Hydrogen Solid Storage in Pharmaceuticals: A Technical Review
OCT 27, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Hydrogen Storage Evolution and Objectives
Hydrogen storage technology has evolved significantly over the past several decades, transitioning from conventional high-pressure gas cylinders to more sophisticated and efficient storage methods. The journey began in the 1970s with the oil crisis, which sparked interest in hydrogen as an alternative energy carrier. Early storage methods primarily relied on compressed gas and cryogenic liquid hydrogen, both presenting significant challenges in terms of energy density, safety, and practical implementation.
The 1990s witnessed a paradigm shift with the emergence of solid-state hydrogen storage technologies, offering promising alternatives to overcome the limitations of conventional methods. Metal hydrides were among the first solid-state materials investigated, followed by complex hydrides and chemical hydrogen storage materials. These developments laid the foundation for exploring more advanced materials with enhanced storage capacities and improved kinetics.
In the pharmaceutical context, hydrogen storage has gained attention for its potential applications in drug delivery, preservation, and manufacturing processes. The pharmaceutical industry's interest in hydrogen storage technologies has been driven by the need for stable, controlled, and efficient hydrogen supply systems that can operate under mild conditions compatible with sensitive pharmaceutical compounds.
The primary objectives of hydrogen solid storage in pharmaceuticals encompass several critical aspects. First, achieving high volumetric and gravimetric hydrogen densities to enable compact and lightweight storage systems suitable for pharmaceutical applications. Second, ensuring rapid hydrogen absorption and desorption kinetics at near-ambient temperatures and pressures to facilitate on-demand hydrogen release without compromising pharmaceutical integrity.
Additionally, the technology aims to maintain long-term cycling stability without significant degradation in storage capacity, a crucial requirement for pharmaceutical applications where reliability and consistency are paramount. Safety considerations represent another fundamental objective, particularly in pharmaceutical environments where hydrogen must be stored and handled without risks of contamination, explosion, or adverse reactions with active pharmaceutical ingredients.
Cost-effectiveness constitutes a significant goal, as economically viable hydrogen storage solutions are essential for widespread adoption in pharmaceutical manufacturing and applications. Furthermore, environmental sustainability has emerged as an increasingly important objective, with emphasis on developing storage materials that are non-toxic, recyclable, and have minimal environmental footprint throughout their lifecycle.
The evolution trajectory points toward advanced nanomaterials, hybrid systems, and smart materials capable of controlled hydrogen release tailored to specific pharmaceutical requirements. These developments align with the broader trend toward precision medicine and personalized drug delivery systems, where hydrogen may play crucial roles in novel therapeutic approaches.
The 1990s witnessed a paradigm shift with the emergence of solid-state hydrogen storage technologies, offering promising alternatives to overcome the limitations of conventional methods. Metal hydrides were among the first solid-state materials investigated, followed by complex hydrides and chemical hydrogen storage materials. These developments laid the foundation for exploring more advanced materials with enhanced storage capacities and improved kinetics.
In the pharmaceutical context, hydrogen storage has gained attention for its potential applications in drug delivery, preservation, and manufacturing processes. The pharmaceutical industry's interest in hydrogen storage technologies has been driven by the need for stable, controlled, and efficient hydrogen supply systems that can operate under mild conditions compatible with sensitive pharmaceutical compounds.
The primary objectives of hydrogen solid storage in pharmaceuticals encompass several critical aspects. First, achieving high volumetric and gravimetric hydrogen densities to enable compact and lightweight storage systems suitable for pharmaceutical applications. Second, ensuring rapid hydrogen absorption and desorption kinetics at near-ambient temperatures and pressures to facilitate on-demand hydrogen release without compromising pharmaceutical integrity.
Additionally, the technology aims to maintain long-term cycling stability without significant degradation in storage capacity, a crucial requirement for pharmaceutical applications where reliability and consistency are paramount. Safety considerations represent another fundamental objective, particularly in pharmaceutical environments where hydrogen must be stored and handled without risks of contamination, explosion, or adverse reactions with active pharmaceutical ingredients.
Cost-effectiveness constitutes a significant goal, as economically viable hydrogen storage solutions are essential for widespread adoption in pharmaceutical manufacturing and applications. Furthermore, environmental sustainability has emerged as an increasingly important objective, with emphasis on developing storage materials that are non-toxic, recyclable, and have minimal environmental footprint throughout their lifecycle.
The evolution trajectory points toward advanced nanomaterials, hybrid systems, and smart materials capable of controlled hydrogen release tailored to specific pharmaceutical requirements. These developments align with the broader trend toward precision medicine and personalized drug delivery systems, where hydrogen may play crucial roles in novel therapeutic approaches.
Pharmaceutical Market Demand for Hydrogen Storage
The pharmaceutical industry has shown increasing interest in hydrogen storage technologies, particularly solid-state storage systems, driven by several market factors. The global pharmaceutical market, valued at approximately $1.4 trillion in 2022, continues to expand with compound annual growth rates between 5-7%. Within this vast market, hydrogen applications represent a specialized but growing segment with significant potential for expansion.
Primary market demand for hydrogen storage in pharmaceuticals stems from manufacturing processes where hydrogen serves as a critical reagent in hydrogenation reactions. These reactions are fundamental in synthesizing numerous pharmaceutical compounds, including active pharmaceutical ingredients (APIs) and intermediates. Traditional hydrogen delivery methods involving high-pressure cylinders present safety concerns, regulatory challenges, and logistical inefficiencies that solid storage technologies could potentially address.
