Amide vs Ester: Evaluating Hydrolysis Rates for Stability
FEB 28, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Hydrolysis Bond Stability Background and Research Objectives
Chemical bond stability represents a fundamental aspect of molecular design and pharmaceutical development, with hydrolysis reactions serving as critical determinants of compound longevity in biological and environmental systems. The comparative analysis of amide and ester bond hydrolysis rates has emerged as a pivotal research area, directly impacting drug formulation strategies, biodegradable material design, and chemical synthesis optimization.
Amide and ester bonds constitute two of the most prevalent linkages in organic chemistry, yet they exhibit markedly different hydrolytic behaviors under identical conditions. Ester bonds, characterized by their carbonyl carbon bonded to an oxygen atom, typically demonstrate higher susceptibility to hydrolysis compared to amide bonds, where the carbonyl carbon connects to a nitrogen atom. This fundamental difference stems from the distinct electronic properties and resonance stabilization mechanisms inherent to each bond type.
The pharmaceutical industry has increasingly recognized the significance of hydrolysis rate evaluation in drug development pipelines. Prodrug design strategies frequently exploit controlled hydrolysis mechanisms to achieve targeted drug release profiles, while stability assessments guide formulation scientists in predicting shelf-life and storage requirements. Similarly, the growing emphasis on sustainable materials has intensified interest in understanding hydrolytic degradation patterns for biodegradable polymers and packaging materials.
Current analytical methodologies for hydrolysis rate determination encompass diverse approaches, ranging from traditional spectroscopic techniques to advanced chromatographic methods. However, standardized protocols for comparative stability assessment remain fragmented across different research domains, creating challenges in data interpretation and cross-study comparisons.
The primary objective of this research initiative centers on establishing comprehensive evaluation frameworks for quantitative comparison of amide and ester bond hydrolysis rates under controlled conditions. This encompasses developing standardized testing protocols that account for pH variations, temperature dependencies, and catalytic influences. Additionally, the research aims to elucidate mechanistic pathways governing hydrolytic processes, enabling predictive modeling capabilities for bond stability assessment.
Secondary objectives include creating databases of hydrolysis rate constants for various amide and ester derivatives, facilitating structure-activity relationship analyses. The research also seeks to identify optimal conditions for accelerated stability testing while maintaining correlation with real-world degradation scenarios, ultimately supporting rational molecular design strategies across pharmaceutical and materials science applications.
Amide and ester bonds constitute two of the most prevalent linkages in organic chemistry, yet they exhibit markedly different hydrolytic behaviors under identical conditions. Ester bonds, characterized by their carbonyl carbon bonded to an oxygen atom, typically demonstrate higher susceptibility to hydrolysis compared to amide bonds, where the carbonyl carbon connects to a nitrogen atom. This fundamental difference stems from the distinct electronic properties and resonance stabilization mechanisms inherent to each bond type.
The pharmaceutical industry has increasingly recognized the significance of hydrolysis rate evaluation in drug development pipelines. Prodrug design strategies frequently exploit controlled hydrolysis mechanisms to achieve targeted drug release profiles, while stability assessments guide formulation scientists in predicting shelf-life and storage requirements. Similarly, the growing emphasis on sustainable materials has intensified interest in understanding hydrolytic degradation patterns for biodegradable polymers and packaging materials.
Current analytical methodologies for hydrolysis rate determination encompass diverse approaches, ranging from traditional spectroscopic techniques to advanced chromatographic methods. However, standardized protocols for comparative stability assessment remain fragmented across different research domains, creating challenges in data interpretation and cross-study comparisons.
The primary objective of this research initiative centers on establishing comprehensive evaluation frameworks for quantitative comparison of amide and ester bond hydrolysis rates under controlled conditions. This encompasses developing standardized testing protocols that account for pH variations, temperature dependencies, and catalytic influences. Additionally, the research aims to elucidate mechanistic pathways governing hydrolytic processes, enabling predictive modeling capabilities for bond stability assessment.
