Optimize Pulsed Electromagnetic Field for Sleep Improvement
MAR 7, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
PEMF Sleep Enhancement Background and Objectives
Sleep disorders represent one of the most pervasive health challenges in modern society, affecting approximately 30-40% of adults globally. The increasing prevalence of insomnia, sleep fragmentation, and circadian rhythm disruptions has created an urgent need for non-pharmacological therapeutic interventions. Traditional sleep medications often carry risks of dependency, tolerance, and adverse side effects, driving the search for alternative treatment modalities.
Pulsed Electromagnetic Field (PEMF) therapy has emerged as a promising non-invasive approach for sleep enhancement, leveraging the body's natural bioelectrical processes to promote restorative sleep patterns. The technology operates on the principle that controlled electromagnetic pulses can influence cellular membrane potential, neurotransmitter regulation, and brainwave entrainment, potentially addressing the underlying physiological mechanisms of sleep dysfunction.
The historical development of PEMF technology traces back to the 1970s when researchers first observed the biological effects of electromagnetic fields on tissue healing and cellular regeneration. Early investigations revealed that specific frequency ranges and pulse characteristics could modulate various physiological processes, including those governing sleep-wake cycles. The discovery that extremely low-frequency electromagnetic fields could influence melatonin production and circadian rhythm regulation marked a pivotal moment in sleep research applications.
Current market demands reflect a growing consumer preference for drug-free sleep solutions, particularly among populations concerned about long-term medication use. The global sleep aids market, valued at over $15 billion annually, shows increasing interest in technology-based interventions that can provide sustained benefits without chemical dependency risks.
The primary objective of optimizing PEMF for sleep improvement centers on identifying the most effective combination of frequency parameters, pulse duration, intensity levels, and treatment protocols. Research aims to establish standardized therapeutic windows that can consistently induce sleep onset, enhance sleep depth, and improve overall sleep architecture while maintaining safety profiles suitable for home-use applications.
Secondary objectives include developing personalized treatment algorithms that can adapt to individual sleep patterns, circadian preferences, and specific sleep disorder types. The integration of real-time sleep monitoring with adaptive PEMF delivery systems represents a key technological goal, enabling dynamic optimization based on physiological feedback during treatment sessions.
Pulsed Electromagnetic Field (PEMF) therapy has emerged as a promising non-invasive approach for sleep enhancement, leveraging the body's natural bioelectrical processes to promote restorative sleep patterns. The technology operates on the principle that controlled electromagnetic pulses can influence cellular membrane potential, neurotransmitter regulation, and brainwave entrainment, potentially addressing the underlying physiological mechanisms of sleep dysfunction.
The historical development of PEMF technology traces back to the 1970s when researchers first observed the biological effects of electromagnetic fields on tissue healing and cellular regeneration. Early investigations revealed that specific frequency ranges and pulse characteristics could modulate various physiological processes, including those governing sleep-wake cycles. The discovery that extremely low-frequency electromagnetic fields could influence melatonin production and circadian rhythm regulation marked a pivotal moment in sleep research applications.
Current market demands reflect a growing consumer preference for drug-free sleep solutions, particularly among populations concerned about long-term medication use. The global sleep aids market, valued at over $15 billion annually, shows increasing interest in technology-based interventions that can provide sustained benefits without chemical dependency risks.
The primary objective of optimizing PEMF for sleep improvement centers on identifying the most effective combination of frequency parameters, pulse duration, intensity levels, and treatment protocols. Research aims to establish standardized therapeutic windows that can consistently induce sleep onset, enhance sleep depth, and improve overall sleep architecture while maintaining safety profiles suitable for home-use applications.
Secondary objectives include developing personalized treatment algorithms that can adapt to individual sleep patterns, circadian preferences, and specific sleep disorder types. The integration of real-time sleep monitoring with adaptive PEMF delivery systems represents a key technological goal, enabling dynamic optimization based on physiological feedback during treatment sessions.
Market Demand for Sleep Improvement Technologies
The global sleep improvement technology market has experienced substantial growth driven by increasing awareness of sleep disorders and their impact on overall health and productivity. Sleep-related issues affect millions of individuals worldwide, with insomnia, sleep apnea, and circadian rhythm disorders representing the most prevalent conditions requiring technological intervention.
Consumer demand for non-pharmaceutical sleep solutions has intensified significantly as people seek alternatives to traditional sleep medications due to concerns about dependency and side effects. This shift has created a favorable environment for innovative technologies like pulsed electromagnetic field therapy, which offers a drug-free approach to sleep enhancement.
