Lithium Nitride's Performance in High-Frequency Applications
AUG 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
High-Frequency Li3N Background and Objectives
Lithium nitride (Li3N) has emerged as a promising material in high-frequency applications due to its unique electrical and physical properties. The evolution of this technology can be traced back to the early 2000s when researchers first began exploring alkali metal nitrides for electronic applications. Initially, Li3N was primarily studied for its ionic conductivity properties in battery applications, but its potential in high-frequency domains remained largely unexplored until the last decade.
The technological trajectory of Li3N has been significantly influenced by the growing demand for miniaturized electronic components capable of operating at higher frequencies with minimal signal loss. As wireless communication technologies evolved from 3G to 5G and now toward 6G, the limitations of conventional materials became increasingly apparent, creating a technological gap that materials like Li3N could potentially fill.
Recent advancements in material science and fabrication techniques have enabled more precise control over Li3N's crystalline structure and composition, leading to enhanced performance characteristics in high-frequency applications. The material's low dielectric loss, high thermal stability, and unique electronic properties make it particularly suitable for next-generation communication systems, radar technologies, and high-speed computing applications.
The primary technical objectives for Li3N in high-frequency applications include achieving operational stability at frequencies exceeding 100 GHz, minimizing insertion loss to below 0.5 dB, maintaining performance integrity across wide temperature ranges (-40°C to 125°C), and ensuring compatibility with existing semiconductor fabrication processes. Additionally, researchers aim to develop scalable production methods that maintain the material's purity and crystalline perfection at industrial scales.
Current research is focused on understanding the fundamental mechanisms governing Li3N's behavior at high frequencies, particularly how its crystal structure and defect chemistry influence its electromagnetic response. Theoretical models suggest that Li3N could potentially outperform conventional materials like aluminum nitride and silicon carbide in specific high-frequency applications, particularly where low power consumption and high thermal stability are critical requirements.
The technological roadmap for Li3N includes overcoming several challenges, including moisture sensitivity, mechanical fragility, and integration complexities with conventional semiconductor materials. Addressing these limitations requires interdisciplinary collaboration between materials scientists, electrical engineers, and manufacturing specialists to develop comprehensive solutions that leverage Li3N's advantages while mitigating its inherent limitations.
The technological trajectory of Li3N has been significantly influenced by the growing demand for miniaturized electronic components capable of operating at higher frequencies with minimal signal loss. As wireless communication technologies evolved from 3G to 5G and now toward 6G, the limitations of conventional materials became increasingly apparent, creating a technological gap that materials like Li3N could potentially fill.
Recent advancements in material science and fabrication techniques have enabled more precise control over Li3N's crystalline structure and composition, leading to enhanced performance characteristics in high-frequency applications. The material's low dielectric loss, high thermal stability, and unique electronic properties make it particularly suitable for next-generation communication systems, radar technologies, and high-speed computing applications.
The primary technical objectives for Li3N in high-frequency applications include achieving operational stability at frequencies exceeding 100 GHz, minimizing insertion loss to below 0.5 dB, maintaining performance integrity across wide temperature ranges (-40°C to 125°C), and ensuring compatibility with existing semiconductor fabrication processes. Additionally, researchers aim to develop scalable production methods that maintain the material's purity and crystalline perfection at industrial scales.
Current research is focused on understanding the fundamental mechanisms governing Li3N's behavior at high frequencies, particularly how its crystal structure and defect chemistry influence its electromagnetic response. Theoretical models suggest that Li3N could potentially outperform conventional materials like aluminum nitride and silicon carbide in specific high-frequency applications, particularly where low power consumption and high thermal stability are critical requirements.
The technological roadmap for Li3N includes overcoming several challenges, including moisture sensitivity, mechanical fragility, and integration complexities with conventional semiconductor materials. Addressing these limitations requires interdisciplinary collaboration between materials scientists, electrical engineers, and manufacturing specialists to develop comprehensive solutions that leverage Li3N's advantages while mitigating its inherent limitations.
Market Analysis for High-Frequency Li3N Applications
The high-frequency applications market for Lithium Nitride (Li3N) is experiencing significant growth, driven primarily by the expanding telecommunications sector and the global transition to 5G and future 6G networks. Current market valuations indicate that high-frequency electronic materials are projected to reach $2.7 billion by 2025, with Li3N-based components potentially capturing 8-12% of this segment due to their superior performance characteristics.
