Compare Pulsed Electromagnetic Field and Cold Compression Therapy
MAR 7, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
PEMF vs Cold Therapy Background and Objectives
Pulsed Electromagnetic Field (PEMF) therapy and Cold Compression Therapy represent two distinct therapeutic modalities that have gained significant attention in rehabilitation medicine, sports recovery, and pain management. Both technologies emerged from different scientific foundations yet share common objectives in promoting tissue healing, reducing inflammation, and managing pain. The comparative analysis of these therapies has become increasingly relevant as healthcare providers seek evidence-based treatment options for various musculoskeletal conditions.
PEMF therapy traces its origins to the early 20th century when researchers first discovered the biological effects of electromagnetic fields on living tissues. The technology evolved from basic electromagnetic research into sophisticated medical devices capable of delivering precisely controlled electromagnetic pulses to targeted body regions. Modern PEMF systems utilize low-frequency electromagnetic fields, typically ranging from 1 to 100 Hz, to stimulate cellular processes and promote healing at the molecular level.
Cold Compression Therapy, conversely, builds upon the well-established principles of cryotherapy and mechanical compression. This approach combines the vasoconstricting effects of cold temperatures with the circulatory benefits of intermittent compression. The technology has evolved from simple ice applications to sophisticated pneumatic compression systems that deliver controlled cold temperatures while providing rhythmic compression patterns to enhance lymphatic drainage and reduce tissue swelling.
The primary objective of comparing these two therapeutic approaches centers on understanding their respective mechanisms of action, clinical efficacy, and optimal application scenarios. PEMF therapy aims to enhance cellular metabolism, improve microcirculation, and modulate inflammatory responses through electromagnetic field interactions with cellular structures. The therapy targets mitochondrial function, calcium ion channels, and cellular membrane potential to accelerate natural healing processes.
Cold Compression Therapy objectives focus on immediate symptom relief through vasoconstriction, metabolic rate reduction, and mechanical enhancement of fluid dynamics. The combined cold and compression effects work synergistically to minimize secondary tissue damage, reduce pain perception through gate control mechanisms, and facilitate faster recovery from acute injuries or post-surgical conditions.
Understanding the comparative effectiveness of these technologies requires examining their applications across different clinical scenarios, patient populations, and treatment protocols. Both therapies demonstrate unique advantages in specific contexts, making their comparative analysis essential for developing comprehensive treatment strategies that optimize patient outcomes while considering factors such as treatment duration, cost-effectiveness, and patient compliance.
PEMF therapy traces its origins to the early 20th century when researchers first discovered the biological effects of electromagnetic fields on living tissues. The technology evolved from basic electromagnetic research into sophisticated medical devices capable of delivering precisely controlled electromagnetic pulses to targeted body regions. Modern PEMF systems utilize low-frequency electromagnetic fields, typically ranging from 1 to 100 Hz, to stimulate cellular processes and promote healing at the molecular level.
Cold Compression Therapy, conversely, builds upon the well-established principles of cryotherapy and mechanical compression. This approach combines the vasoconstricting effects of cold temperatures with the circulatory benefits of intermittent compression. The technology has evolved from simple ice applications to sophisticated pneumatic compression systems that deliver controlled cold temperatures while providing rhythmic compression patterns to enhance lymphatic drainage and reduce tissue swelling.
The primary objective of comparing these two therapeutic approaches centers on understanding their respective mechanisms of action, clinical efficacy, and optimal application scenarios. PEMF therapy aims to enhance cellular metabolism, improve microcirculation, and modulate inflammatory responses through electromagnetic field interactions with cellular structures. The therapy targets mitochondrial function, calcium ion channels, and cellular membrane potential to accelerate natural healing processes.
Cold Compression Therapy objectives focus on immediate symptom relief through vasoconstriction, metabolic rate reduction, and mechanical enhancement of fluid dynamics. The combined cold and compression effects work synergistically to minimize secondary tissue damage, reduce pain perception through gate control mechanisms, and facilitate faster recovery from acute injuries or post-surgical conditions.
Understanding the comparative effectiveness of these technologies requires examining their applications across different clinical scenarios, patient populations, and treatment protocols. Both therapies demonstrate unique advantages in specific contexts, making their comparative analysis essential for developing comprehensive treatment strategies that optimize patient outcomes while considering factors such as treatment duration, cost-effectiveness, and patient compliance.
Market Demand for Non-Invasive Recovery Therapies
The global healthcare landscape is experiencing a significant shift toward non-invasive therapeutic modalities, driven by increasing patient preference for treatments that minimize discomfort and reduce recovery time. This trend has created substantial market opportunities for technologies like Pulsed Electromagnetic Field therapy and Cold Compression therapy, both of which address the growing demand for effective pain management and recovery solutions without surgical intervention.
