Ion Selective Electrode vs. FTIR: Detection Capabilities Compared
MAR 8, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Ion Selective Electrode vs FTIR Detection Background and Goals
Ion selective electrodes and Fourier Transform Infrared spectroscopy represent two fundamentally different analytical approaches that have evolved to address diverse detection challenges across multiple industries. ISE technology emerged in the 1960s as an extension of pH measurement principles, utilizing selective membrane chemistry to quantify specific ionic species in solution. This electrochemical approach revolutionized water quality monitoring, clinical diagnostics, and environmental analysis by providing real-time, cost-effective measurements of target ions.
FTIR spectroscopy developed from classical infrared spectroscopy through the integration of interferometry and computational processing, achieving commercial viability in the 1970s. This molecular spectroscopic technique identifies and quantifies compounds based on their unique vibrational fingerprints, offering broad-spectrum analytical capabilities across gas, liquid, and solid phases. The technology has become indispensable in pharmaceutical analysis, materials characterization, and chemical process monitoring.
The convergence of these technologies in comparative studies reflects the analytical chemistry field's evolution toward method validation and optimization. Modern analytical challenges increasingly demand multi-technique approaches, where understanding the complementary strengths and limitations of different detection methods becomes crucial for method selection and analytical strategy development.
Current technological trends emphasize miniaturization, automation, and real-time monitoring capabilities. ISE technology has progressed toward solid-state sensors, wireless monitoring systems, and improved selectivity through advanced membrane materials. Meanwhile, FTIR has evolved to include portable instruments, enhanced sensitivity through advanced detector technologies, and expanded sampling accessories for challenging matrices.
The primary objective of comparing ISE and FTIR detection capabilities centers on establishing clear guidelines for analytical method selection based on specific application requirements. This comparison aims to define the optimal operational parameters, detection limits, selectivity profiles, and matrix compatibility for each technique across various analytical scenarios.
Furthermore, this comparative analysis seeks to identify potential synergistic applications where both techniques could complement each other, providing enhanced analytical confidence through orthogonal measurement principles. Understanding these complementary aspects enables the development of hybrid analytical strategies that leverage the strengths of both electrochemical and spectroscopic detection methods.
FTIR spectroscopy developed from classical infrared spectroscopy through the integration of interferometry and computational processing, achieving commercial viability in the 1970s. This molecular spectroscopic technique identifies and quantifies compounds based on their unique vibrational fingerprints, offering broad-spectrum analytical capabilities across gas, liquid, and solid phases. The technology has become indispensable in pharmaceutical analysis, materials characterization, and chemical process monitoring.
The convergence of these technologies in comparative studies reflects the analytical chemistry field's evolution toward method validation and optimization. Modern analytical challenges increasingly demand multi-technique approaches, where understanding the complementary strengths and limitations of different detection methods becomes crucial for method selection and analytical strategy development.
Current technological trends emphasize miniaturization, automation, and real-time monitoring capabilities. ISE technology has progressed toward solid-state sensors, wireless monitoring systems, and improved selectivity through advanced membrane materials. Meanwhile, FTIR has evolved to include portable instruments, enhanced sensitivity through advanced detector technologies, and expanded sampling accessories for challenging matrices.
The primary objective of comparing ISE and FTIR detection capabilities centers on establishing clear guidelines for analytical method selection based on specific application requirements. This comparison aims to define the optimal operational parameters, detection limits, selectivity profiles, and matrix compatibility for each technique across various analytical scenarios.
Furthermore, this comparative analysis seeks to identify potential synergistic applications where both techniques could complement each other, providing enhanced analytical confidence through orthogonal measurement principles. Understanding these complementary aspects enables the development of hybrid analytical strategies that leverage the strengths of both electrochemical and spectroscopic detection methods.
Market Demand for Advanced Chemical Detection Technologies
The global chemical detection technology market is experiencing unprecedented growth driven by stringent regulatory requirements across multiple industries. Environmental monitoring agencies worldwide are implementing more rigorous standards for water quality assessment, air pollution control, and soil contamination detection. These regulatory frameworks mandate the use of precise analytical instruments capable of detecting trace-level contaminants, creating substantial demand for both ion selective electrodes and FTIR spectroscopy systems.
Industrial process control represents another significant demand driver, particularly in pharmaceutical manufacturing, petrochemical processing, and food production. Manufacturing facilities require real-time monitoring capabilities to ensure product quality and compliance with safety standards. The pharmaceutical industry specifically demands analytical technologies that can provide rapid, accurate measurements of ionic concentrations and molecular compositions during drug formulation and quality control processes.
The water treatment and environmental remediation sectors constitute major market segments for chemical detection technologies. Municipal water treatment facilities, industrial wastewater management systems, and groundwater monitoring programs require continuous monitoring of various chemical parameters. Ion selective electrodes excel in applications requiring real-time monitoring of specific ions such as fluoride, nitrate, and heavy metals, while FTIR systems provide comprehensive molecular fingerprinting capabilities for complex contamination scenarios.
