How to Validate Aromatic Compounds’ Purity for Lab Applications
MAR 5, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Aromatic Compound Purity Validation Background and Objectives
Aromatic compounds represent a fundamental class of organic molecules characterized by their distinctive ring structures and delocalized electron systems, playing crucial roles across pharmaceutical, chemical, and materials science industries. These compounds, ranging from simple benzene derivatives to complex polycyclic structures, serve as essential building blocks in drug synthesis, specialty chemicals production, and advanced materials development. The validation of their purity has emerged as a critical quality control parameter that directly impacts research outcomes, product efficacy, and regulatory compliance.
The historical development of aromatic compound purity validation traces back to the early 20th century when basic colorimetric and melting point determinations were the primary assessment methods. The evolution accelerated significantly with the introduction of chromatographic techniques in the 1950s, followed by spectroscopic methods in subsequent decades. Modern analytical chemistry has transformed this field through the integration of high-resolution mass spectrometry, advanced NMR techniques, and sophisticated separation technologies, enabling detection of impurities at parts-per-million levels.
Contemporary laboratory applications demand increasingly stringent purity standards, particularly in pharmaceutical research where trace impurities can significantly affect biological activity and safety profiles. The pharmaceutical industry typically requires purity levels exceeding 95% for research compounds and 99% for clinical candidates. Similarly, electronic materials applications necessitate ultra-high purity standards, often demanding 99.9% or higher purity levels to ensure optimal performance in semiconductor and optoelectronic devices.
The primary objective of modern aromatic compound purity validation encompasses multiple dimensions beyond simple percentage determination. These include comprehensive impurity profiling to identify specific contaminants, quantitative assessment of related substances and degradation products, and validation of structural integrity through orthogonal analytical approaches. Additionally, the validation process must address stability considerations, ensuring that purity assessments remain accurate throughout storage and handling periods.
Current technological objectives focus on developing rapid, cost-effective validation methods that can provide real-time purity assessment without compromising accuracy. This includes the integration of automated analytical platforms, development of portable analytical devices for field applications, and implementation of artificial intelligence algorithms for pattern recognition and impurity identification. The ultimate goal is establishing standardized validation protocols that ensure reproducibility across different laboratories while maintaining the flexibility to address diverse compound classes and application requirements.
The historical development of aromatic compound purity validation traces back to the early 20th century when basic colorimetric and melting point determinations were the primary assessment methods. The evolution accelerated significantly with the introduction of chromatographic techniques in the 1950s, followed by spectroscopic methods in subsequent decades. Modern analytical chemistry has transformed this field through the integration of high-resolution mass spectrometry, advanced NMR techniques, and sophisticated separation technologies, enabling detection of impurities at parts-per-million levels.
Contemporary laboratory applications demand increasingly stringent purity standards, particularly in pharmaceutical research where trace impurities can significantly affect biological activity and safety profiles. The pharmaceutical industry typically requires purity levels exceeding 95% for research compounds and 99% for clinical candidates. Similarly, electronic materials applications necessitate ultra-high purity standards, often demanding 99.9% or higher purity levels to ensure optimal performance in semiconductor and optoelectronic devices.
The primary objective of modern aromatic compound purity validation encompasses multiple dimensions beyond simple percentage determination. These include comprehensive impurity profiling to identify specific contaminants, quantitative assessment of related substances and degradation products, and validation of structural integrity through orthogonal analytical approaches. Additionally, the validation process must address stability considerations, ensuring that purity assessments remain accurate throughout storage and handling periods.
Current technological objectives focus on developing rapid, cost-effective validation methods that can provide real-time purity assessment without compromising accuracy. This includes the integration of automated analytical platforms, development of portable analytical devices for field applications, and implementation of artificial intelligence algorithms for pattern recognition and impurity identification. The ultimate goal is establishing standardized validation protocols that ensure reproducibility across different laboratories while maintaining the flexibility to address diverse compound classes and application requirements.
Market Demand for High-Purity Aromatic Compounds in Labs
The global laboratory chemicals market has experienced substantial growth driven by expanding research activities across pharmaceutical, biotechnology, and academic institutions. High-purity aromatic compounds represent a critical segment within this market, as these materials serve as essential building blocks for drug synthesis, analytical standards, and advanced materials research. The increasing complexity of modern pharmaceutical development, particularly in areas such as precision medicine and novel therapeutic modalities, has intensified demand for aromatic compounds with exceptional purity levels.
Pharmaceutical research laboratories constitute the largest consumer segment for high-purity aromatic compounds. These facilities require materials with purity levels exceeding 99.5% for critical applications including active pharmaceutical ingredient synthesis, impurity profiling studies, and regulatory compliance testing. The stringent quality requirements imposed by regulatory agencies such as FDA and EMA have further amplified demand for validated high-purity materials, as any impurities can significantly impact drug safety and efficacy profiles.
