Optimizing Amide Reaction Pathways for Lower Energy Outputs
FEB 28, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Amide Synthesis Background and Energy Reduction Goals
Amide bonds represent one of the most fundamental linkages in organic chemistry, serving as the backbone of proteins and forming critical connections in pharmaceuticals, polymers, and advanced materials. The formation of these bonds through amide synthesis has been a cornerstone of chemical manufacturing for over a century, with applications spanning from life-saving medications to high-performance engineering plastics.
Traditional amide synthesis methods, particularly those involving carboxylic acid activation with coupling reagents like EDC, DCC, or HATU, typically require elevated temperatures ranging from 60-120°C and extended reaction times of 12-48 hours. These energy-intensive processes contribute significantly to manufacturing costs and environmental impact, with energy consumption often accounting for 30-40% of total production expenses in pharmaceutical and specialty chemical manufacturing.
The pharmaceutical industry alone consumes approximately 2.3 billion kWh annually for amide bond formation processes, representing a substantial opportunity for energy optimization. Current industrial practices frequently rely on thermal activation to overcome the inherent kinetic barriers associated with amide formation, where activation energies typically range from 80-120 kJ/mol depending on the specific substrates and conditions employed.
Recent advances in catalytic systems, including organocatalysis and transition metal catalysis, have demonstrated potential for reducing these energy requirements by 20-50%. Enzymatic approaches using engineered amide synthetases have shown particular promise, operating effectively at ambient temperatures while maintaining high selectivity and yield. Additionally, microwave-assisted synthesis and continuous flow technologies have emerged as viable alternatives for energy reduction.
The primary energy reduction goals center on developing reaction pathways that operate at temperatures below 40°C while maintaining reaction times under 4 hours and achieving yields exceeding 85%. Secondary objectives include minimizing solvent usage, reducing waste generation, and eliminating the need for high-energy purification steps. These targets align with broader sustainability initiatives and regulatory pressures driving the chemical industry toward more environmentally responsible manufacturing processes.
Achieving these energy optimization goals requires addressing fundamental challenges in reaction kinetics, catalyst design, and process intensification, while ensuring that product quality and economic viability remain uncompromised.
Traditional amide synthesis methods, particularly those involving carboxylic acid activation with coupling reagents like EDC, DCC, or HATU, typically require elevated temperatures ranging from 60-120°C and extended reaction times of 12-48 hours. These energy-intensive processes contribute significantly to manufacturing costs and environmental impact, with energy consumption often accounting for 30-40% of total production expenses in pharmaceutical and specialty chemical manufacturing.
The pharmaceutical industry alone consumes approximately 2.3 billion kWh annually for amide bond formation processes, representing a substantial opportunity for energy optimization. Current industrial practices frequently rely on thermal activation to overcome the inherent kinetic barriers associated with amide formation, where activation energies typically range from 80-120 kJ/mol depending on the specific substrates and conditions employed.
Recent advances in catalytic systems, including organocatalysis and transition metal catalysis, have demonstrated potential for reducing these energy requirements by 20-50%. Enzymatic approaches using engineered amide synthetases have shown particular promise, operating effectively at ambient temperatures while maintaining high selectivity and yield. Additionally, microwave-assisted synthesis and continuous flow technologies have emerged as viable alternatives for energy reduction.
The primary energy reduction goals center on developing reaction pathways that operate at temperatures below 40°C while maintaining reaction times under 4 hours and achieving yields exceeding 85%. Secondary objectives include minimizing solvent usage, reducing waste generation, and eliminating the need for high-energy purification steps. These targets align with broader sustainability initiatives and regulatory pressures driving the chemical industry toward more environmentally responsible manufacturing processes.
Achieving these energy optimization goals requires addressing fundamental challenges in reaction kinetics, catalyst design, and process intensification, while ensuring that product quality and economic viability remain uncompromised.
