Ion Selective Electrode in Biotechnology: Performance Verification
MAR 8, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
ISE Biotechnology Background and Performance Goals
Ion selective electrodes have emerged as pivotal analytical tools in biotechnology applications, representing a convergence of electrochemical sensing principles with biological system requirements. These electrochemical sensors utilize selective membrane technologies to detect specific ionic species in complex biological matrices, offering real-time monitoring capabilities essential for modern biotechnological processes. The fundamental principle relies on the development of a measurable potential difference across a selective membrane when exposed to target ions, following the Nernst equation relationship.
The biotechnology sector has witnessed exponential growth in demand for precise ionic monitoring across diverse applications including cell culture optimization, fermentation process control, and bioprocess validation. Traditional analytical methods often require sample preparation, laboratory analysis, and significant time delays, creating bottlenecks in time-sensitive biotechnological operations. Ion selective electrodes address these limitations by providing continuous, in-situ monitoring capabilities that align with the industry's shift toward real-time process analytics and Quality by Design principles.
Current biotechnology applications span multiple domains, from pharmaceutical manufacturing where precise pH and ionic strength control ensures product quality, to environmental biotechnology where nutrient monitoring optimizes bioremediation processes. Cell culture applications particularly benefit from ISE technology, as maintaining optimal ionic concentrations directly impacts cell viability, growth rates, and product expression levels. The technology has evolved from simple glass pH electrodes to sophisticated multi-ion sensors capable of simultaneous detection of critical parameters.
The primary performance goals for ISE implementation in biotechnology center on achieving analytical reliability comparable to traditional laboratory methods while maintaining the advantages of continuous monitoring. Selectivity coefficients must demonstrate minimal interference from competing ions commonly present in biological systems, including proteins, metabolites, and buffer components. Response time requirements typically demand sub-minute equilibration to support real-time process control decisions.
Stability and drift characteristics represent critical performance parameters, as biotechnological processes often extend over days or weeks. Long-term stability goals target drift rates below 1% per day for critical applications, with calibration intervals extending beyond 72 hours to minimize process interruptions. Temperature compensation mechanisms must function effectively across the typical bioprocess range of 25-40°C, ensuring measurement accuracy regardless of thermal fluctuations.
Detection limits and linear response ranges must accommodate the diverse ionic concentrations encountered in biotechnology applications, from millimolar levels in culture media to micromolar concentrations in purified products. Biocompatibility requirements demand materials that resist fouling from biological components while maintaining measurement integrity throughout extended exposure periods.
The biotechnology sector has witnessed exponential growth in demand for precise ionic monitoring across diverse applications including cell culture optimization, fermentation process control, and bioprocess validation. Traditional analytical methods often require sample preparation, laboratory analysis, and significant time delays, creating bottlenecks in time-sensitive biotechnological operations. Ion selective electrodes address these limitations by providing continuous, in-situ monitoring capabilities that align with the industry's shift toward real-time process analytics and Quality by Design principles.
Current biotechnology applications span multiple domains, from pharmaceutical manufacturing where precise pH and ionic strength control ensures product quality, to environmental biotechnology where nutrient monitoring optimizes bioremediation processes. Cell culture applications particularly benefit from ISE technology, as maintaining optimal ionic concentrations directly impacts cell viability, growth rates, and product expression levels. The technology has evolved from simple glass pH electrodes to sophisticated multi-ion sensors capable of simultaneous detection of critical parameters.
The primary performance goals for ISE implementation in biotechnology center on achieving analytical reliability comparable to traditional laboratory methods while maintaining the advantages of continuous monitoring. Selectivity coefficients must demonstrate minimal interference from competing ions commonly present in biological systems, including proteins, metabolites, and buffer components. Response time requirements typically demand sub-minute equilibration to support real-time process control decisions.
Stability and drift characteristics represent critical performance parameters, as biotechnological processes often extend over days or weeks. Long-term stability goals target drift rates below 1% per day for critical applications, with calibration intervals extending beyond 72 hours to minimize process interruptions. Temperature compensation mechanisms must function effectively across the typical bioprocess range of 25-40°C, ensuring measurement accuracy regardless of thermal fluctuations.
Detection limits and linear response ranges must accommodate the diverse ionic concentrations encountered in biotechnology applications, from millimolar levels in culture media to micromolar concentrations in purified products. Biocompatibility requirements demand materials that resist fouling from biological components while maintaining measurement integrity throughout extended exposure periods.
Market Demand for ISE-Based Biotech Applications
The biotechnology sector demonstrates substantial demand for ion selective electrode applications across multiple domains, driven by the critical need for precise, real-time monitoring of ionic species in biological systems. Clinical diagnostics represents the largest market segment, where ISE technology enables rapid measurement of electrolytes including sodium, potassium, chloride, and calcium in blood, serum, and urine samples. Point-of-care testing devices incorporating ISE sensors have gained significant traction due to their ability to deliver immediate results in emergency departments and intensive care units.
