Ion Selective Electrode vs. Spectrophotometry: Detection Accuracy
MAR 8, 20268 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Ion Electrode vs Spectrophotometry Background and Goals
Ion selective electrodes (ISEs) and spectrophotometry represent two fundamental analytical approaches that have evolved significantly since their inception in the early 20th century. ISEs emerged from the pioneering work on glass electrodes in the 1900s, with the first practical pH electrode developed in 1909. The technology expanded rapidly through the development of membrane-based sensors in the 1960s and 1970s, incorporating various ionophores and selective materials to detect specific ions with remarkable precision.
Spectrophotometry traces its origins to the Beer-Lambert law established in the mid-1800s, with practical instruments becoming available in the 1940s. The technique has undergone continuous refinement, progressing from simple colorimetric measurements to sophisticated UV-Vis, atomic absorption, and emission spectroscopy systems. Modern spectrophotometric instruments now feature advanced optics, CCD detectors, and automated sample handling capabilities.
The convergence of these technologies in analytical chemistry has created a compelling need to evaluate their relative detection accuracy across various applications. Current market demands increasingly require analytical methods that can deliver both high precision and cost-effectiveness, particularly in environmental monitoring, clinical diagnostics, pharmaceutical quality control, and industrial process monitoring.
The primary technical objective centers on establishing comprehensive accuracy benchmarks for both methodologies across different analyte concentrations, matrix compositions, and operational conditions. This evaluation aims to determine optimal detection limits, precision ranges, and interference susceptibility for each approach. Understanding the fundamental accuracy limitations of ISEs, including electrode drift, membrane selectivity coefficients, and temperature dependencies, versus spectrophotometric challenges such as matrix effects, spectral interferences, and baseline stability, forms the core technical goal.
Strategic objectives include developing decision frameworks for method selection based on specific analytical requirements, sample characteristics, and operational constraints. The research seeks to identify complementary applications where hybrid approaches might enhance overall analytical performance, while establishing clear guidelines for when each method provides superior accuracy advantages in real-world analytical scenarios.
Spectrophotometry traces its origins to the Beer-Lambert law established in the mid-1800s, with practical instruments becoming available in the 1940s. The technique has undergone continuous refinement, progressing from simple colorimetric measurements to sophisticated UV-Vis, atomic absorption, and emission spectroscopy systems. Modern spectrophotometric instruments now feature advanced optics, CCD detectors, and automated sample handling capabilities.
The convergence of these technologies in analytical chemistry has created a compelling need to evaluate their relative detection accuracy across various applications. Current market demands increasingly require analytical methods that can deliver both high precision and cost-effectiveness, particularly in environmental monitoring, clinical diagnostics, pharmaceutical quality control, and industrial process monitoring.
The primary technical objective centers on establishing comprehensive accuracy benchmarks for both methodologies across different analyte concentrations, matrix compositions, and operational conditions. This evaluation aims to determine optimal detection limits, precision ranges, and interference susceptibility for each approach. Understanding the fundamental accuracy limitations of ISEs, including electrode drift, membrane selectivity coefficients, and temperature dependencies, versus spectrophotometric challenges such as matrix effects, spectral interferences, and baseline stability, forms the core technical goal.
Strategic objectives include developing decision frameworks for method selection based on specific analytical requirements, sample characteristics, and operational constraints. The research seeks to identify complementary applications where hybrid approaches might enhance overall analytical performance, while establishing clear guidelines for when each method provides superior accuracy advantages in real-world analytical scenarios.
Market Demand for Accurate Ion Detection Methods
The global market for accurate ion detection methods is experiencing robust growth driven by expanding applications across multiple industries and increasingly stringent regulatory requirements. Environmental monitoring represents one of the largest demand segments, where precise measurement of ionic species in water bodies, soil samples, and atmospheric conditions has become critical for compliance with environmental protection standards. Industrial process control applications generate substantial demand as manufacturers seek to optimize production efficiency and ensure product quality through real-time ionic concentration monitoring.
Healthcare and clinical diagnostics constitute another major market driver, with hospitals and laboratories requiring reliable ion detection for blood gas analysis, electrolyte monitoring, and pharmaceutical quality control. The pharmaceutical industry specifically demands high-precision analytical methods for drug development, formulation testing, and regulatory compliance, creating sustained market pressure for improved detection accuracy and reliability.
Food and beverage industries increasingly rely on accurate ion detection for quality assurance, safety testing, and nutritional labeling compliance. Agricultural applications drive demand through soil analysis, fertilizer optimization, and crop monitoring programs that require precise measurement of nutrient ions and contaminants.
