CO₂ Capture Sorbent Material Development for Recycling Processes
SEP 28, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
CO₂ Capture Materials Background and Objectives
Carbon dioxide (CO₂) capture technology has evolved significantly over the past several decades, driven by the urgent need to mitigate climate change and reduce greenhouse gas emissions. Initially focused on large-scale industrial applications like power plants, CO₂ capture technologies have gradually expanded to address emissions from various sectors including transportation, manufacturing, and waste management. The development trajectory has moved from first-generation absorption-based systems using liquid amines to more advanced sorbent materials with enhanced capture efficiency and reduced energy penalties.
The evolution of CO₂ capture sorbent materials has progressed through several distinct phases. Early research concentrated on liquid solvents such as monoethanolamine (MEA), which despite their effectiveness, suffered from high regeneration energy requirements and degradation issues. This led to the exploration of solid sorbents including activated carbons, zeolites, and metal-organic frameworks (MOFs), which offered improved stability and lower energy demands for regeneration.
Recent technological advancements have focused on developing specialized materials specifically designed for recycling processes, where CO₂ capture conditions differ significantly from traditional industrial settings. These materials must function effectively under variable temperature and pressure conditions, demonstrate resilience to contaminants common in recycling streams, and maintain performance over numerous capture-release cycles.
The primary objective of current CO₂ capture sorbent material development for recycling processes is to create cost-effective, highly selective, and durable materials that can efficiently capture CO₂ from diverse waste streams. These materials must operate with minimal energy input while maintaining high capture capacity and selectivity even in the presence of moisture and other contaminants typical in recycling environments.
Another critical goal is to develop sorbents that integrate seamlessly with existing recycling infrastructure, minimizing the need for extensive system redesigns or capital investments. This includes materials that can function effectively at near-ambient conditions, reducing the energy penalty associated with traditional capture technologies.
Looking forward, the field is trending toward multifunctional sorbent materials that not only capture CO₂ but also facilitate its conversion into valuable products, creating closed-loop systems within recycling operations. This approach aligns with circular economy principles and enhances the economic viability of carbon capture implementation in recycling facilities.
The development of these advanced materials represents a convergence of materials science, chemical engineering, and environmental technology, with potential applications extending beyond traditional recycling to emerging fields such as direct air capture and distributed carbon management systems.
The evolution of CO₂ capture sorbent materials has progressed through several distinct phases. Early research concentrated on liquid solvents such as monoethanolamine (MEA), which despite their effectiveness, suffered from high regeneration energy requirements and degradation issues. This led to the exploration of solid sorbents including activated carbons, zeolites, and metal-organic frameworks (MOFs), which offered improved stability and lower energy demands for regeneration.
Recent technological advancements have focused on developing specialized materials specifically designed for recycling processes, where CO₂ capture conditions differ significantly from traditional industrial settings. These materials must function effectively under variable temperature and pressure conditions, demonstrate resilience to contaminants common in recycling streams, and maintain performance over numerous capture-release cycles.
The primary objective of current CO₂ capture sorbent material development for recycling processes is to create cost-effective, highly selective, and durable materials that can efficiently capture CO₂ from diverse waste streams. These materials must operate with minimal energy input while maintaining high capture capacity and selectivity even in the presence of moisture and other contaminants typical in recycling environments.
Another critical goal is to develop sorbents that integrate seamlessly with existing recycling infrastructure, minimizing the need for extensive system redesigns or capital investments. This includes materials that can function effectively at near-ambient conditions, reducing the energy penalty associated with traditional capture technologies.
Looking forward, the field is trending toward multifunctional sorbent materials that not only capture CO₂ but also facilitate its conversion into valuable products, creating closed-loop systems within recycling operations. This approach aligns with circular economy principles and enhances the economic viability of carbon capture implementation in recycling facilities.
The development of these advanced materials represents a convergence of materials science, chemical engineering, and environmental technology, with potential applications extending beyond traditional recycling to emerging fields such as direct air capture and distributed carbon management systems.