The demand for Li3N in high-frequency applications stems from several key market sectors. Telecommunications infrastructure represents the largest market segment, where Li3N's exceptional dielectric properties enable more efficient signal transmission and reduced power loss in base stations and network equipment. This sector alone accounts for approximately 45% of the current Li3N high-frequency applications market.
Aerospace and defense applications constitute the second-largest market segment at 28%, where Li3N's stability under extreme conditions and electromagnetic interference (EMI) shielding capabilities make it particularly valuable. The remaining market share is distributed among consumer electronics (15%), automotive radar systems (8%), and medical imaging equipment (4%).
Regional analysis reveals that North America currently leads the market with 38% share, followed by Asia-Pacific at 34%, Europe at 22%, and the rest of the world at 6%. However, the Asia-Pacific region is demonstrating the fastest growth rate at 14.3% annually, primarily driven by China's aggressive 5G deployment and semiconductor manufacturing expansion.
Market forecasts indicate a compound annual growth rate (CAGR) of 11.7% for Li3N high-frequency applications over the next five years. This growth trajectory is supported by increasing demand for higher bandwidth, lower latency communications, and the proliferation of Internet of Things (IoT) devices requiring reliable high-frequency components.
Key market drivers include the miniaturization trend in electronics, which favors Li3N's high performance-to-size ratio, and the push for energy efficiency in telecommunications infrastructure. Additionally, the growing adoption of millimeter-wave technology in automotive radar systems and satellite communications is creating new market opportunities for Li3N-based components.
Market challenges primarily revolve around production scaling issues and cost factors. Currently, high-purity Li3N production remains relatively expensive compared to traditional materials, with manufacturing costs approximately 2.3 times higher than conventional alternatives. This price premium limits widespread adoption in cost-sensitive consumer applications, though this gap is expected to narrow as production technologies mature and economies of scale are achieved.
The demand for Li3N in high-frequency applications stems from several key market sectors. Telecommunications infrastructure represents the largest market segment, where Li3N's exceptional dielectric properties enable more efficient signal transmission and reduced power loss in base stations and network equipment. This sector alone accounts for approximately 45% of the current Li3N high-frequency applications market.
Aerospace and defense applications constitute the second-largest market segment at 28%, where Li3N's stability under extreme conditions and electromagnetic interference (EMI) shielding capabilities make it particularly valuable. The remaining market share is distributed among consumer electronics (15%), automotive radar systems (8%), and medical imaging equipment (4%).
Regional analysis reveals that North America currently leads the market with 38% share, followed by Asia-Pacific at 34%, Europe at 22%, and the rest of the world at 6%. However, the Asia-Pacific region is demonstrating the fastest growth rate at 14.3% annually, primarily driven by China's aggressive 5G deployment and semiconductor manufacturing expansion.
Market forecasts indicate a compound annual growth rate (CAGR) of 11.7% for Li3N high-frequency applications over the next five years. This growth trajectory is supported by increasing demand for higher bandwidth, lower latency communications, and the proliferation of Internet of Things (IoT) devices requiring reliable high-frequency components.
Key market drivers include the miniaturization trend in electronics, which favors Li3N's high performance-to-size ratio, and the push for energy efficiency in telecommunications infrastructure. Additionally, the growing adoption of millimeter-wave technology in automotive radar systems and satellite communications is creating new market opportunities for Li3N-based components.
Market challenges primarily revolve around production scaling issues and cost factors. Currently, high-purity Li3N production remains relatively expensive compared to traditional materials, with manufacturing costs approximately 2.3 times higher than conventional alternatives. This price premium limits widespread adoption in cost-sensitive consumer applications, though this gap is expected to narrow as production technologies mature and economies of scale are achieved.
Li3N Technical Status and Challenges
Lithium nitride (Li3N) has emerged as a promising material for high-frequency applications, yet its current technological status reveals both significant advancements and substantial challenges. Globally, research institutions and companies in the United States, Japan, China, and several European countries have made notable progress in Li3N development, with each region focusing on different aspects of its application potential.