Market Demand for Energy-Efficient Amide Production
The pharmaceutical industry represents the largest consumer segment for energy-efficient amide production technologies, driven by the extensive use of amide bonds in drug molecules and active pharmaceutical ingredients. Traditional amide synthesis methods, particularly those involving high-temperature coupling reactions, contribute significantly to manufacturing costs and environmental impact. Pharmaceutical companies are increasingly prioritizing green chemistry initiatives and sustainable manufacturing processes to meet regulatory requirements and corporate sustainability goals.
Chemical manufacturing sectors, including specialty chemicals, agrochemicals, and polymer production, demonstrate substantial demand for optimized amide reaction pathways. These industries face mounting pressure to reduce energy consumption while maintaining product quality and yield. The growing emphasis on process intensification and continuous manufacturing has created opportunities for innovative amide synthesis technologies that operate under milder conditions.
The global push toward carbon neutrality and stricter environmental regulations has intensified market demand for energy-efficient chemical processes. Regulatory frameworks in major markets, including the European Union's Green Deal and various national carbon reduction policies, are driving chemical manufacturers to adopt lower-energy production methods. This regulatory environment creates a compelling business case for investing in optimized amide reaction technologies.
Market dynamics reveal a significant cost advantage potential for energy-efficient amide production methods. Energy costs typically represent a substantial portion of total manufacturing expenses in chemical processes, particularly for reactions requiring elevated temperatures or extended reaction times. Companies implementing optimized pathways can achieve competitive advantages through reduced operational costs and improved profit margins.
The biotechnology and materials science sectors are emerging as high-growth markets for energy-efficient amide production. Advanced materials applications, including high-performance polymers and biomaterials, require precise control over amide formation while minimizing energy input. These applications often involve complex molecular architectures where traditional high-energy methods may compromise product integrity.
Contract manufacturing organizations and custom synthesis providers increasingly seek differentiated capabilities in energy-efficient processes to attract environmentally conscious clients. This trend creates market opportunities for technology providers offering optimized amide reaction solutions that can be readily integrated into existing manufacturing infrastructure while delivering measurable energy savings and environmental benefits.
Chemical manufacturing sectors, including specialty chemicals, agrochemicals, and polymer production, demonstrate substantial demand for optimized amide reaction pathways. These industries face mounting pressure to reduce energy consumption while maintaining product quality and yield. The growing emphasis on process intensification and continuous manufacturing has created opportunities for innovative amide synthesis technologies that operate under milder conditions.
The global push toward carbon neutrality and stricter environmental regulations has intensified market demand for energy-efficient chemical processes. Regulatory frameworks in major markets, including the European Union's Green Deal and various national carbon reduction policies, are driving chemical manufacturers to adopt lower-energy production methods. This regulatory environment creates a compelling business case for investing in optimized amide reaction technologies.
Market dynamics reveal a significant cost advantage potential for energy-efficient amide production methods. Energy costs typically represent a substantial portion of total manufacturing expenses in chemical processes, particularly for reactions requiring elevated temperatures or extended reaction times. Companies implementing optimized pathways can achieve competitive advantages through reduced operational costs and improved profit margins.
The biotechnology and materials science sectors are emerging as high-growth markets for energy-efficient amide production. Advanced materials applications, including high-performance polymers and biomaterials, require precise control over amide formation while minimizing energy input. These applications often involve complex molecular architectures where traditional high-energy methods may compromise product integrity.
Contract manufacturing organizations and custom synthesis providers increasingly seek differentiated capabilities in energy-efficient processes to attract environmentally conscious clients. This trend creates market opportunities for technology providers offering optimized amide reaction solutions that can be readily integrated into existing manufacturing infrastructure while delivering measurable energy savings and environmental benefits.
Current Challenges in High-Energy Amide Formation
Traditional amide formation reactions face significant thermodynamic and kinetic barriers that result in high energy consumption and reduced process efficiency. The conventional coupling methods, particularly those involving carboxylic acids and amines, typically require elevated temperatures ranging from 150-200°C and extended reaction times of 12-24 hours to achieve acceptable conversion rates. These harsh conditions not only increase operational costs but also limit the scope of substrates that can be employed, especially those containing thermally sensitive functional groups.