Secondary objectives include creating databases of hydrolysis rate constants for various amide and ester derivatives, facilitating structure-activity relationship analyses. The research also seeks to identify optimal conditions for accelerated stability testing while maintaining correlation with real-world degradation scenarios, ultimately supporting rational molecular design strategies across pharmaceutical and materials science applications.
Market Demand for Stable Amide and Ester Compounds
The pharmaceutical industry represents the largest market segment for stable amide and ester compounds, driven by the critical need for drug molecules with predictable degradation profiles. Pharmaceutical companies require precise understanding of hydrolysis rates to optimize drug formulations, establish shelf-life parameters, and ensure therapeutic efficacy throughout product lifecycles. The demand extends across various therapeutic areas including oncology, cardiovascular diseases, and central nervous system disorders, where molecular stability directly impacts patient safety and treatment outcomes.
Biotechnology and biopharmaceutical sectors demonstrate rapidly expanding demand for compounds with controlled hydrolysis characteristics. These industries particularly value amide bonds for their enhanced stability in biological environments, making them essential for peptide-based therapeutics, protein conjugates, and advanced drug delivery systems. The growing emphasis on personalized medicine and targeted therapies has intensified requirements for molecules with specific stability profiles tailored to individual patient needs.
The agrochemical industry constitutes another significant market driver, where pesticides, herbicides, and fertilizers require carefully engineered stability characteristics. Agricultural applications demand compounds that maintain efficacy under diverse environmental conditions while ensuring controlled degradation to minimize ecological impact. The balance between functional longevity and environmental safety creates substantial market opportunities for optimized amide and ester formulations.
Cosmetics and personal care industries increasingly seek stable compounds for active ingredient delivery systems, anti-aging formulations, and long-lasting product performance. Consumer expectations for product durability and consistent performance drive demand for molecules with well-characterized hydrolysis behavior, particularly in moisture-rich environments typical of skincare applications.
Industrial applications spanning polymer manufacturing, specialty chemicals, and materials science require compounds with tailored stability profiles for specific operational conditions. These sectors value the ability to predict and control degradation rates for quality assurance, process optimization, and product development initiatives.
The market demand is further amplified by regulatory requirements across multiple industries mandating comprehensive stability testing and documentation. Regulatory agencies worldwide require detailed hydrolysis data for product approvals, creating sustained demand for analytical services and research capabilities in this field.
Biotechnology and biopharmaceutical sectors demonstrate rapidly expanding demand for compounds with controlled hydrolysis characteristics. These industries particularly value amide bonds for their enhanced stability in biological environments, making them essential for peptide-based therapeutics, protein conjugates, and advanced drug delivery systems. The growing emphasis on personalized medicine and targeted therapies has intensified requirements for molecules with specific stability profiles tailored to individual patient needs.
The agrochemical industry constitutes another significant market driver, where pesticides, herbicides, and fertilizers require carefully engineered stability characteristics. Agricultural applications demand compounds that maintain efficacy under diverse environmental conditions while ensuring controlled degradation to minimize ecological impact. The balance between functional longevity and environmental safety creates substantial market opportunities for optimized amide and ester formulations.
Cosmetics and personal care industries increasingly seek stable compounds for active ingredient delivery systems, anti-aging formulations, and long-lasting product performance. Consumer expectations for product durability and consistent performance drive demand for molecules with well-characterized hydrolysis behavior, particularly in moisture-rich environments typical of skincare applications.
Industrial applications spanning polymer manufacturing, specialty chemicals, and materials science require compounds with tailored stability profiles for specific operational conditions. These sectors value the ability to predict and control degradation rates for quality assurance, process optimization, and product development initiatives.
The market demand is further amplified by regulatory requirements across multiple industries mandating comprehensive stability testing and documentation. Regulatory agencies worldwide require detailed hydrolysis data for product approvals, creating sustained demand for analytical services and research capabilities in this field.