The healthcare sector represents a primary market segment, with sleep clinics and medical facilities increasingly adopting advanced therapeutic technologies to treat patients with chronic sleep disorders. Healthcare providers are particularly interested in evidence-based solutions that can complement existing treatment protocols while reducing reliance on pharmaceutical interventions.
Corporate wellness programs have emerged as another significant market driver, as employers recognize the connection between employee sleep quality and workplace performance. Companies are investing in sleep improvement technologies to reduce absenteeism, enhance productivity, and lower healthcare costs associated with sleep-related health issues.
The consumer electronics market has witnessed growing demand for personal sleep optimization devices, with individuals seeking convenient home-based solutions. This segment includes health-conscious consumers, shift workers, frequent travelers, and aging populations who experience natural changes in sleep patterns.
Market research indicates strong growth potential in emerging economies where rising disposable incomes and increasing health awareness are driving adoption of wellness technologies. The integration of sleep improvement devices with smartphone applications and wearable technology has further expanded market accessibility and appeal.
Regulatory acceptance of electromagnetic field therapies for sleep applications has strengthened market confidence, with several jurisdictions approving specific devices for therapeutic use. This regulatory support has encouraged investment in research and development while providing consumers with validated treatment options.
The market demonstrates particular strength in developed regions where healthcare infrastructure supports advanced therapeutic technologies and reimbursement systems may cover approved treatments. However, expanding global awareness of sleep health importance suggests significant untapped potential across diverse geographic markets.
Consumer demand for non-pharmaceutical sleep solutions has intensified significantly as people seek alternatives to traditional sleep medications due to concerns about dependency and side effects. This shift has created a favorable environment for innovative technologies like pulsed electromagnetic field therapy, which offers a drug-free approach to sleep enhancement.
The healthcare sector represents a primary market segment, with sleep clinics and medical facilities increasingly adopting advanced therapeutic technologies to treat patients with chronic sleep disorders. Healthcare providers are particularly interested in evidence-based solutions that can complement existing treatment protocols while reducing reliance on pharmaceutical interventions.
Corporate wellness programs have emerged as another significant market driver, as employers recognize the connection between employee sleep quality and workplace performance. Companies are investing in sleep improvement technologies to reduce absenteeism, enhance productivity, and lower healthcare costs associated with sleep-related health issues.
The consumer electronics market has witnessed growing demand for personal sleep optimization devices, with individuals seeking convenient home-based solutions. This segment includes health-conscious consumers, shift workers, frequent travelers, and aging populations who experience natural changes in sleep patterns.
Market research indicates strong growth potential in emerging economies where rising disposable incomes and increasing health awareness are driving adoption of wellness technologies. The integration of sleep improvement devices with smartphone applications and wearable technology has further expanded market accessibility and appeal.
Regulatory acceptance of electromagnetic field therapies for sleep applications has strengthened market confidence, with several jurisdictions approving specific devices for therapeutic use. This regulatory support has encouraged investment in research and development while providing consumers with validated treatment options.
The market demonstrates particular strength in developed regions where healthcare infrastructure supports advanced therapeutic technologies and reimbursement systems may cover approved treatments. However, expanding global awareness of sleep health importance suggests significant untapped potential across diverse geographic markets.
Current PEMF Sleep Applications Status and Challenges
Current PEMF sleep applications demonstrate significant promise yet face substantial implementation challenges across clinical and consumer markets. The technology has gained traction primarily through medical device manufacturers and wellness companies targeting sleep disorders, with applications ranging from clinical insomnia treatment to consumer sleep enhancement products.
The medical sector represents the most established application domain, where PEMF devices are utilized in sleep clinics and hospitals for treating chronic insomnia, circadian rhythm disorders, and sleep-related anxiety conditions. Clinical studies have shown measurable improvements in sleep onset time and sleep quality metrics, with some devices receiving regulatory approval for therapeutic use. However, the high cost of medical-grade equipment and lengthy treatment protocols limit widespread adoption.
Consumer-oriented PEMF sleep devices have emerged as a growing market segment, featuring portable units designed for home use. These products typically operate at lower field intensities and offer simplified control interfaces, making them accessible to general consumers seeking sleep improvement solutions. Major challenges include inconsistent product quality, lack of standardized protocols, and varying efficacy claims across different manufacturers.
