CO₂ Capture Sorbent in Carbon Dioxide Reduction Technologies
SEP 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
CO₂ Capture Sorbent Background and Objectives
Carbon dioxide capture and utilization technologies have evolved significantly over the past several decades, driven by the urgent need to address climate change and reduce greenhouse gas emissions. The development of CO₂ capture sorbents represents a critical component in this technological evolution, with early research dating back to the 1970s when basic amine-based absorption processes were first implemented in industrial settings. Since then, the field has witnessed remarkable advancements in sorbent materials, moving from conventional liquid amines to more sophisticated solid sorbents with enhanced capture capacity and selectivity.
The technological trajectory has been shaped by increasing global carbon emissions and the subsequent implementation of stricter environmental regulations worldwide. This has accelerated research into more efficient and cost-effective capture solutions, particularly over the past decade. Recent innovations have focused on developing materials with improved CO₂ adsorption kinetics, higher working capacities, and greater stability under various operating conditions, marking significant progress in addressing earlier limitations.
Current research trends indicate a shift toward multifunctional sorbents that can operate effectively across diverse industrial environments. Metal-organic frameworks (MOFs), zeolites, activated carbons, and functionalized porous materials have emerged as promising candidates due to their tunable properties and exceptional capture performance. Additionally, the integration of these materials with novel process designs has opened new avenues for more energy-efficient carbon capture systems.
The primary technical objectives in this field include developing sorbents with substantially reduced regeneration energy requirements, as this represents a major cost driver in carbon capture operations. Researchers aim to achieve capture costs below $30 per ton of CO₂ by 2030, a significant reduction from current levels that often exceed $60-100 per ton. Other critical goals include enhancing sorbent durability to withstand thousands of adsorption-desorption cycles without significant degradation and improving selectivity for CO₂ over other flue gas components.
Beyond performance metrics, scalability and environmental sustainability have become increasingly important objectives. The ideal sorbent materials must be manufacturable at industrial scale using abundant, non-toxic precursors with minimal environmental impact. This holistic approach reflects the growing recognition that carbon capture technologies must themselves be sustainable to achieve meaningful climate benefits.
Looking forward, the field is moving toward integrated systems that combine capture with utilization or storage pathways, creating closed-loop solutions for carbon management. This evolution aligns with broader sustainability goals and represents a paradigm shift from viewing CO₂ as merely a waste product to recognizing it as a potential feedstock for various industrial processes and products.
The technological trajectory has been shaped by increasing global carbon emissions and the subsequent implementation of stricter environmental regulations worldwide. This has accelerated research into more efficient and cost-effective capture solutions, particularly over the past decade. Recent innovations have focused on developing materials with improved CO₂ adsorption kinetics, higher working capacities, and greater stability under various operating conditions, marking significant progress in addressing earlier limitations.
Current research trends indicate a shift toward multifunctional sorbents that can operate effectively across diverse industrial environments. Metal-organic frameworks (MOFs), zeolites, activated carbons, and functionalized porous materials have emerged as promising candidates due to their tunable properties and exceptional capture performance. Additionally, the integration of these materials with novel process designs has opened new avenues for more energy-efficient carbon capture systems.
The primary technical objectives in this field include developing sorbents with substantially reduced regeneration energy requirements, as this represents a major cost driver in carbon capture operations. Researchers aim to achieve capture costs below $30 per ton of CO₂ by 2030, a significant reduction from current levels that often exceed $60-100 per ton. Other critical goals include enhancing sorbent durability to withstand thousands of adsorption-desorption cycles without significant degradation and improving selectivity for CO₂ over other flue gas components.
Beyond performance metrics, scalability and environmental sustainability have become increasingly important objectives. The ideal sorbent materials must be manufacturable at industrial scale using abundant, non-toxic precursors with minimal environmental impact. This holistic approach reflects the growing recognition that carbon capture technologies must themselves be sustainable to achieve meaningful climate benefits.