Market Analysis for CO₂ Capture Technologies
The global CO₂ capture market is experiencing significant growth, driven by increasing environmental regulations and corporate sustainability commitments. Current market valuations place the carbon capture, utilization, and storage (CCUS) sector at approximately $2.5 billion in 2023, with projections indicating expansion to reach $7-9 billion by 2030, representing a compound annual growth rate (CAGR) of 15-20%.
Industrial sectors contribute nearly 25% of global CO₂ emissions, with cement, steel, and chemical manufacturing being the largest emitters. These hard-to-abate sectors present substantial market opportunities for specialized CO₂ capture solutions, particularly those involving advanced sorbent materials designed for recycling processes.
Regional market analysis reveals varying adoption rates and regulatory frameworks. Europe leads in market maturity due to stringent carbon pricing mechanisms and the EU Emissions Trading System (ETS), which has established carbon prices exceeding €80 per ton. North America follows with growing momentum, particularly after the introduction of the Inflation Reduction Act in the US, which increased the 45Q tax credit for carbon sequestration to $85 per ton for industrial applications.
Asia-Pacific represents the fastest-growing market segment, with China announcing ambitious carbon neutrality targets by 2060 and implementing regional carbon trading systems. Japan and South Korea have similarly established concrete decarbonization roadmaps that specifically include carbon capture technologies.
Market segmentation by technology type shows post-combustion capture dominating with approximately 60% market share, followed by pre-combustion (25%) and oxy-fuel combustion (15%). Within the sorbent material segment, amine-based solutions currently hold the largest market share at 45%, though novel materials including metal-organic frameworks (MOFs), zeolites, and carbon-based sorbents are gaining traction due to their enhanced recyclability and lower regeneration energy requirements.
Customer demand analysis indicates shifting priorities, with end-users increasingly valuing total cost of ownership over initial capital expenditure. Energy efficiency during regeneration cycles and sorbent longevity have become critical purchasing factors, with industries willing to pay premiums of 15-20% for solutions that demonstrate superior recyclability and reduced operational costs.
Competitive pricing analysis shows current carbon capture costs ranging from $40-100 per ton of CO₂, with next-generation sorbent materials targeting cost reductions to $30-50 per ton by 2030. This price point is considered the threshold for widespread commercial adoption beyond regulatory compliance.
Market barriers include high initial capital requirements, uncertain regulatory landscapes in developing economies, and competition from alternative decarbonization strategies. However, the increasing implementation of carbon pricing mechanisms globally and growing corporate net-zero commitments are expected to accelerate market penetration of advanced CO₂ capture technologies.
Industrial sectors contribute nearly 25% of global CO₂ emissions, with cement, steel, and chemical manufacturing being the largest emitters. These hard-to-abate sectors present substantial market opportunities for specialized CO₂ capture solutions, particularly those involving advanced sorbent materials designed for recycling processes.
Regional market analysis reveals varying adoption rates and regulatory frameworks. Europe leads in market maturity due to stringent carbon pricing mechanisms and the EU Emissions Trading System (ETS), which has established carbon prices exceeding €80 per ton. North America follows with growing momentum, particularly after the introduction of the Inflation Reduction Act in the US, which increased the 45Q tax credit for carbon sequestration to $85 per ton for industrial applications.
Asia-Pacific represents the fastest-growing market segment, with China announcing ambitious carbon neutrality targets by 2060 and implementing regional carbon trading systems. Japan and South Korea have similarly established concrete decarbonization roadmaps that specifically include carbon capture technologies.
Market segmentation by technology type shows post-combustion capture dominating with approximately 60% market share, followed by pre-combustion (25%) and oxy-fuel combustion (15%). Within the sorbent material segment, amine-based solutions currently hold the largest market share at 45%, though novel materials including metal-organic frameworks (MOFs), zeolites, and carbon-based sorbents are gaining traction due to their enhanced recyclability and lower regeneration energy requirements.
Customer demand analysis indicates shifting priorities, with end-users increasingly valuing total cost of ownership over initial capital expenditure. Energy efficiency during regeneration cycles and sorbent longevity have become critical purchasing factors, with industries willing to pay premiums of 15-20% for solutions that demonstrate superior recyclability and reduced operational costs.