The market exhibits strong preference for detection methods that combine high accuracy with operational efficiency. End-users consistently prioritize technologies offering rapid analysis capabilities, minimal sample preparation requirements, and robust performance across diverse sample matrices. Cost-effectiveness remains a crucial consideration, particularly for routine monitoring applications where high sample throughput is essential.
Emerging applications in biotechnology, nanotechnology research, and advanced materials development are creating new market segments with specialized accuracy requirements. These sectors often demand detection limits and precision levels that challenge conventional analytical approaches, driving innovation in both ion-selective electrode and spectrophotometric technologies.
Regional market dynamics show particularly strong growth in Asia-Pacific regions due to industrial expansion and environmental monitoring initiatives, while established markets in North America and Europe focus on upgrading existing analytical capabilities to meet evolving regulatory standards and technological advancement opportunities.
Healthcare and clinical diagnostics constitute another major market driver, with hospitals and laboratories requiring reliable ion detection for blood gas analysis, electrolyte monitoring, and pharmaceutical quality control. The pharmaceutical industry specifically demands high-precision analytical methods for drug development, formulation testing, and regulatory compliance, creating sustained market pressure for improved detection accuracy and reliability.
Food and beverage industries increasingly rely on accurate ion detection for quality assurance, safety testing, and nutritional labeling compliance. Agricultural applications drive demand through soil analysis, fertilizer optimization, and crop monitoring programs that require precise measurement of nutrient ions and contaminants.
The market exhibits strong preference for detection methods that combine high accuracy with operational efficiency. End-users consistently prioritize technologies offering rapid analysis capabilities, minimal sample preparation requirements, and robust performance across diverse sample matrices. Cost-effectiveness remains a crucial consideration, particularly for routine monitoring applications where high sample throughput is essential.
Emerging applications in biotechnology, nanotechnology research, and advanced materials development are creating new market segments with specialized accuracy requirements. These sectors often demand detection limits and precision levels that challenge conventional analytical approaches, driving innovation in both ion-selective electrode and spectrophotometric technologies.
Regional market dynamics show particularly strong growth in Asia-Pacific regions due to industrial expansion and environmental monitoring initiatives, while established markets in North America and Europe focus on upgrading existing analytical capabilities to meet evolving regulatory standards and technological advancement opportunities.
Current Detection Accuracy Challenges and Limitations
Ion selective electrodes face significant accuracy limitations stemming from electrode drift, temperature sensitivity, and interference from competing ions. Drift phenomena occur due to gradual changes in electrode membrane properties, leading to baseline shifts that can accumulate over time and compromise long-term measurement reliability. Temperature fluctuations directly affect the Nernst equation response, requiring constant temperature compensation or controlled measurement environments to maintain accuracy.
Interference represents a critical challenge for ISE systems, as the presence of ions with similar charge characteristics can trigger cross-sensitivity responses. This selectivity coefficient variation becomes particularly problematic in complex sample matrices where multiple ionic species coexist. Additionally, electrode conditioning requirements and membrane aging effects introduce systematic errors that demand frequent calibration protocols.
Spectrophotometric detection encounters distinct accuracy challenges related to optical path variations, light source stability, and sample matrix effects. Beer's law deviations occur at higher concentrations, limiting the linear detection range and requiring dilution procedures that introduce additional measurement uncertainty. Turbidity and particulate matter in samples cause light scattering artifacts that significantly impact photometric readings.
Wavelength accuracy and bandwidth limitations in spectrophotometric instruments create systematic errors, particularly when analyzing compounds with overlapping absorption spectra. Temperature-dependent absorption coefficients and pH-sensitive chromophore behavior further complicate accurate quantification. Reagent stability issues and color development kinetics introduce time-dependent variables that affect measurement reproducibility.
Both methodologies struggle with matrix-dependent calibration requirements, where sample composition variations necessitate method-specific validation procedures. Environmental factors including humidity, electromagnetic interference, and mechanical vibrations can introduce measurement artifacts in both electrode and optical systems.
Detection limit constraints represent another shared challenge, where trace-level analyte quantification approaches the noise threshold of both techniques. Signal-to-noise ratio optimization becomes critical for maintaining accuracy at lower concentration ranges, often requiring signal averaging or extended measurement protocols that impact analytical throughput.
Standardization and traceability issues persist across both platforms, as reference material availability and inter-laboratory comparison studies reveal systematic biases that highlight fundamental accuracy limitations inherent to each detection principle.