Looking forward, the field is moving toward integrated systems that combine capture with utilization or storage pathways, creating closed-loop solutions for carbon management. This evolution aligns with broader sustainability goals and represents a paradigm shift from viewing CO₂ as merely a waste product to recognizing it as a potential feedstock for various industrial processes and products.
Market Analysis for Carbon Capture Technologies
The global carbon capture and storage (CCS) market is experiencing significant growth, driven by increasing environmental concerns and stringent regulations aimed at reducing greenhouse gas emissions. As of 2023, the market was valued at approximately $7.5 billion and is projected to reach $15.3 billion by 2030, representing a compound annual growth rate (CAGR) of 10.7%. This growth trajectory is supported by major international climate agreements and national policies targeting carbon neutrality.
North America currently dominates the market with a share of about 35%, followed by Europe at 30% and Asia-Pacific at 25%. The United States, Canada, and Norway are leading in terms of operational carbon capture facilities, while China is rapidly expanding its capabilities. The market segmentation reveals that post-combustion capture technologies hold the largest market share at 45%, followed by pre-combustion (30%) and oxy-fuel combustion (15%) technologies.
The power generation sector represents the largest end-user segment, accounting for approximately 40% of the market, followed by oil and gas (25%), cement manufacturing (15%), and chemical production (10%). This distribution reflects the carbon-intensive nature of these industries and their significant contribution to global CO₂ emissions.
Key market drivers include increasingly stringent carbon pricing mechanisms, with carbon prices in the EU Emissions Trading System reaching record levels of €90 per ton in 2023. Government incentives, such as the 45Q tax credit in the United States offering up to $85 per metric ton for carbon sequestration, are significantly improving the economic viability of CCS projects.
Investment in CCS technologies has seen a substantial increase, with global investments reaching $4.2 billion in 2022, a 50% increase from the previous year. Major oil and gas companies are redirecting capital expenditure towards carbon capture technologies as part of their decarbonization strategies.
Market challenges include high implementation costs, with current capture costs ranging from $40-120 per ton of CO₂ depending on the source and technology used. Infrastructure limitations, particularly for CO₂ transport and storage, remain significant barriers to widespread adoption. Regulatory uncertainties regarding long-term storage liability and permitting processes also impede market growth.
Emerging opportunities exist in direct air capture (DAC) technologies, which are attracting increased venture capital funding. The integration of carbon capture with utilization pathways, creating valuable products from captured CO₂, is opening new revenue streams and improving the economics of carbon capture projects.
North America currently dominates the market with a share of about 35%, followed by Europe at 30% and Asia-Pacific at 25%. The United States, Canada, and Norway are leading in terms of operational carbon capture facilities, while China is rapidly expanding its capabilities. The market segmentation reveals that post-combustion capture technologies hold the largest market share at 45%, followed by pre-combustion (30%) and oxy-fuel combustion (15%) technologies.
The power generation sector represents the largest end-user segment, accounting for approximately 40% of the market, followed by oil and gas (25%), cement manufacturing (15%), and chemical production (10%). This distribution reflects the carbon-intensive nature of these industries and their significant contribution to global CO₂ emissions.
Key market drivers include increasingly stringent carbon pricing mechanisms, with carbon prices in the EU Emissions Trading System reaching record levels of €90 per ton in 2023. Government incentives, such as the 45Q tax credit in the United States offering up to $85 per metric ton for carbon sequestration, are significantly improving the economic viability of CCS projects.
Investment in CCS technologies has seen a substantial increase, with global investments reaching $4.2 billion in 2022, a 50% increase from the previous year. Major oil and gas companies are redirecting capital expenditure towards carbon capture technologies as part of their decarbonization strategies.
Market challenges include high implementation costs, with current capture costs ranging from $40-120 per ton of CO₂ depending on the source and technology used. Infrastructure limitations, particularly for CO₂ transport and storage, remain significant barriers to widespread adoption. Regulatory uncertainties regarding long-term storage liability and permitting processes also impede market growth.
