduction of nitro groups to amino groups through nitroreductase enzymes. This process typically occurs in oxygen-rich environments where microorganisms such as Pseudomonas, Rhodococcus, and Bacillus species utilize nitroaromatic compounds as carbon or nitrogen sources. The pathway proceeds through sequential reduction steps, converting nitro groups to nitroso, hydroxylamine, and finally amino derivatives, which can then undergo ring cleavage through conventional aromatic degradation mechanisms.
Anaerobic biodegradation pathways offer alternative routes particularly effective for highly substituted nitroaromatic compounds that resist aerobic treatment. Under oxygen-limited conditions, specialized anaerobic bacteria employ reductive dehalogenation and nitro group reduction mechanisms. These pathways often involve complex electron transport chains and specialized enzymes capable of handling recalcitrant compounds that remain stable under aerobic conditions.
Co-metabolic degradation represents another significant pathway where nitroaromatic compounds are transformed by enzymes produced for the metabolism of other substrates. This process is particularly relevant for compounds that cannot serve as sole carbon sources but can be degraded when suitable co-substrates are present. Methanotrophic and ammonia-oxidizing bacteria frequently demonstrate co-metabolic capabilities for nitroaromatic transformation.
The effectiveness of biodegradation pathways depends critically on environmental factors including pH, temperature, nutrient availability, and the presence of inhibitory substances. Bioaugmentation strategies involving the introduction of specialized microbial consortia and biostimulation through nutrient supplementation have shown promising results in enhancing natural biodegradation rates. Recent advances in genetic engineering have also enabled the development of enhanced microbial strains with improved degradation capabilities for specific nitroaromatic compounds.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!