Healthcare systems worldwide are grappling with rising costs and patient volume pressures, making non-invasive recovery therapies increasingly attractive from both clinical and economic perspectives. These treatments offer reduced hospitalization requirements, lower risk profiles, and improved patient compliance compared to traditional invasive procedures. The aging global population, particularly in developed markets, has intensified the need for accessible recovery solutions that can be administered in outpatient settings or even at home.
The sports medicine and rehabilitation sectors represent particularly robust growth areas for non-invasive recovery therapies. Professional sports organizations, fitness centers, and rehabilitation clinics are actively seeking evidence-based treatments that can accelerate recovery while maintaining safety standards. This demand extends beyond elite athletics to include recreational sports participants and weekend warriors who seek professional-grade recovery solutions.
Chronic pain management represents another significant market driver, with millions of patients seeking alternatives to pharmaceutical interventions due to concerns about long-term medication dependency and side effects. Non-invasive therapies offer compelling value propositions for conditions such as arthritis, fibromyalgia, and musculoskeletal disorders, where traditional treatment options may have limited effectiveness or undesirable consequences.
The post-surgical recovery market has also embraced non-invasive modalities as complementary treatments that can enhance healing outcomes and reduce complications. Orthopedic procedures, in particular, have shown strong adoption rates for both electromagnetic field and cold compression therapies as standard components of recovery protocols.
Technological advancement and miniaturization have made these therapies more accessible and user-friendly, expanding their market reach beyond clinical settings into home healthcare applications. This accessibility factor has broadened the potential customer base significantly, creating opportunities for direct-to-consumer markets and telemedicine integration.
Regulatory acceptance and clinical validation continue to strengthen market confidence in non-invasive recovery therapies. As more peer-reviewed studies demonstrate efficacy and safety profiles, healthcare providers are increasingly incorporating these modalities into standard treatment protocols, further driving market expansion and establishing sustainable demand patterns across multiple therapeutic categories.
Healthcare systems worldwide are grappling with rising costs and patient volume pressures, making non-invasive recovery therapies increasingly attractive from both clinical and economic perspectives. These treatments offer reduced hospitalization requirements, lower risk profiles, and improved patient compliance compared to traditional invasive procedures. The aging global population, particularly in developed markets, has intensified the need for accessible recovery solutions that can be administered in outpatient settings or even at home.
The sports medicine and rehabilitation sectors represent particularly robust growth areas for non-invasive recovery therapies. Professional sports organizations, fitness centers, and rehabilitation clinics are actively seeking evidence-based treatments that can accelerate recovery while maintaining safety standards. This demand extends beyond elite athletics to include recreational sports participants and weekend warriors who seek professional-grade recovery solutions.
Chronic pain management represents another significant market driver, with millions of patients seeking alternatives to pharmaceutical interventions due to concerns about long-term medication dependency and side effects. Non-invasive therapies offer compelling value propositions for conditions such as arthritis, fibromyalgia, and musculoskeletal disorders, where traditional treatment options may have limited effectiveness or undesirable consequences.
The post-surgical recovery market has also embraced non-invasive modalities as complementary treatments that can enhance healing outcomes and reduce complications. Orthopedic procedures, in particular, have shown strong adoption rates for both electromagnetic field and cold compression therapies as standard components of recovery protocols.
Technological advancement and miniaturization have made these therapies more accessible and user-friendly, expanding their market reach beyond clinical settings into home healthcare applications. This accessibility factor has broadened the potential customer base significantly, creating opportunities for direct-to-consumer markets and telemedicine integration.
Regulatory acceptance and clinical validation continue to strengthen market confidence in non-invasive recovery therapies. As more peer-reviewed studies demonstrate efficacy and safety profiles, healthcare providers are increasingly incorporating these modalities into standard treatment protocols, further driving market expansion and establishing sustainable demand patterns across multiple therapeutic categories.
Current Status of PEMF and Cold Compression Technologies
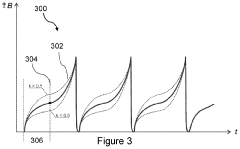
Pulsed Electromagnetic Field (PEMF) therapy has evolved significantly since its initial development in the 1970s. Currently, PEMF devices operate across various frequency ranges, typically from 1 Hz to 10,000 Hz, with magnetic field intensities ranging from 0.1 to 5,000 Gauss. Modern PEMF systems utilize sophisticated waveform generators that can produce sinusoidal, square, or sawtooth wave patterns, allowing for precise therapeutic targeting. The technology has achieved regulatory approval in multiple jurisdictions, with FDA clearance for specific applications including bone healing, depression treatment, and pain management.
Contemporary PEMF devices demonstrate remarkable versatility in their delivery mechanisms. Portable units weighing less than 2 pounds offer localized treatment through applicator coils, while full-body mat systems provide comprehensive exposure. Advanced models incorporate programmable protocols with variable intensity, frequency, and duration settings, enabling personalized treatment regimens. Battery-powered devices now offer 8-12 hours of continuous operation, significantly enhancing patient compliance and treatment accessibility.