Competitive pricing analysis shows current carbon capture costs ranging from $40-100 per ton of CO₂, with next-generation sorbent materials targeting cost reductions to $30-50 per ton by 2030. This price point is considered the threshold for widespread commercial adoption beyond regulatory compliance.
Market barriers include high initial capital requirements, uncertain regulatory landscapes in developing economies, and competition from alternative decarbonization strategies. However, the increasing implementation of carbon pricing mechanisms globally and growing corporate net-zero commitments are expected to accelerate market penetration of advanced CO₂ capture technologies.
Current Sorbent Technologies and Challenges
Carbon dioxide capture technologies have evolved significantly over the past decades, with various sorbent materials being developed to address the growing need for efficient CO₂ removal. Currently, the most widely deployed sorbent technologies include amine-based solvents, solid adsorbents, membrane systems, and cryogenic separation methods. Among these, amine-based technologies dominate the commercial landscape, particularly monoethanolamine (MEA) solutions, which can achieve capture efficiencies of 85-95% in optimal conditions.
Solid sorbents represent another important category, including activated carbons, zeolites, metal-organic frameworks (MOFs), and amine-functionalized silica materials. These materials offer advantages in terms of lower regeneration energy requirements compared to liquid amine systems, with some advanced MOFs demonstrating CO₂ uptake capacities exceeding 1.5 g CO₂/g sorbent under specific conditions.
Despite these advancements, significant challenges persist in the development of CO₂ capture sorbents for recycling processes. A primary concern is the high energy penalty associated with sorbent regeneration, which can consume 20-30% of a power plant's energy output when implemented at scale. This energy requirement substantially impacts the economic viability of carbon capture technologies in industrial applications.
Sorbent degradation presents another critical challenge, particularly in recycling contexts where materials must withstand numerous adsorption-desorption cycles. Current amine-based sorbents typically lose 1-3% of their capacity per cycle due to thermal and oxidative degradation, necessitating regular replenishment and increasing operational costs.
Selectivity remains problematic in real-world applications where gas streams contain multiple components. Most existing sorbents exhibit reduced CO₂ capture performance in the presence of water vapor, SOx, NOx, and other contaminants commonly found in industrial emissions, limiting their practical utility in recycling processes.
Scale-up challenges further complicate implementation, as materials that perform well in laboratory settings often face manufacturing constraints, mechanical stability issues, and increased costs when produced at industrial scales. The cost of current advanced sorbent materials can range from $20-500/kg, compared to conventional MEA solutions at $1-2/kg.
Water consumption represents an additional concern, particularly in regions facing water scarcity. Traditional amine scrubbing systems require significant water inputs, with consumption rates of 1-2 tons of water per ton of CO₂ captured, creating sustainability challenges for widespread deployment.
Addressing these technical limitations requires interdisciplinary approaches combining materials science, chemical engineering, and process optimization to develop next-generation sorbents specifically designed for the unique demands of recycling processes.
Solid sorbents represent another important category, including activated carbons, zeolites, metal-organic frameworks (MOFs), and amine-functionalized silica materials. These materials offer advantages in terms of lower regeneration energy requirements compared to liquid amine systems, with some advanced MOFs demonstrating CO₂ uptake capacities exceeding 1.5 g CO₂/g sorbent under specific conditions.
Despite these advancements, significant challenges persist in the development of CO₂ capture sorbents for recycling processes. A primary concern is the high energy penalty associated with sorbent regeneration, which can consume 20-30% of a power plant's energy output when implemented at scale. This energy requirement substantially impacts the economic viability of carbon capture technologies in industrial applications.
Sorbent degradation presents another critical challenge, particularly in recycling contexts where materials must withstand numerous adsorption-desorption cycles. Current amine-based sorbents typically lose 1-3% of their capacity per cycle due to thermal and oxidative degradation, necessitating regular replenishment and increasing operational costs.
Selectivity remains problematic in real-world applications where gas streams contain multiple components. Most existing sorbents exhibit reduced CO₂ capture performance in the presence of water vapor, SOx, NOx, and other contaminants commonly found in industrial emissions, limiting their practical utility in recycling processes.
