Raman Spectroscopy vs Other Diagnostic Methods: Critical Analysis
SEP 19, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Raman Spectroscopy Evolution and Objectives
Raman spectroscopy has evolved significantly since its discovery by C.V. Raman in 1928, who observed the inelastic scattering of light by molecules, a phenomenon that later earned him the Nobel Prize in Physics. The initial applications were limited to fundamental research in molecular physics and chemistry due to technological constraints. The development trajectory changed dramatically with the invention of the laser in the 1960s, providing the intense monochromatic light source necessary for efficient Raman scattering detection.
The 1970s and 1980s witnessed the emergence of Fourier Transform Raman spectroscopy, which improved signal-to-noise ratios and expanded the technique's applicability. By the 1990s, the introduction of charge-coupled device (CCD) detectors and holographic notch filters revolutionized the field, making Raman systems more compact, sensitive, and user-friendly. These advancements paved the way for the technique's transition from research laboratories to clinical settings.
The early 2000s marked a significant turning point with the development of surface-enhanced Raman spectroscopy (SERS), which amplifies Raman signals by factors of 10^6 to 10^14 through interactions with metallic nanostructures. This breakthrough dramatically improved detection sensitivity, enabling the analysis of trace substances and even single molecules. Concurrently, tip-enhanced Raman spectroscopy (TERS) emerged, combining Raman spectroscopy with scanning probe microscopy to achieve nanoscale spatial resolution.
Recent years have seen the integration of Raman technology with artificial intelligence and machine learning algorithms, enhancing data interpretation capabilities and enabling real-time analysis. Portable and handheld Raman devices have also been developed, facilitating point-of-care diagnostics and field applications. The miniaturization trend continues with the development of chip-based Raman systems that promise to further democratize access to this powerful analytical technique.
The primary objective of modern Raman spectroscopy in diagnostic applications is to provide non-invasive, label-free molecular characterization with high specificity and sensitivity. It aims to detect disease biomarkers at earlier stages than conventional methods, potentially improving patient outcomes through earlier intervention. Additionally, the technique seeks to offer real-time feedback during surgical procedures, helping surgeons distinguish between healthy and pathological tissues with greater precision.
Another key goal is to develop standardized protocols and reference databases that enable reliable comparison of results across different instruments and clinical settings. The field also strives to overcome remaining technical challenges, such as fluorescence interference and limited penetration depth, through innovative approaches like spatially offset Raman spectroscopy (SORS) and transmission Raman spectroscopy, which allow for deeper tissue analysis.
The 1970s and 1980s witnessed the emergence of Fourier Transform Raman spectroscopy, which improved signal-to-noise ratios and expanded the technique's applicability. By the 1990s, the introduction of charge-coupled device (CCD) detectors and holographic notch filters revolutionized the field, making Raman systems more compact, sensitive, and user-friendly. These advancements paved the way for the technique's transition from research laboratories to clinical settings.
The early 2000s marked a significant turning point with the development of surface-enhanced Raman spectroscopy (SERS), which amplifies Raman signals by factors of 10^6 to 10^14 through interactions with metallic nanostructures. This breakthrough dramatically improved detection sensitivity, enabling the analysis of trace substances and even single molecules. Concurrently, tip-enhanced Raman spectroscopy (TERS) emerged, combining Raman spectroscopy with scanning probe microscopy to achieve nanoscale spatial resolution.
Recent years have seen the integration of Raman technology with artificial intelligence and machine learning algorithms, enhancing data interpretation capabilities and enabling real-time analysis. Portable and handheld Raman devices have also been developed, facilitating point-of-care diagnostics and field applications. The miniaturization trend continues with the development of chip-based Raman systems that promise to further democratize access to this powerful analytical technique.
The primary objective of modern Raman spectroscopy in diagnostic applications is to provide non-invasive, label-free molecular characterization with high specificity and sensitivity. It aims to detect disease biomarkers at earlier stages than conventional methods, potentially improving patient outcomes through earlier intervention. Additionally, the technique seeks to offer real-time feedback during surgical procedures, helping surgeons distinguish between healthy and pathological tissues with greater precision.
Another key goal is to develop standardized protocols and reference databases that enable reliable comparison of results across different instruments and clinical settings. The field also strives to overcome remaining technical challenges, such as fluorescence interference and limited penetration depth, through innovative approaches like spatially offset Raman spectroscopy (SORS) and transmission Raman spectroscopy, which allow for deeper tissue analysis.
Clinical Diagnostic Market Needs Analysis
The global clinical diagnostics market is experiencing significant growth, driven by increasing prevalence of chronic and infectious diseases, aging populations, and technological advancements in diagnostic methodologies. Current market valuation exceeds $70 billion with projected annual growth rates between 5-7% through 2028, creating substantial opportunities for innovative diagnostic technologies like Raman spectroscopy.