Academic and government research institutions represent another significant demand driver, particularly in fields such as organic chemistry, materials science, and environmental analysis. These organizations increasingly require certified reference materials and high-purity reagents to ensure reproducibility and reliability of research outcomes. The growing emphasis on publication quality and research integrity has led to more stringent internal quality standards, driving adoption of premium-grade aromatic compounds.
The analytical testing and quality control sector has emerged as a rapidly expanding market segment. Contract research organizations, analytical service providers, and in-house quality control laboratories require high-purity aromatic compounds as reference standards for method validation, instrument calibration, and sample analysis. The proliferation of advanced analytical techniques such as ultra-high-performance liquid chromatography and high-resolution mass spectrometry has created demand for increasingly pure reference materials.
Market growth is further supported by expanding applications in emerging fields such as green chemistry, sustainable materials development, and advanced manufacturing processes. These applications often require aromatic compounds with specific purity profiles and minimal environmental impact, creating opportunities for specialized suppliers who can provide comprehensive purity validation documentation and sustainable sourcing practices.
Pharmaceutical research laboratories constitute the largest consumer segment for high-purity aromatic compounds. These facilities require materials with purity levels exceeding 99.5% for critical applications including active pharmaceutical ingredient synthesis, impurity profiling studies, and regulatory compliance testing. The stringent quality requirements imposed by regulatory agencies such as FDA and EMA have further amplified demand for validated high-purity materials, as any impurities can significantly impact drug safety and efficacy profiles.