Cold compression therapy has simultaneously advanced through integration of precise temperature control and pneumatic compression systems. Current devices maintain therapeutic temperatures between 35-50°F (1.7-10°C) while delivering intermittent compression pressures of 20-80 mmHg. Modern units feature microprocessor-controlled cooling systems that eliminate temperature fluctuations, ensuring consistent therapeutic delivery. The technology has expanded beyond traditional ice packs to include circulating cold water systems, thermoelectric cooling, and phase-change material applications.
Recent innovations in cold compression include anatomically designed wraps with targeted compression zones and integrated temperature sensors for real-time monitoring. Professional-grade systems now offer simultaneous multi-site treatment capabilities, allowing comprehensive post-surgical or injury management. Portable units weighing under 5 pounds provide clinical-grade performance in home settings, featuring rechargeable batteries and smartphone connectivity for treatment tracking.
Both technologies face distinct implementation challenges. PEMF therapy encounters variability in treatment protocols and limited standardization across devices, while cold compression therapy must balance effective cooling with patient comfort and safety considerations. Ongoing research focuses on optimizing treatment parameters and developing combination therapies that leverage the complementary mechanisms of both modalities.
Contemporary PEMF devices demonstrate remarkable versatility in their delivery mechanisms. Portable units weighing less than 2 pounds offer localized treatment through applicator coils, while full-body mat systems provide comprehensive exposure. Advanced models incorporate programmable protocols with variable intensity, frequency, and duration settings, enabling personalized treatment regimens. Battery-powered devices now offer 8-12 hours of continuous operation, significantly enhancing patient compliance and treatment accessibility.
Cold compression therapy has simultaneously advanced through integration of precise temperature control and pneumatic compression systems. Current devices maintain therapeutic temperatures between 35-50°F (1.7-10°C) while delivering intermittent compression pressures of 20-80 mmHg. Modern units feature microprocessor-controlled cooling systems that eliminate temperature fluctuations, ensuring consistent therapeutic delivery. The technology has expanded beyond traditional ice packs to include circulating cold water systems, thermoelectric cooling, and phase-change material applications.
Recent innovations in cold compression include anatomically designed wraps with targeted compression zones and integrated temperature sensors for real-time monitoring. Professional-grade systems now offer simultaneous multi-site treatment capabilities, allowing comprehensive post-surgical or injury management. Portable units weighing under 5 pounds provide clinical-grade performance in home settings, featuring rechargeable batteries and smartphone connectivity for treatment tracking.
Both technologies face distinct implementation challenges. PEMF therapy encounters variability in treatment protocols and limited standardization across devices, while cold compression therapy must balance effective cooling with patient comfort and safety considerations. Ongoing research focuses on optimizing treatment parameters and developing combination therapies that leverage the complementary mechanisms of both modalities.
Existing PEMF and Cold Compression Solutions
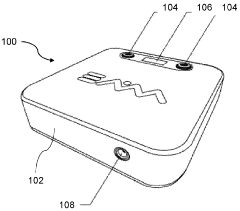
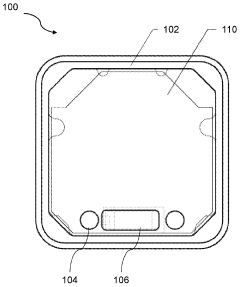
01 Combined pulsed electromagnetic field and cold compression therapy devices
Therapeutic devices that integrate both pulsed electromagnetic field (PEMF) technology and cold compression therapy into a single system for enhanced treatment outcomes. These combined modalities work synergistically to reduce inflammation, pain, and swelling while promoting tissue healing and recovery. The devices typically feature controllable electromagnetic field generators coupled with cooling mechanisms that can be applied simultaneously or sequentially to the treatment area.- Combined pulsed electromagnetic field and cold compression therapy devices: Therapeutic devices that integrate both pulsed electromagnetic field (PEMF) technology and cold compression therapy into a single system for enhanced treatment outcomes. These combined modalities work synergistically to reduce inflammation, pain, and swelling while promoting tissue healing and recovery. The devices typically feature controllable electromagnetic field generators coupled with cooling mechanisms that can be applied simultaneously or sequentially to the treatment area.
- Pulsed electromagnetic field therapy systems and methods: Devices and methods specifically designed to deliver pulsed electromagnetic fields for therapeutic purposes. These systems generate controlled electromagnetic pulses at specific frequencies and intensities to stimulate cellular activity, enhance blood circulation, and promote tissue regeneration. The technology can be applied to various body parts for treating musculoskeletal conditions, wound healing, and pain management.
- Cold compression therapy apparatus and control systems: Therapeutic devices that apply controlled cold temperatures combined with compression to injured or inflamed tissues. These systems typically include cooling units, compression garments or wraps, and control mechanisms to regulate temperature and pressure levels. The therapy helps reduce swelling, minimize pain, and accelerate recovery by controlling tissue temperature and promoting fluid drainage from the affected area.