Healthcare systems worldwide face mounting pressure to reduce costs while improving diagnostic accuracy and patient outcomes. This has created a critical need for diagnostic methods that offer rapid results, high sensitivity and specificity, minimal invasiveness, and cost-effectiveness. Traditional diagnostic approaches often require lengthy laboratory processing times, invasive sampling procedures, or expose patients to radiation, creating substantial market gaps for alternative technologies.
Point-of-care testing represents one of the fastest-growing segments within clinical diagnostics, with healthcare providers increasingly demanding solutions that enable immediate decision-making at patient bedsides, emergency departments, and outpatient settings. Raman spectroscopy's potential for real-time, non-destructive analysis positions it favorably within this expanding market segment.
Oncology diagnostics present particularly compelling opportunities, as current methods for cancer detection and characterization often involve invasive biopsies, expensive imaging, or laboratory tests with limited sensitivity. The market actively seeks technologies capable of earlier detection, improved staging accuracy, and better treatment monitoring capabilities - areas where Raman spectroscopy shows considerable promise.
Infectious disease diagnostics have gained renewed focus following global health crises, with heightened demand for rapid pathogen identification and antimicrobial resistance detection. Current methods frequently require culture growth or complex molecular techniques, creating market openings for spectroscopic approaches offering faster results with comparable accuracy.
Personalized medicine trends are reshaping diagnostic requirements, with growing emphasis on technologies capable of detecting molecular and metabolic biomarkers that inform individualized treatment decisions. This shift favors advanced spectroscopic methods that can analyze biological samples at molecular levels without extensive sample preparation.
Regulatory and reimbursement landscapes significantly influence market adoption of new diagnostic technologies. Successful market penetration requires demonstrating clear clinical utility, cost-effectiveness, and compatibility with existing healthcare workflows. Technologies offering multiple diagnostic applications across different medical specialties present particularly attractive investment opportunities due to their broader market potential.
Healthcare systems worldwide face mounting pressure to reduce costs while improving diagnostic accuracy and patient outcomes. This has created a critical need for diagnostic methods that offer rapid results, high sensitivity and specificity, minimal invasiveness, and cost-effectiveness. Traditional diagnostic approaches often require lengthy laboratory processing times, invasive sampling procedures, or expose patients to radiation, creating substantial market gaps for alternative technologies.
Point-of-care testing represents one of the fastest-growing segments within clinical diagnostics, with healthcare providers increasingly demanding solutions that enable immediate decision-making at patient bedsides, emergency departments, and outpatient settings. Raman spectroscopy's potential for real-time, non-destructive analysis positions it favorably within this expanding market segment.
Oncology diagnostics present particularly compelling opportunities, as current methods for cancer detection and characterization often involve invasive biopsies, expensive imaging, or laboratory tests with limited sensitivity. The market actively seeks technologies capable of earlier detection, improved staging accuracy, and better treatment monitoring capabilities - areas where Raman spectroscopy shows considerable promise.
Infectious disease diagnostics have gained renewed focus following global health crises, with heightened demand for rapid pathogen identification and antimicrobial resistance detection. Current methods frequently require culture growth or complex molecular techniques, creating market openings for spectroscopic approaches offering faster results with comparable accuracy.
Personalized medicine trends are reshaping diagnostic requirements, with growing emphasis on technologies capable of detecting molecular and metabolic biomarkers that inform individualized treatment decisions. This shift favors advanced spectroscopic methods that can analyze biological samples at molecular levels without extensive sample preparation.
Regulatory and reimbursement landscapes significantly influence market adoption of new diagnostic technologies. Successful market penetration requires demonstrating clear clinical utility, cost-effectiveness, and compatibility with existing healthcare workflows. Technologies offering multiple diagnostic applications across different medical specialties present particularly attractive investment opportunities due to their broader market potential.
Current Limitations of Raman vs Traditional Diagnostics
Despite the promising capabilities of Raman spectroscopy in diagnostic applications, several limitations currently hinder its widespread adoption compared to traditional diagnostic methods. One significant challenge is the inherently weak Raman signal, which is approximately 10^-6 times weaker than Rayleigh scattering. This fundamental limitation necessitates longer acquisition times and sophisticated detection systems, making real-time analysis challenging in clinical settings where rapid results are often crucial.
The high cost of Raman instrumentation presents another substantial barrier. High-quality spectrometers, lasers, and detectors required for reliable Raman analysis can cost between $50,000 to $200,000, significantly exceeding the price of many conventional diagnostic tools. This cost factor severely limits accessibility, particularly in resource-constrained healthcare environments and developing regions.