Technical implementation faces several critical obstacles. Parameter optimization remains highly individualized, with optimal frequency ranges, intensity levels, and exposure durations varying significantly among users. Current devices often employ fixed parameter sets that may not address individual physiological differences or specific sleep disorder characteristics. Additionally, the lack of real-time feedback mechanisms prevents dynamic adjustment based on user response.
Regulatory compliance presents another significant challenge, particularly in distinguishing between medical devices and wellness products. Different jurisdictions maintain varying approval requirements, creating market fragmentation and limiting international product distribution. Safety concerns regarding long-term electromagnetic exposure, while generally considered minimal at therapeutic levels, continue to influence regulatory decisions and consumer acceptance.
Manufacturing scalability and cost reduction remain persistent challenges for broader market penetration. High-quality electromagnetic field generators require precision components and sophisticated control systems, resulting in elevated production costs that limit affordability for mass market applications. Furthermore, the absence of standardized testing protocols complicates product validation and comparison across different manufacturers.
The medical sector represents the most established application domain, where PEMF devices are utilized in sleep clinics and hospitals for treating chronic insomnia, circadian rhythm disorders, and sleep-related anxiety conditions. Clinical studies have shown measurable improvements in sleep onset time and sleep quality metrics, with some devices receiving regulatory approval for therapeutic use. However, the high cost of medical-grade equipment and lengthy treatment protocols limit widespread adoption.
Consumer-oriented PEMF sleep devices have emerged as a growing market segment, featuring portable units designed for home use. These products typically operate at lower field intensities and offer simplified control interfaces, making them accessible to general consumers seeking sleep improvement solutions. Major challenges include inconsistent product quality, lack of standardized protocols, and varying efficacy claims across different manufacturers.
Technical implementation faces several critical obstacles. Parameter optimization remains highly individualized, with optimal frequency ranges, intensity levels, and exposure durations varying significantly among users. Current devices often employ fixed parameter sets that may not address individual physiological differences or specific sleep disorder characteristics. Additionally, the lack of real-time feedback mechanisms prevents dynamic adjustment based on user response.
Regulatory compliance presents another significant challenge, particularly in distinguishing between medical devices and wellness products. Different jurisdictions maintain varying approval requirements, creating market fragmentation and limiting international product distribution. Safety concerns regarding long-term electromagnetic exposure, while generally considered minimal at therapeutic levels, continue to influence regulatory decisions and consumer acceptance.
Manufacturing scalability and cost reduction remain persistent challenges for broader market penetration. High-quality electromagnetic field generators require precision components and sophisticated control systems, resulting in elevated production costs that limit affordability for mass market applications. Furthermore, the absence of standardized testing protocols complicates product validation and comparison across different manufacturers.
Existing PEMF Sleep Optimization Solutions
01 PEMF devices for sleep induction and improvement
Pulsed electromagnetic field devices can be specifically designed to induce and improve sleep quality by applying controlled electromagnetic pulses at specific frequencies and intensities. These devices typically operate at low frequencies that correspond to natural brain wave patterns during sleep stages, helping users transition into deeper sleep states. The systems may include programmable controllers that adjust pulse parameters based on sleep phase detection or predetermined sleep protocols.- PEMF devices for sleep induction and improvement: Pulsed electromagnetic field devices can be specifically designed to induce sleep and improve sleep quality by applying electromagnetic pulses at frequencies that promote relaxation and natural sleep cycles. These devices typically operate at low frequencies and specific pulse patterns that align with brain wave patterns associated with sleep states. The systems may include control mechanisms to adjust pulse duration, intensity, and frequency to optimize sleep induction effects.
- Wearable PEMF sleep therapy systems: Wearable devices incorporating pulsed electromagnetic field technology can be worn during sleep to provide continuous therapeutic electromagnetic stimulation. These systems are designed for comfort and portability, allowing users to receive PEMF therapy throughout the night. The wearable format enables consistent treatment positioning and can include sensors to monitor sleep patterns and adjust electromagnetic field parameters accordingly.
- PEMF sleep systems with biofeedback monitoring: Advanced pulsed electromagnetic field systems integrate biofeedback mechanisms to monitor physiological parameters during sleep and automatically adjust electromagnetic field characteristics. These systems can detect sleep stages, heart rate, breathing patterns, and other biomarkers to optimize the electromagnetic pulse delivery in real-time. The integration of monitoring and treatment allows for personalized therapy that adapts to individual sleep patterns and needs.