The activation energy requirements for direct amide bond formation present a fundamental challenge, with typical activation barriers exceeding 25-30 kcal/mol in uncatalyzed systems. This high energy threshold necessitates the use of expensive coupling reagents such as HATU, HBTU, or EDC, which generate significant amounts of chemical waste and require costly purification steps. Additionally, these reagents often lead to racemization of chiral centers and formation of unwanted side products, further complicating the synthetic process.
Catalyst deactivation represents another critical obstacle in current amide synthesis methodologies. Many promising catalytic systems suffer from rapid deactivation due to product inhibition, where the formed amide coordinates strongly to the metal center, effectively poisoning the catalyst. This phenomenon is particularly pronounced in homogeneous catalytic systems using precious metals like palladium, rhodium, or ruthenium, where catalyst turnover numbers rarely exceed 100-500 cycles before significant activity loss occurs.
Selectivity issues plague many existing amide formation protocols, particularly when dealing with polyfunctional substrates. Competing reactions such as ester formation, substrate decomposition, or unwanted cyclization reactions often occur under the harsh conditions required for amide bond formation. The challenge becomes more acute in pharmaceutical applications where regioselectivity and chemoselectivity are paramount for maintaining the integrity of complex molecular architectures.
Scalability constraints further limit the practical application of many promising laboratory-scale amide formation methods. Reactions that perform well on milligram scales often encounter heat and mass transfer limitations when scaled to industrial volumes, leading to decreased yields and increased energy consumption per unit product. The heterogeneous nature of many solid-supported catalysts exacerbates these issues, creating hot spots and uneven reaction profiles in large-scale reactors.
The activation energy requirements for direct amide bond formation present a fundamental challenge, with typical activation barriers exceeding 25-30 kcal/mol in uncatalyzed systems. This high energy threshold necessitates the use of expensive coupling reagents such as HATU, HBTU, or EDC, which generate significant amounts of chemical waste and require costly purification steps. Additionally, these reagents often lead to racemization of chiral centers and formation of unwanted side products, further complicating the synthetic process.
Catalyst deactivation represents another critical obstacle in current amide synthesis methodologies. Many promising catalytic systems suffer from rapid deactivation due to product inhibition, where the formed amide coordinates strongly to the metal center, effectively poisoning the catalyst. This phenomenon is particularly pronounced in homogeneous catalytic systems using precious metals like palladium, rhodium, or ruthenium, where catalyst turnover numbers rarely exceed 100-500 cycles before significant activity loss occurs.
Selectivity issues plague many existing amide formation protocols, particularly when dealing with polyfunctional substrates. Competing reactions such as ester formation, substrate decomposition, or unwanted cyclization reactions often occur under the harsh conditions required for amide bond formation. The challenge becomes more acute in pharmaceutical applications where regioselectivity and chemoselectivity are paramount for maintaining the integrity of complex molecular architectures.
Scalability constraints further limit the practical application of many promising laboratory-scale amide formation methods. Reactions that perform well on milligram scales often encounter heat and mass transfer limitations when scaled to industrial volumes, leading to decreased yields and increased energy consumption per unit product. The heterogeneous nature of many solid-supported catalysts exacerbates these issues, creating hot spots and uneven reaction profiles in large-scale reactors.
Existing Low-Energy Amide Synthesis Solutions
01 Amide formation through direct condensation reactions
Direct condensation methods for amide synthesis involve the reaction between carboxylic acids and amines, often requiring activation agents or catalysts to facilitate the formation of the amide bond. These pathways typically involve dehydration steps and can be optimized for energy efficiency by controlling reaction conditions such as temperature, pressure, and the use of coupling reagents. The energy output and thermodynamics of these reactions are influenced by the choice of substrates and reaction conditions.- Amide formation through direct condensation reactions: Direct condensation methods for amide synthesis involve the reaction between carboxylic acids and amines, often requiring activation agents or catalysts to facilitate the bond formation. These pathways typically involve dehydration steps and can be optimized for energy efficiency through the use of coupling reagents or enzymatic catalysis. The energy output and thermodynamics of these reactions depend on the specific substrates and reaction conditions employed.