Technical complexity also impedes widespread implementation. Raman systems require precise alignment, calibration, and maintenance by trained personnel. The interpretation of Raman spectra demands specialized expertise that many clinical laboratories currently lack, creating a steep learning curve for adoption compared to well-established diagnostic protocols.
Sample preparation challenges further complicate Raman applications in diagnostics. Biological samples often exhibit autofluorescence that can overwhelm the weak Raman signals. While techniques like surface-enhanced Raman spectroscopy (SERS) can enhance sensitivity, they introduce additional complexity and variability that affect reproducibility and standardization across different laboratories.
Standardization itself remains problematic. Unlike many traditional diagnostic methods with established protocols and reference standards, Raman spectroscopy lacks universally accepted procedures for sample preparation, data acquisition, and analysis. This absence of standardization complicates multi-center studies and regulatory approval processes.
The limited penetration depth of conventional Raman techniques (typically a few hundred micrometers) restricts its application for deep tissue diagnostics without invasive procedures. While techniques like spatially offset Raman spectroscopy (SORS) show promise for deeper penetration, they remain less developed than established imaging modalities like MRI or CT scanning.
Regulatory hurdles present additional challenges. The novel nature of Raman-based diagnostics means fewer approved clinical applications compared to traditional methods with established regulatory pathways. The validation process for new Raman applications requires extensive clinical trials and comparison studies against gold standard methods, creating a significant time and resource investment before clinical implementation.
The high cost of Raman instrumentation presents another substantial barrier. High-quality spectrometers, lasers, and detectors required for reliable Raman analysis can cost between $50,000 to $200,000, significantly exceeding the price of many conventional diagnostic tools. This cost factor severely limits accessibility, particularly in resource-constrained healthcare environments and developing regions.
Technical complexity also impedes widespread implementation. Raman systems require precise alignment, calibration, and maintenance by trained personnel. The interpretation of Raman spectra demands specialized expertise that many clinical laboratories currently lack, creating a steep learning curve for adoption compared to well-established diagnostic protocols.
Sample preparation challenges further complicate Raman applications in diagnostics. Biological samples often exhibit autofluorescence that can overwhelm the weak Raman signals. While techniques like surface-enhanced Raman spectroscopy (SERS) can enhance sensitivity, they introduce additional complexity and variability that affect reproducibility and standardization across different laboratories.
Standardization itself remains problematic. Unlike many traditional diagnostic methods with established protocols and reference standards, Raman spectroscopy lacks universally accepted procedures for sample preparation, data acquisition, and analysis. This absence of standardization complicates multi-center studies and regulatory approval processes.
The limited penetration depth of conventional Raman techniques (typically a few hundred micrometers) restricts its application for deep tissue diagnostics without invasive procedures. While techniques like spatially offset Raman spectroscopy (SORS) show promise for deeper penetration, they remain less developed than established imaging modalities like MRI or CT scanning.
Regulatory hurdles present additional challenges. The novel nature of Raman-based diagnostics means fewer approved clinical applications compared to traditional methods with established regulatory pathways. The validation process for new Raman applications requires extensive clinical trials and comparison studies against gold standard methods, creating a significant time and resource investment before clinical implementation.
Comparative Analysis of Diagnostic Methodologies
01 Raman spectroscopy instrumentation and apparatus design
Various designs and configurations of Raman spectroscopy instruments that improve measurement accuracy, sensitivity, and reliability. These include specialized optical components, laser sources, detectors, and overall system architectures that enhance the collection of Raman scattered light while minimizing interference. Innovations focus on making instruments more compact, robust, and suitable for specific applications.- Raman spectroscopy instrumentation and apparatus design: Various designs and configurations of Raman spectroscopy instruments are disclosed, including improvements in optical components, detection systems, and overall apparatus architecture. These innovations focus on enhancing sensitivity, resolution, and reliability of Raman measurements while reducing interference from background signals. The designs incorporate specialized light sources, filters, detectors, and optical arrangements to optimize the collection of Raman scattered light from samples.
- Biomedical and clinical applications of Raman spectroscopy: Raman spectroscopy techniques applied to medical diagnostics, clinical analysis, and biological sample characterization. These applications include non-invasive disease detection, tissue analysis, blood component measurement, and in vivo monitoring of biological processes. The technology enables rapid identification of biomarkers, pathogens, and cellular components without requiring extensive sample preparation, offering potential for point-of-care diagnostics and personalized medicine approaches.