- Wearable therapeutic devices with electromagnetic and thermal capabilities: Portable and wearable therapeutic devices designed for convenient application of electromagnetic field therapy and temperature control. These devices feature flexible designs that conform to body contours, allowing patients to receive treatment while maintaining mobility. The systems often include adjustable straps, rechargeable power sources, and programmable treatment protocols for personalized therapy sessions.
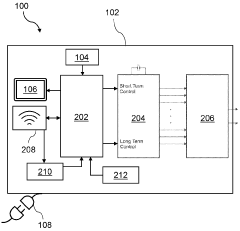
- Control and monitoring systems for combination therapy: Advanced control systems and monitoring technologies for managing combined electromagnetic field and cold compression therapies. These systems feature programmable controllers, sensors for monitoring treatment parameters, user interfaces for adjusting therapy settings, and safety mechanisms to ensure proper treatment delivery. The technology enables precise control of treatment duration, intensity, temperature, and compression levels based on patient needs and treatment protocols.
02 Pulsed electromagnetic field therapy systems for tissue treatment
Devices and methods utilizing pulsed electromagnetic fields to stimulate cellular activity and promote healing in various tissues. These systems generate controlled electromagnetic pulses at specific frequencies and intensities to penetrate tissue and influence cellular processes such as ion transport, protein synthesis, and blood circulation. The therapy can be applied for pain management, bone healing, wound recovery, and inflammation reduction.Expand Specific Solutions03 Cold compression therapy apparatus and methods
Therapeutic systems that apply controlled cooling and compression to body parts for reducing swelling, pain, and inflammation. These devices typically include cooling units, compression garments or wraps, and control systems that regulate temperature and pressure levels. The therapy is commonly used for post-surgical recovery, sports injuries, and acute trauma management by constricting blood vessels and reducing metabolic activity in the treated area.Expand Specific Solutions04 Wearable therapeutic devices with electromagnetic and thermal modalities
Portable and wearable treatment devices designed for convenient application of electromagnetic field therapy and temperature-based treatments. These devices feature flexible designs that conform to body contours, allowing patients to receive therapy while maintaining mobility. They incorporate power sources, control electronics, and therapeutic elements in compact configurations suitable for home use or ambulatory treatment settings.Expand Specific Solutions05 Control systems and treatment protocols for multimodal therapy
Advanced control mechanisms and programmable treatment protocols for managing combined therapeutic modalities. These systems feature microprocessors, sensors, and user interfaces that allow precise adjustment of electromagnetic field parameters, cooling temperatures, compression levels, and treatment duration. The control systems may include preset programs for different conditions, real-time monitoring capabilities, and safety features to ensure optimal therapeutic outcomes while preventing adverse effects.Expand Specific Solutions
Major Players in PEMF and Cold Therapy Markets
The competitive landscape for Pulsed Electromagnetic Field (PEMF) and Cold Compression Therapy represents a mature yet evolving medical technology sector with significant market potential. The industry has progressed beyond early adoption, with established players like Medtronic, Boston Scientific's Cardiac Pacemakers, and Smith + Nephew demonstrating proven commercial viability. Market size spans multiple therapeutic areas including cardiovascular, orthopedic, and aesthetic medicine, supported by companies such as Venus Concept and Regenesis Biomedical. Technology maturity varies across applications, with cardiac devices showing high sophistication through St. Jude Medical and Zoll Circulation, while emerging players like Galvanize Therapeutics and Node Co. Ltd. indicate continued innovation opportunities. The presence of both established medical device giants and specialized startups suggests a dynamic competitive environment with room for technological advancement and market expansion.
Zoll Circulation, Inc.
Technical Solution: Zoll Circulation develops therapeutic hypothermia and circulation management systems that incorporate cold compression therapy principles for cardiac and neurological applications. Their technology provides precise temperature control and graduated compression to improve circulation and reduce metabolic demand during critical care scenarios. The systems feature automated temperature regulation, continuous monitoring capabilities, and integration with existing medical infrastructure for comprehensive patient management.
Strengths: Critical care specialization, proven clinical outcomes, robust monitoring systems. Weaknesses: Limited to hospital settings, high acquisition and maintenance costs.
Medtronic, Inc.
Technical Solution: Medtronic has developed comprehensive pulsed electromagnetic field (PEMF) therapy systems that utilize low-frequency electromagnetic pulses to stimulate cellular repair and reduce inflammation. Their technology incorporates precise frequency modulation ranging from 1-100 Hz to target specific tissue types and conditions. The company's PEMF devices feature advanced control algorithms that adjust pulse intensity and duration based on treatment protocols, enabling personalized therapy for bone healing, wound care, and pain management applications.
Strengths: Extensive clinical validation, FDA approval for multiple indications, strong market presence. Weaknesses: High cost, limited portability of some systems.