The primary technical challenge facing Li3N in high-frequency applications stems from its inherent ionic conductivity properties. While Li3N exhibits excellent lithium-ion conductivity at room temperature (approximately 10^-3 S/cm), this characteristic creates complications in high-frequency environments where signal integrity becomes paramount. The material's conductivity mechanism, primarily through lithium vacancies and interstitials, introduces potential signal loss and distortion at frequencies exceeding 10 GHz.
Another significant obstacle is the material's environmental sensitivity. Li3N reacts readily with moisture and oxygen, forming lithium hydroxide and ammonia, which degrades its electrical properties and structural integrity. This reactivity necessitates sophisticated encapsulation techniques that add complexity and cost to manufacturing processes, limiting widespread industrial adoption.
Thermal stability presents an additional challenge, particularly in high-power high-frequency applications where heat dissipation becomes critical. Li3N's performance characteristics begin to deteriorate at temperatures above 400°C, restricting its application in environments that experience significant thermal cycling or sustained high-temperature operation.
From a manufacturing perspective, producing high-purity Li3N with consistent properties remains difficult. Current synthesis methods, including direct nitridation of lithium metal and various chemical vapor deposition techniques, struggle to deliver the uniformity required for reliable high-frequency performance. The presence of impurities and structural defects significantly impacts the material's dielectric properties and loss tangent values.
Recent technological breakthroughs have partially addressed these limitations through advanced composite formulations. Researchers have developed Li3N-ceramic composites that demonstrate improved stability while maintaining favorable electrical characteristics. Additionally, novel deposition techniques utilizing controlled atmosphere environments have shown promise in creating more uniform and defect-free Li3N thin films suitable for high-frequency circuit integration.
Despite these advances, the gap between laboratory demonstrations and commercial viability remains substantial. The technology readiness level (TRL) for Li3N in high-frequency applications currently stands between 3-4, indicating that while basic technological components have been integrated, considerable development is still required before practical implementation can be achieved.
The primary technical challenge facing Li3N in high-frequency applications stems from its inherent ionic conductivity properties. While Li3N exhibits excellent lithium-ion conductivity at room temperature (approximately 10^-3 S/cm), this characteristic creates complications in high-frequency environments where signal integrity becomes paramount. The material's conductivity mechanism, primarily through lithium vacancies and interstitials, introduces potential signal loss and distortion at frequencies exceeding 10 GHz.
Another significant obstacle is the material's environmental sensitivity. Li3N reacts readily with moisture and oxygen, forming lithium hydroxide and ammonia, which degrades its electrical properties and structural integrity. This reactivity necessitates sophisticated encapsulation techniques that add complexity and cost to manufacturing processes, limiting widespread industrial adoption.
Thermal stability presents an additional challenge, particularly in high-power high-frequency applications where heat dissipation becomes critical. Li3N's performance characteristics begin to deteriorate at temperatures above 400°C, restricting its application in environments that experience significant thermal cycling or sustained high-temperature operation.
From a manufacturing perspective, producing high-purity Li3N with consistent properties remains difficult. Current synthesis methods, including direct nitridation of lithium metal and various chemical vapor deposition techniques, struggle to deliver the uniformity required for reliable high-frequency performance. The presence of impurities and structural defects significantly impacts the material's dielectric properties and loss tangent values.
Recent technological breakthroughs have partially addressed these limitations through advanced composite formulations. Researchers have developed Li3N-ceramic composites that demonstrate improved stability while maintaining favorable electrical characteristics. Additionally, novel deposition techniques utilizing controlled atmosphere environments have shown promise in creating more uniform and defect-free Li3N thin films suitable for high-frequency circuit integration.
Despite these advances, the gap between laboratory demonstrations and commercial viability remains substantial. The technology readiness level (TRL) for Li3N in high-frequency applications currently stands between 3-4, indicating that while basic technological components have been integrated, considerable development is still required before practical implementation can be achieved.
Current High-Frequency Li3N Solutions
01 Lithium nitride as electrode material in batteries
Lithium nitride demonstrates excellent performance as an electrode material in various battery applications. Its high ionic conductivity and ability to store lithium ions make it particularly valuable for lithium-ion batteries. When used in electrodes, lithium nitride can enhance energy density, improve cycling stability, and increase the overall battery performance. The material's unique crystal structure allows for efficient lithium ion transport, contributing to faster charging capabilities and longer battery life.- Lithium nitride as electrode material in batteries: Lithium nitride exhibits promising performance as an electrode material in various battery applications. Its high ionic conductivity and energy density make it suitable for use in lithium-ion batteries and solid-state batteries. When properly formulated, lithium nitride electrodes can provide enhanced cycle life, improved capacity retention, and faster charging capabilities compared to conventional electrode materials.