Current Hydrolysis Rate Evaluation Methods and Challenges
Current hydrolysis rate evaluation methods encompass several established analytical approaches, each with distinct advantages and limitations. Spectrophotometric techniques represent the most widely adopted methodology, utilizing UV-Vis spectroscopy to monitor substrate depletion or product formation over time. This approach offers real-time monitoring capabilities and relatively straightforward implementation, making it suitable for both amide and ester bond analysis.
High-performance liquid chromatography (HPLC) serves as another cornerstone method, providing superior separation capabilities and quantitative precision. HPLC-based approaches excel in complex sample matrices where multiple hydrolysis products may interfere with spectrophotometric measurements. However, this method requires extensive sample preparation and cannot provide continuous real-time data, necessitating discrete time-point sampling.
Nuclear magnetic resonance (NMR) spectroscopy offers unique advantages for hydrolysis rate determination, particularly for amide bonds where traditional methods may lack sensitivity. NMR enables direct observation of bond-breaking events and provides structural information about hydrolysis intermediates. Despite these benefits, NMR methods face limitations in sensitivity and require specialized expertise for data interpretation.
Potentiometric titration methods provide cost-effective alternatives for hydrolysis studies, particularly when pH changes accompany bond cleavage. These techniques offer excellent precision for ester hydrolysis under basic conditions but demonstrate limited applicability for amide bonds due to minimal pH changes during neutral hydrolysis.
Significant challenges persist across all current methodologies. Temperature control represents a critical factor, as hydrolysis rates exhibit strong temperature dependence, requiring precise thermal regulation to ensure reproducible results. Buffer selection and ionic strength maintenance pose additional complications, particularly when comparing amide and ester hydrolysis under physiologically relevant conditions.
Sensitivity limitations become particularly pronounced when evaluating amide bonds, which typically exhibit hydrolysis rates several orders of magnitude slower than corresponding esters. This disparity necessitates extended monitoring periods for amides, increasing experimental complexity and potential for systematic errors.
Matrix effects present another substantial challenge, especially in biological or pharmaceutical applications where complex sample compositions can interfere with analytical measurements. Protein binding, metal ion catalysis, and competing reactions frequently complicate rate determinations and compromise comparative studies between amide and ester systems.
Standardization issues further complicate cross-laboratory comparisons, as variations in experimental protocols, buffer systems, and analytical conditions can significantly impact measured hydrolysis rates. The absence of universally accepted reference standards for both amide and ester hydrolysis studies limits the reliability of comparative stability assessments across different research groups.
High-performance liquid chromatography (HPLC) serves as another cornerstone method, providing superior separation capabilities and quantitative precision. HPLC-based approaches excel in complex sample matrices where multiple hydrolysis products may interfere with spectrophotometric measurements. However, this method requires extensive sample preparation and cannot provide continuous real-time data, necessitating discrete time-point sampling.
Nuclear magnetic resonance (NMR) spectroscopy offers unique advantages for hydrolysis rate determination, particularly for amide bonds where traditional methods may lack sensitivity. NMR enables direct observation of bond-breaking events and provides structural information about hydrolysis intermediates. Despite these benefits, NMR methods face limitations in sensitivity and require specialized expertise for data interpretation.
Potentiometric titration methods provide cost-effective alternatives for hydrolysis studies, particularly when pH changes accompany bond cleavage. These techniques offer excellent precision for ester hydrolysis under basic conditions but demonstrate limited applicability for amide bonds due to minimal pH changes during neutral hydrolysis.
Significant challenges persist across all current methodologies. Temperature control represents a critical factor, as hydrolysis rates exhibit strong temperature dependence, requiring precise thermal regulation to ensure reproducible results. Buffer selection and ionic strength maintenance pose additional complications, particularly when comparing amide and ester hydrolysis under physiologically relevant conditions.
Sensitivity limitations become particularly pronounced when evaluating amide bonds, which typically exhibit hydrolysis rates several orders of magnitude slower than corresponding esters. This disparity necessitates extended monitoring periods for amides, increasing experimental complexity and potential for systematic errors.