Core Technologies in Electromagnetic Field Generation
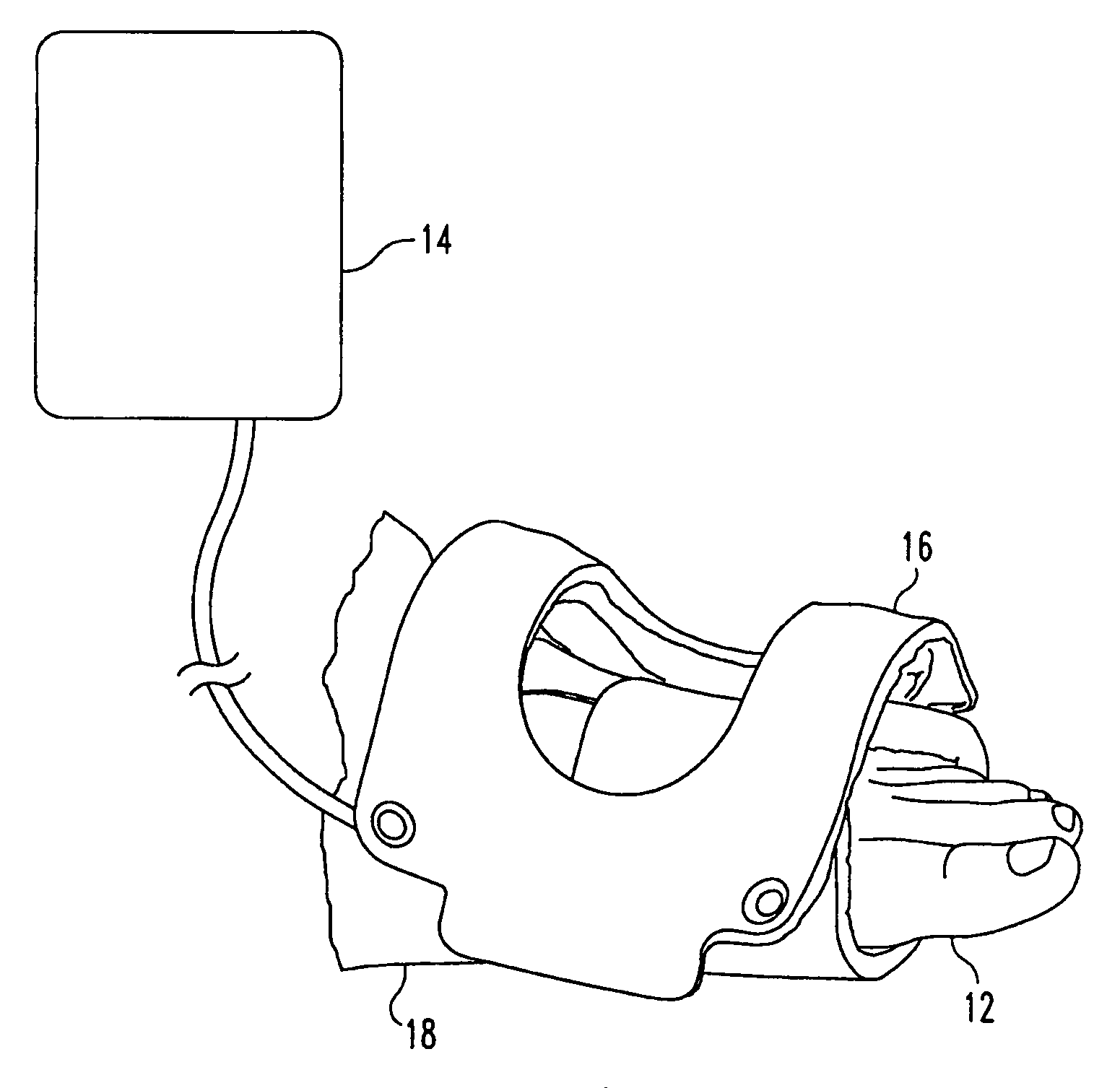
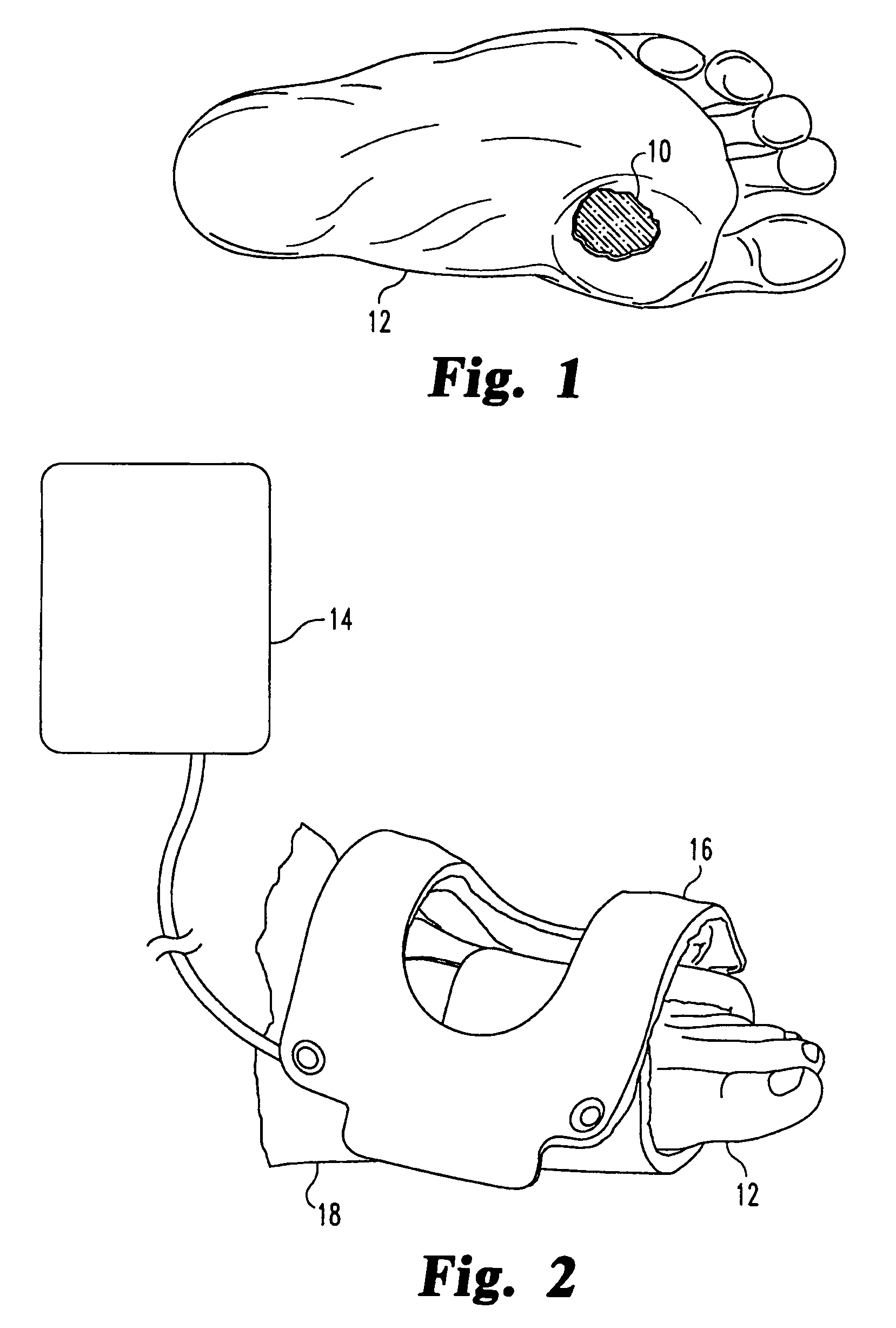
Pulsed electromagnetic field method of treating soft tissue wounds
PatentActiveUS7520849B1
Innovation
- The use of a pulsed electromagnetic field (PEMF) with specific signal characteristics, including repetitive pulse bursts and unipolar magnetic field pulses, is applied externally to the wound area without direct contact, using a treatment coil and signal generator to promote wound healing.
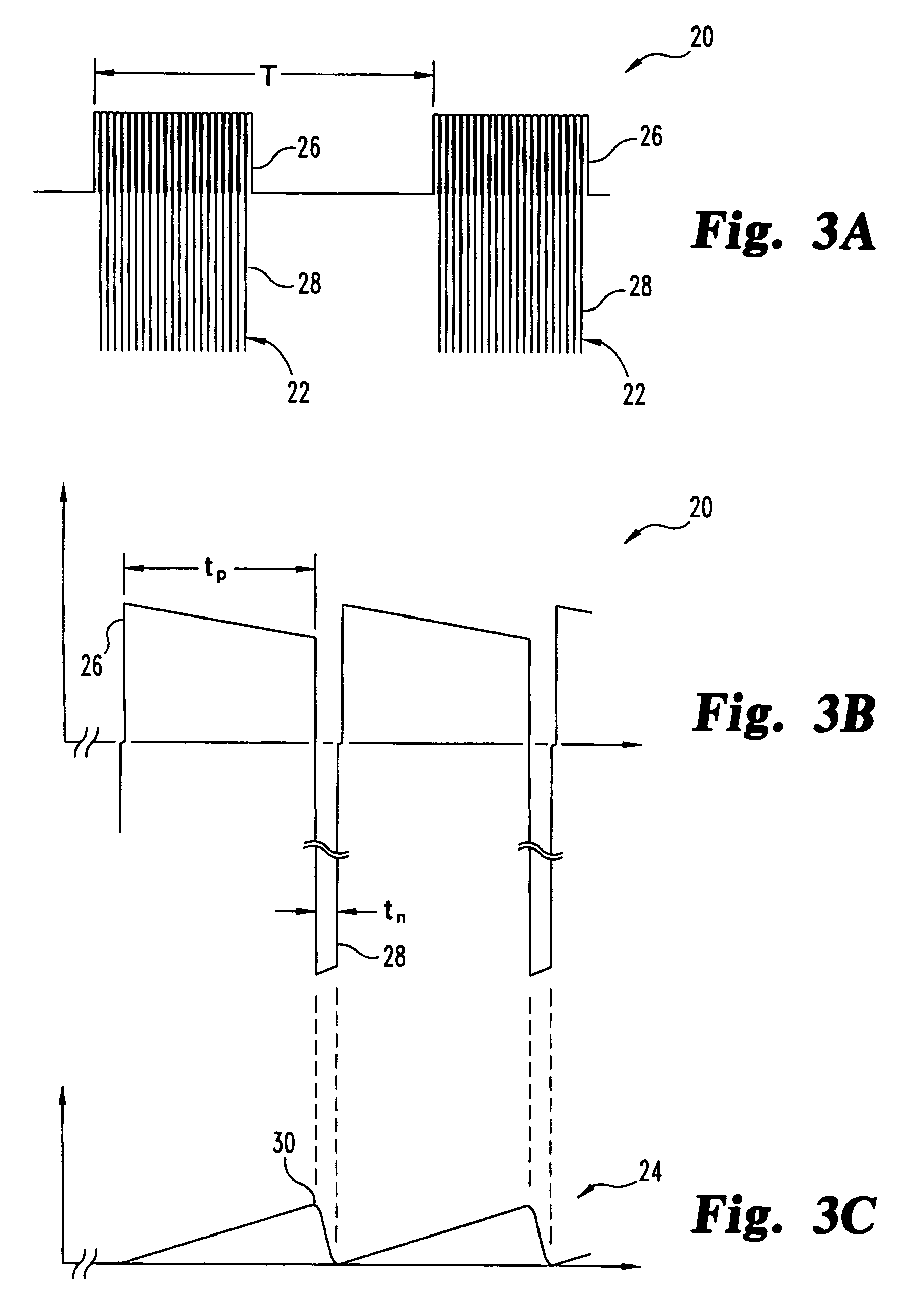
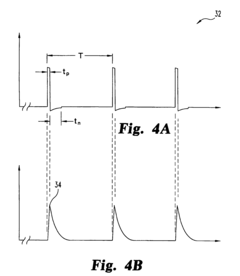
A pulsed electromagnetic field apparatus and method for generating frequencies
PatentWO2024127242A1
Innovation
- A PEMF apparatus with a pulse generator and electromagnetic field generation means that uses modified sawtooth waveforms with pre-stress and relaxation periods, and quasi-sine signals with pulse width modulation, along with a feedback circuit for frequency stability and precision, and a bifilar antenna for scalar wave generation.
Clinical Trial Requirements for Therapeutic Devices