Laboratory automation trends are reshaping market demand patterns, with increasing preference for integrated analytical systems that combine multiple detection methodologies. Research institutions and commercial laboratories seek versatile platforms capable of handling diverse analytical requirements while maintaining high throughput and reliability. This trend favors hybrid approaches that leverage the complementary strengths of electrochemical and spectroscopic detection methods.
Emerging applications in biotechnology and personalized medicine are creating new market opportunities for portable and miniaturized chemical detection systems. Point-of-care diagnostics, field-deployable environmental monitoring, and in-situ industrial process monitoring require compact, robust analytical instruments with simplified operation protocols.
The market landscape is increasingly characterized by demand for cost-effective solutions that balance analytical performance with operational efficiency. End-users prioritize technologies offering lower maintenance requirements, reduced consumable costs, and simplified calibration procedures while maintaining analytical accuracy and precision standards.
Industrial process control represents another significant demand driver, particularly in pharmaceutical manufacturing, petrochemical processing, and food production. Manufacturing facilities require real-time monitoring capabilities to ensure product quality and compliance with safety standards. The pharmaceutical industry specifically demands analytical technologies that can provide rapid, accurate measurements of ionic concentrations and molecular compositions during drug formulation and quality control processes.
The water treatment and environmental remediation sectors constitute major market segments for chemical detection technologies. Municipal water treatment facilities, industrial wastewater management systems, and groundwater monitoring programs require continuous monitoring of various chemical parameters. Ion selective electrodes excel in applications requiring real-time monitoring of specific ions such as fluoride, nitrate, and heavy metals, while FTIR systems provide comprehensive molecular fingerprinting capabilities for complex contamination scenarios.
Laboratory automation trends are reshaping market demand patterns, with increasing preference for integrated analytical systems that combine multiple detection methodologies. Research institutions and commercial laboratories seek versatile platforms capable of handling diverse analytical requirements while maintaining high throughput and reliability. This trend favors hybrid approaches that leverage the complementary strengths of electrochemical and spectroscopic detection methods.
Emerging applications in biotechnology and personalized medicine are creating new market opportunities for portable and miniaturized chemical detection systems. Point-of-care diagnostics, field-deployable environmental monitoring, and in-situ industrial process monitoring require compact, robust analytical instruments with simplified operation protocols.
The market landscape is increasingly characterized by demand for cost-effective solutions that balance analytical performance with operational efficiency. End-users prioritize technologies offering lower maintenance requirements, reduced consumable costs, and simplified calibration procedures while maintaining analytical accuracy and precision standards.
Current State and Challenges of ISE and FTIR Detection Methods
Ion Selective Electrodes represent a mature electrochemical detection technology that has been widely deployed across analytical laboratories and field applications for several decades. Current ISE systems demonstrate exceptional selectivity for specific ionic species, with detection limits reaching nanomolar concentrations for optimized electrode designs. The technology excels in aqueous environments and offers real-time monitoring capabilities with relatively simple instrumentation requirements. However, ISE methods face significant limitations in complex sample matrices where interfering ions can compromise measurement accuracy.
Modern ISE implementations struggle with drift phenomena, requiring frequent calibration procedures that limit their autonomous operation capabilities. The electrode membrane degradation over time presents ongoing maintenance challenges, particularly in harsh environmental conditions. Additionally, the technology shows limited multiplexing capabilities, typically requiring separate electrodes for each target analyte, which increases system complexity and cost for multi-parameter analysis.
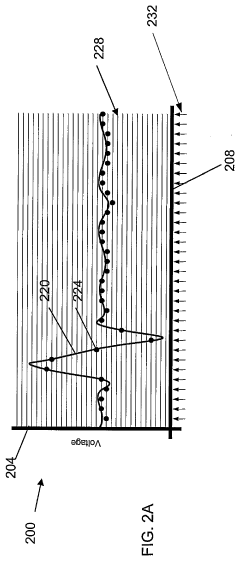
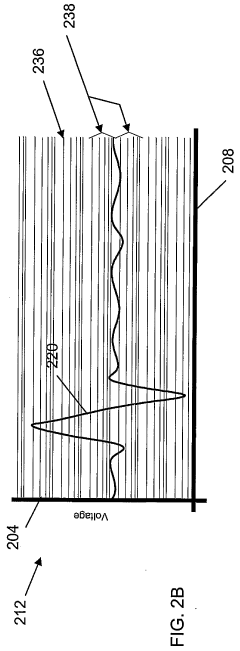
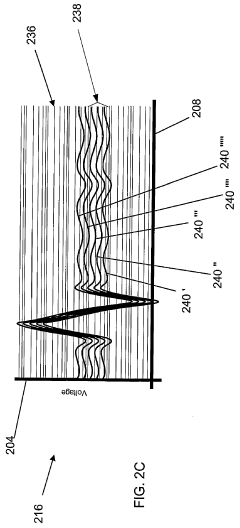
Fourier Transform Infrared spectroscopy has evolved into a sophisticated analytical platform capable of simultaneous detection and identification of multiple chemical species. Contemporary FTIR systems achieve remarkable spectral resolution and sensitivity through advanced detector technologies and signal processing algorithms. The technique provides comprehensive molecular fingerprinting capabilities, enabling both qualitative identification and quantitative analysis across diverse sample types including gases, liquids, and solids.