- Enhanced Raman techniques and signal processing methods: Advanced Raman spectroscopy techniques including surface-enhanced Raman spectroscopy (SERS), resonance Raman, and spatially offset Raman spectroscopy (SORS), along with sophisticated signal processing algorithms. These methods significantly improve detection sensitivity, allowing for identification of trace substances and analysis through packaging or tissue. The innovations include specialized substrates, nanostructures, and computational approaches that enhance Raman signals and extract meaningful data from complex spectra.
- Portable and miniaturized Raman systems: Development of compact, portable, and field-deployable Raman spectroscopy systems for on-site analysis applications. These innovations focus on miniaturization of components, integration with mobile devices, power efficiency, and ruggedization for field use. The systems enable real-time analysis in diverse environments including industrial settings, security checkpoints, environmental monitoring, and remote locations where traditional laboratory equipment is impractical.
- Industrial and material analysis applications: Raman spectroscopy applications for industrial quality control, material identification, and process monitoring. These innovations enable non-destructive analysis of pharmaceuticals, polymers, chemicals, semiconductors, and nanomaterials. The technology allows for rapid verification of material composition, detection of contaminants, monitoring of chemical reactions, and characterization of material properties in manufacturing environments, improving production efficiency and product quality.
02 Raman spectroscopy for biological and medical applications
Applications of Raman spectroscopy in medical diagnostics, biological sample analysis, and healthcare. These technologies enable non-invasive or minimally invasive detection of biomarkers, disease indicators, and physiological parameters. Implementations include in vivo measurements, tissue characterization, blood analysis, and integration with medical devices for real-time monitoring during procedures.Expand Specific Solutions03 Enhanced Raman techniques and signal processing methods
Advanced techniques that improve the sensitivity and specificity of Raman spectroscopy, including surface-enhanced Raman spectroscopy (SERS), tip-enhanced Raman spectroscopy (TERS), and specialized signal processing algorithms. These methods overcome traditional limitations of Raman spectroscopy by enhancing signal strength, reducing noise, and enabling detection of trace substances or specific molecular signatures.Expand Specific Solutions04 Raman spectroscopy for material identification and characterization
Applications focused on identifying, analyzing, and characterizing materials using Raman spectroscopy. These include systems for detecting counterfeit products, analyzing pharmaceutical compounds, identifying hazardous substances, characterizing nanomaterials, and quality control in manufacturing. The technologies enable rapid, non-destructive analysis of chemical composition and molecular structure.Expand Specific Solutions05 Portable and field-deployable Raman systems
Miniaturized and ruggedized Raman spectroscopy systems designed for use outside laboratory environments. These innovations focus on creating portable, handheld, or field-deployable instruments that maintain high performance while being suitable for on-site analysis. Applications include environmental monitoring, security screening, geological surveys, and point-of-care diagnostics in remote locations.Expand Specific Solutions
Leading Companies in Spectroscopic Diagnostics
Raman spectroscopy technology is currently in a growth phase within the medical diagnostics market, with an expanding global footprint estimated at $1.5 billion and projected annual growth of 6-8%. The competitive landscape features established scientific instrumentation companies like ChemImage Corp., Nikon Corp., and Excelitas Technologies alongside diversified technology giants such as Intel Corp., Sony Group Corp., and Koninklijke Philips NV. Academic institutions including MIT, National University of Singapore, and Vanderbilt University are driving innovation through research partnerships. The technology demonstrates moderate maturity in laboratory settings but remains in early clinical adoption stages, with companies like Synaptive Medical and Perceptronix Medical working to bridge this gap through specialized medical applications that leverage Raman's non-invasive molecular analysis capabilities against traditional diagnostic methods.
ChemImage Corp.
Technical Solution: ChemImage has developed advanced Raman spectroscopy systems specifically designed for medical diagnostics and forensic applications. Their proprietary Molecular Chemical Imaging (MCI) technology combines Raman spectroscopy with digital imaging to provide both spatial and spectral information simultaneously[1]. Their systems employ confocal Raman microscopy with automated sample handling and sophisticated chemometric analysis algorithms to identify molecular signatures in complex biological samples. ChemImage's technology utilizes multimodal imaging that combines Raman with fluorescence and near-infrared imaging to enhance diagnostic capabilities[3]. Their systems feature high-throughput screening capabilities with automated pattern recognition software that can identify specific molecular markers associated with disease states. The company has also developed portable Raman devices for point-of-care diagnostics that maintain high sensitivity while reducing form factor.
Strengths: Superior molecular specificity compared to conventional imaging; non-destructive tissue analysis without contrast agents; multimodal capabilities that enhance diagnostic accuracy through complementary data streams. Weaknesses: Higher cost compared to traditional diagnostic methods; longer acquisition times for high-resolution molecular maps; requires specialized training for operation and data interpretation.