Academic and government research institutions represent another significant demand driver, particularly in fields such as organic chemistry, materials science, and environmental analysis. These organizations increasingly require certified reference materials and high-purity reagents to ensure reproducibility and reliability of research outcomes. The growing emphasis on publication quality and research integrity has led to more stringent internal quality standards, driving adoption of premium-grade aromatic compounds.
The analytical testing and quality control sector has emerged as a rapidly expanding market segment. Contract research organizations, analytical service providers, and in-house quality control laboratories require high-purity aromatic compounds as reference standards for method validation, instrument calibration, and sample analysis. The proliferation of advanced analytical techniques such as ultra-high-performance liquid chromatography and high-resolution mass spectrometry has created demand for increasingly pure reference materials.
Market growth is further supported by expanding applications in emerging fields such as green chemistry, sustainable materials development, and advanced manufacturing processes. These applications often require aromatic compounds with specific purity profiles and minimal environmental impact, creating opportunities for specialized suppliers who can provide comprehensive purity validation documentation and sustainable sourcing practices.
Current Challenges in Aromatic Compound Purity Analysis
The validation of aromatic compounds' purity in laboratory applications faces numerous analytical challenges that significantly impact research accuracy and reproducibility. Traditional analytical methods often struggle with the complex molecular structures and similar physicochemical properties inherent to aromatic compounds, creating substantial obstacles for precise purity determination.
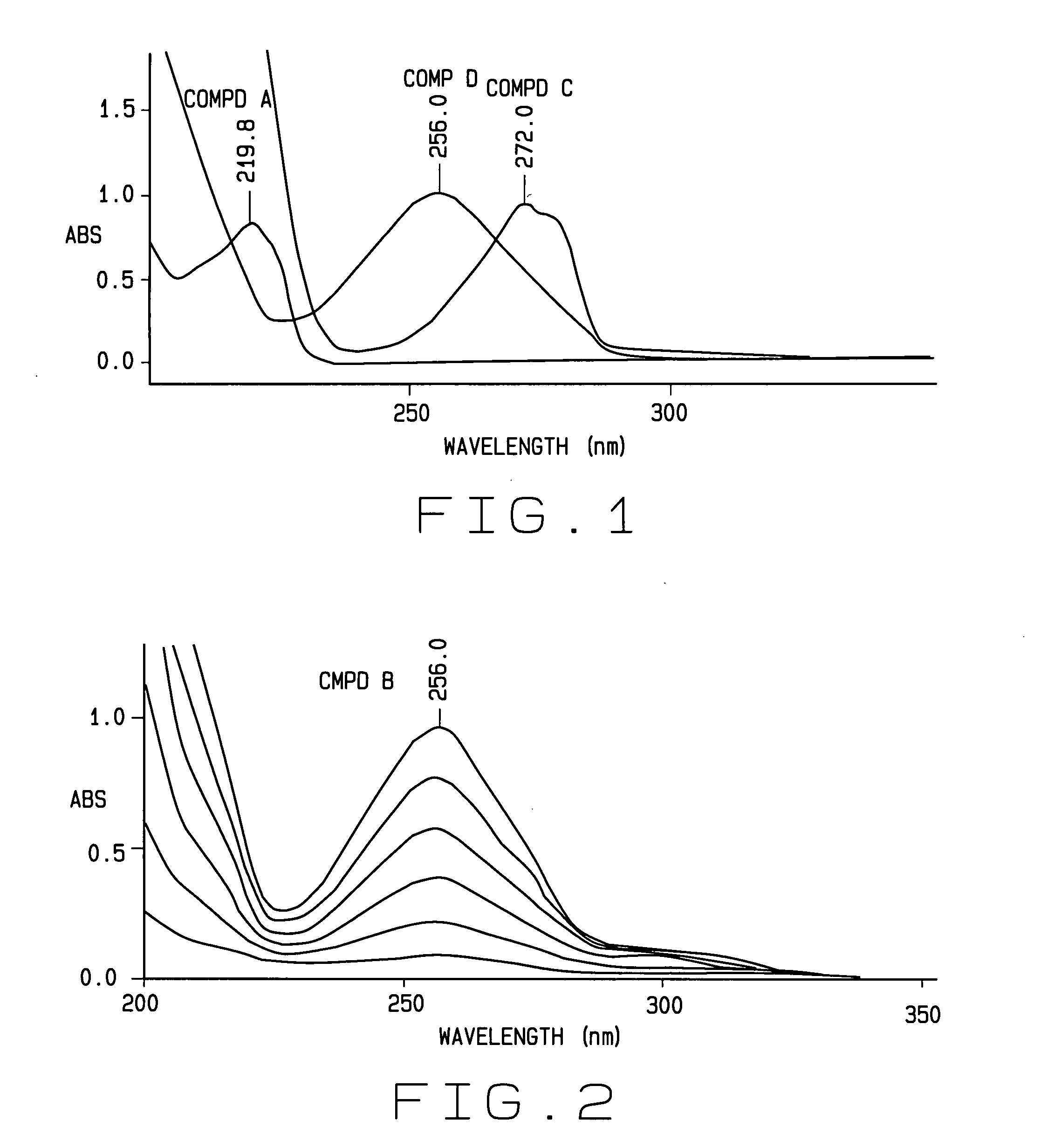
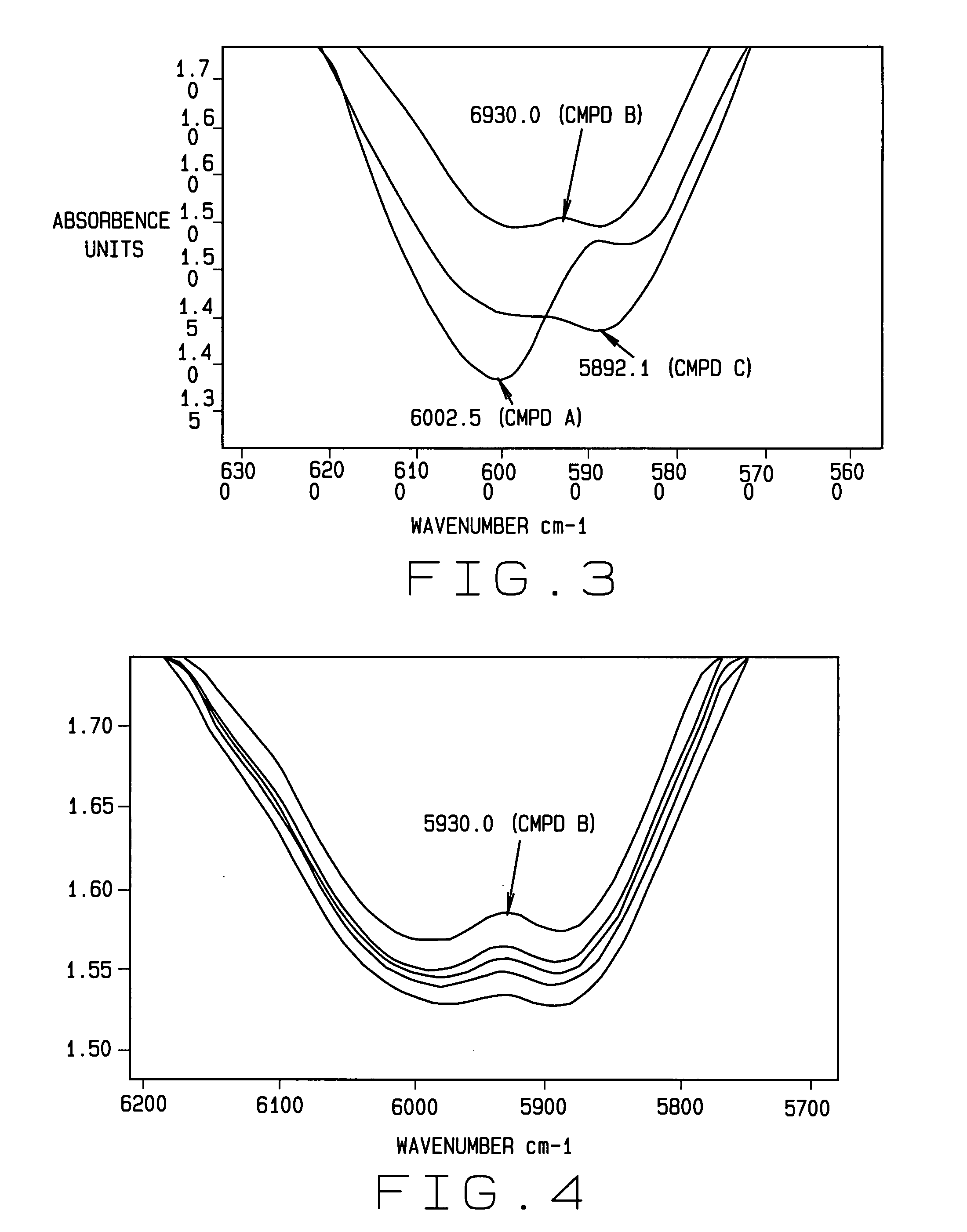
Spectroscopic interference represents a primary challenge in aromatic compound analysis. The overlapping absorption bands and similar spectral signatures of structurally related aromatic compounds frequently lead to misidentification and inaccurate quantification. Nuclear magnetic resonance spectroscopy, while powerful, encounters difficulties when distinguishing between isomeric forms or compounds with minor structural variations, particularly in complex mixtures containing multiple aromatic species.