Current FTIR technology faces challenges related to water interference, which significantly complicates analysis in aqueous systems due to strong water absorption bands that mask target analyte signals. Sample preparation requirements often introduce complexity and potential contamination risks, particularly for trace-level detection applications. The interpretation of complex spectra requires sophisticated chemometric approaches and extensive spectral libraries, demanding higher operator expertise compared to ISE methods.
Both technologies encounter limitations in dynamic range optimization, where ISE systems may suffer from non-linear responses at extreme concentrations, while FTIR methods face challenges in maintaining linearity across wide concentration ranges due to Beer's law deviations. Environmental factors such as temperature fluctuations and electromagnetic interference affect both detection methods, though through different mechanisms and to varying degrees.
The integration of artificial intelligence and machine learning algorithms presents emerging opportunities for both technologies to overcome traditional limitations through enhanced data processing and predictive maintenance capabilities.
Modern ISE implementations struggle with drift phenomena, requiring frequent calibration procedures that limit their autonomous operation capabilities. The electrode membrane degradation over time presents ongoing maintenance challenges, particularly in harsh environmental conditions. Additionally, the technology shows limited multiplexing capabilities, typically requiring separate electrodes for each target analyte, which increases system complexity and cost for multi-parameter analysis.
Fourier Transform Infrared spectroscopy has evolved into a sophisticated analytical platform capable of simultaneous detection and identification of multiple chemical species. Contemporary FTIR systems achieve remarkable spectral resolution and sensitivity through advanced detector technologies and signal processing algorithms. The technique provides comprehensive molecular fingerprinting capabilities, enabling both qualitative identification and quantitative analysis across diverse sample types including gases, liquids, and solids.
Current FTIR technology faces challenges related to water interference, which significantly complicates analysis in aqueous systems due to strong water absorption bands that mask target analyte signals. Sample preparation requirements often introduce complexity and potential contamination risks, particularly for trace-level detection applications. The interpretation of complex spectra requires sophisticated chemometric approaches and extensive spectral libraries, demanding higher operator expertise compared to ISE methods.
Both technologies encounter limitations in dynamic range optimization, where ISE systems may suffer from non-linear responses at extreme concentrations, while FTIR methods face challenges in maintaining linearity across wide concentration ranges due to Beer's law deviations. Environmental factors such as temperature fluctuations and electromagnetic interference affect both detection methods, though through different mechanisms and to varying degrees.
The integration of artificial intelligence and machine learning algorithms presents emerging opportunities for both technologies to overcome traditional limitations through enhanced data processing and predictive maintenance capabilities.
Existing ISE and FTIR Detection Solutions Comparison
01 Ion-selective electrode design and construction
Ion-selective electrodes are designed with specific membrane materials and configurations to selectively detect target ions in solution. The electrode construction includes membrane composition, internal reference solutions, and electrode body design that enables selective ion detection through potential measurements. Various membrane materials such as polymers, glass, or crystalline materials are utilized to achieve ion selectivity and sensitivity.- Ion-selective electrode design and construction: Ion-selective electrodes are designed with specific membrane materials and configurations to detect target ions in solution. The electrode construction includes ion-selective membranes, reference electrodes, and internal filling solutions that enable selective detection of specific ions such as sodium, potassium, calcium, or chloride. The membrane composition and electrode geometry are optimized to achieve high selectivity and sensitivity for the target analyte.
- FTIR spectroscopy detection methods and applications: Fourier Transform Infrared spectroscopy is utilized for molecular identification and structural analysis through the detection of characteristic absorption bands. The technique involves measuring infrared radiation absorption by samples to identify functional groups and chemical bonds. FTIR detection capabilities include qualitative and quantitative analysis of organic and inorganic compounds, with applications in material characterization and chemical composition analysis.
- Combined analytical systems integrating multiple detection techniques: Integrated analytical systems combine ion-selective electrodes with spectroscopic methods to provide comprehensive chemical analysis. These hybrid systems enable simultaneous or sequential measurements using different detection principles, enhancing analytical capabilities and providing complementary information about sample composition. The integration allows for cross-validation of results and expanded detection ranges.
- Calibration and measurement optimization for electrochemical sensors: Calibration procedures and measurement optimization techniques are essential for accurate ion detection using selective electrodes. Methods include standard addition techniques, multi-point calibration curves, and compensation for interfering ions. Signal processing and data analysis algorithms improve measurement accuracy and reduce noise, while temperature compensation and drift correction enhance long-term stability and reliability of measurements.
- Sample preparation and analysis protocols for detection systems: Sample preparation methods are developed to optimize detection performance for both ion-selective electrodes and FTIR spectroscopy. Protocols include sample pretreatment, matrix modification, and interference removal techniques. Standardized procedures ensure reproducible measurements and accurate quantification, with specific considerations for sample pH adjustment, ionic strength control, and elimination of spectral interferences.