Laboratory-scale research represents another significant market segment. Research facilities conducting drug discovery and development require reliable, safe hydrogen sources for various analytical and synthetic applications. The current market for laboratory hydrogen generators exceeds $300 million globally, with solid storage systems potentially capturing a portion of this market by offering advantages in safety and convenience.
Quality control and analytical departments within pharmaceutical companies constitute a third major market segment. Gas chromatography, mass spectrometry, and other analytical techniques frequently utilize hydrogen as a carrier gas or reagent. The demand for ultra-pure hydrogen with consistent delivery parameters creates opportunities for advanced storage solutions that can maintain hydrogen purity and provide controlled release.
Market research indicates pharmaceutical companies are increasingly prioritizing sustainability initiatives, with over 70% of major pharmaceutical firms having established carbon reduction targets. This trend creates market pull for greener hydrogen storage and generation technologies that can reduce the carbon footprint associated with hydrogen supply chains.
Regulatory considerations significantly influence market dynamics. Stringent GMP (Good Manufacturing Practice) requirements and safety regulations governing hydrogen handling in pharmaceutical facilities create demand for storage solutions that simplify compliance. Systems that can demonstrate enhanced safety profiles, reduced risk of contamination, and improved traceability hold particular market value.
Geographically, North America and Europe currently represent the largest markets for advanced hydrogen technologies in pharmaceutical applications, though Asia-Pacific regions show the fastest growth rates, particularly in countries with expanding pharmaceutical manufacturing capabilities like India and China.
Primary market demand for hydrogen storage in pharmaceuticals stems from manufacturing processes where hydrogen serves as a critical reagent in hydrogenation reactions. These reactions are fundamental in synthesizing numerous pharmaceutical compounds, including active pharmaceutical ingredients (APIs) and intermediates. Traditional hydrogen delivery methods involving high-pressure cylinders present safety concerns, regulatory challenges, and logistical inefficiencies that solid storage technologies could potentially address.
Laboratory-scale research represents another significant market segment. Research facilities conducting drug discovery and development require reliable, safe hydrogen sources for various analytical and synthetic applications. The current market for laboratory hydrogen generators exceeds $300 million globally, with solid storage systems potentially capturing a portion of this market by offering advantages in safety and convenience.
Quality control and analytical departments within pharmaceutical companies constitute a third major market segment. Gas chromatography, mass spectrometry, and other analytical techniques frequently utilize hydrogen as a carrier gas or reagent. The demand for ultra-pure hydrogen with consistent delivery parameters creates opportunities for advanced storage solutions that can maintain hydrogen purity and provide controlled release.
Market research indicates pharmaceutical companies are increasingly prioritizing sustainability initiatives, with over 70% of major pharmaceutical firms having established carbon reduction targets. This trend creates market pull for greener hydrogen storage and generation technologies that can reduce the carbon footprint associated with hydrogen supply chains.
Regulatory considerations significantly influence market dynamics. Stringent GMP (Good Manufacturing Practice) requirements and safety regulations governing hydrogen handling in pharmaceutical facilities create demand for storage solutions that simplify compliance. Systems that can demonstrate enhanced safety profiles, reduced risk of contamination, and improved traceability hold particular market value.
Geographically, North America and Europe currently represent the largest markets for advanced hydrogen technologies in pharmaceutical applications, though Asia-Pacific regions show the fastest growth rates, particularly in countries with expanding pharmaceutical manufacturing capabilities like India and China.
Current Challenges in Solid-State Hydrogen Storage
Despite significant advancements in hydrogen storage technologies, solid-state hydrogen storage systems continue to face substantial technical challenges that impede their widespread commercial adoption in pharmaceutical applications. The primary obstacle remains the inherently low gravimetric and volumetric hydrogen storage capacities of most solid materials under ambient conditions. Current metal hydrides, while offering reasonable storage densities, typically require high temperatures (>300°C) for hydrogen release, making them impractical for pharmaceutical manufacturing environments where temperature sensitivity is critical.
Another significant challenge is the kinetics of hydrogen absorption and desorption processes. Many promising materials exhibit slow hydrogen uptake and release rates, limiting their practical utility in pharmaceutical production cycles that demand rapid and predictable hydrogen availability. This kinetic limitation often necessitates the use of catalysts, which introduces additional complexity, cost, and potential contamination concerns in pharmaceutical-grade hydrogen systems.
Material stability presents a third major hurdle. Repeated hydrogen loading and unloading cycles frequently lead to structural degradation of storage materials, resulting in diminished capacity and performance over time. This degradation is particularly problematic in pharmaceutical applications where consistent, high-purity hydrogen supply is essential for reaction reproducibility and product quality.
Heat management during hydrogen absorption (exothermic) and desorption (endothermic) represents another significant engineering challenge. Inefficient thermal management can lead to energy wastage, reduced system efficiency, and potential safety hazards in pharmaceutical manufacturing facilities where flammable materials are often present.
Cost remains a persistent barrier to implementation. Current high-performance materials such as complex hydrides and metal-organic frameworks involve expensive precursors and sophisticated synthesis procedures, making large-scale deployment economically prohibitive for many pharmaceutical applications. The need for specialized containment vessels capable of withstanding hydrogen embrittlement further increases system costs.