Chromatographic separation limitations pose another significant hurdle. High-performance liquid chromatography and gas chromatography methods often fail to achieve adequate resolution between closely related aromatic compounds. Co-elution of impurities with target compounds remains a persistent problem, especially when dealing with synthetic aromatic compounds that may contain structurally similar by-products or starting materials.
Sample preparation complexities further compound these analytical challenges. Aromatic compounds frequently exhibit poor solubility in conventional solvents, requiring specialized extraction and dissolution techniques that may introduce additional impurities or cause sample degradation. The volatility of certain aromatic compounds also presents storage and handling difficulties that can compromise sample integrity during analysis.
Matrix effects significantly impact quantitative analysis accuracy. The presence of trace metals, residual solvents, or other organic impurities can interfere with analytical measurements, leading to systematic errors in purity assessment. These matrix interferences are particularly problematic in mass spectrometry applications, where ion suppression or enhancement effects can dramatically alter analytical results.
Detection sensitivity limitations restrict the ability to identify and quantify trace-level impurities that may be critical for specific laboratory applications. Many conventional analytical techniques lack sufficient sensitivity to detect impurities at parts-per-million or parts-per-billion levels, which may be necessary for pharmaceutical or electronic material applications.
Standardization and reference material availability present ongoing challenges for method validation and quality assurance. The lack of certified reference materials for many aromatic compounds makes it difficult to establish reliable calibration standards and validate analytical methods, ultimately affecting the reliability of purity determinations across different laboratories and analytical platforms.
Spectroscopic interference represents a primary challenge in aromatic compound analysis. The overlapping absorption bands and similar spectral signatures of structurally related aromatic compounds frequently lead to misidentification and inaccurate quantification. Nuclear magnetic resonance spectroscopy, while powerful, encounters difficulties when distinguishing between isomeric forms or compounds with minor structural variations, particularly in complex mixtures containing multiple aromatic species.
Chromatographic separation limitations pose another significant hurdle. High-performance liquid chromatography and gas chromatography methods often fail to achieve adequate resolution between closely related aromatic compounds. Co-elution of impurities with target compounds remains a persistent problem, especially when dealing with synthetic aromatic compounds that may contain structurally similar by-products or starting materials.
Sample preparation complexities further compound these analytical challenges. Aromatic compounds frequently exhibit poor solubility in conventional solvents, requiring specialized extraction and dissolution techniques that may introduce additional impurities or cause sample degradation. The volatility of certain aromatic compounds also presents storage and handling difficulties that can compromise sample integrity during analysis.
Matrix effects significantly impact quantitative analysis accuracy. The presence of trace metals, residual solvents, or other organic impurities can interfere with analytical measurements, leading to systematic errors in purity assessment. These matrix interferences are particularly problematic in mass spectrometry applications, where ion suppression or enhancement effects can dramatically alter analytical results.
Detection sensitivity limitations restrict the ability to identify and quantify trace-level impurities that may be critical for specific laboratory applications. Many conventional analytical techniques lack sufficient sensitivity to detect impurities at parts-per-million or parts-per-billion levels, which may be necessary for pharmaceutical or electronic material applications.
Standardization and reference material availability present ongoing challenges for method validation and quality assurance. The lack of certified reference materials for many aromatic compounds makes it difficult to establish reliable calibration standards and validate analytical methods, ultimately affecting the reliability of purity determinations across different laboratories and analytical platforms.