- Catalytic amide synthesis with metal complexes: Metal-catalyzed pathways for amide formation utilize transition metal complexes to activate carbonyl groups or facilitate C-N bond formation. These catalytic systems can significantly reduce the activation energy required for amide synthesis and improve reaction selectivity. The energy profiles of these reactions are influenced by the choice of metal catalyst, ligands, and reaction parameters such as temperature and pressure.
- Enzymatic amide bond formation: Biocatalytic approaches to amide synthesis employ enzymes such as peptide synthetases or lipases to catalyze bond formation under mild conditions. These enzymatic pathways offer advantages in terms of selectivity and environmental sustainability, with energy requirements typically lower than traditional chemical methods. The reaction energetics are governed by enzyme-substrate interactions and can be modulated through protein engineering or reaction medium optimization.
- Microwave-assisted and flow chemistry amide synthesis: Advanced reaction technologies including microwave irradiation and continuous flow systems enable rapid amide formation with improved energy efficiency. These methods provide enhanced heat transfer and mixing, leading to reduced reaction times and better control over reaction exotherms. The energy output profiles in these systems can be precisely monitored and optimized through real-time process analytics.
- Solid-phase and polymer-supported amide synthesis: Solid-phase methodologies for amide bond formation involve the use of polymer supports or solid matrices to facilitate synthesis and purification. These approaches can improve reaction efficiency by enabling excess reagent use and simplifying workup procedures. The energetics of these heterogeneous systems differ from solution-phase reactions due to mass transfer considerations and surface interactions that affect activation energies.
02 Catalytic amide synthesis with energy optimization
Catalytic approaches to amide formation utilize various catalysts including metal complexes, enzymes, or organocatalysts to lower activation energy barriers and improve reaction efficiency. These methods focus on reducing energy consumption while maintaining high yields and selectivity. The catalytic pathways can involve different mechanisms such as acyl transfer or direct coupling, with energy outputs being optimized through catalyst design and reaction parameter control.Expand Specific Solutions03 Electrochemical and photochemical amide formation pathways
Alternative energy input methods for amide synthesis include electrochemical and photochemical approaches that utilize electrical current or light energy to drive the reaction. These methods offer advantages in terms of energy efficiency and environmental sustainability by avoiding traditional heating methods. The energy outputs in these systems are controlled through electrode design, light wavelength selection, and reaction medium optimization.Expand Specific Solutions04 Amide bond formation in continuous flow systems
Continuous flow reactor systems for amide synthesis provide enhanced control over reaction parameters and improved energy efficiency compared to batch processes. These systems allow for precise temperature control, efficient mixing, and reduced reaction times, leading to optimized energy outputs. The continuous nature of these processes enables better heat management and integration of energy recovery systems.Expand Specific Solutions05 Thermodynamic analysis and energy recovery in amide reactions
Understanding the thermodynamic aspects of amide formation reactions is crucial for optimizing energy outputs and developing energy-efficient processes. This includes analyzing reaction enthalpies, activation energies, and equilibrium constants. Energy recovery strategies can be implemented to capture and reuse heat generated during exothermic amide formation reactions, improving overall process efficiency and reducing energy consumption.Expand Specific Solutions
Key Players in Catalytic Amide Formation Industry
The amide reaction pathway optimization field represents an emerging yet rapidly evolving sector within green chemistry and pharmaceutical manufacturing. The industry is transitioning from traditional high-energy synthetic methods toward more sustainable, energy-efficient processes, driven by environmental regulations and cost reduction imperatives. Market growth is substantial, particularly in pharmaceutical and specialty chemical sectors where amide bonds are fundamental. Technology maturity varies significantly across players, with leading research institutions like Institute of Science Tokyo, Tohoku University, and Osaka University advancing fundamental catalytic mechanisms, while industrial giants such as Sumitomo Chemical, Evonik Operations, and Clariant International focus on scalable manufacturing solutions. Academic-industry collaborations through organizations like Wisconsin Alumni Research Foundation and Purdue Research Foundation are accelerating commercialization of novel catalytic systems and process optimization technologies.