Clinical trials for therapeutic devices comparing Pulsed Electromagnetic Field (PEMF) and Cold Compression Therapy must adhere to stringent regulatory frameworks established by agencies such as the FDA, EMA, and other national regulatory bodies. These devices are typically classified as Class II medical devices, requiring 510(k) premarket notification or CE marking depending on the jurisdiction. The regulatory pathway demands comprehensive documentation demonstrating safety and efficacy through well-designed clinical studies.
Study design requirements mandate randomized controlled trials with appropriate control groups, often including sham devices or standard care comparators. For PEMF devices, double-blinding presents unique challenges due to the electromagnetic field generation, necessitating sophisticated sham devices that mimic operational sounds and visual indicators without producing therapeutic fields. Cold compression therapy trials face similar blinding challenges, requiring careful consideration of temperature sensation and device operation feedback.
Primary endpoint selection must align with the intended therapeutic indication, whether pain reduction, inflammation control, or tissue healing acceleration. Validated outcome measures such as Visual Analog Scale for pain, range of motion assessments, and inflammatory biomarkers are essential. Secondary endpoints should include functional improvement metrics, quality of life assessments, and long-term safety monitoring parameters.
Patient population criteria require precise inclusion and exclusion parameters based on the target condition severity, duration, and demographic factors. Contraindications for PEMF therapy include pregnancy, presence of metallic implants, and certain cardiac conditions, while cold compression therapy contraindications encompass circulatory disorders and cold sensitivity conditions. Sample size calculations must account for expected effect sizes, statistical power requirements, and potential dropout rates.
Safety monitoring protocols demand comprehensive adverse event reporting systems, with particular attention to device-specific risks. PEMF devices require monitoring for electromagnetic interference with other medical devices, while cold compression systems necessitate surveillance for tissue damage from excessive cooling or pressure application.
Data collection standards must comply with Good Clinical Practice guidelines, incorporating electronic data capture systems with audit trails and source document verification. Statistical analysis plans should specify primary analysis methods, handling of missing data, and interim analysis procedures for safety monitoring.