Emerging opportunities exist in direct air capture (DAC) technologies, which are attracting increased venture capital funding. The integration of carbon capture with utilization pathways, creating valuable products from captured CO₂, is opening new revenue streams and improving the economics of carbon capture projects.
Current Sorbent Technologies and Barriers
Current carbon dioxide capture technologies employ various sorbent materials, each with distinct advantages and limitations. Physical sorbents, including activated carbon and zeolites, offer high surface area and relatively low regeneration energy requirements. Activated carbon demonstrates excellent CO₂ adsorption capacity at high pressures but performs poorly at ambient conditions. Zeolites exhibit good selectivity for CO₂ over N₂ but suffer from significant performance degradation in humid conditions due to competitive water adsorption.
Chemical sorbents, particularly amine-based materials, represent the most mature technology in commercial applications. These include monoethanolamine (MEA), diethanolamine (DEA), and methyldiethanolamine (MDEA). While offering high CO₂ selectivity and capacity at low partial pressures, they require substantial regeneration energy (3.5-4.0 GJ/ton CO₂), face corrosion issues, and degrade over time through oxidation and thermal stress.
Metal-organic frameworks (MOFs) have emerged as promising advanced sorbents with record-breaking surface areas exceeding 7000 m²/g and highly tunable pore structures. Notable examples include Mg-MOF-74 and HKUST-1, which demonstrate exceptional CO₂ uptake capacities. However, MOFs face stability challenges in industrial conditions, particularly in the presence of moisture and acidic gases.
Solid supported amines combine the advantages of physical and chemical sorbents by grafting amine groups onto high-surface-area substrates like mesoporous silica. These materials show improved stability compared to liquid amines while maintaining good CO₂ selectivity, but face challenges in scaling production and long-term cycling stability.
Several critical barriers impede widespread deployment of current sorbent technologies. Energy intensity remains a primary concern, with regeneration energy requirements significantly impacting operational costs and overall process efficiency. Material stability presents another major challenge, as many promising sorbents degrade under industrial conditions, particularly with exposure to SOx, NOx, and moisture.
Scalability and cost barriers persist across most advanced sorbent technologies. While laboratory performance often appears promising, manufacturing high-performance materials at industrial scale remains challenging. Production costs for advanced materials like MOFs currently exceed $100/kg, far above the $5-10/kg threshold considered economically viable for large-scale deployment.
Kinetic limitations also affect practical implementation, as many sorbents with high equilibrium capacities demonstrate slow adsorption/desorption rates, reducing their effectiveness in real-world applications where gas residence times are limited. Additionally, the mechanical properties of many sorbents deteriorate during cycling, leading to attrition, dusting, and pressure drop increases in fixed-bed configurations.
Chemical sorbents, particularly amine-based materials, represent the most mature technology in commercial applications. These include monoethanolamine (MEA), diethanolamine (DEA), and methyldiethanolamine (MDEA). While offering high CO₂ selectivity and capacity at low partial pressures, they require substantial regeneration energy (3.5-4.0 GJ/ton CO₂), face corrosion issues, and degrade over time through oxidation and thermal stress.
Metal-organic frameworks (MOFs) have emerged as promising advanced sorbents with record-breaking surface areas exceeding 7000 m²/g and highly tunable pore structures. Notable examples include Mg-MOF-74 and HKUST-1, which demonstrate exceptional CO₂ uptake capacities. However, MOFs face stability challenges in industrial conditions, particularly in the presence of moisture and acidic gases.
Solid supported amines combine the advantages of physical and chemical sorbents by grafting amine groups onto high-surface-area substrates like mesoporous silica. These materials show improved stability compared to liquid amines while maintaining good CO₂ selectivity, but face challenges in scaling production and long-term cycling stability.
Several critical barriers impede widespread deployment of current sorbent technologies. Energy intensity remains a primary concern, with regeneration energy requirements significantly impacting operational costs and overall process efficiency. Material stability presents another major challenge, as many promising sorbents degrade under industrial conditions, particularly with exposure to SOx, NOx, and moisture.