Scale-up challenges further complicate implementation, as materials that perform well in laboratory settings often face manufacturing constraints, mechanical stability issues, and increased costs when produced at industrial scales. The cost of current advanced sorbent materials can range from $20-500/kg, compared to conventional MEA solutions at $1-2/kg.
Water consumption represents an additional concern, particularly in regions facing water scarcity. Traditional amine scrubbing systems require significant water inputs, with consumption rates of 1-2 tons of water per ton of CO₂ captured, creating sustainability challenges for widespread deployment.
Addressing these technical limitations requires interdisciplinary approaches combining materials science, chemical engineering, and process optimization to develop next-generation sorbents specifically designed for the unique demands of recycling processes.
Existing CO₂ Sorbent Solutions for Recycling Processes
01 Metal-organic frameworks (MOFs) for CO₂ capture
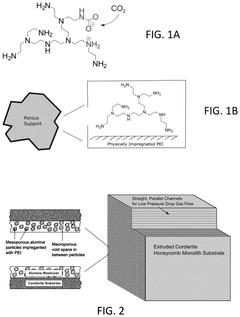
Metal-organic frameworks are advanced porous materials with high surface area and tunable pore structures that demonstrate exceptional CO₂ capture efficiency. These materials can be modified with functional groups to enhance CO₂ selectivity and adsorption capacity. MOFs offer advantages such as low regeneration energy requirements and stability over multiple adsorption-desorption cycles, making them promising candidates for industrial carbon capture applications.- Metal-organic frameworks (MOFs) for CO₂ capture: Metal-organic frameworks represent a class of highly porous materials with exceptional surface areas that can be tailored for selective CO₂ adsorption. These crystalline structures combine metal ions or clusters with organic linkers to create frameworks with tunable pore sizes and functionalities. MOFs demonstrate high CO₂ capture efficiency due to their ability to be chemically modified with specific functional groups that enhance CO₂ affinity. Their regeneration typically requires less energy compared to traditional sorbents, making them promising candidates for industrial carbon capture applications.
- Amine-functionalized sorbent materials: Amine-functionalized materials represent a significant advancement in CO₂ capture technology. These sorbents incorporate various amine groups that chemically bind with CO₂ through carbamate formation. The high selectivity for CO₂ over other gases makes these materials particularly effective for flue gas applications. Solid supports such as silica, polymers, or porous carbons can be impregnated or grafted with amines to create high-capacity sorbents. The capture efficiency can be optimized by controlling amine loading, type of amine groups, and support structure, while regeneration typically occurs at moderate temperatures.
- Zeolite and molecular sieve-based CO₂ capture: Zeolites and molecular sieves are aluminosilicate materials with well-defined pore structures that enable selective adsorption of CO₂. These materials capture carbon dioxide primarily through physical adsorption mechanisms, with efficiency dependent on pore size distribution, Si/Al ratio, and cation type. Modified zeolites with enhanced hydrophobicity show improved performance in humid conditions, addressing a common limitation in flue gas applications. Their high thermal stability allows for multiple adsorption-desorption cycles without significant degradation, though energy requirements for regeneration can be substantial compared to some newer sorbent technologies.
- Carbon-based sorbents for CO₂ capture: Carbon-based materials including activated carbons, carbon nanotubes, and graphene derivatives offer promising CO₂ capture capabilities. These materials can be produced from various precursors including biomass, polymers, or industrial waste, making them potentially cost-effective. Their capture efficiency stems from high surface areas, tunable pore structures, and the ability to incorporate functional groups that enhance CO₂ selectivity. Surface modification techniques such as nitrogen doping or incorporation of basic sites significantly improve adsorption capacity. Carbon-based sorbents generally demonstrate good stability across multiple adsorption-desorption cycles and can operate effectively across a range of temperatures and pressures.