- Bed-integrated PEMF sleep enhancement systems: Electromagnetic field generators can be integrated directly into sleeping surfaces such as mattresses, pillows, or bed frames to provide whole-body or targeted PEMF therapy during sleep. These integrated systems eliminate the need for separate devices and ensure consistent field exposure throughout the sleep period. The bed-integrated approach allows for larger electromagnetic field coverage and can include multiple coil configurations for different body regions.
- PEMF therapy protocols for sleep disorders: Specific treatment protocols utilizing pulsed electromagnetic fields have been developed to address various sleep disorders including insomnia, sleep apnea, and circadian rhythm disruptions. These protocols define optimal frequency ranges, pulse durations, treatment timing, and session lengths for different sleep-related conditions. The therapeutic approaches may combine PEMF with other modalities and include guidelines for treatment schedules that align with natural circadian rhythms.
02 Wearable PEMF sleep therapy systems
Wearable devices incorporating pulsed electromagnetic field technology can be worn during sleep to provide continuous therapeutic electromagnetic stimulation. These systems are designed as comfortable sleep accessories such as headbands, eye masks, or mattress pads that deliver targeted electromagnetic pulses to promote relaxation and sleep. The wearable format allows for convenient home use and can include sensors to monitor sleep patterns and adjust treatment parameters accordingly.Expand Specific Solutions03 PEMF frequency modulation for circadian rhythm regulation
Systems that utilize variable frequency pulsed electromagnetic fields to regulate circadian rhythms and address sleep disorders. The technology involves modulating electromagnetic pulse frequencies throughout the night to align with natural sleep-wake cycles, potentially addressing conditions like insomnia or jet lag. These systems may incorporate timing mechanisms that automatically adjust field parameters based on time of day or detected physiological states.Expand Specific Solutions04 Integrated PEMF sleep monitoring and treatment systems
Combined systems that both monitor sleep parameters and deliver pulsed electromagnetic field therapy based on real-time feedback. These integrated platforms use sensors to detect sleep stages, movement, and other physiological indicators, then automatically adjust electromagnetic field characteristics to optimize sleep quality. The systems may include data logging capabilities and connectivity features for tracking sleep improvement over time.Expand Specific Solutions05 PEMF mattress and bedding systems for sleep enhancement
Electromagnetic field generating components integrated into mattresses, pillows, or bedding materials to provide whole-body or localized sleep therapy. These systems embed coils or electromagnetic generators within sleep surfaces to deliver therapeutic pulsed fields during rest periods. The integration into existing sleep furniture allows for passive treatment without requiring user interaction, and may include zoned treatment areas for targeted therapy.Expand Specific Solutions
Key Players in PEMF Sleep Enhancement Industry
The pulsed electromagnetic field (PEMF) technology for sleep improvement represents an emerging market segment within the broader sleep economy, currently in its early commercialization phase. The industry demonstrates significant growth potential, with established healthcare giants like Koninklijke Philips NV and Medtronic AF Luxembourg SARL leveraging their extensive resources alongside specialized companies such as Neuroenhancement Lab LLC and SofPulse Inc. Technology maturity varies considerably across players, with companies like StimScience Inc. and Wave Neuroscience Inc. developing sophisticated neuromodulation platforms, while others like Urgotech SAS and Bestcare&Sumian Biotech focus on consumer-accessible solutions. The competitive landscape includes traditional medical device manufacturers, innovative startups, and research institutions from universities like Xidian University and University of Electronic Science & Technology of China, indicating strong academic backing. Market fragmentation suggests the technology is still evolving, with regulatory approval processes and clinical validation remaining key barriers to widespread adoption.
Koninklijke Philips NV
Technical Solution: Philips has developed advanced sleep optimization solutions utilizing pulsed electromagnetic field (PEMF) technology integrated with their comprehensive sleep monitoring systems. Their approach combines low-frequency electromagnetic pulses (typically 1-30 Hz) with real-time sleep stage detection to deliver targeted stimulation during specific sleep phases. The system uses proprietary algorithms to adjust pulse intensity and frequency based on individual sleep patterns, optimizing delta wave enhancement during deep sleep stages. Their technology incorporates multi-channel PEMF coils embedded in sleep surfaces, delivering precisely controlled electromagnetic fields that promote natural sleep architecture while minimizing sleep fragmentation.
Strengths: Established healthcare brand with extensive sleep research capabilities and integrated monitoring systems. Weaknesses: Higher cost point and complex setup requirements may limit consumer adoption.
SofPulse, Inc.