Interference represents a critical challenge for ISE systems, as the presence of ions with similar charge characteristics can trigger cross-sensitivity responses. This selectivity coefficient variation becomes particularly problematic in complex sample matrices where multiple ionic species coexist. Additionally, electrode conditioning requirements and membrane aging effects introduce systematic errors that demand frequent calibration protocols.
Spectrophotometric detection encounters distinct accuracy challenges related to optical path variations, light source stability, and sample matrix effects. Beer's law deviations occur at higher concentrations, limiting the linear detection range and requiring dilution procedures that introduce additional measurement uncertainty. Turbidity and particulate matter in samples cause light scattering artifacts that significantly impact photometric readings.
Wavelength accuracy and bandwidth limitations in spectrophotometric instruments create systematic errors, particularly when analyzing compounds with overlapping absorption spectra. Temperature-dependent absorption coefficients and pH-sensitive chromophore behavior further complicate accurate quantification. Reagent stability issues and color development kinetics introduce time-dependent variables that affect measurement reproducibility.
Both methodologies struggle with matrix-dependent calibration requirements, where sample composition variations necessitate method-specific validation procedures. Environmental factors including humidity, electromagnetic interference, and mechanical vibrations can introduce measurement artifacts in both electrode and optical systems.
Detection limit constraints represent another shared challenge, where trace-level analyte quantification approaches the noise threshold of both techniques. Signal-to-noise ratio optimization becomes critical for maintaining accuracy at lower concentration ranges, often requiring signal averaging or extended measurement protocols that impact analytical throughput.
Standardization and traceability issues persist across both platforms, as reference material availability and inter-laboratory comparison studies reveal systematic biases that highlight fundamental accuracy limitations inherent to each detection principle.
Existing Ion Detection Solutions Comparison
01 Ion selective electrode design and membrane optimization
Improvements in ion selective electrode accuracy through optimized membrane compositions and electrode structures. This includes the development of specialized ion-selective membranes with enhanced selectivity coefficients, reduced interference from competing ions, and improved response times. Advanced membrane materials and configurations enable more precise detection of target ions in complex sample matrices.- Ion-selective electrode design and membrane optimization: Improvements in ion-selective electrode accuracy through optimized membrane compositions, electrode structures, and selective ionophores. These enhancements focus on improving selectivity, sensitivity, and response time of the electrodes for specific ion detection. Advanced membrane materials and electrode configurations reduce interference from competing ions and improve measurement precision across various concentration ranges.
- Spectrophotometric detection systems and optical components: Advanced spectrophotometric detection methods utilizing improved optical systems, light sources, and detector configurations to enhance measurement accuracy. These systems incorporate wavelength selection mechanisms, signal processing algorithms, and calibration techniques to minimize optical interference and improve detection limits. The integration of multiple wavelength detection and reference beam compensation further enhances measurement reliability.
- Calibration and standardization methods: Systematic approaches for calibrating both ion-selective electrodes and spectrophotometric instruments to ensure measurement accuracy. These methods include multi-point calibration procedures, standard reference materials, and automated calibration protocols. Temperature compensation, drift correction, and periodic recalibration strategies are employed to maintain long-term measurement stability and accuracy.
- Interference elimination and signal processing: Techniques for reducing measurement errors caused by interfering substances and signal noise in both detection methods. These include mathematical correction algorithms, background subtraction methods, and selective masking agents. Advanced signal processing approaches such as digital filtering, baseline correction, and multivariate analysis improve the accuracy of measurements in complex sample matrices.
- Integrated detection systems and comparative analysis: Combined analytical systems that utilize both ion-selective electrodes and spectrophotometry for cross-validation and enhanced accuracy. These integrated approaches allow for simultaneous or sequential measurements using multiple detection principles, enabling error detection and result verification. Automated systems incorporate data comparison algorithms and quality control protocols to ensure measurement reliability across different analytical techniques.