Pharmaceutical manufacturing constitutes another major demand driver, particularly in bioprocess monitoring and quality control applications. ISE-based systems provide continuous monitoring of pH, dissolved oxygen, and specific ion concentrations during fermentation processes, cell culture operations, and downstream purification steps. The growing biopharmaceutical industry, especially in monoclonal antibody and vaccine production, requires robust analytical tools capable of maintaining product quality and regulatory compliance.
Environmental biotechnology applications present emerging opportunities for ISE technology deployment. Water treatment facilities utilize ISE sensors for monitoring nutrient levels, heavy metal contamination, and treatment efficacy in biological remediation processes. Agricultural biotechnology applications include soil analysis systems that measure nutrient availability and pH levels to optimize crop yields and reduce environmental impact.
Food and beverage industries increasingly adopt ISE-based solutions for quality assurance and safety monitoring. These applications encompass measuring salt content, acidity levels, and preservative concentrations in processed foods, as well as monitoring fermentation parameters in brewing and dairy operations. The demand for rapid, on-site testing capabilities drives adoption of portable ISE devices in food processing facilities.
Research and academic institutions represent a steady demand source for specialized ISE applications in biotechnology research. These include studying cellular metabolism, investigating ion transport mechanisms, and developing novel biosensors. The expanding field of personalized medicine creates additional demand for miniaturized ISE devices capable of monitoring biomarkers in small sample volumes.
Market growth factors include increasing healthcare expenditure, stringent regulatory requirements for pharmaceutical manufacturing, and growing awareness of environmental monitoring needs. The shift toward automation and continuous monitoring in biotechnology processes further accelerates ISE adoption, as these sensors provide reliable, cost-effective solutions for maintaining process control and product quality standards.
Pharmaceutical manufacturing constitutes another major demand driver, particularly in bioprocess monitoring and quality control applications. ISE-based systems provide continuous monitoring of pH, dissolved oxygen, and specific ion concentrations during fermentation processes, cell culture operations, and downstream purification steps. The growing biopharmaceutical industry, especially in monoclonal antibody and vaccine production, requires robust analytical tools capable of maintaining product quality and regulatory compliance.
Environmental biotechnology applications present emerging opportunities for ISE technology deployment. Water treatment facilities utilize ISE sensors for monitoring nutrient levels, heavy metal contamination, and treatment efficacy in biological remediation processes. Agricultural biotechnology applications include soil analysis systems that measure nutrient availability and pH levels to optimize crop yields and reduce environmental impact.
Food and beverage industries increasingly adopt ISE-based solutions for quality assurance and safety monitoring. These applications encompass measuring salt content, acidity levels, and preservative concentrations in processed foods, as well as monitoring fermentation parameters in brewing and dairy operations. The demand for rapid, on-site testing capabilities drives adoption of portable ISE devices in food processing facilities.
Research and academic institutions represent a steady demand source for specialized ISE applications in biotechnology research. These include studying cellular metabolism, investigating ion transport mechanisms, and developing novel biosensors. The expanding field of personalized medicine creates additional demand for miniaturized ISE devices capable of monitoring biomarkers in small sample volumes.
Market growth factors include increasing healthcare expenditure, stringent regulatory requirements for pharmaceutical manufacturing, and growing awareness of environmental monitoring needs. The shift toward automation and continuous monitoring in biotechnology processes further accelerates ISE adoption, as these sensors provide reliable, cost-effective solutions for maintaining process control and product quality standards.
Current ISE Performance Challenges in Biotech
Ion selective electrodes face significant performance challenges when deployed in biotechnology applications, primarily due to the complex nature of biological matrices. The presence of interfering ions, proteins, and other biomolecules creates a hostile environment that compromises electrode selectivity and accuracy. Traditional ISEs designed for simple aqueous solutions often exhibit poor performance when exposed to blood, serum, cell culture media, or fermentation broths.
Drift and stability issues represent another critical challenge in biotech applications. ISEs typically experience baseline drift over time, which becomes particularly problematic during long-term bioprocessing operations or continuous monitoring scenarios. Temperature fluctuations, pH variations, and the accumulation of biological fouling on electrode surfaces exacerbate these stability problems, leading to measurement uncertainties that can compromise process control and product quality.
Membrane fouling constitutes a major technical obstacle that significantly impacts ISE performance in biological systems. Protein adsorption, cell adhesion, and biofilm formation on electrode surfaces create barriers that impede ion transport and alter the electrode's response characteristics. This fouling phenomenon is particularly severe in applications involving whole blood analysis, microbial fermentation monitoring, and tissue engineering environments where high concentrations of organic compounds are present.
Calibration complexity poses substantial operational challenges in biotechnology settings. The dynamic nature of biological systems requires frequent recalibration to maintain measurement accuracy, but standard calibration procedures often prove inadequate for complex biological matrices. Matrix effects can cause significant deviations from expected electrode behavior, making it difficult to establish reliable calibration curves that remain valid throughout extended measurement periods.