02 FTIR spectroscopy detection methods and applications
Fourier Transform Infrared spectroscopy is employed for molecular identification and quantification through infrared absorption measurements. The technique involves analyzing the vibrational modes of molecules to determine chemical composition and structure. FTIR detection capabilities include real-time monitoring, non-destructive analysis, and identification of functional groups in various sample types.Expand Specific Solutions03 Combined analytical systems integrating multiple detection techniques
Integrated analytical platforms combine ion-selective electrodes with spectroscopic methods to provide comprehensive chemical analysis. These systems enable simultaneous or sequential measurements using different detection principles, enhancing analytical capabilities and providing complementary information about sample composition. The integration improves accuracy and expands the range of detectable analytes.Expand Specific Solutions04 Sensor calibration and signal processing methods
Advanced calibration techniques and signal processing algorithms are implemented to improve the accuracy and reliability of ion-selective electrodes and FTIR measurements. These methods include baseline correction, interference compensation, temperature correction, and multivariate data analysis. Signal processing enhances detection limits and reduces measurement errors in complex sample matrices.Expand Specific Solutions05 Application in environmental and industrial monitoring
Ion-selective electrodes and FTIR detection systems are applied for continuous monitoring in environmental and industrial settings. These applications include water quality assessment, process control, contamination detection, and compliance monitoring. The technologies enable real-time analysis of multiple parameters with minimal sample preparation and rapid response times.Expand Specific Solutions
Key Players in ISE and FTIR Analytical Instrument Industry
The ion selective electrode versus FTIR detection capabilities comparison represents a mature analytical technology sector experiencing steady growth driven by increasing demand for precise chemical analysis across healthcare, environmental monitoring, and industrial applications. The market demonstrates significant scale with established players like Siemens Healthcare Diagnostics, Radiometer A/S, and Koninklijke Philips NV dominating clinical diagnostics, while Bruker Daltonics and Thermo Finnigan lead spectroscopy instrumentation. Technology maturity varies significantly between approaches: ion selective electrodes represent well-established, cost-effective solutions with proven reliability, whereas FTIR systems offer advanced molecular identification capabilities but require higher investment. Emerging companies like QuantaRed Technologies and Autonomous Medical Devices are driving innovation through specialized applications and point-of-care solutions, while research institutions including MIT and EPFL continue advancing fundamental detection methodologies, indicating ongoing technological evolution despite market maturity.
Siemens Healthcare Diagnostics, Inc.
Technical Solution: Siemens Healthcare has developed advanced ion selective electrode systems for clinical diagnostics, particularly for blood gas analysis and electrolyte monitoring. Their technology integrates multiple ISE sensors for simultaneous detection of Na+, K+, Cl-, and Ca2+ ions with high precision and rapid response times. The company's ISE platforms feature automated calibration systems and temperature compensation algorithms to ensure measurement accuracy across varying conditions. Their systems are widely deployed in hospital laboratories and point-of-care testing environments, offering real-time monitoring capabilities for critical care applications.
Strengths: Proven clinical reliability, automated calibration, multi-parameter detection. Weaknesses: Limited to ionic species detection, requires regular maintenance and calibration.
Bruker Daltonics GmbH & Co. KG
Technical Solution: Bruker specializes in FTIR spectroscopy systems with advanced detection capabilities for molecular identification and quantitative analysis. Their FTIR instruments utilize sophisticated interferometer designs and MCT detectors to achieve high sensitivity and spectral resolution. The company's technology enables detection of organic compounds, polymers, and trace contaminants through characteristic vibrational fingerprints. Bruker's FTIR systems incorporate automated sample handling, spectral libraries with over 200,000 reference spectra, and advanced chemometric algorithms for complex mixture analysis. Their instruments are extensively used in pharmaceutical, environmental, and materials science applications.
Strengths: High spectral resolution, extensive spectral libraries, versatile molecular detection. Weaknesses: Higher cost, requires skilled operators, limited real-time monitoring capabilities.
Core Innovations in Ion Selective and FTIR Detection Patents
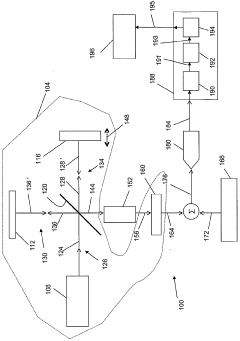
Fourier transform infrared spectrometer
PatentWO2006115490A1
Innovation
- The system employs a Fourier Transform Infrared spectrometer with a path length difference swept in time, combined with a selected signal to reduce ADC nonlinearity, using an analog-to-digital converter that processes the combined signal to minimize nonlinearity effects, and further processes the signal to generate a digital representation that accurately reflects the radiometric signals.