Sumitomo Chemical Co., Ltd.
Technical Solution: Sumitomo Chemical has developed proprietary metal-catalyzed systems for low-energy amide synthesis, particularly focusing on copper and nickel-based catalysts that enable direct amidation reactions under mild conditions. Their technology incorporates microreactor systems and continuous flow processes to optimize heat and mass transfer, resulting in improved energy efficiency. The company has established pilot-scale facilities for testing these optimized pathways, with emphasis on reducing waste generation and energy consumption. Their approach includes innovative ligand design and reaction engineering solutions that have demonstrated significant improvements in both energy efficiency and product quality for pharmaceutical and agrochemical applications.
Strengths: Extensive industrial infrastructure and strong expertise in catalyst development and scale-up processes. Weaknesses: May face challenges in adapting existing production facilities to new catalytic systems.
Evonik Operations GmbH
Technical Solution: Evonik has developed innovative catalytic systems for energy-efficient amide synthesis, focusing on organocatalysis and biocatalytic approaches. Their technology platform includes novel phosphine-based catalysts and enzymatic systems that operate under mild conditions, reducing energy requirements by up to 40% compared to traditional methods. The company has invested heavily in green chemistry initiatives, developing solvent-free and low-temperature amide formation processes. Their approach emphasizes sustainability while maintaining industrial scalability, with particular expertise in specialty chemicals and pharmaceutical intermediates production through optimized amide coupling reactions.
Strengths: Strong industrial experience and proven scalability for commercial applications. Weaknesses: Technology may be limited to specific substrate classes and reaction types.
Core Catalytic Innovations for Energy-Efficient Amidation
Reaction agent for amide reaction and method for producing amide compound using same
PatentInactiveUS20240209020A1
Innovation
- A silane compound with a specific structure is used as a reaction agent, either alone or in combination with a Lewis acid catalyst and/or a phosphorus compound, to facilitate amidation reactions between carboxyl and amino groups, enabling the production of amide compounds with high stereoselectivity and efficiency.
Process for chemical reaction of amino acids and amides yielding selective conversion products
PatentInactiveUS20040225133A1
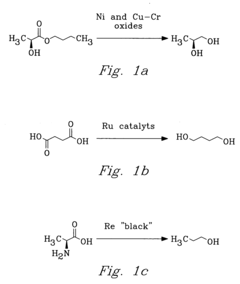
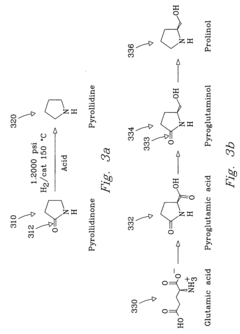
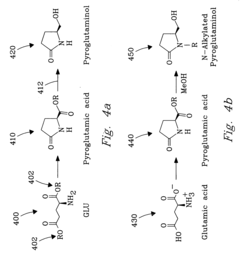
Innovation
- The process involves selecting starting materials such as L-glutamic acid and L-pyroglutamic acid, reacting them under controlled conditions with precious-metal catalysts at elevated temperatures and specific pH levels, and using a reduction catalyst to achieve high yields and selectivity in the formation of desirable products.