Koninklijke Philips NV
Technical Solution: Philips has developed integrated diagnostic platforms that incorporate Raman spectroscopy alongside other imaging modalities for comprehensive tissue analysis. Their systems combine Raman with optical coherence tomography (OCT) to provide both molecular and structural information in a single examination[2]. Philips' technology utilizes fiber-optic Raman probes that can be integrated into existing endoscopic systems, enabling in vivo molecular analysis during standard endoscopic procedures. Their proprietary algorithms perform real-time analysis of Raman spectra to identify abnormal tissue regions with high sensitivity and specificity, particularly in gastrointestinal and pulmonary applications[4]. The company has developed specialized Raman-based contrast agents that selectively bind to cancer cells, enhancing signal differentiation between healthy and malignant tissues. Philips' systems feature automated calibration and background correction mechanisms that compensate for biological autofluorescence, improving the reliability of Raman measurements in clinical settings.
Strengths: Seamless integration with existing clinical workflows and equipment; multimodal capabilities providing complementary diagnostic information; real-time analysis capabilities suitable for intraoperative guidance. Weaknesses: Relatively limited depth penetration compared to MRI or CT; signal interference from ambient light and other optical sources in clinical environments; requires careful calibration to maintain accuracy across different tissue types.
Breakthrough Patents in Raman Diagnostic Applications
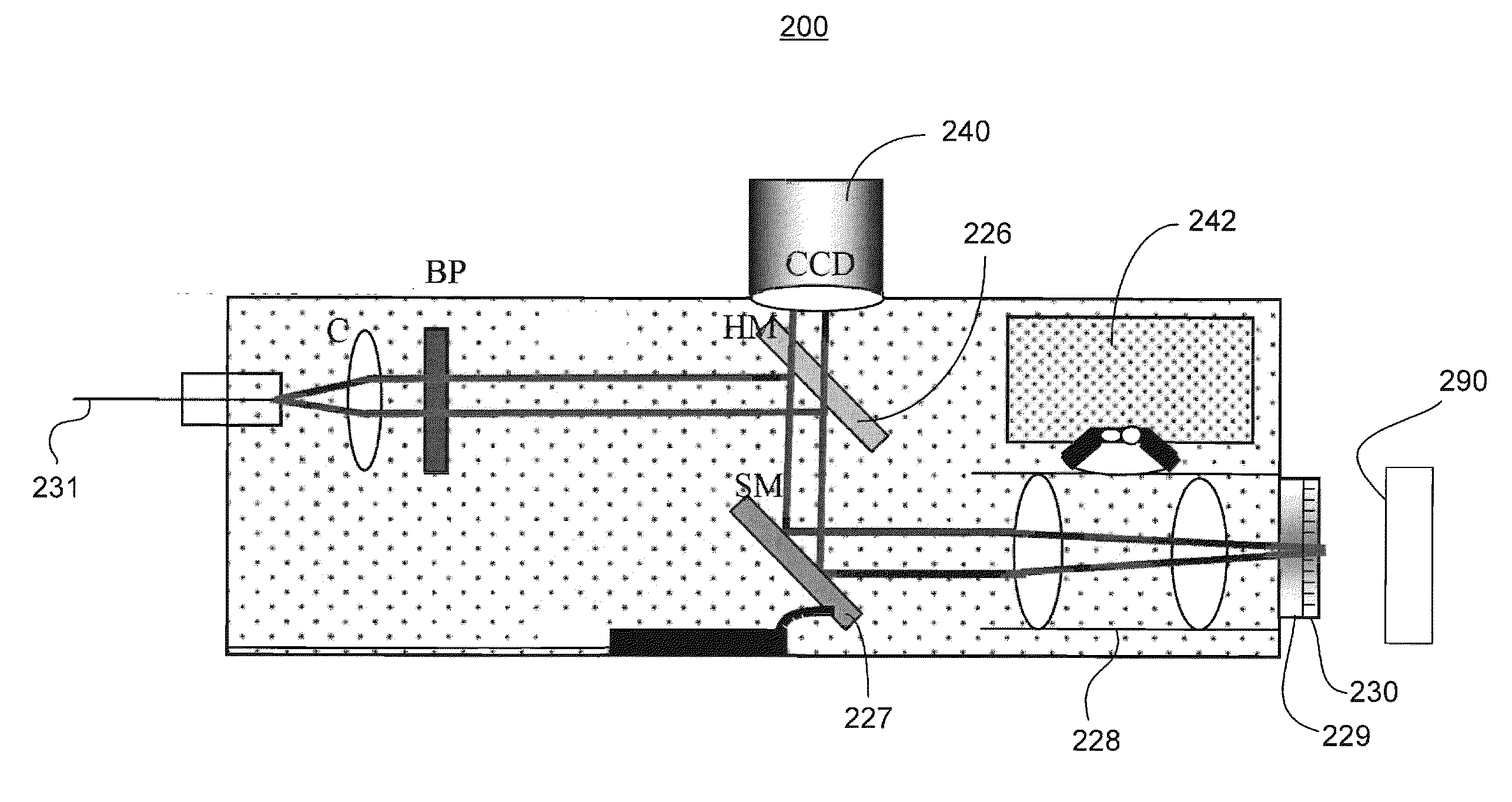
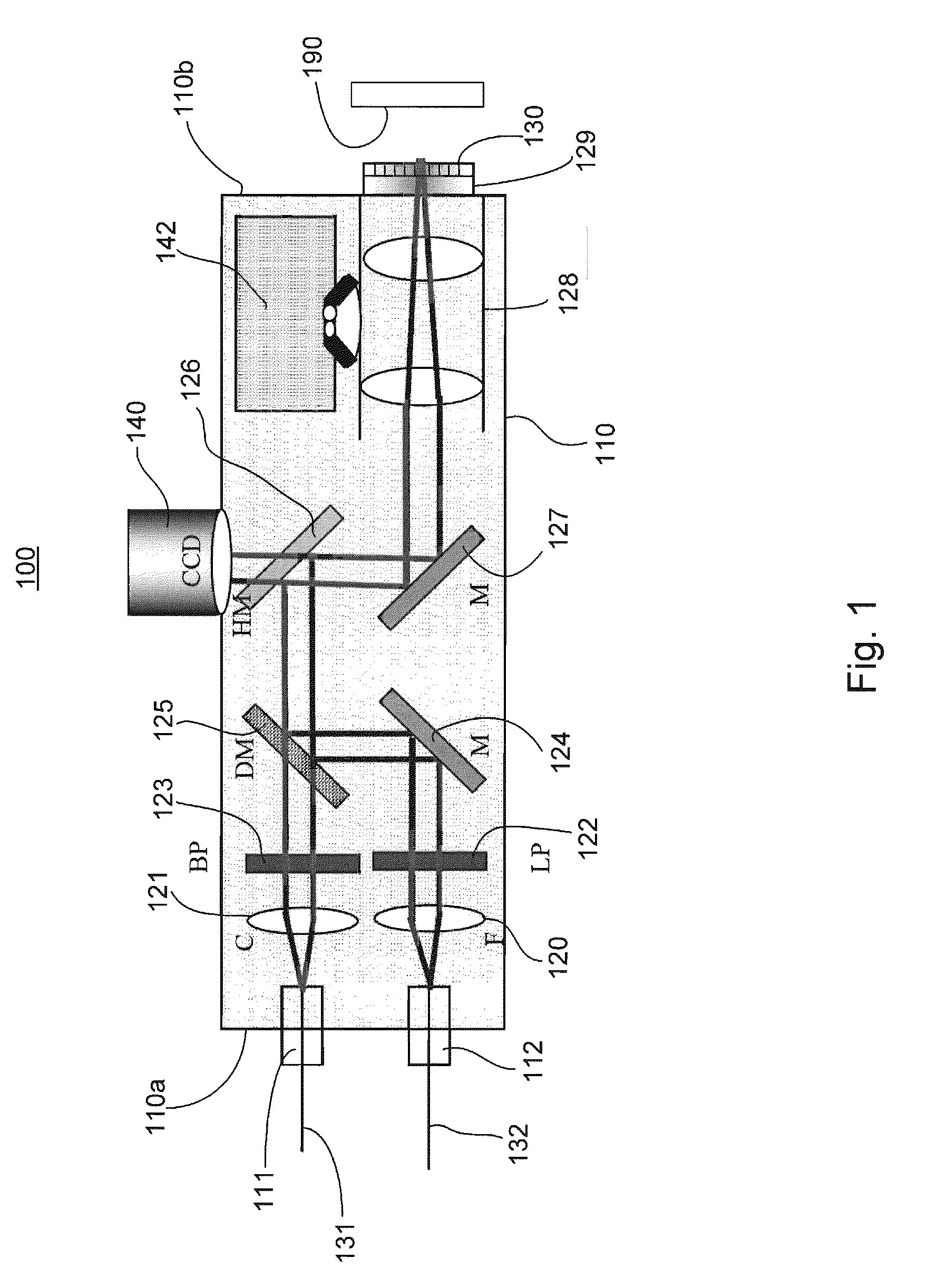
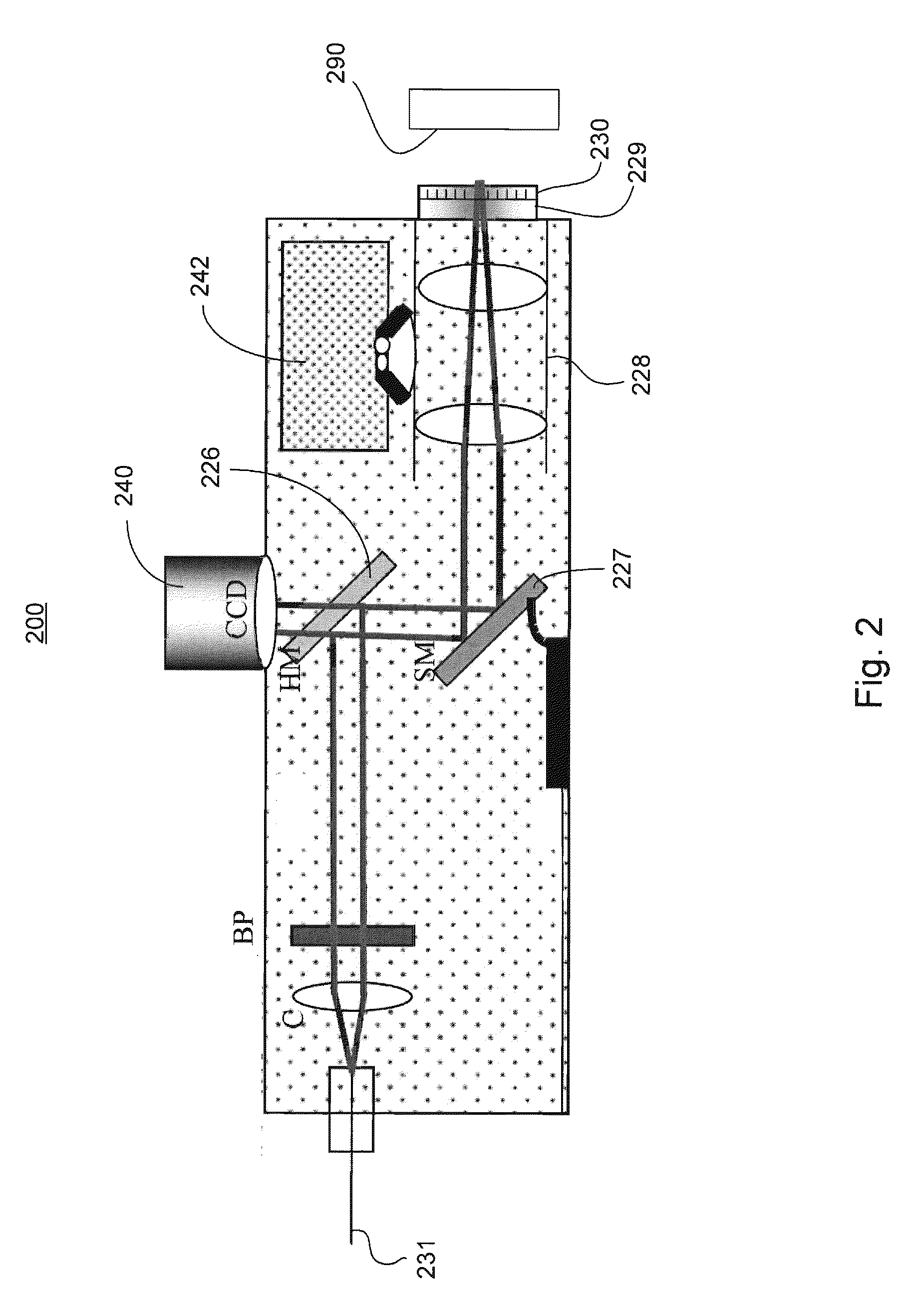
Device and method for non-invasively evaluating a target of interest of a living subject
PatentActiveUS20100214562A1
Innovation
- A handheld probe integrating confocal reflectance imaging, confocal Raman spectroscopy, and gross spatial imaging, which uses a combination of optical paths, lenses, filters, and scanning mirrors to capture both morphologic and biochemical information from skin lesions, allowing for real-time, non-invasive evaluation.
Regulatory Framework for Novel Diagnostic Technologies
The regulatory landscape for novel diagnostic technologies like Raman spectroscopy operates within a complex framework designed to ensure safety, efficacy, and ethical implementation. In the United States, the FDA classifies diagnostic technologies into different risk categories, with Raman spectroscopy typically falling under Class II medical devices requiring 510(k) clearance. This process demands manufacturers demonstrate substantial equivalence to legally marketed devices, presenting a significant hurdle for innovative technologies without clear predecessors.