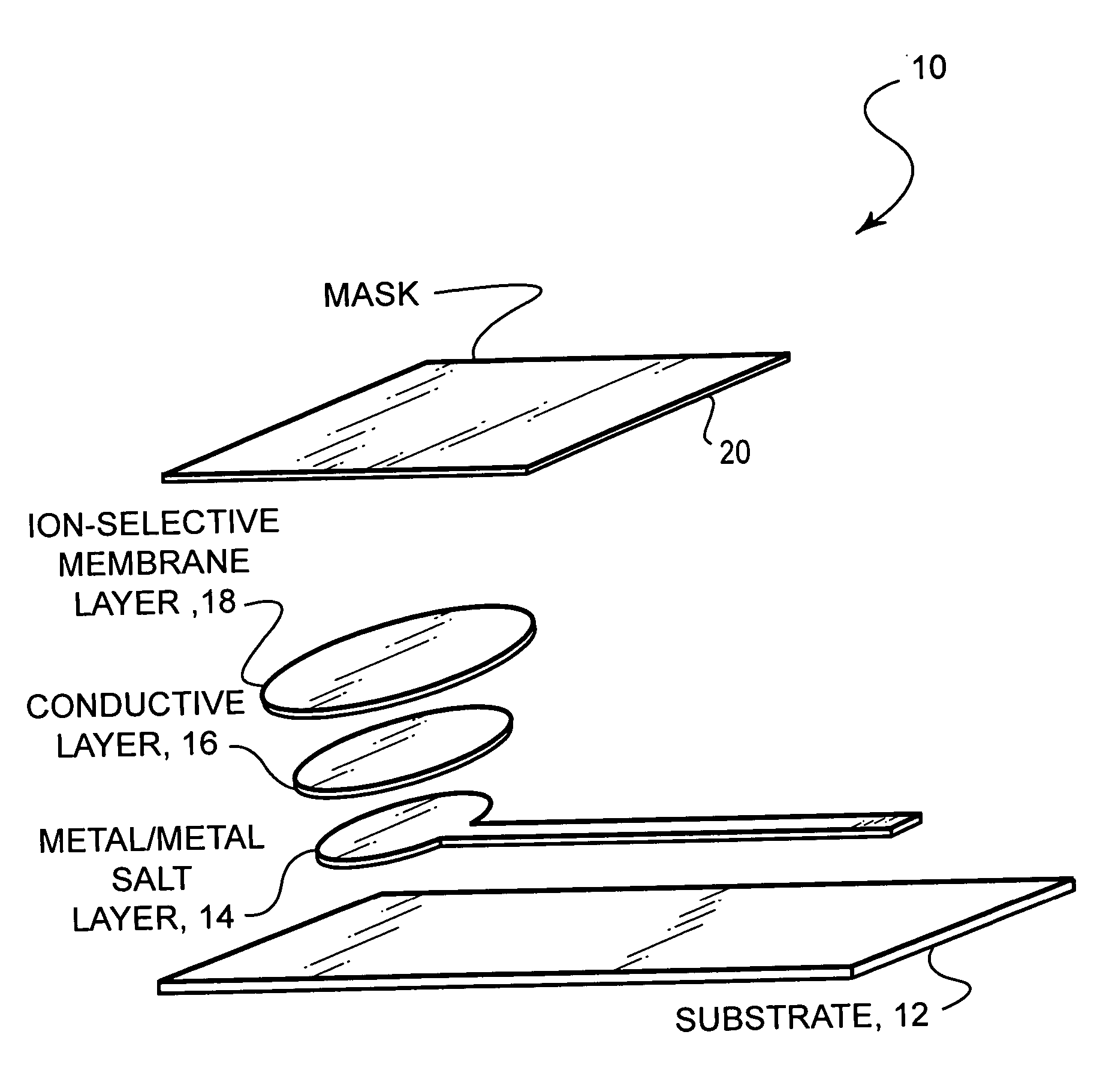
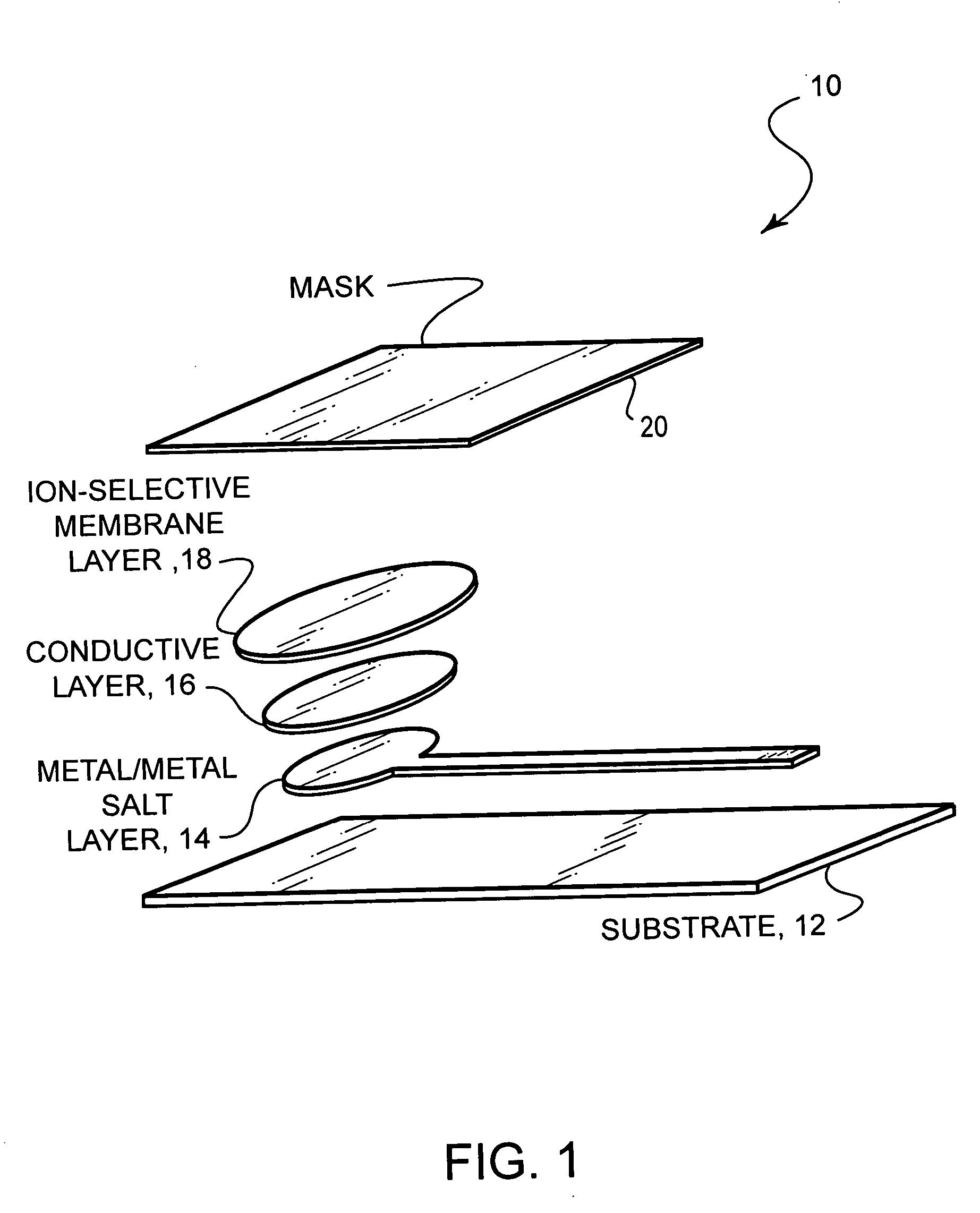
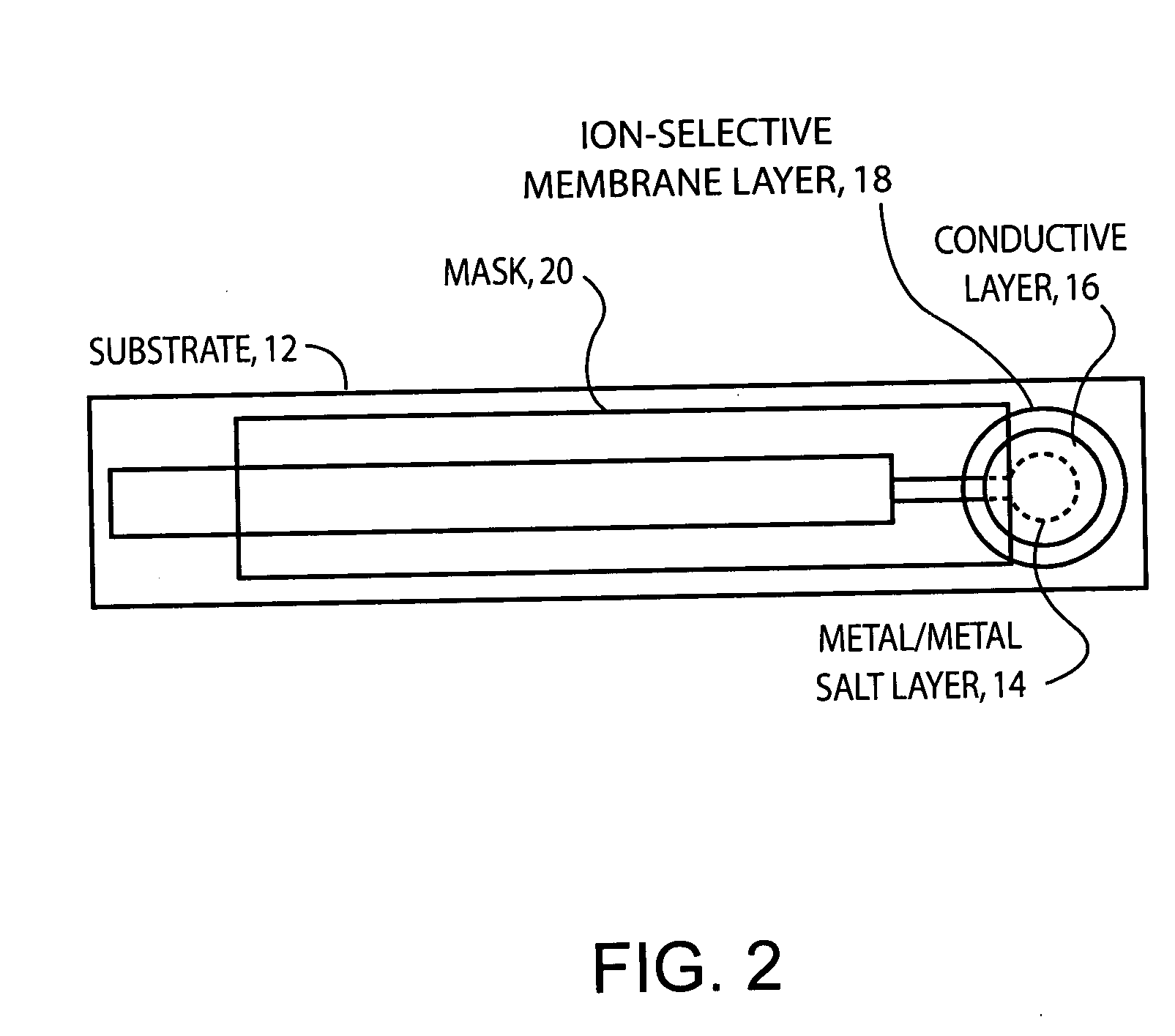
Ion-selective electrodes
PatentInactiveUS20050191428A1
Innovation
- A stable, compact ion-selective electrode design featuring a water-impermeable substrate, an electrically conductive metal/metal salt layer, a hydrophobic conductive layer with ions for stable potential, an ion-selective layer, and a water-impermeable barrier layer to prevent exposure to aqueous solutions, eliminating the need for calibration and reducing equilibration time.
Analytical Method Validation Standards and Regulations
The validation of analytical methods for both Ion Selective Electrodes (ISE) and Fourier Transform Infrared Spectroscopy (FTIR) must comply with internationally recognized standards to ensure reliable and reproducible results. The International Conference on Harmonisation (ICH) Q2(R1) guideline serves as the primary framework for analytical method validation, establishing requirements for accuracy, precision, specificity, detection limit, quantitation limit, linearity, and range. These parameters are equally applicable to both electrochemical and spectroscopic techniques, though their implementation varies significantly between ISE and FTIR methodologies.
For ISE validation, the United States Pharmacopeia (USP) Chapter 1225 and European Pharmacopoeia guidelines provide specific requirements for potentiometric measurements. These standards mandate slope verification within 54-60 mV per decade for monovalent