Current Analytical Solutions for Aromatic Purity Validation
01 Purification methods using distillation and crystallization
Various purification techniques including distillation, fractional distillation, and crystallization processes are employed to achieve high purity levels in aromatic compounds. These methods involve separating impurities based on differences in boiling points, vapor pressures, or solubility characteristics. The processes may include multiple stages of distillation or recrystallization to remove trace contaminants and achieve the desired purity specifications for industrial or pharmaceutical applications.- Purification methods using distillation and crystallization: Various purification techniques including distillation, fractional distillation, and crystallization processes are employed to achieve high purity levels in aromatic compounds. These methods involve separating impurities based on differences in boiling points, vapor pressures, or solubility characteristics. The processes may include multiple stages of distillation or recrystallization to progressively remove contaminants and achieve the desired purity specifications for aromatic compounds.
- Adsorption and filtration purification techniques: Adsorption-based purification methods utilize selective adsorbents to remove impurities from aromatic compounds. These techniques may involve passing the aromatic compound through beds of activated carbon, silica gel, or other adsorbent materials that preferentially bind impurities while allowing the desired aromatic compound to pass through. Filtration methods, including membrane filtration and molecular sieves, are also employed to separate impurities based on molecular size or chemical properties.
- Chemical treatment and washing processes: Chemical purification methods involve treating aromatic compounds with specific reagents to remove or neutralize impurities. These processes may include acid washing, alkali treatment, or reaction with selective chemical agents that convert impurities into easily removable forms. Washing procedures with various solvents or aqueous solutions help eliminate residual contaminants and improve the overall purity of the aromatic compounds.
- Analytical methods for purity determination: Advanced analytical techniques are utilized to measure and verify the purity of aromatic compounds. These methods include chromatographic techniques, spectroscopic analysis, and other instrumental approaches that can detect and quantify trace impurities. The analytical procedures establish quality control standards and ensure that aromatic compounds meet specified purity requirements for their intended applications.
- Catalytic and enzymatic purification approaches: Catalytic processes and enzymatic treatments offer selective purification methods for aromatic compounds. These approaches utilize catalysts or enzymes that specifically target certain impurities or facilitate conversion reactions that enhance purity. The methods may involve selective hydrogenation, oxidation, or other catalytic transformations that preferentially affect impurities while preserving the desired aromatic compound structure.
02 Adsorption and chromatographic separation techniques
Adsorption-based purification methods utilizing various adsorbent materials and chromatographic separation techniques are applied to remove specific impurities from aromatic compounds. These methods involve selective binding of impurities to solid adsorbents or separation based on differential migration rates through chromatographic media. The techniques are particularly effective for removing trace organic impurities and achieving ultra-high purity levels required for specialized applications.Expand Specific Solutions03 Chemical treatment and reactive purification
Chemical treatment methods involve the use of specific reagents or catalysts to convert impurities into removable forms or to selectively react with contaminants in aromatic compounds. These reactive purification processes may include oxidation, reduction, or complexation reactions that target specific impurity types. The treated compounds can then be separated through conventional methods, resulting in enhanced purity levels of the final aromatic product.Expand Specific Solutions04 Membrane separation and filtration technologies
Advanced membrane-based separation technologies and specialized filtration systems are utilized to purify aromatic compounds by selectively permeating desired components while retaining impurities. These methods include pervaporation, nanofiltration, and molecular sieve membranes that operate based on size exclusion, solubility differences, or molecular recognition principles. The membrane processes offer energy-efficient alternatives to traditional thermal separation methods for achieving high purity aromatic compounds.Expand Specific Solutions05 Analytical methods for purity determination and quality control
Comprehensive analytical techniques are employed to determine the purity levels and identify trace impurities in aromatic compounds. These methods include spectroscopic analysis, gas chromatography, liquid chromatography, and mass spectrometry techniques that provide quantitative and qualitative assessment of purity. The analytical protocols establish quality control standards and ensure that aromatic compounds meet specified purity requirements for their intended applications in various industries.Expand Specific Solutions
Key Players in Analytical Instrumentation and Chemical Supply
The aromatic compounds purity validation market is experiencing rapid growth driven by increasing regulatory requirements across pharmaceutical, food, and fragrance industries. The industry is in a mature development stage with established analytical methodologies, yet continues evolving with advanced spectroscopic and chromatographic techniques. Market size reflects substantial investment from major players including pharmaceutical companies like Sunshine Lake Pharma and Purdue Pharma, fragrance leaders such as Givaudan SA, Firmenich SA, and Symrise GmbH, alongside food industry giants Mars Inc. and Ajinomoto Co. Technology maturity varies significantly - while traditional validation methods are well-established, companies like Waters Technology Corp. and Aryballe Technologies SA are pioneering next-generation sensor technologies and analytical instruments. Academic institutions including University of California, Beijing Institute of Technology, and University of Coimbra contribute fundamental research, while specialized testing organizations like Zhejiang Zhengming Testing Co. provide comprehensive validation services, creating a competitive landscape spanning from traditional analytical approaches to innovative digital sensing solutions.