- Hybrid and composite sorbent materials: Hybrid and composite sorbents combine multiple material types to achieve enhanced CO₂ capture performance beyond what individual components can provide. These materials typically integrate complementary capture mechanisms, such as combining physical adsorption properties of porous supports with chemical absorption capabilities of active components. Examples include MOF-polymer composites, amine-grafted silica-carbon hybrids, and mixed matrix materials. The synergistic effects in these composites often result in improved capture capacity, selectivity, stability, and regeneration efficiency. Advanced manufacturing techniques like 3D printing and controlled deposition methods enable precise structural control of these complex materials, optimizing mass transfer and reaction kinetics for maximum capture efficiency.
02 Amine-functionalized sorbents
Amine-functionalized materials represent a significant class of CO₂ capture sorbents with high selectivity and capacity. These materials work through chemical adsorption mechanisms where amine groups react with CO₂ to form carbamates or bicarbonates. Various supports including silica, polymers, and porous carbons can be functionalized with amines to enhance capture efficiency. The capture performance can be optimized by controlling amine loading, type of amine groups, and support characteristics.Expand Specific Solutions03 Zeolite and molecular sieve sorbents
Zeolites and molecular sieves are crystalline aluminosilicate materials with well-defined pore structures that enable selective CO₂ capture. These materials can be tailored by adjusting the silicon-to-aluminum ratio and incorporating various cations to enhance CO₂ adsorption capacity and selectivity. Their high thermal stability allows for efficient regeneration, though their performance may be affected by moisture. Recent developments include modified zeolites with improved hydrothermal stability and reduced regeneration energy requirements.Expand Specific Solutions04 Carbon-based sorbent materials
Carbon-based materials including activated carbons, carbon nanotubes, and graphene derivatives offer promising CO₂ capture capabilities due to their high surface area and pore volume. These materials can be produced from various precursors including biomass, polymers, and industrial waste, making them cost-effective options. Surface modification through nitrogen doping, metal incorporation, or chemical functionalization can significantly enhance their CO₂ capture efficiency. Their hydrophobic nature provides advantages in humid conditions compared to hydrophilic sorbents.Expand Specific Solutions05 Regeneration methods and energy efficiency
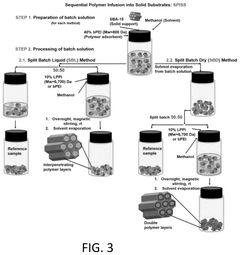
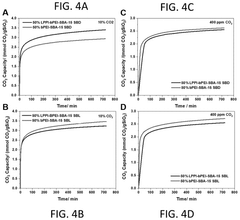
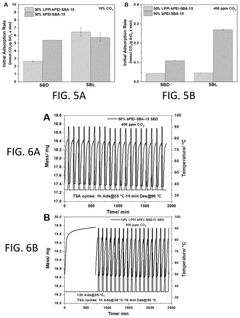
The overall efficiency of CO₂ capture systems depends significantly on the regeneration process of sorbent materials. Various regeneration methods including temperature swing adsorption (TSA), pressure swing adsorption (PSA), and vacuum swing adsorption (VSA) affect the energy requirements and sorbent longevity. Advanced regeneration techniques such as microwave-assisted desorption and electrical swing adsorption can reduce energy consumption. Optimizing the regeneration conditions is crucial for maintaining sorbent performance over multiple cycles and improving the economic viability of carbon capture technologies.Expand Specific Solutions
Leading Organizations in CO₂ Capture Material Development
The CO₂ capture sorbent material market for recycling processes is in an early growth phase, characterized by increasing investments and technological advancements. The market is projected to expand significantly as carbon neutrality goals drive demand for efficient carbon capture solutions. Currently, the technology landscape shows varying maturity levels, with established players like Climeworks AG and Global Thermostat Operations leading commercial direct air capture implementations, while academic institutions (Huazhong University, Rice University, University of Queensland) focus on fundamental research. Energy corporations (ExxonMobil, China Petroleum & Chemical Corp., Equinor) are strategically investing in sorbent technologies to complement their decarbonization strategies. Material specialists (Corning, NGK Insulators, Noritake) are leveraging their expertise to develop advanced ceramic and composite sorbents with enhanced CO₂ selectivity and regeneration capabilities.