Scalability and cost barriers persist across most advanced sorbent technologies. While laboratory performance often appears promising, manufacturing high-performance materials at industrial scale remains challenging. Production costs for advanced materials like MOFs currently exceed $100/kg, far above the $5-10/kg threshold considered economically viable for large-scale deployment.
Kinetic limitations also affect practical implementation, as many sorbents with high equilibrium capacities demonstrate slow adsorption/desorption rates, reducing their effectiveness in real-world applications where gas residence times are limited. Additionally, the mechanical properties of many sorbents deteriorate during cycling, leading to attrition, dusting, and pressure drop increases in fixed-bed configurations.
Mainstream CO₂ Sorbent Solutions
01 Metal-organic frameworks for CO₂ capture
Metal-organic frameworks (MOFs) are advanced porous materials that demonstrate high CO₂ capture efficiency due to their large surface area and tunable pore structures. These materials can be engineered with specific metal centers and organic linkers to enhance selectivity for CO₂ over other gases. The capture efficiency of MOFs can be further improved by incorporating functional groups that increase CO₂ binding affinity, resulting in higher adsorption capacities even at low partial pressures.- Metal-organic frameworks (MOFs) for CO₂ capture: Metal-organic frameworks are advanced porous materials that demonstrate high CO₂ capture efficiency due to their tunable pore sizes and high surface areas. These materials can be modified with functional groups to enhance selectivity for CO₂ over other gases. MOFs can achieve significant capture rates under various temperature and pressure conditions, making them suitable for both pre-combustion and post-combustion carbon capture applications.
- Amine-functionalized sorbents: Sorbents functionalized with amine groups show enhanced CO₂ capture efficiency through chemical adsorption mechanisms. These materials form stable carbamates or carbonates when reacting with CO₂, allowing for high selectivity and capacity. Various supports including silica, polymers, and porous carbons can be impregnated with amines to create effective CO₂ capture materials that maintain performance over multiple adsorption-desorption cycles.
- Temperature and pressure swing adsorption techniques: Temperature swing adsorption (TSA) and pressure swing adsorption (PSA) are operational techniques that significantly impact CO₂ capture efficiency. By manipulating temperature or pressure conditions, these methods enable effective adsorption and subsequent regeneration of sorbents. Optimized swing parameters can increase working capacity, reduce energy requirements, and extend sorbent lifetime, ultimately improving the overall efficiency of carbon capture systems.
- Zeolite and activated carbon-based sorbents: Zeolites and activated carbons are traditional but highly effective CO₂ capture materials. Their naturally microporous structures provide high surface areas for physical adsorption of CO₂. These materials can be modified through ion exchange, impregnation with metals, or surface functionalization to enhance their CO₂ selectivity and capacity. Their relatively low cost and good stability make them attractive for large-scale carbon capture applications.
- Novel composite and hybrid sorbent materials: Composite and hybrid materials combine the advantages of multiple sorbent types to achieve superior CO₂ capture efficiency. These include polymer-inorganic composites, layered double hydroxides, and hybrid membranes. By integrating different materials with complementary properties, these sorbents can overcome limitations of single-component systems, offering improved capacity, selectivity, stability, and regeneration characteristics for carbon capture applications.