Matrix effects present another substantial challenge, especially in biological or pharmaceutical applications where complex sample compositions can interfere with analytical measurements. Protein binding, metal ion catalysis, and competing reactions frequently complicate rate determinations and compromise comparative studies between amide and ester systems.
Standardization issues further complicate cross-laboratory comparisons, as variations in experimental protocols, buffer systems, and analytical conditions can significantly impact measured hydrolysis rates. The absence of universally accepted reference standards for both amide and ester hydrolysis studies limits the reliability of comparative stability assessments across different research groups.
Existing Hydrolysis Rate Measurement Solutions
01 Hydrolysis-resistant amide and ester bond modifications
Certain chemical modifications can be made to amide and ester bonds to increase their resistance to hydrolysis. These modifications include the introduction of steric hindrance around the bond, incorporation of electron-withdrawing groups, or the use of specific protecting groups. Such structural modifications can significantly reduce the hydrolysis rate by making the carbonyl carbon less susceptible to nucleophilic attack, thereby improving the stability of compounds in aqueous environments.- Hydrolysis-resistant amide and ester bond modifications: Certain chemical modifications can be made to amide and ester bonds to increase their resistance to hydrolysis. These modifications include the introduction of steric hindrance around the bond, incorporation of electron-withdrawing groups, or the use of specific substituents that stabilize the bond structure. Such modifications are particularly useful in pharmaceutical applications where prolonged stability is required.
- Comparative hydrolysis rates of amide versus ester bonds: Studies have shown that amide bonds generally exhibit slower hydrolysis rates compared to ester bonds under similar conditions. This difference is attributed to the resonance stabilization of the amide bond and the lower electrophilicity of the carbonyl carbon. Understanding these comparative rates is essential for designing molecules with desired stability profiles in various pH and temperature conditions.
- pH-dependent hydrolysis kinetics: The hydrolysis rates of both amide and ester bonds are significantly influenced by pH conditions. Ester bonds typically hydrolyze faster in alkaline conditions, while amide bonds show increased hydrolysis rates under strongly acidic or basic conditions. The pH-dependent behavior is crucial for applications in drug delivery systems and biodegradable materials where controlled degradation is desired.
- Enzymatic hydrolysis mechanisms: Enzymatic hydrolysis of amide and ester bonds involves specific catalytic mechanisms that differ from chemical hydrolysis. Esterases and proteases exhibit different substrate specificities and catalytic efficiencies. The enzymatic approach offers advantages in terms of selectivity and mild reaction conditions, making it valuable for biotechnological applications and prodrug activation strategies.
- Temperature effects on hydrolysis stability: Temperature plays a critical role in determining the hydrolysis rates of amide and ester bonds. Elevated temperatures generally accelerate hydrolysis reactions by increasing molecular motion and collision frequency. The activation energy and temperature sensitivity differ between amide and ester bonds, which is important for formulation stability and shelf-life predictions in pharmaceutical and chemical products.