Climeworks AG
Technical Solution: Climeworks has developed a Direct Air Capture (DAC) technology using solid sorbent materials that selectively capture CO₂ from ambient air. Their modular collectors contain proprietary amine-functionalized filter materials that bind with CO₂ molecules when air passes through. Once saturated, the filters are heated to approximately 100°C using low-grade waste heat, which releases concentrated CO₂ that can be collected for storage or utilization. Climeworks' technology operates in a cyclical process: adsorption at ambient temperature followed by regeneration at elevated temperatures. Their latest generation plants demonstrate improved energy efficiency, with thermal energy requirements reduced to approximately 2000-2500 kWh per ton of CO₂ captured. The company has successfully deployed commercial plants including Orca in Iceland (4,000 tons/year capacity) and is developing Mammoth (36,000 tons/year) with integration to permanent geological storage solutions.
Strengths: Modular design allows for scalable deployment; operates with relatively low-temperature heat sources (80-100°C); achieves high CO₂ purity (>99%); proven commercial implementation with multiple operational plants. Weaknesses: Still relatively high energy requirements compared to point-source capture; higher cost per ton of CO₂ captured ($600-800/ton) compared to industrial capture; requires significant land area for large-scale deployment.
Global Thermostat Operations LLC
Technical Solution: Global Thermostat has pioneered an advanced CO₂ capture technology using proprietary amine-based solid sorbents deposited on porous, honeycomb ceramic monoliths. Their system operates through a temperature-swing adsorption process where ambient air contacts the sorbent material, allowing CO₂ molecules to bind to the amine groups. The regeneration process uses low-temperature heat (85-100°C) from various sources including industrial waste heat, solar thermal, or geothermal energy to release concentrated CO₂. A key innovation is their "carbon negative" approach, where the heat required for regeneration can be supplied by renewable sources, ensuring the process removes more CO₂ than it emits. Global Thermostat's technology achieves capture costs estimated between $100-250 per ton of CO₂, significantly lower than many competitors. Their modular design allows for integration with existing industrial facilities, enabling both direct air capture and flue gas applications with the same core technology, providing flexibility for various implementation scenarios.
Strengths: Dual-purpose technology applicable to both direct air capture and point-source emissions; relatively low regeneration temperatures reduce energy costs; modular design enables flexible scaling and deployment; potential for integration with existing industrial processes. Weaknesses: Still requires significant energy input for large-scale deployment; ceramic monolith structures may be subject to thermal stress during cycling; technology has been demonstrated at pilot scale but needs further validation at commercial scale.
Key Innovations in CO₂ Capture Material Science
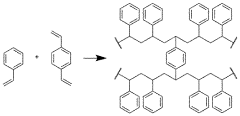
Sorbents, systems including sorbents, and methods using the sorbents
PatentPendingUS20240335784A1
Innovation
- Development of sorbents comprising a CO2-philic phase with a combination of polypropylenimine and polyethylenimine, which provides improved oxidative stability and hydrophilicity, allowing for efficient CO2 capture and regeneration, and are integrated into a structured support for enhanced performance.
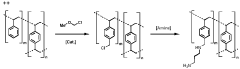
Sorbent materials for co2 capture, uses thereof and methods for making same
PatentWO2024002881A1
Innovation
- A sorbent material comprising primary, secondary, and tertiary amine moieties immobilized on a styrene-divinylbenzene support, functionalized with specific alkylamine groups, is used for cyclic adsorption/desorption, enhancing CO2 capture capacity and kinetic uptake, while maintaining stability through controlled temperature and steam injection.
Environmental Impact Assessment of Sorbent Materials
The environmental impact assessment of sorbent materials for CO₂ capture is a critical component in evaluating their overall sustainability and viability for large-scale implementation. Traditional carbon capture technologies often present significant environmental trade-offs that must be carefully considered against their climate change mitigation benefits.
When examining the life cycle environmental footprint of CO₂ capture sorbent materials, several key impact categories emerge. These include resource depletion, energy consumption, water usage, land use changes, and potential toxicity concerns. Amine-based sorbents, while effective for CO₂ capture, often require energy-intensive regeneration processes and may degrade into potentially harmful compounds over multiple capture-release cycles.