02 Spectrophotometric calibration and measurement techniques
Enhanced spectrophotometry detection accuracy through advanced calibration methods, wavelength optimization, and signal processing algorithms. This involves the use of multi-wavelength analysis, baseline correction techniques, and automated calibration procedures to minimize measurement errors. Improved optical components and detector sensitivity contribute to more accurate quantitative analysis across various concentration ranges.Expand Specific Solutions03 Combined detection systems integrating multiple analytical methods
Integration of ion selective electrodes with spectrophotometric detection to provide complementary analytical data and cross-validation of results. These hybrid systems leverage the strengths of both techniques to achieve superior accuracy through simultaneous or sequential measurements. The combination allows for internal standardization and correction of matrix effects that may affect individual detection methods.Expand Specific Solutions04 Signal processing and interference correction algorithms
Advanced data processing methods to improve detection accuracy by compensating for background noise, drift, and interfering substances. This includes mathematical algorithms for baseline correction, signal averaging, and multivariate calibration techniques. Digital signal processing enhances the precision of both electrochemical and optical measurements by filtering out systematic errors and improving signal-to-noise ratios.Expand Specific Solutions05 Quality control and validation methods for analytical accuracy
Systematic approaches to ensure and validate the accuracy of ion selective electrode and spectrophotometric measurements through standardized protocols, reference materials, and statistical analysis. This encompasses method validation procedures, precision testing, accuracy assessment using certified reference standards, and inter-laboratory comparison studies to establish measurement reliability and traceability.Expand Specific Solutions
Key Players in Analytical Instrumentation Industry
The ion selective electrode versus spectrophotometry detection accuracy landscape represents a mature analytical chemistry market experiencing steady growth driven by increasing demand for precise analytical measurements across pharmaceutical, environmental, and clinical applications. The market demonstrates significant scale with established players like Shimadzu Corp., Thermo Fisher Scientific, and Beckman Coulter dominating through comprehensive analytical instrument portfolios. Technology maturity varies considerably, with spectrophotometry representing well-established methodology while ion selective electrode technology continues advancing through innovations from companies like Unisense A/S and Oxford Biosensors Ltd. Major manufacturers including Hitachi High-Tech America, Roche Diagnostics, and Hamamatsu Photonics are driving technological convergence by integrating both detection methods into hybrid analytical platforms. The competitive landscape shows consolidation around companies offering complete analytical solutions, with emerging players like SIONEX Corp. focusing on specialized sensor technologies, while academic institutions such as EPFL and Fudan University contribute fundamental research advancing detection accuracy capabilities.
Shimadzu Corp.
Technical Solution: Shimadzu has developed sophisticated spectrophotometric systems including UV-Vis spectrophotometers with detection capabilities reaching sub-ppm levels and measurement precision of ±0.02% transmittance. Their systems incorporate advanced optical designs with double-beam configurations to minimize baseline drift and environmental interference. The company also produces ion selective electrodes with proprietary membrane formulations that demonstrate enhanced selectivity coefficients and reduced cross-sensitivity to interfering ions. Their analytical solutions often integrate both methodologies to provide comprehensive detection capabilities, particularly in pharmaceutical quality control and environmental analysis where accuracy validation is critical.
Strengths: High precision optical systems, innovative membrane technology for ISE, strong presence in pharmaceutical markets. Weaknesses: Limited real-time monitoring capabilities, higher maintenance requirements for optical components.
Hitachi High-Tech America, Inc.
Technical Solution: Hitachi High-Tech specializes in advanced spectrophotometric instrumentation with their atomic absorption and UV-Vis systems achieving detection limits in the ppb range for trace metal analysis. Their spectrophotometers feature automated sample handling systems and sophisticated background correction techniques that enhance measurement accuracy to within ±1% for most analytical applications. The company has also developed ion selective electrode arrays for multi-parameter analysis, incorporating microelectronic fabrication techniques to create miniaturized sensor platforms. These systems are particularly effective in semiconductor manufacturing quality control and environmental monitoring where both high accuracy and rapid throughput are essential requirements.
Strengths: Advanced automation capabilities, excellent trace analysis performance, strong semiconductor industry expertise. Weaknesses: Limited portable solutions, higher complexity in operation and maintenance.
Core Innovations in ISE and Spectrophotometry
Electrolyte Concentration Measuring Apparatus and Measuring Method Using Same
PatentActiveUS20160054257A1
Innovation
- An electrolyte concentration measuring apparatus and method that includes multiple ion selective electrodes and a reference electrode, with a potential measuring unit to measure voltage and current, and a resistance measuring unit to determine direct-current resistance, thereby stabilizing the interface potential and reducing errors.