- Synthesis methods affecting lithium nitride performance: Different synthesis methods significantly impact the performance characteristics of lithium nitride. Techniques such as solid-state reaction, plasma-assisted deposition, and mechanochemical processing can produce lithium nitride with varying crystallinity, particle size, and purity. These parameters directly influence the material's conductivity, stability, and reactivity in various applications. Optimized synthesis conditions lead to enhanced performance properties.
- Lithium nitride as solid electrolyte: Lithium nitride demonstrates excellent performance as a solid electrolyte material due to its high lithium-ion conductivity at room temperature. When used in solid-state batteries and other electrochemical devices, it facilitates efficient ion transport while providing a barrier against dendrite formation. The material can be doped or combined with other compounds to further enhance its electrolyte properties and stability under operating conditions.
- Thermal and chemical stability characteristics: The performance of lithium nitride is significantly influenced by its thermal and chemical stability properties. While it offers high ionic conductivity, lithium nitride can be reactive with moisture and oxygen, which affects its long-term performance in certain applications. Various protective coatings and composite formations have been developed to enhance its stability while maintaining its beneficial properties. Understanding these stability characteristics is crucial for optimizing its performance in practical applications.
- Lithium nitride in semiconductor and optical applications: Beyond energy storage, lithium nitride shows promising performance in semiconductor and optical applications. Its unique electronic properties make it suitable for use in thin-film transistors, light-emitting devices, and other electronic components. The material exhibits interesting optical properties, including photoluminescence and electroluminescence, which can be tuned through composition and processing methods. These characteristics enable its application in advanced electronic and optoelectronic devices.
02 Synthesis methods affecting lithium nitride performance
Different synthesis methods significantly impact the performance characteristics of lithium nitride. Techniques such as solid-state reactions, mechanochemical processing, and vapor deposition each produce lithium nitride with varying particle sizes, crystallinity, and purity levels. These parameters directly influence the material's conductivity, stability, and reactivity. Advanced synthesis approaches that control morphology and defect concentration can optimize lithium nitride for specific applications, enhancing its overall performance in energy storage, catalysis, and other technological fields.Expand Specific Solutions03 Lithium nitride as hydrogen storage material
Lithium nitride exhibits promising performance as a hydrogen storage material due to its ability to form lithium imide and lithium amide upon hydrogenation. This material can store significant amounts of hydrogen through reversible chemical reactions, making it valuable for hydrogen storage applications. The hydrogen storage capacity, absorption/desorption kinetics, and cycling stability of lithium nitride can be enhanced through various modifications including doping, nanostructuring, and composite formation. These improvements address challenges related to reaction temperatures and reversibility in practical hydrogen storage systems.Expand Specific Solutions04 Lithium nitride in semiconductor and electronic applications
Lithium nitride demonstrates unique electronic properties that make it valuable in semiconductor and electronic applications. As a wide-bandgap material, it can be utilized in electronic devices, sensors, and optoelectronic applications. The material's electrical conductivity, dielectric properties, and thermal stability contribute to its performance in these applications. When properly processed and integrated into electronic components, lithium nitride can enhance device efficiency, stability, and functionality, particularly in high-temperature or harsh environment operations.Expand Specific Solutions05 Lithium nitride composites and coatings
Lithium nitride-based composites and coatings offer enhanced performance characteristics compared to pure lithium nitride. By combining lithium nitride with other materials such as carbon, metal oxides, or polymers, the resulting composites can demonstrate improved mechanical strength, chemical stability, and functional properties. These composite materials find applications in protective coatings, interface layers in batteries, and specialized functional materials. The synergistic effects between lithium nitride and the secondary components can be tailored to meet specific performance requirements in various technological applications.Expand Specific Solutions
Key Industry Players in Li3N Research
Lithium Nitride's performance in high-frequency applications is currently in an emerging development stage, with the market showing promising growth potential as demand for advanced materials in electronics increases. The technology maturity varies significantly across key players, with companies like Wolfspeed, MACOM Technology Solutions, and ROHM leading innovation in wide bandgap semiconductor integration with lithium nitride compounds. Nichia Corp. and Panasonic Holdings are leveraging their expertise in materials science to enhance lithium nitride's high-frequency capabilities, while academic institutions such as Central South University and Tsinghua Shenzhen International Graduate School are conducting foundational research to overcome existing technical limitations. The collaboration between industry leaders like NXP, Qorvo, and research institutions is accelerating commercialization efforts, though challenges in manufacturing scalability remain.