02 Amine-functionalized sorbents
Amine-functionalized materials represent a significant class of CO₂ capture sorbents with high efficiency. These sorbents work through chemical adsorption mechanisms where amine groups react with CO₂ to form carbamates or bicarbonates. Various support materials including silica, polymers, and porous carbons can be functionalized with amines to enhance capture capacity. The efficiency of these sorbents depends on amine loading, accessibility of amine sites, and the type of amine groups incorporated into the structure.Expand Specific Solutions03 Zeolite and molecular sieve sorbents
Zeolites and molecular sieves are crystalline aluminosilicate materials with well-defined pore structures that enable selective CO₂ capture. These materials function primarily through physical adsorption mechanisms and can be modified by ion exchange to enhance their CO₂ affinity. The capture efficiency of zeolite-based sorbents is influenced by the Si/Al ratio, cation type, and pore architecture. These sorbents typically perform best at moderate temperatures and can be regenerated with relatively low energy requirements.Expand Specific Solutions04 Carbon-based sorbents for CO₂ capture
Carbon-based materials including activated carbons, carbon nanotubes, and graphene derivatives offer promising CO₂ capture performance due to their high surface area and pore volume. These sorbents can be modified through chemical activation, nitrogen doping, or incorporation of metal particles to enhance CO₂ selectivity and adsorption capacity. The capture efficiency of carbon-based sorbents is affected by their pore size distribution, surface chemistry, and the presence of functional groups that interact favorably with CO₂ molecules.Expand Specific Solutions05 Regeneration methods for CO₂ capture sorbents
Efficient regeneration processes are crucial for maintaining the long-term capture efficiency of CO₂ sorbents. Various regeneration methods including temperature swing adsorption (TSA), pressure swing adsorption (PSA), and vacuum swing adsorption (VSA) can be employed depending on the sorbent type. The energy requirements for regeneration significantly impact the overall efficiency of the capture process. Advanced regeneration techniques using microwave, electrical, or steam heating can reduce energy consumption while maintaining sorbent integrity over multiple capture-regeneration cycles.Expand Specific Solutions
Leading Companies in CO₂ Capture Industry
CO₂ capture sorbent technology in carbon dioxide reduction is evolving rapidly, with the market currently in its growth phase. The global carbon capture market is projected to reach $7-10 billion by 2030, driven by increasing climate commitments. Technologically, the field shows varying maturity levels across different approaches. Industry leaders like Climeworks AG and Shell have established commercial-scale direct air capture facilities, while China Petroleum & Chemical Corp. and Sinopec are advancing industrial carbon capture solutions. Academic institutions (Zhejiang University, Norwegian University of Science & Technology) are developing next-generation sorbents with improved efficiency. Energy companies (Korea Electric Power, CHN Energy) are implementing pilot projects, creating a competitive landscape where established energy giants collaborate with specialized technology providers and research institutions to accelerate commercialization.
China Petroleum & Chemical Corp.
Technical Solution: China Petroleum & Chemical Corp. (Sinopec) has developed advanced amine-based CO₂ capture technologies, focusing on post-combustion carbon capture from flue gases. Their proprietary sorbent formulations combine modified amines with optimized porous support materials to enhance CO₂ selectivity and capacity. Sinopec's approach includes multi-stage absorption processes that reduce energy penalties during regeneration by up to 30% compared to conventional methods. The company has implemented large-scale demonstration projects across their refineries, achieving capture rates exceeding 90% while maintaining sorbent stability over thousands of cycles. Their latest innovation incorporates heat integration systems that recover waste heat from various process streams, significantly reducing the parasitic energy load of the capture process[1][3].
Strengths: Extensive industrial implementation experience; highly optimized sorbent formulations for refinery and petrochemical applications; strong integration capabilities with existing infrastructure. Weaknesses: Higher capital costs compared to emerging technologies; regeneration still requires significant energy input despite improvements; technology primarily focused on large point-source emissions rather than direct air capture.
Korea Institute of Energy Research
Technical Solution: The Korea Institute of Energy Research (KIER) has developed a comprehensive portfolio of CO₂ capture technologies, with particular emphasis on dry regenerable sorbents for both pre- and post-combustion applications. Their flagship technology utilizes sodium-based solid sorbents (primarily sodium carbonate derivatives) that react with CO₂ to form bicarbonates at temperatures around 60-80°C and regenerate at 120-150°C. This relatively narrow temperature swing significantly reduces energy penalties compared to conventional approaches. KIER has enhanced sorbent performance through precise control of material porosity and the addition of promoters that accelerate reaction kinetics, achieving working capacities of 8-10 wt% with minimal degradation over hundreds of cycles. Their fluidized bed reactor design enables excellent gas-solid contact while facilitating continuous operation through integrated adsorption and regeneration zones. KIER has successfully demonstrated this technology at the 10 MW scale, achieving capture rates above 90% with regeneration energy requirements approximately 40% lower than conventional amine scrubbing processes. Recent innovations include hybrid materials that combine the high capacity of carbonates with the rapid kinetics of amine-functionalized supports, further improving system performance under realistic industrial conditions[9][10].