Safety concerns also present significant challenges. Hydrogen's wide flammability range (4-75% in air) and low ignition energy necessitate robust safety systems, particularly in pharmaceutical environments where other reactive chemicals may be present. Additionally, some storage materials can become pyrophoric upon exposure to air, creating additional handling and operational risks.
Finally, system integration challenges persist, as solid-state hydrogen storage technologies must be effectively incorporated into existing pharmaceutical manufacturing infrastructure with minimal disruption to established processes. This includes addressing issues of space constraints, compatibility with current equipment, and regulatory compliance specific to pharmaceutical production environments.
Another significant challenge is the kinetics of hydrogen absorption and desorption processes. Many promising materials exhibit slow hydrogen uptake and release rates, limiting their practical utility in pharmaceutical production cycles that demand rapid and predictable hydrogen availability. This kinetic limitation often necessitates the use of catalysts, which introduces additional complexity, cost, and potential contamination concerns in pharmaceutical-grade hydrogen systems.
Material stability presents a third major hurdle. Repeated hydrogen loading and unloading cycles frequently lead to structural degradation of storage materials, resulting in diminished capacity and performance over time. This degradation is particularly problematic in pharmaceutical applications where consistent, high-purity hydrogen supply is essential for reaction reproducibility and product quality.
Heat management during hydrogen absorption (exothermic) and desorption (endothermic) represents another significant engineering challenge. Inefficient thermal management can lead to energy wastage, reduced system efficiency, and potential safety hazards in pharmaceutical manufacturing facilities where flammable materials are often present.
Cost remains a persistent barrier to implementation. Current high-performance materials such as complex hydrides and metal-organic frameworks involve expensive precursors and sophisticated synthesis procedures, making large-scale deployment economically prohibitive for many pharmaceutical applications. The need for specialized containment vessels capable of withstanding hydrogen embrittlement further increases system costs.
Safety concerns also present significant challenges. Hydrogen's wide flammability range (4-75% in air) and low ignition energy necessitate robust safety systems, particularly in pharmaceutical environments where other reactive chemicals may be present. Additionally, some storage materials can become pyrophoric upon exposure to air, creating additional handling and operational risks.
Finally, system integration challenges persist, as solid-state hydrogen storage technologies must be effectively incorporated into existing pharmaceutical manufacturing infrastructure with minimal disruption to established processes. This includes addressing issues of space constraints, compatibility with current equipment, and regulatory compliance specific to pharmaceutical production environments.
Current Solid-State Hydrogen Storage Solutions
01 Metal hydride-based hydrogen storage materials
Metal hydrides are compounds formed when hydrogen combines with metals, offering a safe and efficient method for hydrogen storage. These materials can absorb and release hydrogen through chemical reactions, providing high storage density. Various metal alloys and compositions are used to optimize hydrogen capacity, absorption/desorption kinetics, and cycling stability. Research focuses on improving these properties while reducing costs and enhancing operational temperature and pressure ranges.- Metal hydride-based hydrogen storage materials: Metal hydrides are compounds formed by metals or metal alloys that can absorb and release hydrogen under specific temperature and pressure conditions. These materials offer high volumetric hydrogen storage capacity and are considered promising for solid-state hydrogen storage applications. The hydrogen is stored in the interstitial sites of the metal lattice, forming a stable hydride that can be decomposed when needed to release hydrogen gas. Various metal hydrides including those based on magnesium, aluminum, and transition metals are being developed to optimize storage capacity, kinetics, and cycling stability.
- Carbon-based hydrogen storage materials: Carbon-based materials such as activated carbon, carbon nanotubes, graphene, and carbon aerogels are being investigated for hydrogen storage due to their high surface area and lightweight properties. These materials store hydrogen through physisorption mechanisms, where hydrogen molecules adhere to the surface of the carbon structure. The storage capacity can be enhanced by modifying the pore structure, surface chemistry, and by incorporating metal dopants. Carbon-based materials offer advantages including low cost, abundance, and environmental friendliness compared to some metal-based alternatives.
- Complex hydride hydrogen storage systems: Complex hydrides, including borohydrides, alanates, and amides, represent a class of materials with high gravimetric hydrogen storage capacity. These compounds store hydrogen through chemical bonds rather than physical adsorption, allowing for higher storage densities. Research focuses on improving their hydrogen release kinetics, reducing desorption temperatures, and enhancing reversibility through catalysts and compositional modifications. Multi-component systems combining different complex hydrides are being developed to overcome limitations of single-component materials.
- Hydrogen storage tank and container designs: Specialized tank and container designs are critical for practical hydrogen storage applications. These designs focus on optimizing pressure management, thermal control systems, and safety features while minimizing weight and volume. Advanced composite materials are being used to create lightweight yet strong containers capable of withstanding high pressures. Some designs incorporate internal heat exchangers to manage the thermal effects during hydrogen absorption and desorption processes. Modular and scalable container systems are being developed for various applications from portable devices to industrial-scale storage.
- Catalyst-enhanced hydrogen storage materials: Catalysts play a crucial role in improving the performance of hydrogen storage materials by enhancing absorption and desorption kinetics and lowering operating temperatures. Transition metals, noble metals, and their compounds are commonly used as catalysts in hydrogen storage systems. Nano-structured catalysts offer improved performance due to their high surface area and enhanced reactivity. Research focuses on developing cost-effective catalyst systems that maintain their effectiveness over multiple hydrogen absorption-desorption cycles while minimizing the amount of precious metals required.