ions at 25°C, response time evaluation typically within 1-3 minutes, and selectivity coefficient determination using separate solution or mixed solution methods. The FDA's Analytical Procedures and Methods Validation guidance emphasizes the importance of demonstrating electrode stability and interference studies for complex matrices.
FTIR method validation follows ASTM E1655 standards for infrared qualitative analysis and ASTM E2617 for quantitative applications. The International Organization for Standardization (ISO) 21543 specifically addresses validation requirements for molecular spectroscopy methods. These standards require demonstration of spectral resolution, wavenumber accuracy, photometric accuracy, and baseline stability. Additionally, chemometric model validation becomes crucial when employing multivariate analysis techniques with FTIR data.
Regulatory compliance varies across different industries and geographical regions. The European Medicines Agency (EMA) guidelines align closely with ICH standards but emphasize additional requirements for method robustness and technology transfer protocols. The Association of Official Analytical Chemists (AOAC) International provides specific validation guidelines for food and agricultural applications, which are particularly relevant for ISE methods used in nutritional analysis.
Quality assurance protocols mandate regular system suitability testing for both techniques. ISE systems require daily calibration verification and electrode conditioning protocols, while FTIR instruments need performance qualification including operational qualification and performance verification according to ASTM E2891 standards. Documentation requirements under Good Laboratory Practice (GLP) regulations ensure traceability and reproducibility of validation studies, with specific emphasis on method lifecycle management and change control procedures throughout the analytical method's operational lifetime.
For ISE validation, the United States Pharmacopeia (USP) Chapter 1225 and European Pharmacopoeia guidelines provide specific requirements for potentiometric measurements. These standards mandate slope verification within 54-60 mV per decade for monovalent ions at 25°C, response time evaluation typically within 1-3 minutes, and selectivity coefficient determination using separate solution or mixed solution methods. The FDA's Analytical Procedures and Methods Validation guidance emphasizes the importance of demonstrating electrode stability and interference studies for complex matrices.
FTIR method validation follows ASTM E1655 standards for infrared qualitative analysis and ASTM E2617 for quantitative applications. The International Organization for Standardization (ISO) 21543 specifically addresses validation requirements for molecular spectroscopy methods. These standards require demonstration of spectral resolution, wavenumber accuracy, photometric accuracy, and baseline stability. Additionally, chemometric model validation becomes crucial when employing multivariate analysis techniques with FTIR data.