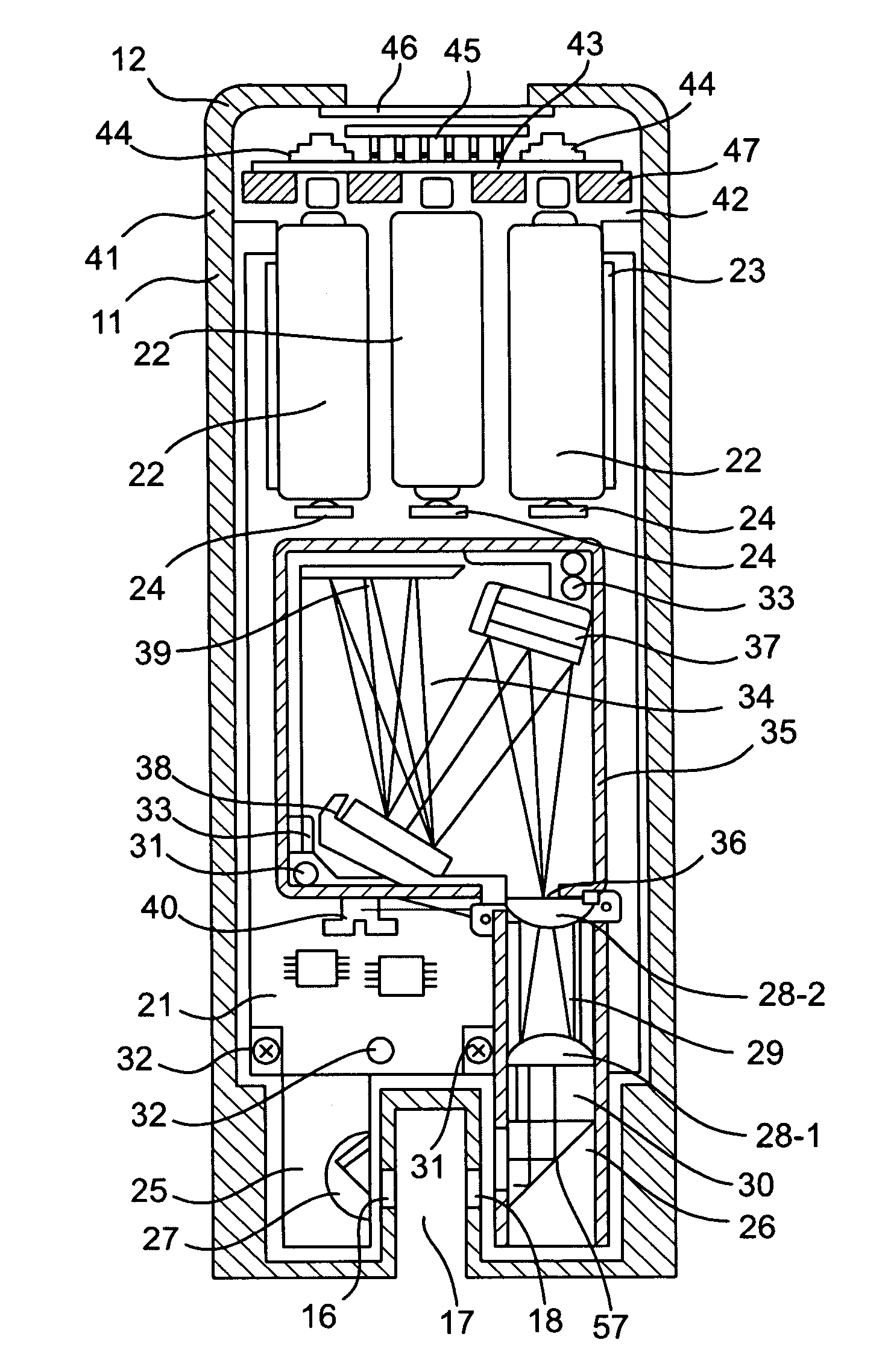
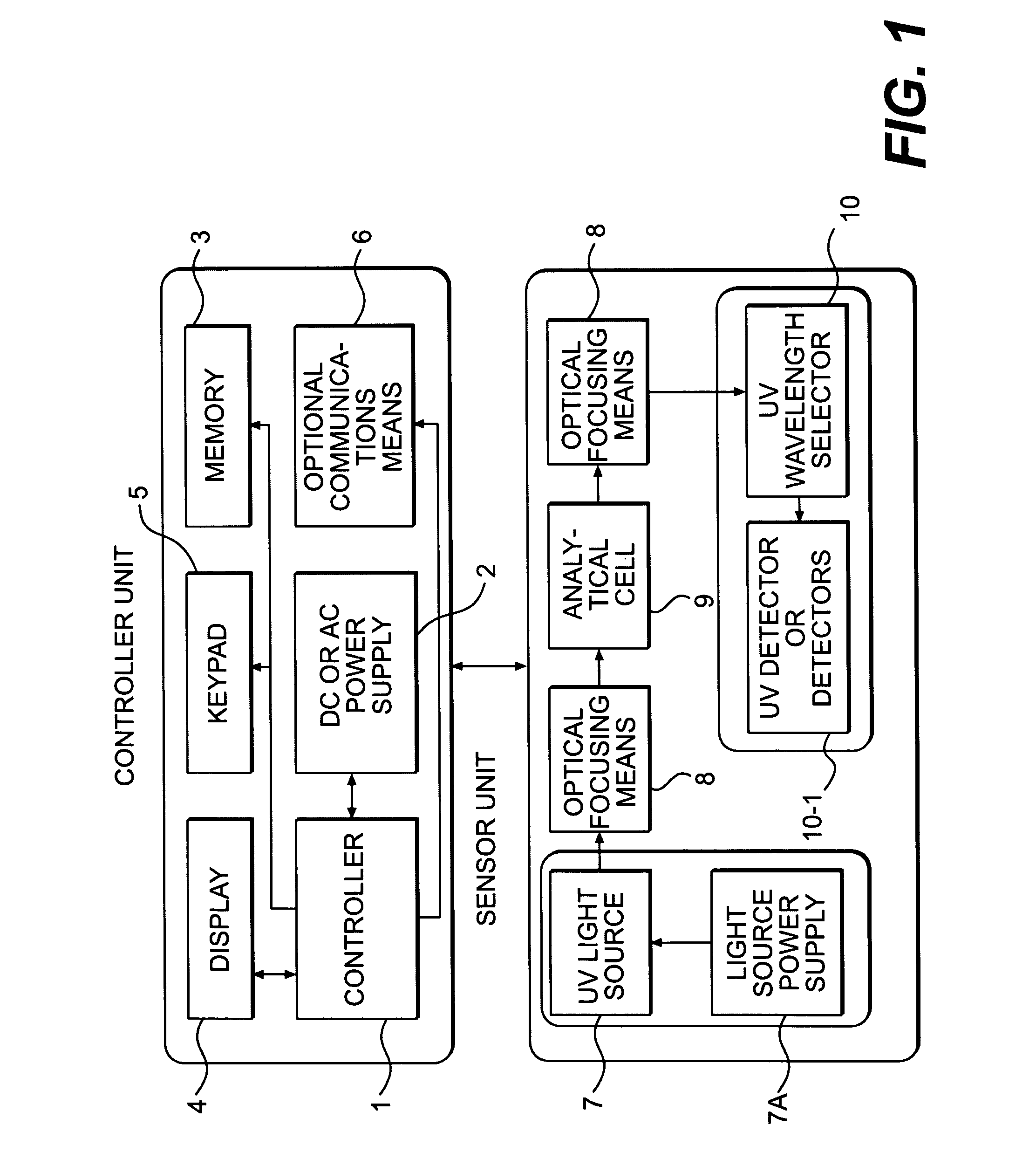
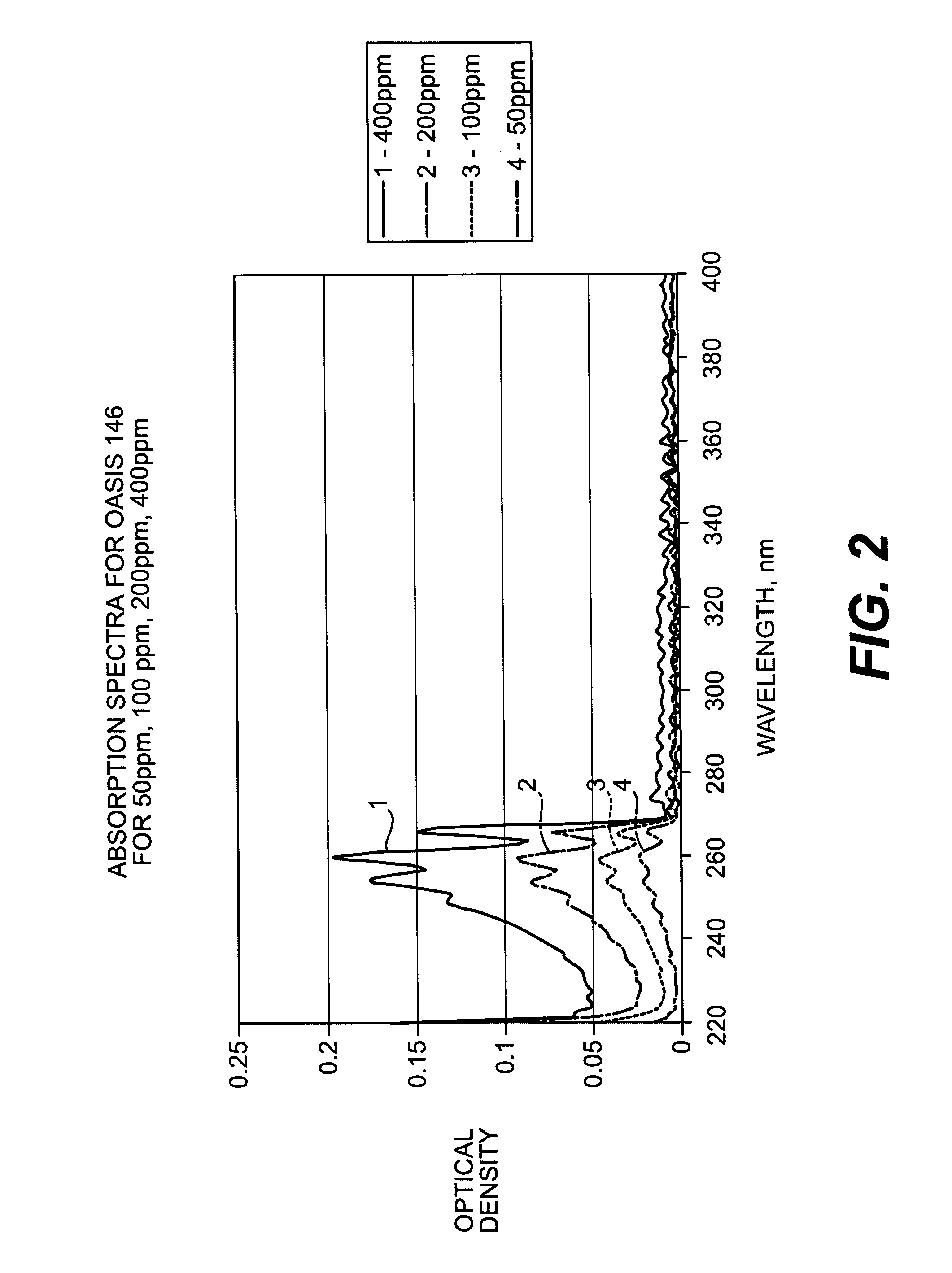
Near UV absorption spectrometer and method for using the same
PatentInactiveUS20070138401A1
Innovation
- A portable near UV absorption spectrometer measures the absorbance properties of quats over specific wavelengths, allowing for direct, automatic, and continuous monitoring of quat concentrations in sanitizing solutions using a handheld device with a UV light source and dispersive system, transforming output signals into absorbance values to calculate concentrations.
Analytical Method Validation Standards
Analytical method validation represents a critical framework for establishing the reliability and accuracy of measurement techniques in analytical chemistry. The validation process encompasses systematic evaluation of method performance characteristics, including precision, accuracy, specificity, linearity, range, detection limits, and robustness. These parameters form the foundation for regulatory compliance and scientific acceptance of analytical procedures.