Technical Solution: SofPulse specializes in targeted PEMF therapy devices that utilize specific frequency ranges (27.12 MHz carrier frequency with low-frequency modulation) for sleep enhancement applications. Their technology employs burst-mode electromagnetic pulses designed to influence circadian rhythm regulation and promote deeper sleep states. The system features adaptive pulse timing that synchronizes with natural sleep cycles, delivering therapeutic electromagnetic fields through compact, wearable devices. Their approach focuses on optimizing melatonin production and reducing cortisol levels through precisely calibrated electromagnetic stimulation, with clinical studies demonstrating improved sleep onset time and sleep quality metrics.
Strengths: Specialized PEMF expertise with clinically validated frequency protocols and portable device design. Weaknesses: Limited market presence and higher regulatory compliance requirements for medical-grade devices.
Core PEMF Sleep Enhancement Innovations
Sleep inducing device
PatentActiveCN107961430A
Innovation
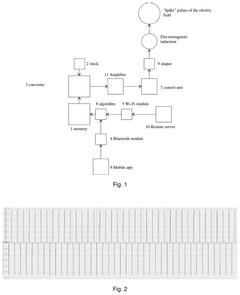
- Design a sleep induction device, which includes a physiological signal acquisition module, a sleep depth monitoring module and a pulsed electromagnetic field induction module. By collecting and processing real-time physiological signals, it obtains the optimal frequency and emits pulsed electromagnetic fields to induce a specific sleep state.
Method for correcting a person’s sleep parameters and system for the implementation thereof
PatentPendingUS20250058135A1
Innovation
- Generating short 'spike' electric field pulses of varying duration, eliminating the constant magnetic field component, and using these pulses to improve sleep parameters through specific algorithms synchronized with sleep stages.
Medical Device Regulations for PEMF Sleep Systems
The regulatory landscape for PEMF sleep systems presents a complex framework that varies significantly across global markets. In the United States, the FDA classifies PEMF devices based on their intended use and risk profile, with sleep improvement applications typically falling under Class II medical devices requiring 510(k) premarket notification. The regulatory pathway demands substantial clinical evidence demonstrating both safety and efficacy for sleep-related claims.
European markets operate under the Medical Device Regulation (MDR), which replaced the Medical Device Directive in 2021. PEMF sleep systems must obtain CE marking through conformity assessment procedures, with most devices requiring involvement of notified bodies. The MDR emphasizes clinical evaluation and post-market surveillance, creating more stringent requirements for manufacturers entering European markets.
Clinical trial requirements represent a critical regulatory hurdle for PEMF sleep systems. Regulatory bodies typically require randomized controlled trials with objective sleep measurements, including polysomnography or actigraphy data. The trials must demonstrate statistically significant improvements in sleep parameters while establishing an acceptable safety profile. Study designs must account for placebo effects, which are particularly pronounced in sleep interventions.
Safety standards for PEMF devices focus on electromagnetic field exposure limits and electrical safety requirements. The International Electrotechnical Commission (IEC) provides relevant standards, including IEC 60601 series for medical electrical equipment. Manufacturers must demonstrate compliance with specific absorption rate (SAR) limits and ensure devices do not interfere with other medical equipment or implanted devices like pacemakers.
Quality management systems under ISO 13485 are mandatory for medical device manufacturers. This standard requires comprehensive documentation of design controls, risk management processes, and manufacturing procedures. For PEMF sleep systems, particular attention must be paid to electromagnetic compatibility testing and software validation if the device includes digital components.
Labeling and marketing claim regulations strictly govern how manufacturers can promote PEMF sleep systems. Claims must be substantiated by clinical evidence accepted by regulatory authorities. The distinction between medical device claims and general wellness statements significantly impacts regulatory pathways and market access strategies.
Post-market surveillance obligations require manufacturers to monitor device performance and report adverse events. This includes establishing complaint handling procedures and conducting periodic safety updates. For PEMF sleep systems, long-term safety data collection becomes particularly important given the chronic nature of sleep disorders and extended device usage patterns.
European markets operate under the Medical Device Regulation (MDR), which replaced the Medical Device Directive in 2021. PEMF sleep systems must obtain CE marking through conformity assessment procedures, with most devices requiring involvement of notified bodies. The MDR emphasizes clinical evaluation and post-market surveillance, creating more stringent requirements for manufacturers entering European markets.
Clinical trial requirements represent a critical regulatory hurdle for PEMF sleep systems. Regulatory bodies typically require randomized controlled trials with objective sleep measurements, including polysomnography or actigraphy data. The trials must demonstrate statistically significant improvements in sleep parameters while establishing an acceptable safety profile. Study designs must account for placebo effects, which are particularly pronounced in sleep interventions.