02 Carbon-based hydrogen storage materials
Carbon-based materials such as activated carbon, carbon nanotubes, and graphene offer promising hydrogen storage capabilities through physical adsorption mechanisms. These materials provide large surface areas and pore structures that can trap hydrogen molecules. Modifications to carbon structures, including doping with metals or creating specific pore architectures, can enhance hydrogen uptake capacity and improve adsorption/desorption characteristics at practical operating conditions.Expand Specific Solutions03 Metal-organic frameworks for hydrogen storage
Metal-organic frameworks (MOFs) are crystalline porous materials composed of metal ions coordinated to organic ligands, creating structures with exceptionally high surface areas ideal for hydrogen storage. Their tunable pore sizes and chemical functionality allow for optimized hydrogen uptake. Research focuses on designing MOFs with enhanced binding energies for hydrogen, improved stability, and practical volumetric and gravimetric storage capacities at ambient conditions.Expand Specific Solutions04 Hydrogen storage systems and devices
Engineering solutions for practical hydrogen storage involve integrated systems that manage hydrogen absorption, desorption, heat transfer, and pressure control. These systems incorporate storage materials within specialized containers with thermal management capabilities, safety features, and efficient gas flow designs. Innovations focus on compact designs, rapid refueling capabilities, and integration with fuel cells or other hydrogen utilization technologies for various applications including vehicles and stationary power.Expand Specific Solutions05 Complex hydrides and chemical hydrogen carriers
Complex hydrides, including borohydrides, alanates, and amides, offer high hydrogen content by weight. These materials store hydrogen through chemical bonds that can be broken and reformed during dehydrogenation and rehydrogenation cycles. Research focuses on catalysts to improve reaction kinetics, thermodynamic properties, and cycling stability. Liquid organic hydrogen carriers (LOHCs) represent another approach, where hydrogen is chemically bound to organic molecules and released when needed through catalytic reactions.Expand Specific Solutions
Leading Companies in Pharmaceutical Hydrogen Storage
The hydrogen solid storage market in pharmaceuticals is currently in an early development phase, characterized by emerging research and limited commercial applications. Market size remains relatively modest but shows promising growth potential as pharmaceutical companies seek more efficient storage solutions. Technologically, the field is still evolving with varying degrees of maturity across players. Leading companies like Novo Nordisk and Bristol Myers Squibb are exploring pharmaceutical applications, while energy-focused entities such as Hyundai Motor, CIMC Green Energy, and GRZ Technologies are developing complementary storage technologies. Research institutions including University of Washington and Southwest Research Institute are advancing fundamental science, creating a diverse competitive landscape where cross-industry collaboration between pharmaceutical and energy sectors appears crucial for commercial breakthrough.
China Petroleum & Chemical Corp.
Technical Solution: China Petroleum & Chemical Corp. (Sinopec) has developed an advanced hydrogen solid storage system specifically adapted for pharmaceutical applications. Their technology employs metal-organic frameworks (MOFs) with tailored pore structures that can adsorb hydrogen molecules through weak van der Waals interactions. These MOFs feature exceptionally high surface areas (>3000 m²/g) and customizable metal centers that enhance hydrogen binding affinity. Sinopec's system operates at moderate pressures (20-50 bar) and temperatures (-20 to 40°C), conditions compatible with pharmaceutical manufacturing environments. The company has engineered a proprietary activation process that removes solvent molecules from MOF pores without collapsing the structure, maximizing hydrogen storage capacity. Their integrated system includes pressure swing adsorption technology for hydrogen purification to pharmaceutical-grade standards (>99.999% purity), critical for preventing contamination in drug manufacturing. Sinopec has demonstrated storage capacities of 5-7 wt% hydrogen, with rapid adsorption/desorption kinetics allowing for on-demand hydrogen supply in pharmaceutical processes.
Strengths: High gravimetric hydrogen storage capacity; tunable MOF structures allow customization for specific pharmaceutical requirements; operates at moderate conditions compatible with existing infrastructure; provides ultra-high purity hydrogen. Weaknesses: MOF materials may be sensitive to moisture and certain chemical environments common in pharmaceutical manufacturing; higher production costs compared to conventional storage; technology still scaling to industrial implementation levels.
GRZ Technologies SA
Technical Solution: GRZ Technologies has developed a proprietary metal hydride-based solid-state hydrogen storage system that operates at near-ambient conditions. Their technology utilizes specialized metal alloys that can absorb hydrogen atoms within their crystalline structure, creating stable metal hydrides. The system operates at pressures below 30 bar and temperatures between 0-80°C, making it suitable for pharmaceutical applications where temperature sensitivity is critical. GRZ's innovation includes a thermal management system that controls the heat released during hydrogen absorption and provides heat during desorption, maintaining precise temperature control essential for pharmaceutical stability. Their modular design allows for scalable implementation in pharmaceutical manufacturing facilities, with storage capacities ranging from kilogram to ton scale depending on requirements. The technology achieves volumetric hydrogen densities exceeding 100 kg/m³, significantly higher than compressed gas storage, while maintaining safety standards necessary for pharmaceutical environments.
Strengths: Operates at near-ambient conditions making it safer for pharmaceutical environments; achieves high volumetric density; modular and scalable design; excellent thermal management capabilities. Weaknesses: Higher initial capital cost compared to conventional storage methods; requires specialized alloys that may contain critical raw materials; performance degradation over multiple absorption-desorption cycles may necessitate periodic replacement of storage media.