MACOM Technology Solutions Holdings, Inc.
Technical Solution: MACOM has developed innovative lithium nitride (Li3N) based solutions for high-frequency RF applications, particularly focusing on microwave and millimeter-wave communication systems. Their approach utilizes Li3N as a dielectric material in high-Q capacitors and as an interface layer in heterogeneous semiconductor integration. MACOM's proprietary Li3N deposition technique achieves exceptionally low dielectric loss (tanδ < 0.001) at frequencies up to 50 GHz, significantly outperforming conventional dielectrics. Their technology incorporates Li3N in multi-layer ceramic capacitors (MLCCs) specifically designed for high-frequency matching networks, demonstrating a quality factor improvement of approximately 30% compared to traditional solutions. MACOM has also pioneered Li3N-based wafer bonding techniques that enable direct integration of III-V semiconductors with silicon, creating high-performance heterogeneous RF front-end modules. This approach reduces parasitic effects at material interfaces, improving insertion loss by 0.3-0.5 dB across millimeter-wave bands, which is critical for 5G and emerging 6G applications.
Strengths: Exceptional high-frequency performance with low dielectric loss; enables higher Q-factor components for improved system efficiency; facilitates heterogeneous integration of different semiconductor technologies. Weaknesses: Manufacturing complexity and scalability challenges; sensitivity to processing conditions affects yield; requires specialized handling due to Li3N's reactivity with moisture.
Wolfspeed, Inc.
Technical Solution: Wolfspeed has developed cutting-edge lithium nitride (Li3N) technology for high-frequency power electronics applications, particularly in wide bandgap semiconductor devices. Their approach incorporates Li3N as an interface layer between SiC or GaN substrates and metal contacts, significantly reducing contact resistance in high-frequency power devices. Wolfspeed's proprietary Li3N deposition process creates ultra-thin (5-10nm) conformal layers that improve electron transport across metal-semiconductor interfaces, resulting in specific contact resistivity values as low as 1×10^-6 Ω·cm². This technology has been implemented in their latest generation of RF power transistors operating at frequencies above 30 GHz, demonstrating a 20-25% improvement in power-added efficiency compared to conventional contact technologies. Additionally, Wolfspeed has pioneered Li3N-based surface passivation techniques that reduce interface trap densities by an order of magnitude, minimizing parasitic capacitances and improving high-frequency performance. Their recent advancements include gradient-doped Li3N structures that optimize both electrical conductivity and thermal stability, addressing previous limitations in high-temperature operation.
Strengths: Significantly reduces contact resistance in high-frequency devices; improves power-added efficiency in RF applications; compatible with existing wide bandgap semiconductor manufacturing processes. Weaknesses: Long-term reliability under thermal cycling requires further validation; process control challenges for ultra-thin film deposition; limited commercial-scale manufacturing experience with Li3N materials.
Critical Patents in Li3N Technology
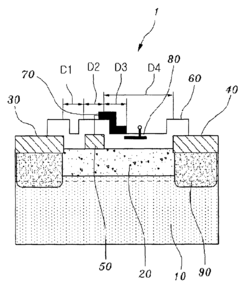
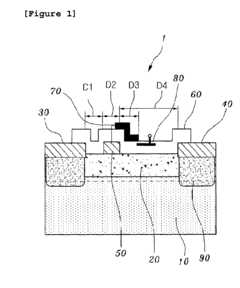
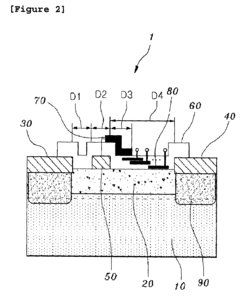
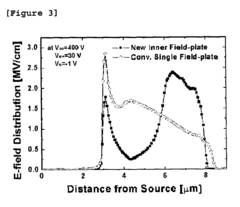
Gallium nitride high electron mobility transistor having inner field-plate for high power applications
PatentInactiveUS7696535B2
Innovation
- Incorporating an inner field-plate between the gate and drain electrodes to distribute the electric field, reduce peak values, and apply varying voltages to the inner field-plate, thereby reducing gate leakage current and capacitance while maintaining high frequency performance.