Response time limitations become critical constraints in biotech applications requiring real-time monitoring and rapid process adjustments. Many ISEs exhibit sluggish response kinetics when measuring low concentrations of target ions in viscous biological media. This delayed response can compromise the effectiveness of automated control systems and limit the ability to detect rapid changes in bioprocess conditions.
Temperature sensitivity represents an additional challenge, as biological systems often operate at elevated temperatures that can affect electrode performance. The temperature coefficients of ISE responses may vary significantly in biological matrices compared to standard solutions, requiring sophisticated temperature compensation algorithms that are not always available in commercial instrumentation.
Drift and stability issues represent another critical challenge in biotech applications. ISEs typically experience baseline drift over time, which becomes particularly problematic during long-term bioprocessing operations or continuous monitoring scenarios. Temperature fluctuations, pH variations, and the accumulation of biological fouling on electrode surfaces exacerbate these stability problems, leading to measurement uncertainties that can compromise process control and product quality.
Membrane fouling constitutes a major technical obstacle that significantly impacts ISE performance in biological systems. Protein adsorption, cell adhesion, and biofilm formation on electrode surfaces create barriers that impede ion transport and alter the electrode's response characteristics. This fouling phenomenon is particularly severe in applications involving whole blood analysis, microbial fermentation monitoring, and tissue engineering environments where high concentrations of organic compounds are present.
Calibration complexity poses substantial operational challenges in biotechnology settings. The dynamic nature of biological systems requires frequent recalibration to maintain measurement accuracy, but standard calibration procedures often prove inadequate for complex biological matrices. Matrix effects can cause significant deviations from expected electrode behavior, making it difficult to establish reliable calibration curves that remain valid throughout extended measurement periods.
Response time limitations become critical constraints in biotech applications requiring real-time monitoring and rapid process adjustments. Many ISEs exhibit sluggish response kinetics when measuring low concentrations of target ions in viscous biological media. This delayed response can compromise the effectiveness of automated control systems and limit the ability to detect rapid changes in bioprocess conditions.
Temperature sensitivity represents an additional challenge, as biological systems often operate at elevated temperatures that can affect electrode performance. The temperature coefficients of ISE responses may vary significantly in biological matrices compared to standard solutions, requiring sophisticated temperature compensation algorithms that are not always available in commercial instrumentation.
Current ISE Performance Verification Methods
01 Ion-selective membrane composition and materials
The performance of ion-selective electrodes is significantly influenced by the composition of the ion-selective membrane. Various materials including polymeric matrices, plasticizers, and ionophores are used to create membranes with specific selectivity characteristics. The choice of membrane materials affects the electrode's sensitivity, selectivity coefficient, and response time. Optimization of membrane composition involves balancing hydrophobicity, ion exchange capacity, and mechanical stability to achieve desired analytical performance.- Ion-selective membrane composition and materials: The performance of ion-selective electrodes is significantly influenced by the composition of the ion-selective membrane. Various materials including polymeric matrices, plasticizers, and ionophores are used to create membranes with specific selectivity characteristics. The choice of membrane materials affects the electrode's sensitivity, selectivity coefficient, and response time. Optimization of membrane composition involves balancing the hydrophobicity, ion exchange capacity, and mechanical stability of the membrane to achieve desired analytical performance.
- Reference electrode design and integration: The reference electrode component is critical for maintaining stable potential measurements in ion-selective electrode systems. Various designs incorporate different reference systems including silver-silver chloride, calomel, or solid-state reference elements. The integration method between the ion-selective membrane and reference electrode affects the overall electrode stability, drift characteristics, and lifetime. Advanced designs focus on miniaturization and compatibility with different sample matrices while maintaining consistent reference potential.
- Signal conditioning and measurement electronics: Electronic circuitry for signal processing and conditioning plays a vital role in ion-selective electrode performance. High-impedance input amplifiers, noise reduction circuits, and temperature compensation systems are employed to improve measurement accuracy and stability. Digital signal processing techniques enable real-time calibration, drift correction, and multi-ion detection capabilities. The integration of microprocessors and advanced electronics allows for automated measurements and enhanced analytical capabilities.
- Electrode surface modification and conditioning: Surface treatment and conditioning procedures are essential for optimizing ion-selective electrode performance. Various modification techniques including chemical treatment, plasma processing, or coating application are used to enhance membrane adhesion, reduce fouling, and improve response characteristics. Conditioning protocols involving pre-treatment solutions and activation procedures help establish stable baseline potentials and consistent sensitivity. These surface modifications can significantly extend electrode lifetime and maintain performance in challenging sample matrices.