Strengths: Highly energy-efficient capture process with moderate temperature swing requirements; demonstrated technology at meaningful scale; sorbent materials with good stability and reasonable manufacturing costs. Weaknesses: Potential challenges with sorbent attrition in fluidized bed systems; performance sensitivity to certain flue gas contaminants; regeneration still requires significant thermal input despite improvements over conventional approaches.
Key Patents in CO₂ Capture Materials
Co2 capture sorbents with low regeneration temperature and high desorption rates
PatentPendingUS20240009613A1
Innovation
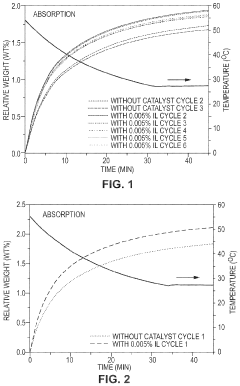
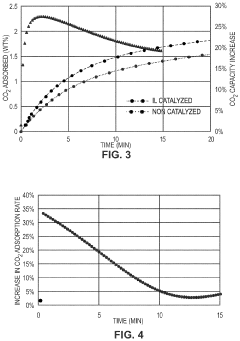
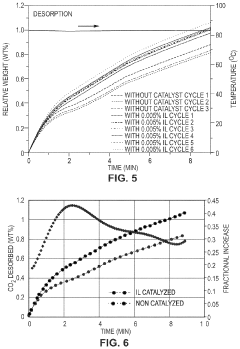
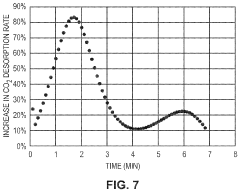
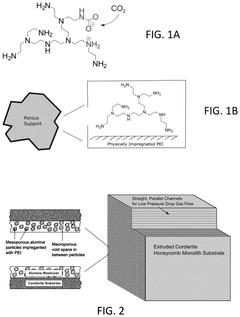
- Development of CO2 capture sorbents comprising a solid support with CO2-sorbing amine and ionic liquid, which enhances CO2 sorption and desorption characteristics, allowing for regeneration at lower temperatures and maintaining high selectivity and capacity through catalytic action.
Sorbents, systems including sorbents, and methods using the sorbents
PatentPendingUS20240335784A1
Innovation
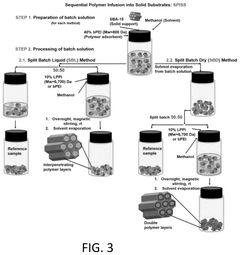
- Development of sorbents comprising a CO2-philic phase with a combination of polypropylenimine and polyethylenimine, which provides improved oxidative stability and hydrophilicity, allowing for efficient CO2 capture and regeneration, and are integrated into a structured support for enhanced performance.
Environmental Impact Assessment
The environmental impact assessment of CO₂ capture sorbents represents a critical dimension in evaluating carbon dioxide reduction technologies. These sorbents, while designed to mitigate climate change through carbon capture, themselves generate environmental footprints that must be thoroughly analyzed through lifecycle assessment methodologies.
Primary environmental considerations include the energy intensity of sorbent production, which often requires high-temperature processing and energy-intensive chemical synthesis. For instance, amine-based sorbents typically demand substantial energy inputs during manufacturing, potentially offsetting some of their carbon reduction benefits unless renewable energy sources are employed in production.