02 pH-dependent hydrolysis kinetics of amide and ester bonds
The hydrolysis rates of amide and ester bonds are highly dependent on pH conditions. Ester bonds typically hydrolyze faster under both acidic and basic conditions compared to amide bonds, which are more stable. The rate of hydrolysis can be controlled by adjusting the pH of the environment, with acidic conditions promoting ester hydrolysis through acid catalysis and basic conditions facilitating both ester and amide hydrolysis through base catalysis. Understanding these pH-dependent mechanisms is crucial for designing stable formulations.Expand Specific Solutions03 Enzymatic hydrolysis of amide and ester bonds
Enzymatic hydrolysis represents a specific pathway for breaking down amide and ester bonds through the action of enzymes such as esterases, lipases, and peptidases. These enzymes can selectively cleave specific bonds under mild physiological conditions. The rate of enzymatic hydrolysis depends on factors including enzyme concentration, substrate specificity, temperature, and the presence of inhibitors or activators. This mechanism is particularly important in biological systems and drug metabolism.Expand Specific Solutions04 Temperature effects on hydrolysis rates
Temperature significantly influences the hydrolysis rates of both amide and ester bonds. Increased temperature generally accelerates hydrolysis reactions by providing more kinetic energy to overcome activation barriers. The relationship between temperature and hydrolysis rate typically follows Arrhenius kinetics, allowing for prediction of stability at different storage conditions. This is particularly important for pharmaceutical formulations and chemical products that must maintain stability across various temperature ranges.Expand Specific Solutions05 Stabilization strategies for hydrolysis-prone bonds
Various stabilization strategies can be employed to reduce the hydrolysis rates of amide and ester bonds in formulations. These include the use of chelating agents to remove catalytic metal ions, addition of antioxidants, incorporation of buffering systems to maintain optimal pH, use of water-miscible co-solvents to reduce water activity, and encapsulation techniques. These approaches can significantly extend the shelf life and stability of products containing hydrolysis-sensitive bonds.Expand Specific Solutions
Key Players in Chemical Stability Testing Industry
The hydrolysis rate evaluation technology for amide versus ester bond stability comparison represents a mature analytical field within the broader pharmaceutical and chemical industries, currently valued at several billion dollars globally. The industry has reached an advanced development stage, with established methodologies and standardized protocols widely adopted across research institutions and commercial entities. Technology maturity is evidenced by the diverse player ecosystem, spanning from pharmaceutical giants like Takeda Pharmaceutical, GlaxoSmithKline, and Allergan focusing on drug stability applications, to specialty chemical companies including DSM IP Assets, Solvay Specialty Polymers, and Eastman Chemical developing advanced materials. Academic institutions such as University of Queensland, Rutgers University, and Donghua University contribute fundamental research, while biotechnology firms like Novozymes leverage enzymatic approaches for hydrolysis studies. The competitive landscape reflects a well-established market with incremental innovations rather than disruptive breakthroughs, indicating technological saturation in core methodologies.
Novozymes A/S
Technical Solution: Novozymes has developed advanced enzymatic hydrolysis technologies for evaluating bond stability, particularly focusing on lipases and esterases for ester bond hydrolysis and proteases for amide bond evaluation. Their enzyme-based analytical platforms enable precise measurement of hydrolysis rates under controlled pH and temperature conditions, providing quantitative data for stability comparisons. The company's proprietary enzyme libraries allow for standardized testing protocols that can differentiate between amide and ester bond susceptibilities to hydrolysis, making them essential tools for pharmaceutical and chemical stability studies.
Strengths: Industry-leading enzyme expertise and comprehensive analytical capabilities. Weaknesses: Limited to enzymatic hydrolysis methods, may not cover all chemical hydrolysis pathways.
Takeda Pharmaceutical Co., Ltd.
Technical Solution: Takeda has developed sophisticated analytical methodologies for evaluating hydrolysis kinetics of drug compounds containing both amide and ester linkages. Their approach combines accelerated stability testing with real-time monitoring using HPLC-MS/MS techniques to quantify degradation products and calculate hydrolysis rate constants. The company employs pH-rate profiles and Arrhenius modeling to predict long-term stability, with particular expertise in comparing the hydrolytic stability of prodrugs and active pharmaceutical ingredients. Their validated protocols enable accurate assessment of bond stability under physiological and storage conditions.
Strengths: Extensive pharmaceutical expertise and validated analytical protocols for drug stability. Weaknesses: Focus primarily on pharmaceutical applications, limited broader chemical industry applications.
Core Innovations in Amide vs Ester Stability Analysis
Compositions and methods comprising prostaglandin related compounds and trefoil factor family peptides for the treatment of glaucoma with reduced hyperemia
PatentInactiveUS20060046958A1
Innovation
- Combining prostaglandin-related compounds with trefoil factor family peptides in dosage forms and methods for topical administration to treat ocular or conjunctival hyperemia, allowing for various administration schedules and compositions to optimize therapeutic effects.