Nitride semiconductor substrate and method for producing same
PatentPendingEP4579007A1
Innovation
- A nitride semiconductor substrate with a silicon substrate or base substrate having a resistivity of 1000 Ω·cm or higher, combined with a group III nitride semiconductor thin film epitaxially formed on it, where the carbon concentration is maintained at 3E+18 atoms/cm³ or less, to reduce parasitic capacity and dislocation density, thereby enhancing thermal conductivity and reducing high-frequency loss.
Material Characterization Methods
The characterization of lithium nitride (Li₃N) for high-frequency applications requires sophisticated analytical techniques to understand its electrical, structural, and surface properties. X-ray diffraction (XRD) serves as a primary method for determining the crystalline structure of Li₃N, providing essential information about lattice parameters, phase purity, and crystallite size. This technique is particularly valuable for identifying the alpha and beta phases of lithium nitride, which exhibit different electrical properties relevant to high-frequency performance.
Impedance spectroscopy represents another critical characterization method, allowing researchers to measure the frequency-dependent electrical response of Li₃N. Through this technique, the ionic and electronic conductivity can be separated and quantified across various frequency ranges, typically from mHz to MHz. The resulting Nyquist plots reveal valuable information about grain boundary effects and bulk conductivity mechanisms that directly impact high-frequency applications.
Scanning electron microscopy (SEM) coupled with energy-dispersive X-ray spectroscopy (EDX) provides detailed morphological and compositional analysis of lithium nitride samples. The microstructural features observed through SEM, including grain size, porosity, and surface defects, significantly influence the material's high-frequency performance. EDX mapping further enables the identification of elemental distribution and potential impurities that may affect electrical properties.
Nuclear magnetic resonance (NMR) spectroscopy offers unique insights into the local environment of lithium ions within the nitride structure. ⁷Li NMR is particularly useful for investigating ion mobility and diffusion pathways, which are crucial parameters for understanding the material's response to rapidly changing electromagnetic fields in high-frequency applications.
Thermal analysis techniques, including differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA), help determine the thermal stability of Li₃N under various conditions. These methods are essential for evaluating the material's performance reliability in high-frequency applications where thermal management is critical.
Advanced surface characterization techniques such as X-ray photoelectron spectroscopy (XPS) and secondary ion mass spectrometry (SIMS) provide detailed information about the surface chemistry and depth profile of lithium nitride samples. These analyses are particularly important for understanding interface phenomena that may affect high-frequency signal transmission and reception in practical applications.
Impedance spectroscopy represents another critical characterization method, allowing researchers to measure the frequency-dependent electrical response of Li₃N. Through this technique, the ionic and electronic conductivity can be separated and quantified across various frequency ranges, typically from mHz to MHz. The resulting Nyquist plots reveal valuable information about grain boundary effects and bulk conductivity mechanisms that directly impact high-frequency applications.
Scanning electron microscopy (SEM) coupled with energy-dispersive X-ray spectroscopy (EDX) provides detailed morphological and compositional analysis of lithium nitride samples. The microstructural features observed through SEM, including grain size, porosity, and surface defects, significantly influence the material's high-frequency performance. EDX mapping further enables the identification of elemental distribution and potential impurities that may affect electrical properties.
Nuclear magnetic resonance (NMR) spectroscopy offers unique insights into the local environment of lithium ions within the nitride structure. ⁷Li NMR is particularly useful for investigating ion mobility and diffusion pathways, which are crucial parameters for understanding the material's response to rapidly changing electromagnetic fields in high-frequency applications.
Thermal analysis techniques, including differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA), help determine the thermal stability of Li₃N under various conditions. These methods are essential for evaluating the material's performance reliability in high-frequency applications where thermal management is critical.
Advanced surface characterization techniques such as X-ray photoelectron spectroscopy (XPS) and secondary ion mass spectrometry (SIMS) provide detailed information about the surface chemistry and depth profile of lithium nitride samples. These analyses are particularly important for understanding interface phenomena that may affect high-frequency signal transmission and reception in practical applications.