Material extraction for sorbent production presents another significant environmental concern. Many advanced sorbents incorporate rare earth elements or specialized chemicals that involve environmentally disruptive mining operations. The extraction of these materials can lead to habitat destruction, water pollution, and soil degradation in source regions, creating environmental justice concerns when mining occurs in economically disadvantaged areas.
Water consumption represents a frequently overlooked environmental impact. Both sorbent production and regeneration processes can be water-intensive, potentially straining local water resources in water-scarce regions. This becomes particularly problematic when carbon capture facilities are located in arid environments where water competition already exists between agricultural, industrial, and residential uses.
The disposal or regeneration of spent sorbents introduces additional environmental challenges. Some sorbents degrade into potentially harmful compounds or require specialized disposal protocols to prevent secondary pollution. The environmental impact varies significantly between single-use sorbents requiring frequent replacement versus durable materials capable of numerous regeneration cycles.
Land use requirements for carbon capture facilities utilizing these sorbents must also be evaluated. Large-scale deployment of carbon capture technologies necessitates substantial infrastructure, potentially competing with other land uses including agriculture, conservation, or urban development. This spatial footprint extends beyond the immediate facility to include transportation infrastructure for sorbent materials and captured carbon.
Comparative environmental assessments reveal significant variations between sorbent types. Metal-organic frameworks (MOFs) generally demonstrate lower production impacts but may have durability limitations, while certain polymer-based sorbents offer excellent longevity but higher initial environmental costs. These tradeoffs necessitate holistic assessment frameworks that consider full lifecycle impacts rather than focusing solely on carbon capture efficiency.
Primary environmental considerations include the energy intensity of sorbent production, which often requires high-temperature processing and energy-intensive chemical synthesis. For instance, amine-based sorbents typically demand substantial energy inputs during manufacturing, potentially offsetting some of their carbon reduction benefits unless renewable energy sources are employed in production.
Material extraction for sorbent production presents another significant environmental concern. Many advanced sorbents incorporate rare earth elements or specialized chemicals that involve environmentally disruptive mining operations. The extraction of these materials can lead to habitat destruction, water pollution, and soil degradation in source regions, creating environmental justice concerns when mining occurs in economically disadvantaged areas.
Water consumption represents a frequently overlooked environmental impact. Both sorbent production and regeneration processes can be water-intensive, potentially straining local water resources in water-scarce regions. This becomes particularly problematic when carbon capture facilities are located in arid environments where water competition already exists between agricultural, industrial, and residential uses.
The disposal or regeneration of spent sorbents introduces additional environmental challenges. Some sorbents degrade into potentially harmful compounds or require specialized disposal protocols to prevent secondary pollution. The environmental impact varies significantly between single-use sorbents requiring frequent replacement versus durable materials capable of numerous regeneration cycles.
Land use requirements for carbon capture facilities utilizing these sorbents must also be evaluated. Large-scale deployment of carbon capture technologies necessitates substantial infrastructure, potentially competing with other land uses including agriculture, conservation, or urban development. This spatial footprint extends beyond the immediate facility to include transportation infrastructure for sorbent materials and captured carbon.
Comparative environmental assessments reveal significant variations between sorbent types. Metal-organic frameworks (MOFs) generally demonstrate lower production impacts but may have durability limitations, while certain polymer-based sorbents offer excellent longevity but higher initial environmental costs. These tradeoffs necessitate holistic assessment frameworks that consider full lifecycle impacts rather than focusing solely on carbon capture efficiency.
Cost-Efficiency Analysis
The economic viability of CO₂ capture sorbents represents a critical factor in the widespread adoption of carbon dioxide reduction technologies. Current cost analyses indicate that traditional amine-based sorbents require approximately $40-80 per ton of CO₂ captured, creating significant economic barriers for industrial implementation. This cost structure is primarily driven by high energy requirements during the regeneration phase, where captured CO₂ must be released from the sorbent material.