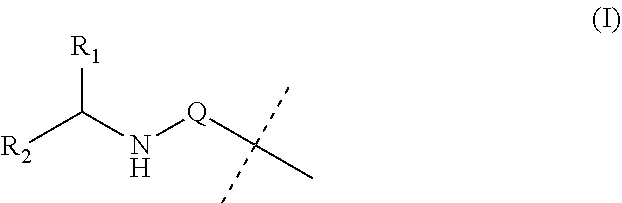
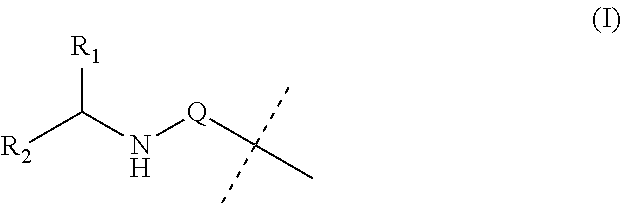
Covalent conjugates
PatentInactiveUS20190142949A1
Innovation
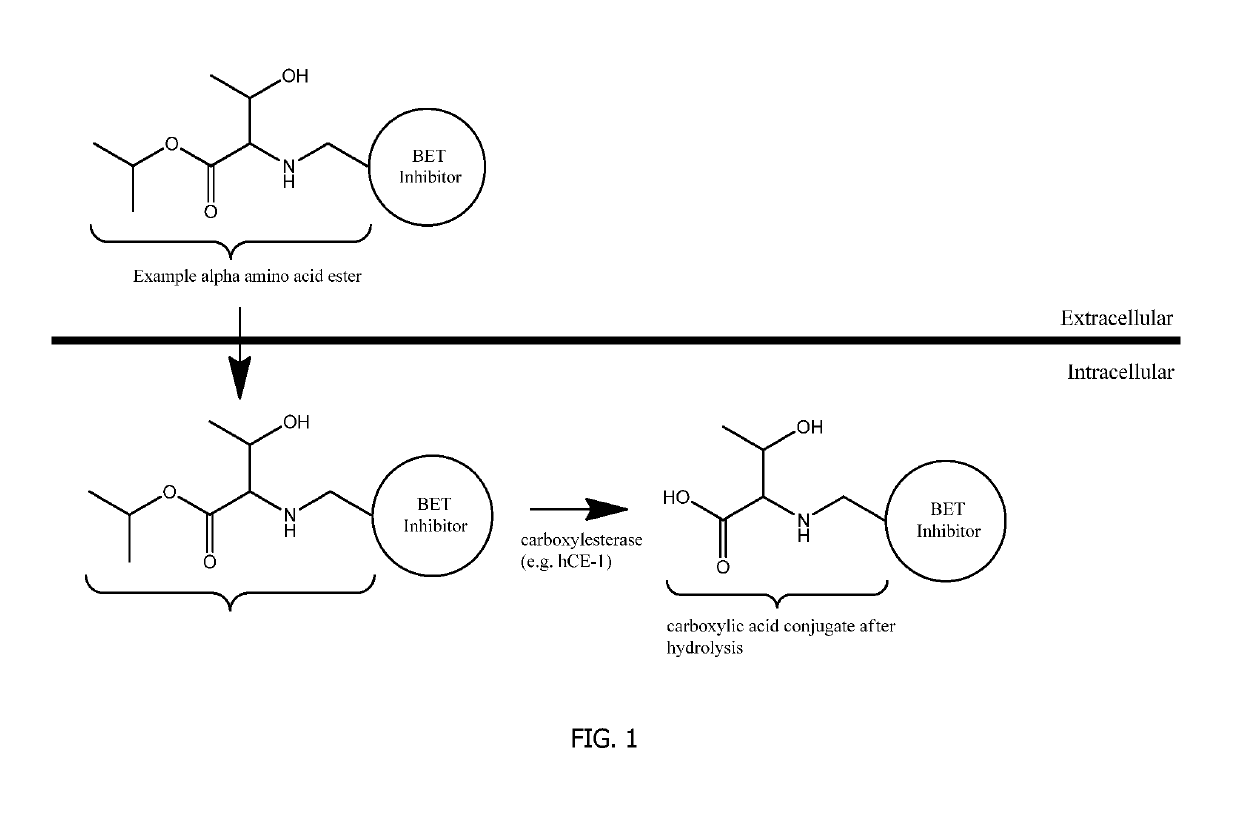
- The development of covalent conjugates of BET inhibitors with alpha amino acid esters, which are hydrolyzed by intracellular carboxylesterases to improve therapeutic profiles by selectively targeting cells expressing hCE-1, such as monocytes and macrophages, thereby enhancing potency and duration of action while reducing systemic exposure.