Solid sorbents such as metal-organic frameworks (MOFs) and zeolites generally demonstrate lower environmental impacts during operation compared to liquid amine systems. However, their production often involves energy-intensive synthesis methods and rare or toxic metal components. Recent research indicates that MOF production can generate up to 40% less greenhouse gas emissions compared to traditional amine scrubbing when evaluated on a life cycle basis.
Water consumption represents another significant environmental consideration. Aqueous amine systems typically require substantial water inputs, with estimates suggesting 1-2 tons of water consumption per ton of CO₂ captured. Novel porous materials and dry sorbent technologies have demonstrated potential to reduce water requirements by up to 80%, though often at the expense of capture efficiency or increased energy demands.
The disposal and end-of-life management of spent sorbent materials present additional environmental challenges. Many advanced sorbents contain metals or compounds that may pose leaching risks in landfill environments. Research into regeneration and recycling pathways for spent sorbents has shown promising results, with some materials maintaining over 85% of their original capture capacity after multiple regeneration cycles.
Emerging biobased sorbent materials derived from agricultural waste streams or sustainable biomass offer potentially lower environmental footprints. These materials leverage natural carbon structures while providing value-added applications for waste products. Life cycle assessments indicate that biobased sorbents can reduce embodied carbon by 30-60% compared to synthetic alternatives, though questions remain regarding their long-term stability and scalability.
The environmental impact of sorbent materials must also consider indirect effects such as reduced air quality from amine emissions, potential soil contamination from disposal, and ecosystem impacts from resource extraction. Comprehensive assessment frameworks that integrate these diverse environmental dimensions are essential for guiding sustainable development of next-generation carbon capture technologies.
When examining the life cycle environmental footprint of CO₂ capture sorbent materials, several key impact categories emerge. These include resource depletion, energy consumption, water usage, land use changes, and potential toxicity concerns. Amine-based sorbents, while effective for CO₂ capture, often require energy-intensive regeneration processes and may degrade into potentially harmful compounds over multiple capture-release cycles.
Solid sorbents such as metal-organic frameworks (MOFs) and zeolites generally demonstrate lower environmental impacts during operation compared to liquid amine systems. However, their production often involves energy-intensive synthesis methods and rare or toxic metal components. Recent research indicates that MOF production can generate up to 40% less greenhouse gas emissions compared to traditional amine scrubbing when evaluated on a life cycle basis.
Water consumption represents another significant environmental consideration. Aqueous amine systems typically require substantial water inputs, with estimates suggesting 1-2 tons of water consumption per ton of CO₂ captured. Novel porous materials and dry sorbent technologies have demonstrated potential to reduce water requirements by up to 80%, though often at the expense of capture efficiency or increased energy demands.
The disposal and end-of-life management of spent sorbent materials present additional environmental challenges. Many advanced sorbents contain metals or compounds that may pose leaching risks in landfill environments. Research into regeneration and recycling pathways for spent sorbents has shown promising results, with some materials maintaining over 85% of their original capture capacity after multiple regeneration cycles.
Emerging biobased sorbent materials derived from agricultural waste streams or sustainable biomass offer potentially lower environmental footprints. These materials leverage natural carbon structures while providing value-added applications for waste products. Life cycle assessments indicate that biobased sorbents can reduce embodied carbon by 30-60% compared to synthetic alternatives, though questions remain regarding their long-term stability and scalability.
The environmental impact of sorbent materials must also consider indirect effects such as reduced air quality from amine emissions, potential soil contamination from disposal, and ecosystem impacts from resource extraction. Comprehensive assessment frameworks that integrate these diverse environmental dimensions are essential for guiding sustainable development of next-generation carbon capture technologies.
Scalability and Economic Viability Analysis
The scalability of CO₂ capture sorbent materials represents a critical factor in determining their practical implementation in industrial recycling processes. Current laboratory-scale developments show promising CO₂ adsorption capacities, but significant challenges emerge when considering industrial-scale production and deployment. The transition from gram-scale synthesis to ton-scale manufacturing introduces complexities in maintaining consistent material properties, structural integrity, and adsorption performance.