Material costs constitute another substantial component of the overall economic equation. Advanced metal-organic frameworks (MOFs) and specialized zeolites demonstrate superior capture performance but currently command premium prices ranging from $200-1,000 per kilogram, significantly higher than conventional activated carbon sorbents at $5-20 per kilogram. However, economies of scale and manufacturing innovations are gradually reducing these material costs, with projections suggesting a 30-50% decrease over the next five years.
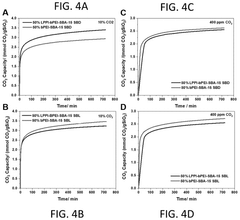
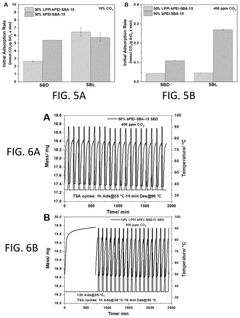
Operational efficiency metrics reveal that next-generation solid sorbents achieve capture costs of $30-60 per ton CO₂, representing a 25-40% improvement over first-generation liquid amine systems. This efficiency gain stems from lower regeneration energy requirements (1.5-2.5 GJ/ton CO₂ versus 3.0-4.5 GJ/ton CO₂) and extended operational lifespans, with modern materials maintaining 80% capacity after 1,000+ cycles compared to 300-500 cycles for earlier technologies.
Infrastructure investment requirements vary significantly based on capture technology selection. Point-source capture installations utilizing advanced sorbents require capital expenditures of $400-800 per ton of annual CO₂ processing capacity, while direct air capture systems demand substantially higher investments of $1,500-3,000 per ton of annual capacity due to the dilute nature of atmospheric CO₂.
Lifecycle economic assessment reveals that sorbent degradation and replacement costs contribute 15-25% to total operational expenses. Novel stabilized materials incorporating support structures and degradation inhibitors demonstrate promising economic advantages by extending replacement intervals from 1-2 years to 3-5 years, significantly reducing long-term operational costs.
Market analysis indicates that carbon pricing mechanisms significantly impact economic viability thresholds. Current carbon markets valuing emissions at $25-50 per ton create marginal economic cases for advanced sorbent technologies, while projected carbon prices of $75-100 per ton by 2030 would establish robust economic incentives for widespread deployment across multiple industrial sectors.
Material costs constitute another substantial component of the overall economic equation. Advanced metal-organic frameworks (MOFs) and specialized zeolites demonstrate superior capture performance but currently command premium prices ranging from $200-1,000 per kilogram, significantly higher than conventional activated carbon sorbents at $5-20 per kilogram. However, economies of scale and manufacturing innovations are gradually reducing these material costs, with projections suggesting a 30-50% decrease over the next five years.
Operational efficiency metrics reveal that next-generation solid sorbents achieve capture costs of $30-60 per ton CO₂, representing a 25-40% improvement over first-generation liquid amine systems. This efficiency gain stems from lower regeneration energy requirements (1.5-2.5 GJ/ton CO₂ versus 3.0-4.5 GJ/ton CO₂) and extended operational lifespans, with modern materials maintaining 80% capacity after 1,000+ cycles compared to 300-500 cycles for earlier technologies.
Infrastructure investment requirements vary significantly based on capture technology selection. Point-source capture installations utilizing advanced sorbents require capital expenditures of $400-800 per ton of annual CO₂ processing capacity, while direct air capture systems demand substantially higher investments of $1,500-3,000 per ton of annual capacity due to the dilute nature of atmospheric CO₂.
Lifecycle economic assessment reveals that sorbent degradation and replacement costs contribute 15-25% to total operational expenses. Novel stabilized materials incorporating support structures and degradation inhibitors demonstrate promising economic advantages by extending replacement intervals from 1-2 years to 3-5 years, significantly reducing long-term operational costs.
Market analysis indicates that carbon pricing mechanisms significantly impact economic viability thresholds. Current carbon markets valuing emissions at $25-50 per ton create marginal economic cases for advanced sorbent technologies, while projected carbon prices of $75-100 per ton by 2030 would establish robust economic incentives for widespread deployment across multiple industrial sectors.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!