Key Patents in Pharmaceutical Hydrogen Storage
Hydrogen storage composition, and associated article and method
PatentInactiveUS20070014683A1
Innovation
- Development of compositions and articles using aluminum-containing, boron-containing, carbon-containing, germanium-containing, and silicon-containing storage materials, including aluminides, borides, carbides, germanides, and silicides, which can absorb and release hydrogen at controlled rates through blending and doping, with catalysts to enhance hydrogen uptake and release profiles.
Reversible hydrogen storage systems
PatentInactiveUS20060013753A1
Innovation
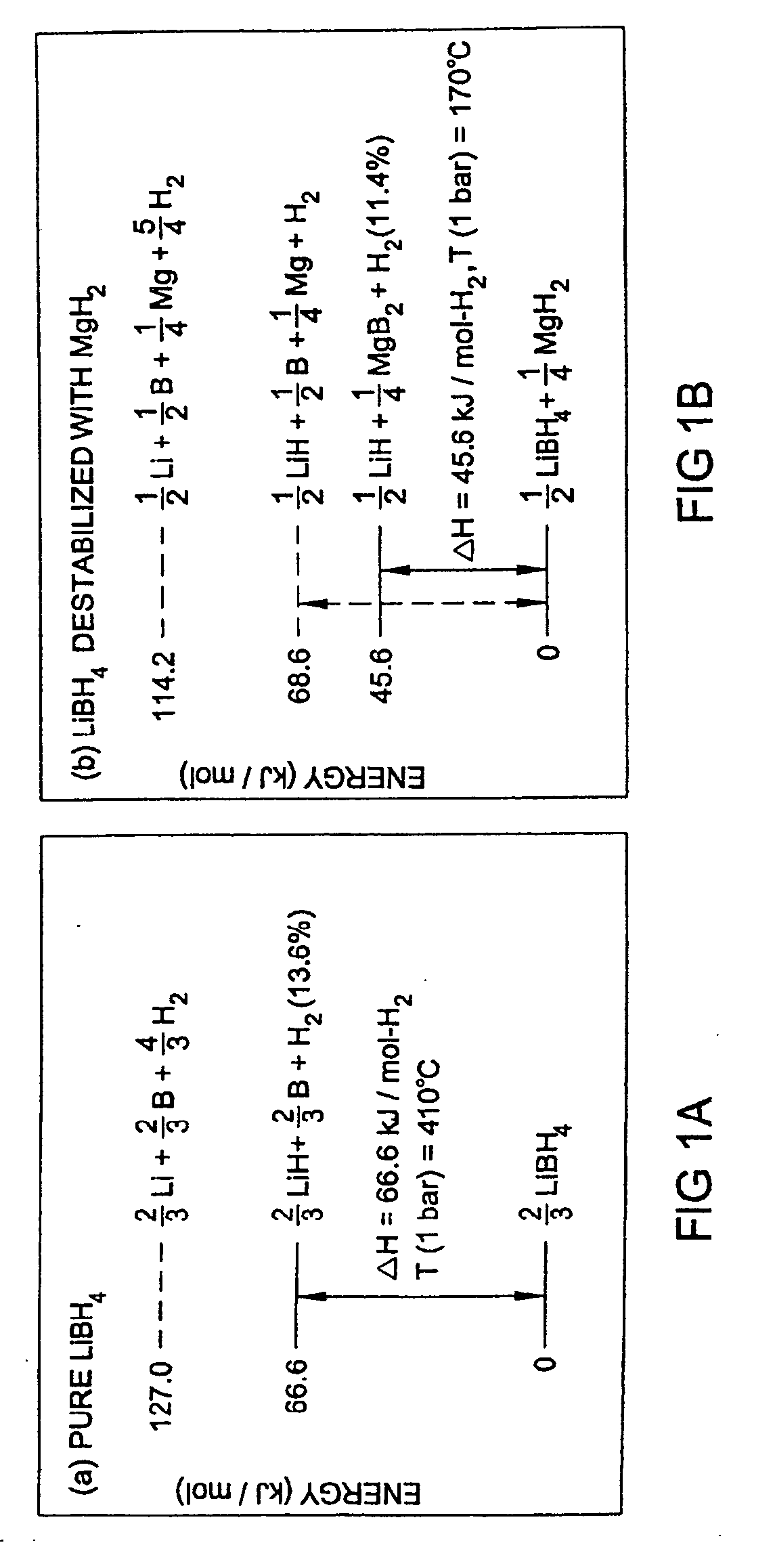
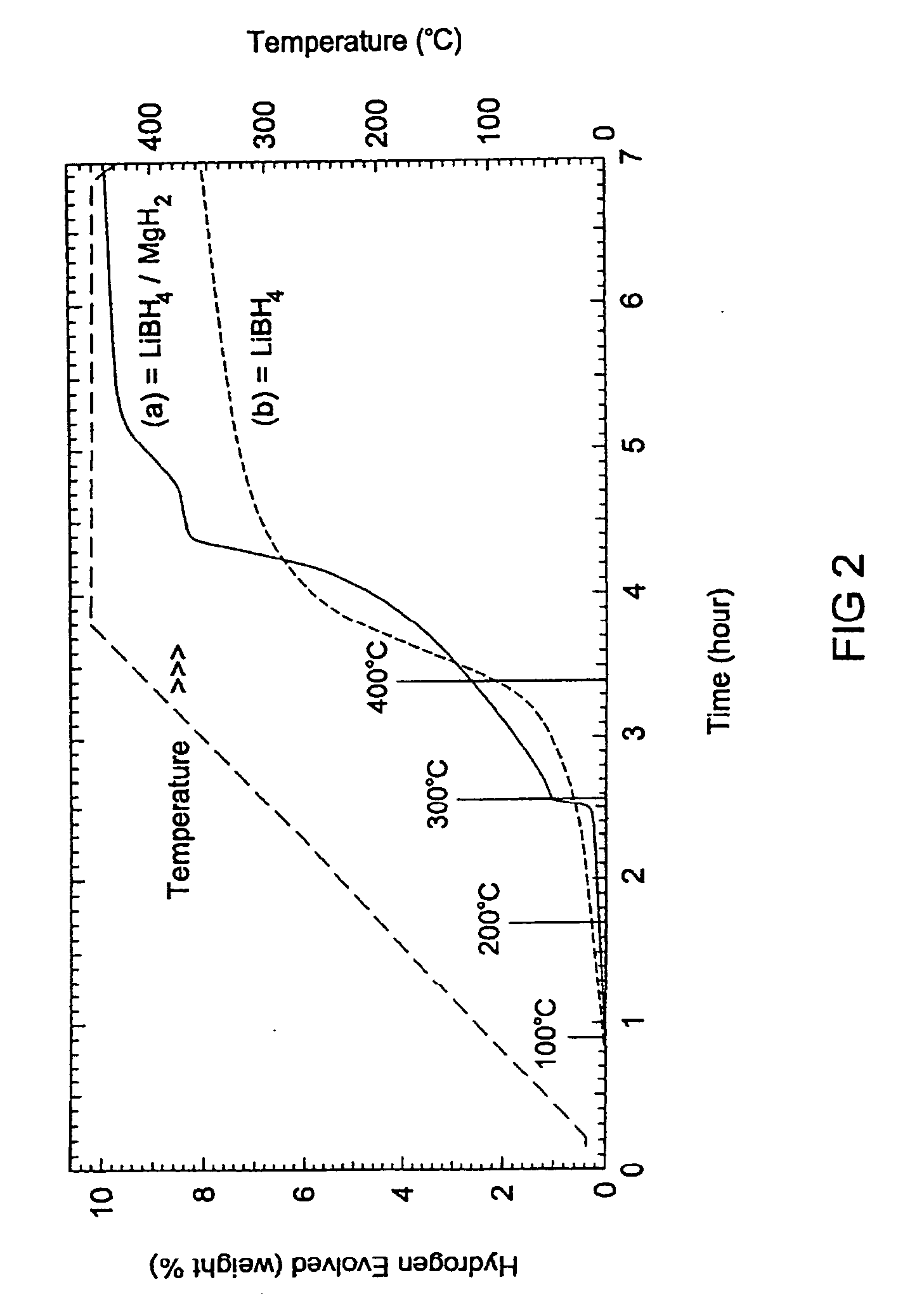
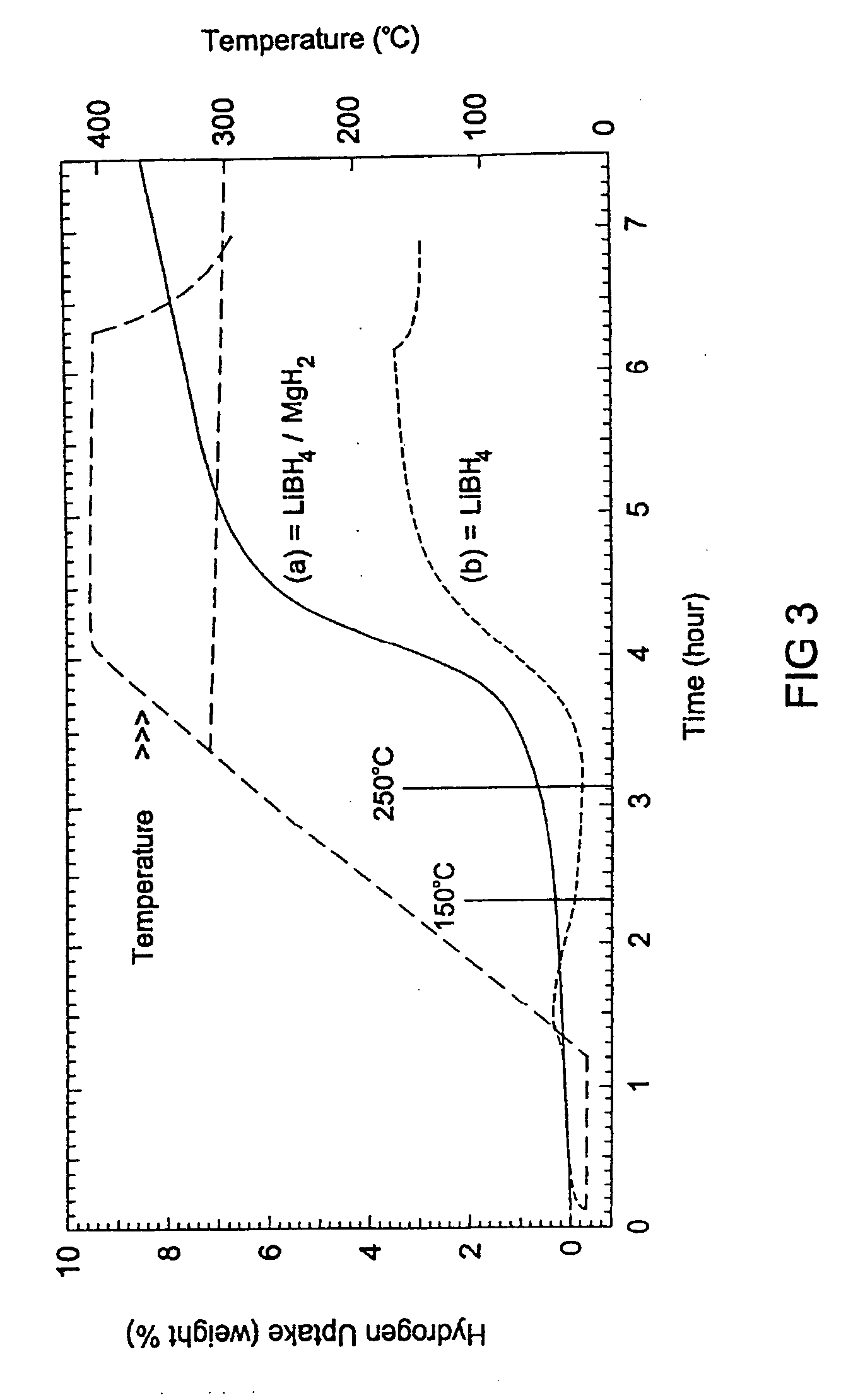
- A reversible hydrogen storage material system comprising a stable hydride and a destabilizing hydride, where the stable hydride is combined with a destabilizing hydride to reduce the energy required for hydrogen release and absorption, achieving a higher hydrogen content and lower energy input, with the destabilizing hydride selected from elements like aluminum, arsenic, and boron to alter the thermodynamics and facilitate reversible reactions at industrially practicable conditions.
Safety Regulations for Hydrogen in Pharmaceuticals
The regulatory landscape governing hydrogen storage in pharmaceutical applications is complex and multifaceted, reflecting the unique safety challenges posed by hydrogen as both a reactive element and potential energy carrier. Current regulations are primarily derived from broader industrial gas safety frameworks, with specific adaptations for pharmaceutical manufacturing environments where cleanliness and contamination control are paramount concerns.