International standards organizations have established comprehensive guidelines for method validation. The International Conference on Harmonisation (ICH) Q2(R1) provides detailed requirements for validation of analytical procedures used in pharmaceutical applications. Similarly, the Association of Official Analytical Chemists (AOAC) International has developed validation protocols specifically addressing various analytical matrices and measurement techniques. The United States Pharmacopeia (USP) and European Pharmacopoeia (EP) maintain stringent validation criteria that must be met before analytical methods can be implemented in regulated environments.
For ion selective electrode methodologies, validation standards emphasize the importance of electrode selectivity coefficients, response time, and interference studies. The electrode's Nernstian response must be demonstrated across the intended concentration range, with particular attention to temperature effects and ionic strength variations. Calibration curve linearity typically requires correlation coefficients exceeding 0.995, while precision studies must demonstrate relative standard deviations below acceptable thresholds.
Spectrophotometric method validation focuses on Beer's law adherence, wavelength accuracy, and photometric linearity. Standards require demonstration of method specificity through spectral purity assessments and interference testing. The validation protocol must address potential matrix effects, solvent compatibility, and instrument-to-instrument variability. Accuracy verification typically involves certified reference materials or standard addition techniques.
Regulatory agencies mandate that validation studies include statistical analysis of results, with appropriate confidence intervals and significance testing. Documentation requirements encompass detailed protocols, raw data, statistical calculations, and comprehensive validation reports. These standards ensure that analytical methods provide reliable, reproducible results suitable for their intended applications, whether in quality control, environmental monitoring, or clinical diagnostics.