Safety standards for PEMF devices focus on electromagnetic field exposure limits and electrical safety requirements. The International Electrotechnical Commission (IEC) provides relevant standards, including IEC 60601 series for medical electrical equipment. Manufacturers must demonstrate compliance with specific absorption rate (SAR) limits and ensure devices do not interfere with other medical equipment or implanted devices like pacemakers.
Quality management systems under ISO 13485 are mandatory for medical device manufacturers. This standard requires comprehensive documentation of design controls, risk management processes, and manufacturing procedures. For PEMF sleep systems, particular attention must be paid to electromagnetic compatibility testing and software validation if the device includes digital components.
Labeling and marketing claim regulations strictly govern how manufacturers can promote PEMF sleep systems. Claims must be substantiated by clinical evidence accepted by regulatory authorities. The distinction between medical device claims and general wellness statements significantly impacts regulatory pathways and market access strategies.
Post-market surveillance obligations require manufacturers to monitor device performance and report adverse events. This includes establishing complaint handling procedures and conducting periodic safety updates. For PEMF sleep systems, long-term safety data collection becomes particularly important given the chronic nature of sleep disorders and extended device usage patterns.
Safety Standards for Electromagnetic Sleep Devices
The safety standards for electromagnetic sleep devices represent a critical regulatory framework that governs the development and deployment of pulsed electromagnetic field (PEMF) technologies for sleep enhancement. Current international standards primarily derive from established electromagnetic compatibility (EMC) guidelines and medical device regulations, with specific attention to exposure limits defined by organizations such as the International Commission on Non-Ionizing Radiation Protection (ICNIRP) and the Federal Communications Commission (FCC).
Existing safety protocols mandate that PEMF sleep devices operate within specific frequency ranges, typically between 0.5 Hz to 100 Hz, with magnetic field intensities not exceeding 1000 microtesla for consumer applications. These parameters ensure that electromagnetic exposure remains well below levels that could potentially cause adverse biological effects while maintaining therapeutic efficacy for sleep improvement.
Regulatory bodies require comprehensive biocompatibility testing for devices intended for prolonged human exposure during sleep cycles. This includes assessment of thermal effects, potential interference with implanted medical devices such as pacemakers, and evaluation of long-term exposure impacts. Manufacturers must demonstrate compliance through rigorous testing protocols that simulate real-world usage patterns over extended periods.
Quality assurance standards encompass electromagnetic field uniformity, signal stability, and device reliability metrics. Safety certifications typically require validation of automatic shut-off mechanisms, temperature monitoring systems, and fail-safe protocols to prevent overexposure scenarios. Additionally, devices must incorporate adequate shielding to prevent electromagnetic interference with other electronic equipment in the sleep environment.
Emerging safety considerations address the unique challenges of overnight exposure, including requirements for continuous monitoring systems and adaptive field modulation based on user physiological responses. Future regulatory developments are expected to establish more specific guidelines for sleep-targeted PEMF applications, potentially incorporating real-time safety monitoring and personalized exposure limits based on individual sensitivity profiles and sleep stage detection.
Existing safety protocols mandate that PEMF sleep devices operate within specific frequency ranges, typically between 0.5 Hz to 100 Hz, with magnetic field intensities not exceeding 1000 microtesla for consumer applications. These parameters ensure that electromagnetic exposure remains well below levels that could potentially cause adverse biological effects while maintaining therapeutic efficacy for sleep improvement.
Regulatory bodies require comprehensive biocompatibility testing for devices intended for prolonged human exposure during sleep cycles. This includes assessment of thermal effects, potential interference with implanted medical devices such as pacemakers, and evaluation of long-term exposure impacts. Manufacturers must demonstrate compliance through rigorous testing protocols that simulate real-world usage patterns over extended periods.
Quality assurance standards encompass electromagnetic field uniformity, signal stability, and device reliability metrics. Safety certifications typically require validation of automatic shut-off mechanisms, temperature monitoring systems, and fail-safe protocols to prevent overexposure scenarios. Additionally, devices must incorporate adequate shielding to prevent electromagnetic interference with other electronic equipment in the sleep environment.
Emerging safety considerations address the unique challenges of overnight exposure, including requirements for continuous monitoring systems and adaptive field modulation based on user physiological responses. Future regulatory developments are expected to establish more specific guidelines for sleep-targeted PEMF applications, potentially incorporating real-time safety monitoring and personalized exposure limits based on individual sensitivity profiles and sleep stage detection.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!