Sustainability Aspects of Li3N Production
The sustainability of Lithium Nitride (Li₃N) production represents a critical consideration in its application for high-frequency electronic components. Current manufacturing processes for Li₃N primarily rely on direct nitridation of lithium metal under nitrogen atmosphere at elevated temperatures, which presents significant energy consumption challenges. The energy-intensive nature of these processes contributes substantially to the carbon footprint associated with Li₃N production, particularly when powered by non-renewable energy sources.
Raw material sourcing presents another sustainability concern, as lithium extraction has well-documented environmental impacts. Traditional lithium mining operations, particularly in salt flats and brine deposits, can lead to water table depletion, habitat disruption, and soil contamination. The growing demand for lithium in battery applications already places pressure on these resources, and increased Li₃N production for high-frequency applications would further exacerbate these challenges.
Waste management throughout the Li₃N lifecycle presents additional sustainability considerations. The highly reactive nature of lithium compounds necessitates careful handling and disposal protocols. End-of-life management for high-frequency components containing Li₃N remains underdeveloped, with limited recycling infrastructure currently available to recover valuable materials from these specialized applications.
Several promising approaches are emerging to address these sustainability challenges. Closed-loop manufacturing systems that recapture and reuse process materials show potential for reducing waste and resource consumption. Additionally, research into lower-temperature synthesis methods could significantly reduce the energy requirements for Li₃N production, with sol-gel and mechanochemical approaches demonstrating particular promise in laboratory settings.
Alternative lithium sourcing strategies may also improve sustainability metrics. Direct lithium extraction (DLE) technologies offer reduced environmental impact compared to traditional mining operations, while lithium recovery from spent batteries represents a circular economy approach that could alleviate pressure on primary lithium resources.
Life cycle assessment (LCA) studies specific to Li₃N in high-frequency applications remain limited, creating a knowledge gap regarding the full environmental implications of these materials. Comprehensive LCA would provide valuable insights into optimization opportunities throughout the production, use, and disposal phases, potentially identifying unexpected environmental hotspots requiring mitigation.
Regulatory frameworks governing sustainable material production continue to evolve globally, with increasing emphasis on extended producer responsibility and material disclosure requirements. Manufacturers incorporating Li₃N into high-frequency applications must anticipate these developing standards to ensure long-term market viability and regulatory compliance.
Raw material sourcing presents another sustainability concern, as lithium extraction has well-documented environmental impacts. Traditional lithium mining operations, particularly in salt flats and brine deposits, can lead to water table depletion, habitat disruption, and soil contamination. The growing demand for lithium in battery applications already places pressure on these resources, and increased Li₃N production for high-frequency applications would further exacerbate these challenges.
Waste management throughout the Li₃N lifecycle presents additional sustainability considerations. The highly reactive nature of lithium compounds necessitates careful handling and disposal protocols. End-of-life management for high-frequency components containing Li₃N remains underdeveloped, with limited recycling infrastructure currently available to recover valuable materials from these specialized applications.
Several promising approaches are emerging to address these sustainability challenges. Closed-loop manufacturing systems that recapture and reuse process materials show potential for reducing waste and resource consumption. Additionally, research into lower-temperature synthesis methods could significantly reduce the energy requirements for Li₃N production, with sol-gel and mechanochemical approaches demonstrating particular promise in laboratory settings.
Alternative lithium sourcing strategies may also improve sustainability metrics. Direct lithium extraction (DLE) technologies offer reduced environmental impact compared to traditional mining operations, while lithium recovery from spent batteries represents a circular economy approach that could alleviate pressure on primary lithium resources.
Life cycle assessment (LCA) studies specific to Li₃N in high-frequency applications remain limited, creating a knowledge gap regarding the full environmental implications of these materials. Comprehensive LCA would provide valuable insights into optimization opportunities throughout the production, use, and disposal phases, potentially identifying unexpected environmental hotspots requiring mitigation.
Regulatory frameworks governing sustainable material production continue to evolve globally, with increasing emphasis on extended producer responsibility and material disclosure requirements. Manufacturers incorporating Li₃N into high-frequency applications must anticipate these developing standards to ensure long-term market viability and regulatory compliance.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!