Environmental Regulations for Chemical Manufacturing
The chemical manufacturing industry faces increasingly stringent environmental regulations that directly impact amide synthesis processes and energy optimization strategies. Regulatory frameworks such as the European Union's REACH regulation, the US Clean Air Act, and emerging carbon emission standards establish strict limits on volatile organic compounds, hazardous air pollutants, and greenhouse gas emissions from chemical production facilities. These regulations particularly affect amide manufacturing due to the traditional use of energy-intensive heating processes and potentially harmful solvents.
Current environmental compliance requirements mandate comprehensive monitoring of reaction byproducts, waste stream management, and energy consumption reporting. The implementation of carbon pricing mechanisms and emission trading systems has created additional economic incentives for manufacturers to pursue lower energy reaction pathways. Regulatory bodies now require detailed environmental impact assessments that include lifecycle analysis of energy consumption, making energy-efficient amide synthesis not just an operational advantage but a compliance necessity.
Recent regulatory developments have introduced specific provisions for green chemistry initiatives, offering expedited approval processes and tax incentives for manufacturers adopting environmentally sustainable reaction pathways. The EPA's Green Chemistry Challenge and similar programs worldwide recognize innovations that reduce energy consumption while maintaining product quality and safety standards. These regulatory incentives align perfectly with the development of optimized amide reaction pathways that operate at lower temperatures and pressures.
Compliance with emerging regulations requires manufacturers to implement real-time monitoring systems for energy consumption and emission tracking throughout amide production processes. New standards mandate the adoption of best available techniques for energy efficiency, pushing the industry toward catalytic processes and alternative reaction conditions that minimize environmental impact. The regulatory landscape continues evolving toward more comprehensive sustainability metrics, including energy intensity per unit of product and overall carbon footprint calculations.
Future regulatory trends indicate increasing focus on circular economy principles and waste minimization in chemical manufacturing. Anticipated regulations will likely establish mandatory energy efficiency benchmarks for amide production, creating market pressure for widespread adoption of optimized reaction pathways that demonstrate measurable reductions in energy consumption and environmental impact.
Current environmental compliance requirements mandate comprehensive monitoring of reaction byproducts, waste stream management, and energy consumption reporting. The implementation of carbon pricing mechanisms and emission trading systems has created additional economic incentives for manufacturers to pursue lower energy reaction pathways. Regulatory bodies now require detailed environmental impact assessments that include lifecycle analysis of energy consumption, making energy-efficient amide synthesis not just an operational advantage but a compliance necessity.
Recent regulatory developments have introduced specific provisions for green chemistry initiatives, offering expedited approval processes and tax incentives for manufacturers adopting environmentally sustainable reaction pathways. The EPA's Green Chemistry Challenge and similar programs worldwide recognize innovations that reduce energy consumption while maintaining product quality and safety standards. These regulatory incentives align perfectly with the development of optimized amide reaction pathways that operate at lower temperatures and pressures.
Compliance with emerging regulations requires manufacturers to implement real-time monitoring systems for energy consumption and emission tracking throughout amide production processes. New standards mandate the adoption of best available techniques for energy efficiency, pushing the industry toward catalytic processes and alternative reaction conditions that minimize environmental impact. The regulatory landscape continues evolving toward more comprehensive sustainability metrics, including energy intensity per unit of product and overall carbon footprint calculations.
Future regulatory trends indicate increasing focus on circular economy principles and waste minimization in chemical manufacturing. Anticipated regulations will likely establish mandatory energy efficiency benchmarks for amide production, creating market pressure for widespread adoption of optimized reaction pathways that demonstrate measurable reductions in energy consumption and environmental impact.
Sustainability Metrics in Pharmaceutical Synthesis
The pharmaceutical industry faces mounting pressure to adopt sustainable practices, particularly in synthetic chemistry where amide bond formation represents one of the most fundamental yet energy-intensive processes. Traditional amide synthesis methods often require harsh reaction conditions, elevated temperatures, and energy-demanding purification steps, contributing significantly to the industry's carbon footprint. As regulatory bodies and stakeholders increasingly demand environmental accountability, establishing comprehensive sustainability metrics for pharmaceutical synthesis has become critical for evaluating and optimizing reaction pathways.