Study design requirements mandate randomized controlled trials with appropriate control groups, often including sham devices or standard care comparators. For PEMF devices, double-blinding presents unique challenges due to the electromagnetic field generation, necessitating sophisticated sham devices that mimic operational sounds and visual indicators without producing therapeutic fields. Cold compression therapy trials face similar blinding challenges, requiring careful consideration of temperature sensation and device operation feedback.
Primary endpoint selection must align with the intended therapeutic indication, whether pain reduction, inflammation control, or tissue healing acceleration. Validated outcome measures such as Visual Analog Scale for pain, range of motion assessments, and inflammatory biomarkers are essential. Secondary endpoints should include functional improvement metrics, quality of life assessments, and long-term safety monitoring parameters.
Patient population criteria require precise inclusion and exclusion parameters based on the target condition severity, duration, and demographic factors. Contraindications for PEMF therapy include pregnancy, presence of metallic implants, and certain cardiac conditions, while cold compression therapy contraindications encompass circulatory disorders and cold sensitivity conditions. Sample size calculations must account for expected effect sizes, statistical power requirements, and potential dropout rates.
Safety monitoring protocols demand comprehensive adverse event reporting systems, with particular attention to device-specific risks. PEMF devices require monitoring for electromagnetic interference with other medical devices, while cold compression systems necessitate surveillance for tissue damage from excessive cooling or pressure application.
Data collection standards must comply with Good Clinical Practice guidelines, incorporating electronic data capture systems with audit trails and source document verification. Statistical analysis plans should specify primary analysis methods, handling of missing data, and interim analysis procedures for safety monitoring.
Safety Standards for Electromagnetic Medical Equipment
The safety standards for electromagnetic medical equipment represent a critical regulatory framework that governs the development, manufacturing, and clinical application of devices utilizing electromagnetic fields for therapeutic purposes. These standards have evolved significantly over the past decades, driven by the increasing adoption of electromagnetic therapies and the need to ensure patient safety while maintaining therapeutic efficacy.
International regulatory bodies, including the International Electrotechnical Commission (IEC) and the Food and Drug Administration (FDA), have established comprehensive guidelines specifically addressing electromagnetic medical devices. The IEC 60601 series serves as the foundational standard for medical electrical equipment, with particular emphasis on electromagnetic compatibility and safety requirements. These standards mandate rigorous testing protocols for electromagnetic emissions, immunity to interference, and biological safety assessments.
For pulsed electromagnetic field devices, safety standards focus on specific absorption rate (SAR) limitations, magnetic field strength thresholds, and exposure duration guidelines. The standards require manufacturers to demonstrate that electromagnetic field intensities remain within established biological safety limits, typically not exceeding 2 watts per kilogram of tissue for localized exposure. Additionally, devices must undergo extensive biocompatibility testing to ensure materials in contact with patients meet ISO 10993 standards.
Regulatory compliance involves multiple phases of testing and documentation. Pre-market submissions must include electromagnetic compatibility testing results, demonstrating that devices neither emit harmful interference nor are susceptible to external electromagnetic disturbances. Clinical safety data requirements mandate comprehensive adverse event reporting and long-term safety monitoring protocols.
Recent regulatory developments have introduced more stringent requirements for electromagnetic medical equipment, particularly regarding cybersecurity and software validation. The FDA's guidance on electromagnetic compatibility emphasizes the importance of risk management throughout the device lifecycle, requiring manufacturers to implement robust quality management systems that continuously monitor safety performance and address emerging electromagnetic interference challenges in modern healthcare environments.
International regulatory bodies, including the International Electrotechnical Commission (IEC) and the Food and Drug Administration (FDA), have established comprehensive guidelines specifically addressing electromagnetic medical devices. The IEC 60601 series serves as the foundational standard for medical electrical equipment, with particular emphasis on electromagnetic compatibility and safety requirements. These standards mandate rigorous testing protocols for electromagnetic emissions, immunity to interference, and biological safety assessments.
For pulsed electromagnetic field devices, safety standards focus on specific absorption rate (SAR) limitations, magnetic field strength thresholds, and exposure duration guidelines. The standards require manufacturers to demonstrate that electromagnetic field intensities remain within established biological safety limits, typically not exceeding 2 watts per kilogram of tissue for localized exposure. Additionally, devices must undergo extensive biocompatibility testing to ensure materials in contact with patients meet ISO 10993 standards.
Regulatory compliance involves multiple phases of testing and documentation. Pre-market submissions must include electromagnetic compatibility testing results, demonstrating that devices neither emit harmful interference nor are susceptible to external electromagnetic disturbances. Clinical safety data requirements mandate comprehensive adverse event reporting and long-term safety monitoring protocols.
Recent regulatory developments have introduced more stringent requirements for electromagnetic medical equipment, particularly regarding cybersecurity and software validation. The FDA's guidance on electromagnetic compatibility emphasizes the importance of risk management throughout the device lifecycle, requiring manufacturers to implement robust quality management systems that continuously monitor safety performance and address emerging electromagnetic interference challenges in modern healthcare environments.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!