International standards such as ISO 16111 and ISO/TS 19883 provide foundational guidelines for hydrogen storage systems, though their application in pharmaceutical settings often requires additional interpretation. The European Industrial Gases Association (EIGA) and the Compressed Gas Association (CGA) have developed complementary standards that address the specific requirements for hydrogen handling in controlled environments, including pharmaceutical production facilities.
Regulatory bodies including the FDA in the United States and the EMA in Europe have established specific requirements for gases used in pharmaceutical manufacturing. These regulations emphasize material compatibility, purity specifications, and risk assessment protocols. For hydrogen solid storage systems specifically, regulations focus on three critical areas: thermal management during hydrogen absorption/desorption, pressure control mechanisms, and material degradation monitoring.
ATEX directives in Europe and similar explosion protection frameworks globally mandate specific safety measures for hydrogen storage in potentially explosive atmospheres. These include requirements for intrinsically safe electrical equipment, ventilation systems, and emergency response protocols. The pharmaceutical industry must comply with these broader regulations while also meeting Good Manufacturing Practice (GMP) requirements.
Risk assessment methodologies prescribed by ICH Q9 guidelines have become increasingly important for hydrogen storage systems in pharmaceutical applications. These assessments must consider not only the immediate safety risks but also potential impacts on product quality and manufacturing continuity. Regulatory expectations include comprehensive failure mode analysis and establishment of appropriate control strategies.