- Calibration methods and performance validation: Accurate calibration procedures and performance validation protocols are fundamental to ensuring reliable ion-selective electrode measurements. Multi-point calibration using standard solutions of known ionic strength establishes the electrode response curve and determines key performance parameters such as slope, detection limit, and linear range. Quality control measures include regular verification of selectivity coefficients, response time assessment, and interference testing. Advanced calibration algorithms account for temperature effects, ionic strength variations, and matrix effects to improve measurement accuracy across diverse applications.
02 Internal reference electrode and electrolyte systems
The internal reference system plays a crucial role in maintaining stable potential and ensuring reliable measurements. Various configurations of internal reference electrodes and electrolyte solutions are employed to minimize drift and improve long-term stability. The design includes considerations for electrolyte composition, concentration, and the interface between the internal solution and the ion-selective membrane. Advanced designs incorporate solid-state or gel-based internal contacts to enhance durability and reduce maintenance requirements.Expand Specific Solutions03 Electrode response characteristics and calibration methods
The analytical performance of ion-selective electrodes is characterized by parameters such as detection limit, linear range, response slope, and selectivity. Calibration procedures and measurement protocols are critical for achieving accurate and reproducible results. Various techniques are employed to optimize response time, reduce interference from competing ions, and extend the operational lifetime. Performance evaluation includes assessment of Nernstian behavior, potential stability, and resistance to fouling under different sample conditions.Expand Specific Solutions04 Miniaturization and solid-state electrode designs
Advances in electrode miniaturization have led to the development of compact, portable, and integrated sensing devices. Solid-state electrode configurations eliminate the need for internal filling solutions, improving robustness and enabling mass production. Microfabrication techniques allow for the creation of electrode arrays and multi-ion sensors with reduced sample volume requirements. These designs are particularly suitable for point-of-care testing, environmental monitoring, and process control applications where conventional electrodes are impractical.Expand Specific Solutions05 Interference reduction and selectivity enhancement
Improving selectivity against interfering ions is essential for accurate measurements in complex sample matrices. Various strategies are employed including the use of selective ionophores, optimization of membrane composition, and implementation of correction algorithms. Surface modification techniques and protective layers can reduce biofouling and extend electrode lifetime in challenging environments. Advanced signal processing methods and multi-sensor arrays with pattern recognition algorithms enable discrimination between target and interfering species, enhancing overall analytical performance.Expand Specific Solutions
Key Players in ISE Biotechnology Market
The ion selective electrode (ISE) biotechnology sector represents a mature yet evolving market experiencing steady growth driven by increasing demand for precise bioanalytical measurements in healthcare and research applications. The industry demonstrates strong technological maturity, evidenced by established players like Beckman Coulter, Roche Diagnostics, and Radiometer who have developed sophisticated ISE-based diagnostic platforms with proven clinical validation. Market dynamics show consolidation among major diagnostic companies while specialized firms like Unisense, Oxford Biosensors, and Kalium Health drive innovation in niche applications. The competitive landscape spans from multinational corporations offering comprehensive diagnostic solutions to focused technology developers creating specialized ISE systems. Research institutions including University of Michigan and Chung Yuan Christian University contribute to advancing electrode materials and sensing mechanisms. The sector benefits from robust regulatory frameworks and established manufacturing capabilities, positioning it for continued expansion as personalized medicine and point-of-care testing demands increase globally.
Beckman Coulter, Inc.
Technical Solution: Beckman Coulter has developed comprehensive ISE solutions for clinical chemistry analyzers, focusing on high-throughput laboratory environments. Their ion selective electrode technology employs polymer membrane sensors with optimized ionophore compositions for enhanced selectivity and stability. The system integrates automated sample handling with multi-ion detection capabilities, enabling simultaneous measurement of multiple electrolytes from small sample volumes. Their ISE modules feature advanced signal processing algorithms to minimize noise and improve measurement precision. The technology includes automated electrode conditioning and maintenance protocols to extend sensor lifetime and reduce operational costs in busy laboratory settings.
Strengths: High-throughput capabilities, excellent automation integration, proven reliability in clinical laboratories. Weaknesses: Limited flexibility for non-standard applications, requires specialized training for operation and maintenance.
F. Hoffmann-La Roche Ltd.
Technical Solution: Roche has developed advanced ion selective electrode systems integrated into their blood gas analyzers and point-of-care testing devices. Their ISE technology focuses on measuring critical electrolytes including sodium, potassium, chloride, and calcium ions in biological samples. The company's ISE sensors utilize solid-state membrane technology with enhanced selectivity coefficients and reduced interference from proteins and other biological matrices. Their systems incorporate automated calibration protocols and real-time quality control measures to ensure accurate measurements in clinical environments. Roche's ISE technology features temperature compensation algorithms and drift correction mechanisms to maintain measurement precision over extended operational periods.
Strengths: Market-leading accuracy and reliability in clinical diagnostics, extensive regulatory approvals, robust quality control systems. Weaknesses: High cost of implementation, complex maintenance requirements, limited customization for specialized research applications.