Chemical Safety Regulations for Stability Testing
Chemical safety regulations for stability testing of pharmaceutical compounds have evolved significantly to address the unique challenges posed by different chemical bond types, particularly when evaluating hydrolysis rates between amide and ester bonds. Regulatory frameworks established by the International Council for Harmonisation (ICH) provide comprehensive guidelines that mandate specific testing protocols for assessing molecular stability under various environmental conditions.
The ICH Q1A(R2) guidelines establish fundamental requirements for stability testing, emphasizing the need for accelerated and long-term studies that can accurately predict degradation pathways. These regulations specifically address hydrolytic degradation mechanisms, requiring pharmaceutical companies to conduct systematic evaluations under controlled temperature and humidity conditions. The guidelines mandate testing at 40°C ± 2°C with 75% ± 5% relative humidity for accelerated studies, conditions that effectively differentiate hydrolysis susceptibility between amide and ester functionalities.
Regulatory authorities have implemented stringent documentation requirements for hydrolysis rate determinations, necessitating validated analytical methods that can quantify degradation products with high precision. The FDA's guidance on analytical procedures and methods validation requires that stability-indicating assays demonstrate specificity, accuracy, and precision when measuring hydrolytic breakdown of different bond types. These regulations ensure that comparative studies between amide and ester stability are conducted using standardized methodologies.
Safety assessment protocols mandated by regulatory bodies require comprehensive evaluation of hydrolysis products, as degradation pathways may generate compounds with different toxicological profiles. The European Medicines Agency (EMA) guidelines emphasize the importance of identifying and characterizing all degradation products exceeding 0.1% threshold levels, with particular attention to those arising from differential hydrolysis rates between structural analogs.
Current regulations also address the statistical requirements for stability data analysis, mandating appropriate sample sizes and statistical methods for comparing hydrolysis rates across different molecular structures. These frameworks ensure that conclusions regarding relative stability between amide and ester bonds are supported by robust statistical evidence, enabling informed decisions in pharmaceutical development and regulatory approval processes.
The ICH Q1A(R2) guidelines establish fundamental requirements for stability testing, emphasizing the need for accelerated and long-term studies that can accurately predict degradation pathways. These regulations specifically address hydrolytic degradation mechanisms, requiring pharmaceutical companies to conduct systematic evaluations under controlled temperature and humidity conditions. The guidelines mandate testing at 40°C ± 2°C with 75% ± 5% relative humidity for accelerated studies, conditions that effectively differentiate hydrolysis susceptibility between amide and ester functionalities.
Regulatory authorities have implemented stringent documentation requirements for hydrolysis rate determinations, necessitating validated analytical methods that can quantify degradation products with high precision. The FDA's guidance on analytical procedures and methods validation requires that stability-indicating assays demonstrate specificity, accuracy, and precision when measuring hydrolytic breakdown of different bond types. These regulations ensure that comparative studies between amide and ester stability are conducted using standardized methodologies.
Safety assessment protocols mandated by regulatory bodies require comprehensive evaluation of hydrolysis products, as degradation pathways may generate compounds with different toxicological profiles. The European Medicines Agency (EMA) guidelines emphasize the importance of identifying and characterizing all degradation products exceeding 0.1% threshold levels, with particular attention to those arising from differential hydrolysis rates between structural analogs.