Givaudan SA
Technical Solution: Givaudan employs multi-dimensional analytical approaches for aromatic compound purity validation, combining gas chromatography-mass spectrometry (GC-MS) with sensory evaluation techniques. Their proprietary validation protocols integrate quantitative analysis using internal standards with organoleptic assessment to ensure both chemical purity and sensory quality. The company utilizes advanced headspace sampling techniques and solid-phase microextraction (SPME) methods for volatile aromatic compounds, achieving detection accuracies exceeding 99.5% for key flavor and fragrance molecules used in consumer products.
Strengths: Expertise in sensory-chemical correlation and specialized aromatic compound knowledge. Weaknesses: Methods primarily optimized for fragrance applications rather than general laboratory use.
Firmenich SA
Technical Solution: Firmenich utilizes advanced chromatographic techniques combined with spectroscopic methods for aromatic compound purity validation. Their analytical protocols employ two-dimensional gas chromatography (GCxGC) systems with time-of-flight mass spectrometry, enabling comprehensive separation and identification of aromatic impurities and isomers. The company's validation framework includes standardized reference materials and certified analytical procedures that comply with international pharmaceutical and food safety standards, achieving quantification limits in the low ppm range for critical aromatic compounds used in flavor and fragrance applications.
Strengths: Specialized expertise in complex aromatic mixtures and regulatory compliance. Weaknesses: Focus primarily on flavor and fragrance applications may limit broader laboratory applicability.
Advanced Spectroscopic and Chromatographic Innovations
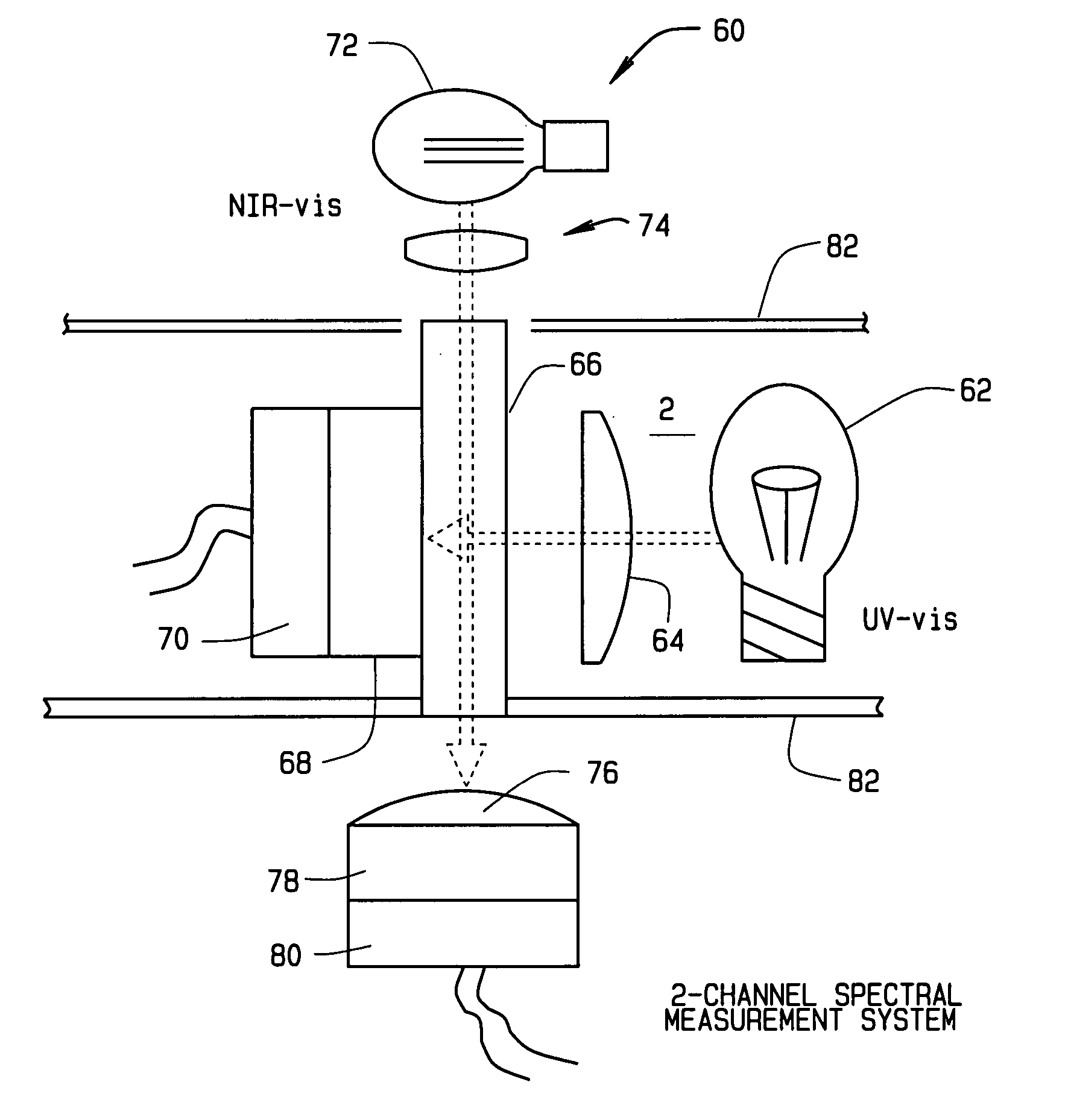
On-site method of providing analysis of potency and purity of pharmaceutical compounds
PatentInactiveUS20070239367A1
Innovation
- A method and system utilizing portable analytical instruments that employ near-infrared (NIR) and ultraviolet-visible (UV-vis) light absorption technologies for on-site analysis of small sample volumes, integrating spectral data with a database for comparison and confirmation of CSPs' purity and potency, enabling real-time verification and quality control.