International standards organizations have established comprehensive guidelines for method validation. The International Conference on Harmonisation (ICH) Q2(R1) provides detailed requirements for validation of analytical procedures used in pharmaceutical applications. Similarly, the Association of Official Analytical Chemists (AOAC) International has developed validation protocols specifically addressing various analytical matrices and measurement techniques. The United States Pharmacopeia (USP) and European Pharmacopoeia (EP) maintain stringent validation criteria that must be met before analytical methods can be implemented in regulated environments.
For ion selective electrode methodologies, validation standards emphasize the importance of electrode selectivity coefficients, response time, and interference studies. The electrode's Nernstian response must be demonstrated across the intended concentration range, with particular attention to temperature effects and ionic strength variations. Calibration curve linearity typically requires correlation coefficients exceeding 0.995, while precision studies must demonstrate relative standard deviations below acceptable thresholds.
Spectrophotometric method validation focuses on Beer's law adherence, wavelength accuracy, and photometric linearity. Standards require demonstration of method specificity through spectral purity assessments and interference testing. The validation protocol must address potential matrix effects, solvent compatibility, and instrument-to-instrument variability. Accuracy verification typically involves certified reference materials or standard addition techniques.
Regulatory agencies mandate that validation studies include statistical analysis of results, with appropriate confidence intervals and significance testing. Documentation requirements encompass detailed protocols, raw data, statistical calculations, and comprehensive validation reports. These standards ensure that analytical methods provide reliable, reproducible results suitable for their intended applications, whether in quality control, environmental monitoring, or clinical diagnostics.
Cost-Benefit Analysis of Detection Technologies
The economic evaluation of ion selective electrode (ISE) and spectrophotometry technologies reveals significant differences in initial capital requirements and operational expenditures. ISE systems typically demand lower upfront investments, with basic electrode setups ranging from $2,000 to $15,000 depending on the target analyte and precision requirements. In contrast, spectrophotometric instruments, particularly advanced UV-Vis and atomic absorption systems, require initial investments between $20,000 and $150,000 for high-end configurations.
Operational cost structures demonstrate contrasting patterns between these technologies. ISE systems incur relatively low maintenance costs but require periodic electrode replacement every 6-24 months, with replacement costs varying from $200 to $2,000 per electrode. Calibration procedures are straightforward and consume minimal reagents, resulting in annual operational costs typically under $5,000 for routine applications.
Spectrophotometry presents higher operational complexity with substantial reagent consumption, specialized sample preparation requirements, and more frequent calibration needs. Annual operational costs often exceed $15,000 when factoring in consumables, maintenance contracts, and skilled operator requirements. However, the versatility of spectrophotometric systems enables multi-analyte detection capabilities, potentially improving cost-effectiveness in laboratories requiring diverse analytical capabilities.
Labor cost considerations favor ISE technology for routine monitoring applications. The simplified operation protocols and minimal sample preparation reduce skilled technician requirements, translating to lower personnel costs. Spectrophotometry demands more specialized training and longer analysis times, increasing labor-related expenses by approximately 40-60% compared to ISE implementations.
Long-term economic sustainability analysis indicates that ISE systems achieve cost-effectiveness advantages in high-throughput, single-analyte applications within 18-24 months of deployment. Spectrophotometry demonstrates superior economic value in research environments and multi-parameter analysis scenarios, where the higher initial investment is offset by analytical versatility and broader application scope over 3-5 year operational periods.
Operational cost structures demonstrate contrasting patterns between these technologies. ISE systems incur relatively low maintenance costs but require periodic electrode replacement every 6-24 months, with replacement costs varying from $200 to $2,000 per electrode. Calibration procedures are straightforward and consume minimal reagents, resulting in annual operational costs typically under $5,000 for routine applications.
Spectrophotometry presents higher operational complexity with substantial reagent consumption, specialized sample preparation requirements, and more frequent calibration needs. Annual operational costs often exceed $15,000 when factoring in consumables, maintenance contracts, and skilled operator requirements. However, the versatility of spectrophotometric systems enables multi-analyte detection capabilities, potentially improving cost-effectiveness in laboratories requiring diverse analytical capabilities.
Labor cost considerations favor ISE technology for routine monitoring applications. The simplified operation protocols and minimal sample preparation reduce skilled technician requirements, translating to lower personnel costs. Spectrophotometry demands more specialized training and longer analysis times, increasing labor-related expenses by approximately 40-60% compared to ISE implementations.
Long-term economic sustainability analysis indicates that ISE systems achieve cost-effectiveness advantages in high-throughput, single-analyte applications within 18-24 months of deployment. Spectrophotometry demonstrates superior economic value in research environments and multi-parameter analysis scenarios, where the higher initial investment is offset by analytical versatility and broader application scope over 3-5 year operational periods.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!