Core ISE Performance Enhancement Patents
Ion-selective electrodes
PatentInactiveUS20050191428A1
Innovation
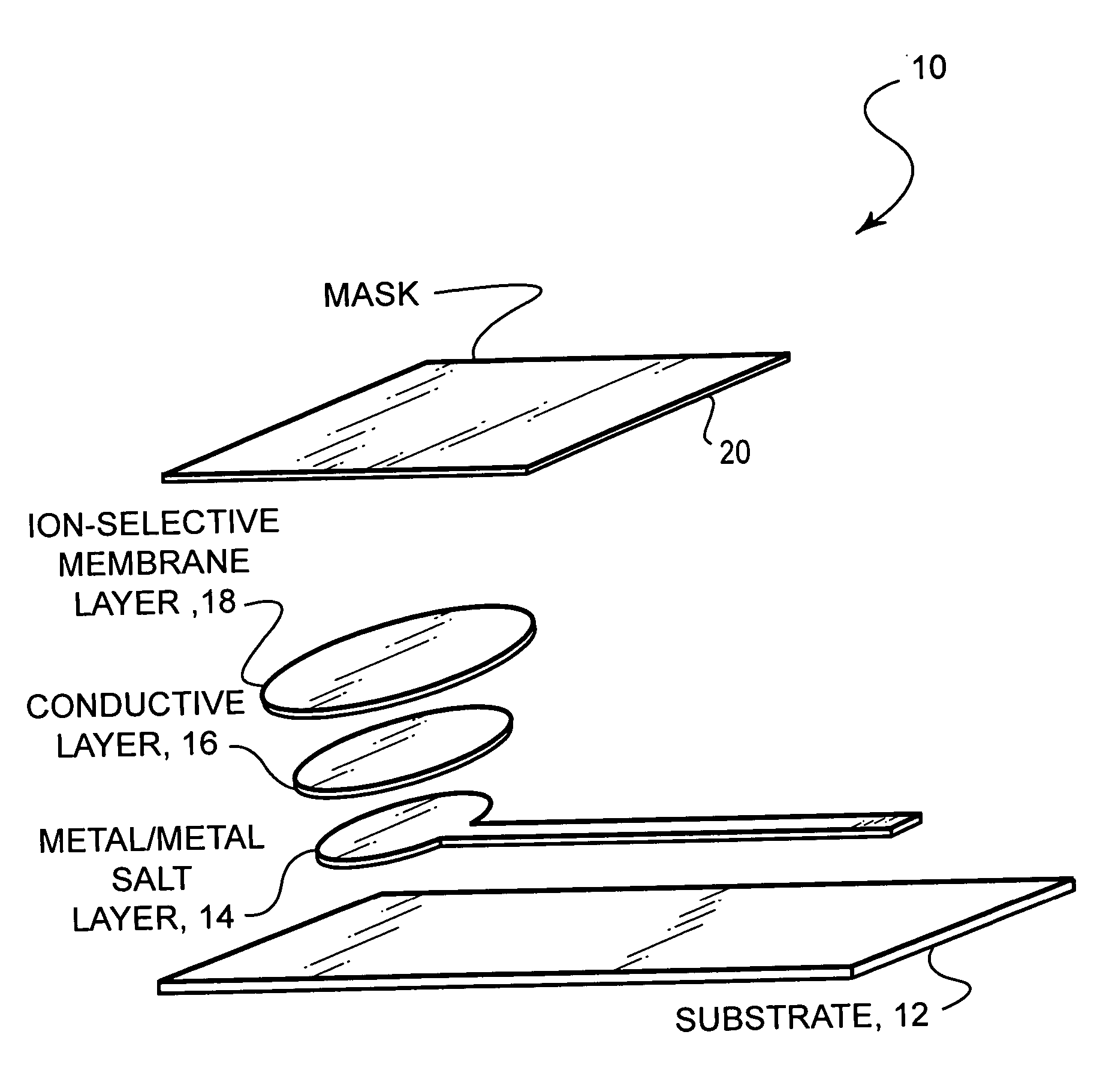
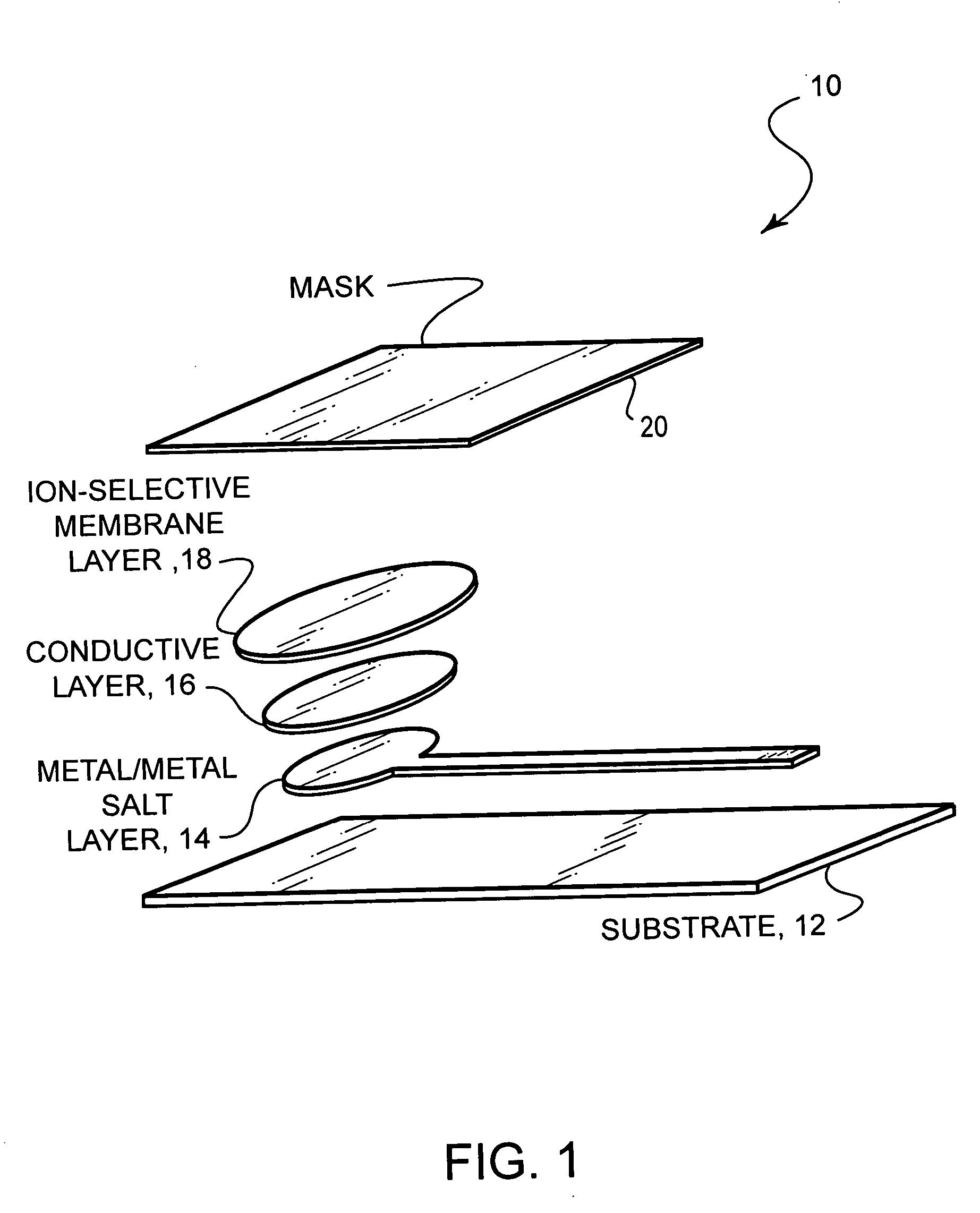
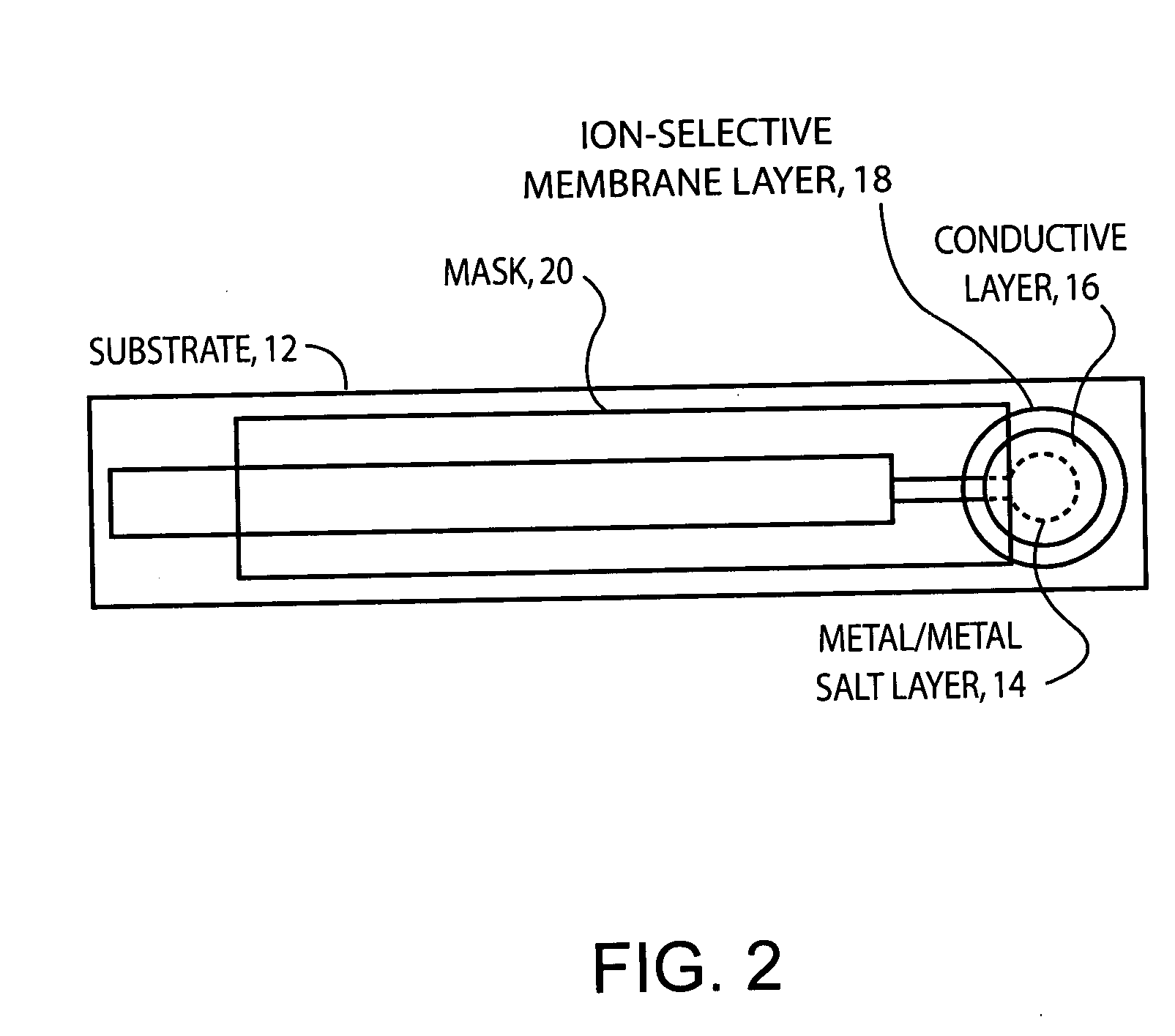
- A stable, compact ion-selective electrode design featuring a water-impermeable substrate, an electrically conductive metal/metal salt layer, a hydrophobic conductive layer with ions for stable potential, an ion-selective layer, and a water-impermeable barrier layer to prevent exposure to aqueous solutions, eliminating the need for calibration and reducing equilibration time.
Ion selective electrode
PatentWO2012067490A1
Innovation
- A solution-cast conductive polymer transducer layer comprising bisphenol A propoxylate diglycidyl ether, a purified diamine binder, and a polar solvent is used, which includes polypyrrole as the primary polymer, minimizing the need for continuous stirring and allowing for a homogenous layer formation, even on small electrodes, with a retaining dam to contain the sensing membrane.
Biotech ISE Regulatory Compliance Standards