Reagent kit for LC-ms analysis of aromatic compounds
PatentInactiveUS20220128530A1
Innovation
- A method involving the use of a diazonium reagent for derivatization of aromatic compounds, allowing for azo coupling reactions in aqueous samples, enhancing ionization efficiency in LC-MS analysis without the need for sample drying, and providing a reagent kit for LC-MS analysis that includes a diazonium reagent and quenchers for unreacted diazonium reagent.
Quality Standards and Regulatory Requirements for Lab Chemicals
The validation of aromatic compounds' purity in laboratory applications is governed by a comprehensive framework of quality standards and regulatory requirements that ensure analytical accuracy and safety. These standards establish the foundation for reliable chemical analysis and maintain consistency across different laboratory environments and applications.
International standards organizations play a crucial role in defining purity requirements for laboratory chemicals. The International Organization for Standardization (ISO) provides fundamental guidelines through ISO 17025, which establishes general requirements for testing and calibration laboratories. This standard mandates that laboratories use chemicals of appropriate purity levels and maintain traceability to recognized reference materials. Additionally, ISO Guide 34 specifically addresses the production of reference materials, ensuring that aromatic compound standards meet stringent purity criteria.
The American Chemical Society (ACS) has established reagent-grade specifications that serve as industry benchmarks for chemical purity. ACS reagent-grade aromatic compounds must meet specific purity thresholds, typically exceeding 99% for most applications, with detailed specifications for maximum allowable impurities. These standards define acceptable limits for water content, residue after ignition, and specific contaminants that could interfere with analytical procedures.
Regulatory agencies impose additional requirements that laboratories must comply with when validating aromatic compound purity. The United States Pharmacopeia (USP) provides monographs for pharmaceutical-grade aromatic compounds, establishing identity tests, purity requirements, and analytical methods. Similarly, the European Pharmacopoeia (Ph. Eur.) sets comparable standards for European markets, often requiring more stringent purity criteria for compounds used in pharmaceutical applications.
Good Laboratory Practice (GLP) regulations, enforced by agencies such as the FDA and EPA, mandate specific documentation and validation procedures for chemical purity assessment. These regulations require laboratories to maintain detailed records of chemical procurement, storage conditions, and purity verification results. Laboratories must also implement quality assurance programs that include regular verification of chemical purity through independent analytical methods.
Specialized applications may require adherence to additional regulatory frameworks. For instance, aromatic compounds used in environmental analysis must comply with EPA Method 8000 series requirements, which specify purity criteria for reference standards and internal standards. Similarly, compounds used in food analysis must meet FDA guidelines for analytical grade chemicals, ensuring that impurities do not interfere with trace-level determinations of contaminants or additives.
International standards organizations play a crucial role in defining purity requirements for laboratory chemicals. The International Organization for Standardization (ISO) provides fundamental guidelines through ISO 17025, which establishes general requirements for testing and calibration laboratories. This standard mandates that laboratories use chemicals of appropriate purity levels and maintain traceability to recognized reference materials. Additionally, ISO Guide 34 specifically addresses the production of reference materials, ensuring that aromatic compound standards meet stringent purity criteria.
The American Chemical Society (ACS) has established reagent-grade specifications that serve as industry benchmarks for chemical purity. ACS reagent-grade aromatic compounds must meet specific purity thresholds, typically exceeding 99% for most applications, with detailed specifications for maximum allowable impurities. These standards define acceptable limits for water content, residue after ignition, and specific contaminants that could interfere with analytical procedures.
Regulatory agencies impose additional requirements that laboratories must comply with when validating aromatic compound purity. The United States Pharmacopeia (USP) provides monographs for pharmaceutical-grade aromatic compounds, establishing identity tests, purity requirements, and analytical methods. Similarly, the European Pharmacopoeia (Ph. Eur.) sets comparable standards for European markets, often requiring more stringent purity criteria for compounds used in pharmaceutical applications.