Current regulations also address the statistical requirements for stability data analysis, mandating appropriate sample sizes and statistical methods for comparing hydrolysis rates across different molecular structures. These frameworks ensure that conclusions regarding relative stability between amide and ester bonds are supported by robust statistical evidence, enabling informed decisions in pharmaceutical development and regulatory approval processes.
Environmental Impact of Hydrolysis Testing Procedures
The environmental implications of hydrolysis testing procedures for evaluating amide and ester bond stability present multifaceted challenges that require careful consideration in laboratory design and regulatory compliance. Traditional hydrolysis testing methods often involve the use of organic solvents, acidic or basic catalysts, and elevated temperatures, which collectively contribute to significant environmental burdens through chemical waste generation and energy consumption.
Solvent usage represents one of the primary environmental concerns in hydrolysis rate evaluation protocols. Conventional testing procedures frequently employ chlorinated solvents, aromatic hydrocarbons, and other volatile organic compounds that pose risks to both human health and environmental systems. The disposal of these solvents requires specialized treatment facilities, and their production contributes to carbon emissions and resource depletion.
Buffer systems utilized in pH-controlled hydrolysis experiments generate substantial volumes of aqueous waste containing various salts and organic compounds. These waste streams often require neutralization and specialized treatment before disposal, particularly when heavy metal catalysts or toxic organic additives are present. The cumulative effect of multiple testing cycles can result in significant waste volumes that challenge laboratory waste management systems.
Energy consumption associated with temperature-controlled hydrolysis studies contributes to the overall carbon footprint of stability testing procedures. Extended incubation periods at elevated temperatures, often ranging from 40°C to 80°C over weeks or months, require continuous heating systems that consume substantial electrical energy. This energy demand becomes particularly significant when conducting comprehensive stability studies across multiple temperature conditions.
Emerging green chemistry approaches are beginning to address these environmental challenges through the development of more sustainable testing methodologies. Water-based testing systems, microwave-assisted hydrolysis techniques, and miniaturized reaction vessels offer promising alternatives that reduce solvent consumption and energy requirements while maintaining analytical precision.
The implementation of automated testing platforms and real-time monitoring systems can further minimize environmental impact by optimizing reaction conditions and reducing the need for repetitive experiments. These technological advances enable more efficient use of resources while generating higher-quality data for stability comparisons between amide and ester bonds.
Solvent usage represents one of the primary environmental concerns in hydrolysis rate evaluation protocols. Conventional testing procedures frequently employ chlorinated solvents, aromatic hydrocarbons, and other volatile organic compounds that pose risks to both human health and environmental systems. The disposal of these solvents requires specialized treatment facilities, and their production contributes to carbon emissions and resource depletion.
Buffer systems utilized in pH-controlled hydrolysis experiments generate substantial volumes of aqueous waste containing various salts and organic compounds. These waste streams often require neutralization and specialized treatment before disposal, particularly when heavy metal catalysts or toxic organic additives are present. The cumulative effect of multiple testing cycles can result in significant waste volumes that challenge laboratory waste management systems.
Energy consumption associated with temperature-controlled hydrolysis studies contributes to the overall carbon footprint of stability testing procedures. Extended incubation periods at elevated temperatures, often ranging from 40°C to 80°C over weeks or months, require continuous heating systems that consume substantial electrical energy. This energy demand becomes particularly significant when conducting comprehensive stability studies across multiple temperature conditions.
Emerging green chemistry approaches are beginning to address these environmental challenges through the development of more sustainable testing methodologies. Water-based testing systems, microwave-assisted hydrolysis techniques, and miniaturized reaction vessels offer promising alternatives that reduce solvent consumption and energy requirements while maintaining analytical precision.
The implementation of automated testing platforms and real-time monitoring systems can further minimize environmental impact by optimizing reaction conditions and reducing the need for repetitive experiments. These technological advances enable more efficient use of resources while generating higher-quality data for stability comparisons between amide and ester bonds.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!