The regulatory landscape for ion selective electrodes in biotechnology applications is governed by multiple international and regional standards that ensure device safety, accuracy, and reliability. The International Organization for Standardization (ISO) provides foundational guidelines through ISO 14155 for clinical investigation of medical devices and ISO 13485 for quality management systems specific to medical devices. These standards establish comprehensive frameworks for ISE development, testing, and deployment in biomedical environments.
In the United States, the Food and Drug Administration (FDA) classifies biotechnology ISEs under medical device regulations, typically falling under Class II devices requiring 510(k) premarket notification. The FDA's Quality System Regulation (QSR) mandates rigorous design controls, risk management protocols, and clinical validation studies. European markets operate under the Medical Device Regulation (MDR) 2017/745, which replaced the previous Medical Device Directive and introduced stricter requirements for clinical evidence and post-market surveillance.
Performance verification standards specifically address analytical measurement requirements through Clinical and Laboratory Standards Institute (CLSI) guidelines, particularly EP15-A3 for user verification of precision and estimation of bias. The International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) provides additional protocols for electrode standardization and calibration procedures. These standards mandate specific acceptance criteria for precision, accuracy, linearity, and measurement range validation.
Quality assurance protocols require comprehensive documentation of electrode performance characteristics, including response time, selectivity coefficients, detection limits, and interference studies. Regulatory bodies demand statistical validation using appropriate sample sizes and control materials that reflect actual clinical specimens. Traceability requirements necessitate calibration against certified reference materials and participation in external quality assessment programs.
Emerging regulatory trends focus on software validation for digital ISE systems, cybersecurity requirements for connected devices, and enhanced post-market surveillance obligations. Regulatory harmonization efforts through the International Medical Device Regulators Forum (IMDRF) are establishing global convergence toward risk-based approaches and streamlined approval pathways for innovative ISE technologies in biotechnology applications.