Good Laboratory Practice (GLP) regulations, enforced by agencies such as the FDA and EPA, mandate specific documentation and validation procedures for chemical purity assessment. These regulations require laboratories to maintain detailed records of chemical procurement, storage conditions, and purity verification results. Laboratories must also implement quality assurance programs that include regular verification of chemical purity through independent analytical methods.
Specialized applications may require adherence to additional regulatory frameworks. For instance, aromatic compounds used in environmental analysis must comply with EPA Method 8000 series requirements, which specify purity criteria for reference standards and internal standards. Similarly, compounds used in food analysis must meet FDA guidelines for analytical grade chemicals, ensuring that impurities do not interfere with trace-level determinations of contaminants or additives.
Green Chemistry Approaches in Aromatic Compound Analysis
Green chemistry principles have revolutionized aromatic compound analysis by introducing environmentally sustainable methodologies that minimize hazardous waste generation while maintaining analytical accuracy. Traditional purity validation methods often rely on toxic organic solvents and generate substantial chemical waste, prompting the development of eco-friendly alternatives that align with the twelve principles of green chemistry.
Supercritical fluid chromatography (SFC) has emerged as a leading green analytical technique for aromatic compound purity assessment. This method utilizes supercritical carbon dioxide as the primary mobile phase, significantly reducing organic solvent consumption by up to 90% compared to conventional HPLC methods. SFC demonstrates exceptional separation efficiency for aromatic compounds while offering rapid analysis times and minimal environmental impact through CO2 recycling systems.
Microextraction techniques represent another significant advancement in sustainable aromatic analysis. Solid-phase microextraction (SPME) and dispersive liquid-liquid microextraction (DLLME) require minimal sample volumes and virtually eliminate solvent waste. These methods enable direct extraction and concentration of aromatic compounds from complex matrices, achieving detection limits comparable to traditional extraction methods while reducing reagent consumption by orders of magnitude.
Ionic liquids have gained prominence as green solvents for aromatic compound extraction and purification processes. These designer solvents exhibit negligible vapor pressure, thermal stability, and tunable selectivity properties that make them ideal for aromatic compound isolation. Task-specific ionic liquids can be synthesized to enhance selectivity for specific aromatic structures, improving purity validation accuracy while maintaining environmental compatibility.
Biocatalytic approaches utilizing engineered enzymes offer selective transformation and purification pathways for aromatic compounds. Enzyme-mediated reactions operate under mild conditions, eliminate harsh chemical reagents, and provide high specificity for target compounds. These biotechnological methods enable both purification and real-time purity monitoring through coupled enzymatic assays.
Water-based analytical systems have been developed to replace organic solvents in aromatic compound analysis. Micellar electrokinetic chromatography and aqueous two-phase extraction systems demonstrate effective separation capabilities while utilizing water as the primary medium. These approaches significantly reduce environmental impact and operational costs associated with solvent disposal and safety measures.
Supercritical fluid chromatography (SFC) has emerged as a leading green analytical technique for aromatic compound purity assessment. This method utilizes supercritical carbon dioxide as the primary mobile phase, significantly reducing organic solvent consumption by up to 90% compared to conventional HPLC methods. SFC demonstrates exceptional separation efficiency for aromatic compounds while offering rapid analysis times and minimal environmental impact through CO2 recycling systems.
Microextraction techniques represent another significant advancement in sustainable aromatic analysis. Solid-phase microextraction (SPME) and dispersive liquid-liquid microextraction (DLLME) require minimal sample volumes and virtually eliminate solvent waste. These methods enable direct extraction and concentration of aromatic compounds from complex matrices, achieving detection limits comparable to traditional extraction methods while reducing reagent consumption by orders of magnitude.
Ionic liquids have gained prominence as green solvents for aromatic compound extraction and purification processes. These designer solvents exhibit negligible vapor pressure, thermal stability, and tunable selectivity properties that make them ideal for aromatic compound isolation. Task-specific ionic liquids can be synthesized to enhance selectivity for specific aromatic structures, improving purity validation accuracy while maintaining environmental compatibility.
Biocatalytic approaches utilizing engineered enzymes offer selective transformation and purification pathways for aromatic compounds. Enzyme-mediated reactions operate under mild conditions, eliminate harsh chemical reagents, and provide high specificity for target compounds. These biotechnological methods enable both purification and real-time purity monitoring through coupled enzymatic assays.
Water-based analytical systems have been developed to replace organic solvents in aromatic compound analysis. Micellar electrokinetic chromatography and aqueous two-phase extraction systems demonstrate effective separation capabilities while utilizing water as the primary medium. These approaches significantly reduce environmental impact and operational costs associated with solvent disposal and safety measures.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!