Production scalability analysis reveals that metal-organic frameworks (MOFs) and amine-functionalized materials face substantial cost barriers when scaled up, with production costs estimated at $200-500/kg for specialized MOFs compared to $20-50/kg for conventional zeolites. This cost differential significantly impacts the economic viability of implementing these advanced materials in large-scale carbon capture systems.
Energy requirements for sorbent regeneration constitute a major economic consideration. Traditional temperature swing adsorption (TSA) processes require 2.5-4.0 GJ/ton CO₂ captured, while newer materials utilizing pressure swing approaches may reduce this to 1.8-2.5 GJ/ton CO₂. This energy demand directly translates to operational costs, which must be balanced against carbon pricing mechanisms to determine economic feasibility.
Lifecycle economic assessment indicates that sorbent durability significantly impacts long-term viability. Materials demonstrating stability over 1,000+ adsorption-desorption cycles show substantially improved cost profiles compared to those requiring replacement after 100-300 cycles. Current advanced sorbents typically maintain 80-90% of their original capacity after 500 cycles, suggesting replacement intervals of 1-2 years in continuous operation scenarios.
Market analysis projects that for widespread industrial adoption, the total cost of CO₂ capture using sorbent-based systems needs to fall below $40-60 per ton of CO₂. Current estimates place most advanced sorbent technologies at $70-120 per ton, indicating the need for further material optimization and process engineering improvements to achieve economic viability.
Infrastructure compatibility represents another crucial economic factor. Retrofitting existing industrial facilities with new sorbent-based capture systems requires capital investments ranging from $500-1,500 per ton of annual CO₂ capture capacity. This investment threshold necessitates sorbent materials that can integrate with minimal modifications to existing processes, particularly in energy-intensive industries like cement and steel manufacturing.
The economic viability ultimately depends on regulatory frameworks and carbon pricing mechanisms. Analysis suggests that carbon prices exceeding $50-75 per ton would make most advanced sorbent technologies economically viable, while current global carbon prices average only $15-30 per ton, creating a significant gap that requires either technological advancement or policy intervention to bridge.
Production scalability analysis reveals that metal-organic frameworks (MOFs) and amine-functionalized materials face substantial cost barriers when scaled up, with production costs estimated at $200-500/kg for specialized MOFs compared to $20-50/kg for conventional zeolites. This cost differential significantly impacts the economic viability of implementing these advanced materials in large-scale carbon capture systems.
Energy requirements for sorbent regeneration constitute a major economic consideration. Traditional temperature swing adsorption (TSA) processes require 2.5-4.0 GJ/ton CO₂ captured, while newer materials utilizing pressure swing approaches may reduce this to 1.8-2.5 GJ/ton CO₂. This energy demand directly translates to operational costs, which must be balanced against carbon pricing mechanisms to determine economic feasibility.
Lifecycle economic assessment indicates that sorbent durability significantly impacts long-term viability. Materials demonstrating stability over 1,000+ adsorption-desorption cycles show substantially improved cost profiles compared to those requiring replacement after 100-300 cycles. Current advanced sorbents typically maintain 80-90% of their original capacity after 500 cycles, suggesting replacement intervals of 1-2 years in continuous operation scenarios.
Market analysis projects that for widespread industrial adoption, the total cost of CO₂ capture using sorbent-based systems needs to fall below $40-60 per ton of CO₂. Current estimates place most advanced sorbent technologies at $70-120 per ton, indicating the need for further material optimization and process engineering improvements to achieve economic viability.
Infrastructure compatibility represents another crucial economic factor. Retrofitting existing industrial facilities with new sorbent-based capture systems requires capital investments ranging from $500-1,500 per ton of annual CO₂ capture capacity. This investment threshold necessitates sorbent materials that can integrate with minimal modifications to existing processes, particularly in energy-intensive industries like cement and steel manufacturing.
The economic viability ultimately depends on regulatory frameworks and carbon pricing mechanisms. Analysis suggests that carbon prices exceeding $50-75 per ton would make most advanced sorbent technologies economically viable, while current global carbon prices average only $15-30 per ton, creating a significant gap that requires either technological advancement or policy intervention to bridge.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!