In the United States, the Food and Drug Administration (FDA) classifies biotechnology ISEs under medical device regulations, typically falling under Class II devices requiring 510(k) premarket notification. The FDA's Quality System Regulation (QSR) mandates rigorous design controls, risk management protocols, and clinical validation studies. European markets operate under the Medical Device Regulation (MDR) 2017/745, which replaced the previous Medical Device Directive and introduced stricter requirements for clinical evidence and post-market surveillance.
Performance verification standards specifically address analytical measurement requirements through Clinical and Laboratory Standards Institute (CLSI) guidelines, particularly EP15-A3 for user verification of precision and estimation of bias. The International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) provides additional protocols for electrode standardization and calibration procedures. These standards mandate specific acceptance criteria for precision, accuracy, linearity, and measurement range validation.
Quality assurance protocols require comprehensive documentation of electrode performance characteristics, including response time, selectivity coefficients, detection limits, and interference studies. Regulatory bodies demand statistical validation using appropriate sample sizes and control materials that reflect actual clinical specimens. Traceability requirements necessitate calibration against certified reference materials and participation in external quality assessment programs.
Emerging regulatory trends focus on software validation for digital ISE systems, cybersecurity requirements for connected devices, and enhanced post-market surveillance obligations. Regulatory harmonization efforts through the International Medical Device Regulators Forum (IMDRF) are establishing global convergence toward risk-based approaches and streamlined approval pathways for innovative ISE technologies in biotechnology applications.
Quality Assurance Protocols for ISE Verification
Quality assurance protocols for ion selective electrode verification in biotechnology applications require systematic approaches to ensure measurement reliability and regulatory compliance. These protocols establish standardized procedures that validate electrode performance across diverse biological matrices and operational conditions.
Calibration verification protocols form the foundation of ISE quality assurance systems. Multi-point calibration procedures using certified reference materials must demonstrate linear response characteristics within specified concentration ranges. Standard operating procedures typically require minimum three-point calibrations with bracketing standards that encompass the expected sample concentration range. Calibration acceptance criteria include correlation coefficients exceeding 0.995 and slope values within manufacturer specifications.
Precision and accuracy assessment protocols involve replicate measurements of quality control samples at low, medium, and high concentration levels. Statistical evaluation requires coefficient of variation values below predetermined thresholds, typically 5% for routine applications and 2% for critical measurements. Accuracy verification employs certified reference materials or spiked samples with known analyte concentrations, requiring recovery percentages within 95-105% acceptance limits.
Interference testing protocols evaluate electrode selectivity in complex biological matrices. Systematic evaluation of common interferents including proteins, lipids, and competing ions ensures measurement specificity. Protocol design incorporates separate solution method testing and mixed solution evaluations to quantify selectivity coefficients according to IUPAC recommendations.
Stability monitoring protocols track electrode performance over extended periods through control chart methodologies. Daily quality control measurements using stable reference solutions identify drift patterns and performance degradation. Acceptance criteria establish control limits based on historical performance data, triggering corrective actions when measurements exceed established boundaries.
Documentation and traceability protocols ensure comprehensive record keeping for regulatory compliance. Electronic data management systems capture calibration parameters, quality control results, and maintenance activities with audit trail capabilities. Periodic protocol reviews incorporate performance trending analysis and continuous improvement initiatives to optimize verification procedures based on operational experience and evolving regulatory requirements.
Calibration verification protocols form the foundation of ISE quality assurance systems. Multi-point calibration procedures using certified reference materials must demonstrate linear response characteristics within specified concentration ranges. Standard operating procedures typically require minimum three-point calibrations with bracketing standards that encompass the expected sample concentration range. Calibration acceptance criteria include correlation coefficients exceeding 0.995 and slope values within manufacturer specifications.
Precision and accuracy assessment protocols involve replicate measurements of quality control samples at low, medium, and high concentration levels. Statistical evaluation requires coefficient of variation values below predetermined thresholds, typically 5% for routine applications and 2% for critical measurements. Accuracy verification employs certified reference materials or spiked samples with known analyte concentrations, requiring recovery percentages within 95-105% acceptance limits.
Interference testing protocols evaluate electrode selectivity in complex biological matrices. Systematic evaluation of common interferents including proteins, lipids, and competing ions ensures measurement specificity. Protocol design incorporates separate solution method testing and mixed solution evaluations to quantify selectivity coefficients according to IUPAC recommendations.
Stability monitoring protocols track electrode performance over extended periods through control chart methodologies. Daily quality control measurements using stable reference solutions identify drift patterns and performance degradation. Acceptance criteria establish control limits based on historical performance data, triggering corrective actions when measurements exceed established boundaries.
Documentation and traceability protocols ensure comprehensive record keeping for regulatory compliance. Electronic data management systems capture calibration parameters, quality control results, and maintenance activities with audit trail capabilities. Periodic protocol reviews incorporate performance trending analysis and continuous improvement initiatives to optimize verification procedures based on operational experience and evolving regulatory requirements.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!