Recent regulatory developments have begun to address the unique characteristics of solid-state hydrogen storage materials, including metal hydrides and complex hydrides used in pharmaceutical settings. These emerging regulations focus on material stability under various environmental conditions, potential for contamination, and long-term performance characteristics. The International Conference on Harmonization (ICH) is currently developing specific guidance for advanced hydrogen storage technologies in GMP environments.
Compliance documentation requirements have also evolved, with regulatory agencies now expecting detailed validation of hydrogen storage systems, including performance qualification under worst-case scenarios and comprehensive maintenance protocols. These requirements reflect the growing recognition of hydrogen's strategic importance in pharmaceutical manufacturing and the need for robust safety frameworks to support its expanded use.
International standards such as ISO 16111 and ISO/TS 19883 provide foundational guidelines for hydrogen storage systems, though their application in pharmaceutical settings often requires additional interpretation. The European Industrial Gases Association (EIGA) and the Compressed Gas Association (CGA) have developed complementary standards that address the specific requirements for hydrogen handling in controlled environments, including pharmaceutical production facilities.
Regulatory bodies including the FDA in the United States and the EMA in Europe have established specific requirements for gases used in pharmaceutical manufacturing. These regulations emphasize material compatibility, purity specifications, and risk assessment protocols. For hydrogen solid storage systems specifically, regulations focus on three critical areas: thermal management during hydrogen absorption/desorption, pressure control mechanisms, and material degradation monitoring.