Energy consumption serves as a primary sustainability indicator in amide synthesis optimization. This metric encompasses direct energy inputs such as heating, cooling, and mixing requirements, as well as indirect energy costs associated with solvent production, catalyst manufacturing, and waste treatment processes. Advanced energy monitoring systems now enable real-time tracking of kilowatt-hours per kilogram of product, providing quantitative baselines for improvement initiatives. Companies are increasingly adopting energy intensity ratios that compare total energy consumption against product yield, allowing for standardized benchmarking across different synthetic routes.
Carbon footprint assessment represents another crucial sustainability dimension, extending beyond immediate energy consumption to encompass the entire lifecycle of amide synthesis. This includes upstream emissions from raw material extraction and processing, transportation-related emissions, and downstream impacts from waste disposal or recycling. Life cycle assessment methodologies specifically tailored for pharmaceutical processes now incorporate sophisticated modeling tools that can predict carbon emissions for alternative reaction pathways before implementation.
Waste generation metrics provide essential insights into process efficiency and environmental impact. These measurements include both chemical waste streams and packaging materials, with particular attention to hazardous waste categories requiring specialized disposal methods. Modern sustainability frameworks emphasize waste-to-product ratios and implement circular economy principles where reaction byproducts can be repurposed or recycled within the manufacturing ecosystem.
Water usage efficiency has emerged as a critical sustainability parameter, particularly given the water-intensive nature of pharmaceutical purification processes. Metrics include total water consumption per unit of product, water recycling rates, and wastewater treatment efficiency. Advanced process design now incorporates water footprint analysis to identify opportunities for closed-loop systems and reduced freshwater dependency.
Solvent sustainability metrics address one of the most significant environmental challenges in amide synthesis. These assessments evaluate solvent toxicity profiles, biodegradability, renewable content, and recycling potential. Green chemistry principles guide the selection of alternative solvents with lower environmental impact while maintaining reaction efficiency and product quality standards.
Energy consumption serves as a primary sustainability indicator in amide synthesis optimization. This metric encompasses direct energy inputs such as heating, cooling, and mixing requirements, as well as indirect energy costs associated with solvent production, catalyst manufacturing, and waste treatment processes. Advanced energy monitoring systems now enable real-time tracking of kilowatt-hours per kilogram of product, providing quantitative baselines for improvement initiatives. Companies are increasingly adopting energy intensity ratios that compare total energy consumption against product yield, allowing for standardized benchmarking across different synthetic routes.
Carbon footprint assessment represents another crucial sustainability dimension, extending beyond immediate energy consumption to encompass the entire lifecycle of amide synthesis. This includes upstream emissions from raw material extraction and processing, transportation-related emissions, and downstream impacts from waste disposal or recycling. Life cycle assessment methodologies specifically tailored for pharmaceutical processes now incorporate sophisticated modeling tools that can predict carbon emissions for alternative reaction pathways before implementation.
Waste generation metrics provide essential insights into process efficiency and environmental impact. These measurements include both chemical waste streams and packaging materials, with particular attention to hazardous waste categories requiring specialized disposal methods. Modern sustainability frameworks emphasize waste-to-product ratios and implement circular economy principles where reaction byproducts can be repurposed or recycled within the manufacturing ecosystem.
Water usage efficiency has emerged as a critical sustainability parameter, particularly given the water-intensive nature of pharmaceutical purification processes. Metrics include total water consumption per unit of product, water recycling rates, and wastewater treatment efficiency. Advanced process design now incorporates water footprint analysis to identify opportunities for closed-loop systems and reduced freshwater dependency.
Solvent sustainability metrics address one of the most significant environmental challenges in amide synthesis. These assessments evaluate solvent toxicity profiles, biodegradability, renewable content, and recycling potential. Green chemistry principles guide the selection of alternative solvents with lower environmental impact while maintaining reaction efficiency and product quality standards.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!