Regulatory compliance varies across different industries and geographical regions. The European Medicines Agency (EMA) guidelines align closely with ICH standards but emphasize additional requirements for method robustness and technology transfer protocols. The Association of Official Analytical Chemists (AOAC) International provides specific validation guidelines for food and agricultural applications, which are particularly relevant for ISE methods used in nutritional analysis.
Quality assurance protocols mandate regular system suitability testing for both techniques. ISE systems require daily calibration verification and electrode conditioning protocols, while FTIR instruments need performance qualification including operational qualification and performance verification according to ASTM E2891 standards. Documentation requirements under Good Laboratory Practice (GLP) regulations ensure traceability and reproducibility of validation studies, with specific emphasis on method lifecycle management and change control procedures throughout the analytical method's operational lifetime.
Environmental Impact of Chemical Detection Technologies
The environmental implications of chemical detection technologies have become increasingly significant as analytical methods proliferate across industrial, research, and monitoring applications. Both Ion Selective Electrodes (ISE) and Fourier Transform Infrared Spectroscopy (FTIR) present distinct environmental footprints that must be evaluated comprehensively to understand their sustainability profiles.
Energy consumption patterns differ substantially between these technologies. ISE systems typically operate with minimal power requirements, utilizing simple electronic circuits and requiring no complex optical components or cooling systems. The low-power nature of electrochemical sensors makes them particularly suitable for remote monitoring applications powered by solar panels or batteries. In contrast, FTIR spectrometers demand significantly higher energy inputs due to their sophisticated optical systems, interferometers, and often require temperature-controlled environments to maintain measurement precision.
Waste generation and material lifecycle considerations reveal contrasting environmental impacts. ISE technology produces minimal electronic waste during operation, though electrode replacement generates small quantities of specialized materials containing heavy metals or rare earth elements. The disposal of ion-selective membranes and reference electrodes requires careful handling due to potential toxic components. FTIR systems generate different waste streams, primarily consisting of optical components, electronic assemblies, and occasional desiccant materials used for moisture control.
Chemical reagent consumption represents another critical environmental factor. ISE measurements often require calibration solutions and ionic strength adjusters, though quantities are typically minimal. Buffer solutions and standard reference materials constitute the primary chemical inputs. FTIR spectroscopy generally operates without consumable chemicals during routine analysis, though sample preparation may require solvents or other processing agents depending on the application.
Manufacturing environmental costs vary significantly between technologies. ISE production involves relatively simple fabrication processes with lower energy intensity, though specialized membrane materials may require complex synthesis procedures. FTIR instrument manufacturing demands precision optical components, sophisticated electronics, and high-purity materials, resulting in higher embodied energy and carbon footprint per unit.
Operational longevity and maintenance requirements influence long-term environmental impact. ISE systems typically require periodic electrode replacement and calibration, generating ongoing material consumption. However, their robust design often enables extended field deployment with minimal intervention. FTIR instruments generally offer longer operational lifespans but require more intensive maintenance procedures, including optical alignment, component replacement, and environmental control systems that contribute to their overall environmental burden throughout the operational lifecycle.
Energy consumption patterns differ substantially between these technologies. ISE systems typically operate with minimal power requirements, utilizing simple electronic circuits and requiring no complex optical components or cooling systems. The low-power nature of electrochemical sensors makes them particularly suitable for remote monitoring applications powered by solar panels or batteries. In contrast, FTIR spectrometers demand significantly higher energy inputs due to their sophisticated optical systems, interferometers, and often require temperature-controlled environments to maintain measurement precision.
Waste generation and material lifecycle considerations reveal contrasting environmental impacts. ISE technology produces minimal electronic waste during operation, though electrode replacement generates small quantities of specialized materials containing heavy metals or rare earth elements. The disposal of ion-selective membranes and reference electrodes requires careful handling due to potential toxic components. FTIR systems generate different waste streams, primarily consisting of optical components, electronic assemblies, and occasional desiccant materials used for moisture control.
Chemical reagent consumption represents another critical environmental factor. ISE measurements often require calibration solutions and ionic strength adjusters, though quantities are typically minimal. Buffer solutions and standard reference materials constitute the primary chemical inputs. FTIR spectroscopy generally operates without consumable chemicals during routine analysis, though sample preparation may require solvents or other processing agents depending on the application.
Manufacturing environmental costs vary significantly between technologies. ISE production involves relatively simple fabrication processes with lower energy intensity, though specialized membrane materials may require complex synthesis procedures. FTIR instrument manufacturing demands precision optical components, sophisticated electronics, and high-purity materials, resulting in higher embodied energy and carbon footprint per unit.
Operational longevity and maintenance requirements influence long-term environmental impact. ISE systems typically require periodic electrode replacement and calibration, generating ongoing material consumption. However, their robust design often enables extended field deployment with minimal intervention. FTIR instruments generally offer longer operational lifespans but require more intensive maintenance procedures, including optical alignment, component replacement, and environmental control systems that contribute to their overall environmental burden throughout the operational lifecycle.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!