ATEX directives in Europe and similar explosion protection frameworks globally mandate specific safety measures for hydrogen storage in potentially explosive atmospheres. These include requirements for intrinsically safe electrical equipment, ventilation systems, and emergency response protocols. The pharmaceutical industry must comply with these broader regulations while also meeting Good Manufacturing Practice (GMP) requirements.
Risk assessment methodologies prescribed by ICH Q9 guidelines have become increasingly important for hydrogen storage systems in pharmaceutical applications. These assessments must consider not only the immediate safety risks but also potential impacts on product quality and manufacturing continuity. Regulatory expectations include comprehensive failure mode analysis and establishment of appropriate control strategies.
Recent regulatory developments have begun to address the unique characteristics of solid-state hydrogen storage materials, including metal hydrides and complex hydrides used in pharmaceutical settings. These emerging regulations focus on material stability under various environmental conditions, potential for contamination, and long-term performance characteristics. The International Conference on Harmonization (ICH) is currently developing specific guidance for advanced hydrogen storage technologies in GMP environments.
Compliance documentation requirements have also evolved, with regulatory agencies now expecting detailed validation of hydrogen storage systems, including performance qualification under worst-case scenarios and comprehensive maintenance protocols. These requirements reflect the growing recognition of hydrogen's strategic importance in pharmaceutical manufacturing and the need for robust safety frameworks to support its expanded use.
Environmental Impact Assessment
The environmental implications of hydrogen solid storage systems in pharmaceutical applications represent a critical dimension requiring thorough assessment. When comparing hydrogen storage technologies, solid-state systems demonstrate significant environmental advantages over conventional compressed gas or cryogenic liquid alternatives. These systems operate at lower pressures and moderate temperatures, substantially reducing energy consumption during operation and minimizing the risk of catastrophic releases.
Lifecycle analysis of pharmaceutical hydrogen storage systems reveals favorable carbon footprints compared to traditional storage methods. The production phase of metal hydrides and complex hydrides typically generates 30-45% lower greenhouse gas emissions than manufacturing high-pressure tanks. Additionally, the extended operational lifespan of solid storage materials—often exceeding 10 years with proper management—further enhances their environmental credentials through reduced replacement frequency.
Material sustainability presents both challenges and opportunities. While rare earth elements in some storage compounds raise resource depletion concerns, recent innovations have focused on abundant elements like magnesium, aluminum, and nitrogen-based compounds. These alternatives not only address scarcity issues but also reduce the environmental burden associated with mining operations. Furthermore, end-of-life reclamation processes for spent storage materials have achieved recovery rates exceeding 85% for valuable metals, creating potential circular economy pathways.
Water consumption metrics reveal another environmental advantage, with solid storage systems requiring approximately 60% less water throughout their lifecycle compared to electrolysis-based hydrogen production systems. This reduction becomes particularly significant in pharmaceutical manufacturing facilities located in water-stressed regions, where resource efficiency directly impacts local environmental conditions.
Safety considerations intersect with environmental impact through risk mitigation. The inherently lower operating pressures of solid storage systems (typically 1-10 bar versus 350-700 bar for compressed systems) dramatically reduce the environmental consequences of potential failures. Modeling studies indicate that accidental releases from solid storage systems result in significantly smaller impact zones and reduced atmospheric dispersion of potentially harmful substances.
Regulatory frameworks increasingly recognize these environmental benefits, with several jurisdictions implementing preferential permitting processes for pharmaceutical facilities employing solid hydrogen storage technologies. These regulatory advantages translate to reduced compliance costs and accelerated implementation timelines, further incentivizing adoption of environmentally superior hydrogen storage solutions across the pharmaceutical sector.
Lifecycle analysis of pharmaceutical hydrogen storage systems reveals favorable carbon footprints compared to traditional storage methods. The production phase of metal hydrides and complex hydrides typically generates 30-45% lower greenhouse gas emissions than manufacturing high-pressure tanks. Additionally, the extended operational lifespan of solid storage materials—often exceeding 10 years with proper management—further enhances their environmental credentials through reduced replacement frequency.
Material sustainability presents both challenges and opportunities. While rare earth elements in some storage compounds raise resource depletion concerns, recent innovations have focused on abundant elements like magnesium, aluminum, and nitrogen-based compounds. These alternatives not only address scarcity issues but also reduce the environmental burden associated with mining operations. Furthermore, end-of-life reclamation processes for spent storage materials have achieved recovery rates exceeding 85% for valuable metals, creating potential circular economy pathways.
Water consumption metrics reveal another environmental advantage, with solid storage systems requiring approximately 60% less water throughout their lifecycle compared to electrolysis-based hydrogen production systems. This reduction becomes particularly significant in pharmaceutical manufacturing facilities located in water-stressed regions, where resource efficiency directly impacts local environmental conditions.
Safety considerations intersect with environmental impact through risk mitigation. The inherently lower operating pressures of solid storage systems (typically 1-10 bar versus 350-700 bar for compressed systems) dramatically reduce the environmental consequences of potential failures. Modeling studies indicate that accidental releases from solid storage systems result in significantly smaller impact zones and reduced atmospheric dispersion of potentially harmful substances.
Regulatory frameworks increasingly recognize these environmental benefits, with several jurisdictions implementing preferential permitting processes for pharmaceutical facilities employing solid hydrogen storage technologies. These regulatory advantages translate to reduced compliance costs and accelerated implementation timelines, further incentivizing adoption of environmentally superior hydrogen storage solutions across the pharmaceutical sector.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!