The European Union's regulatory approach differs through its Medical Device Regulation (MDR), which emphasizes risk-based classification and requires conformity assessment procedures. Raman spectroscopy devices must meet essential requirements for safety and performance, with clinical evidence supporting their intended purpose. The CE marking process involves notified bodies that evaluate technical documentation and quality management systems.
Regulatory frameworks in Asia vary considerably, with Japan's Pharmaceuticals and Medical Devices Agency (PMDA) implementing stringent requirements similar to FDA standards, while China's National Medical Products Administration (NMPA) has recently streamlined approval processes for innovative diagnostics while maintaining strict oversight.
Global harmonization efforts through the International Medical Device Regulators Forum (IMDRF) aim to standardize requirements across jurisdictions, potentially easing the regulatory burden for Raman spectroscopy and other novel diagnostic technologies. However, significant regional differences persist, creating challenges for global market access.
Reimbursement policies represent another critical regulatory consideration. In many healthcare systems, novel diagnostic technologies must demonstrate not only clinical validity but also cost-effectiveness to secure coverage. Raman spectroscopy faces particular challenges in this domain as health technology assessment bodies often require substantial evidence of improved patient outcomes compared to established diagnostic methods.
Privacy and data protection regulations add another layer of complexity, especially for diagnostic technologies that generate large datasets. GDPR in Europe and HIPAA in the US impose strict requirements on data handling, storage, and patient consent that developers must address during technology implementation.
Emerging regulatory trends include adaptive licensing pathways that allow earlier market access with continued post-market surveillance, and increased focus on real-world evidence to supplement traditional clinical trials. These developments may facilitate faster adoption of promising technologies like Raman spectroscopy while maintaining appropriate safeguards.
The European Union's regulatory approach differs through its Medical Device Regulation (MDR), which emphasizes risk-based classification and requires conformity assessment procedures. Raman spectroscopy devices must meet essential requirements for safety and performance, with clinical evidence supporting their intended purpose. The CE marking process involves notified bodies that evaluate technical documentation and quality management systems.
Regulatory frameworks in Asia vary considerably, with Japan's Pharmaceuticals and Medical Devices Agency (PMDA) implementing stringent requirements similar to FDA standards, while China's National Medical Products Administration (NMPA) has recently streamlined approval processes for innovative diagnostics while maintaining strict oversight.
Global harmonization efforts through the International Medical Device Regulators Forum (IMDRF) aim to standardize requirements across jurisdictions, potentially easing the regulatory burden for Raman spectroscopy and other novel diagnostic technologies. However, significant regional differences persist, creating challenges for global market access.
Reimbursement policies represent another critical regulatory consideration. In many healthcare systems, novel diagnostic technologies must demonstrate not only clinical validity but also cost-effectiveness to secure coverage. Raman spectroscopy faces particular challenges in this domain as health technology assessment bodies often require substantial evidence of improved patient outcomes compared to established diagnostic methods.
Privacy and data protection regulations add another layer of complexity, especially for diagnostic technologies that generate large datasets. GDPR in Europe and HIPAA in the US impose strict requirements on data handling, storage, and patient consent that developers must address during technology implementation.
Emerging regulatory trends include adaptive licensing pathways that allow earlier market access with continued post-market surveillance, and increased focus on real-world evidence to supplement traditional clinical trials. These developments may facilitate faster adoption of promising technologies like Raman spectroscopy while maintaining appropriate safeguards.
Cost-Benefit Analysis of Implementing Raman Diagnostics
The implementation of Raman spectroscopy as a diagnostic tool requires careful financial consideration beyond its technical capabilities. Initial capital expenditure for Raman systems ranges from $50,000 to $250,000, depending on resolution, sensitivity, and automation features. This represents a significant investment compared to conventional diagnostic methods like PCR ($15,000-$30,000) or immunoassay platforms ($10,000-$25,000).
Operational costs must also be evaluated comprehensively. While Raman systems have minimal reagent requirements, reducing recurring expenses by approximately 30-40% compared to chemical-based diagnostics, they demand specialized maintenance. Annual maintenance contracts typically cost 8-12% of the initial investment, and specialized technicians command higher salaries than conventional laboratory personnel.
Training represents another substantial cost factor. Laboratory staff require extensive training (40-60 hours initially) to operate Raman equipment effectively and interpret spectral data accurately. This training investment exceeds that of conventional diagnostic methods by approximately 200%, though this gap narrows with time as expertise develops.
The return on investment timeline differs significantly from other diagnostic approaches. Raman diagnostics typically achieve financial break-even in 3-5 years, compared to 1-2 years for conventional methods. However, long-term economic benefits emerge through reduced per-test costs (approximately $2-5 per Raman test versus $8-15 for comparable molecular tests) and minimal consumable requirements.
Scalability economics favor Raman technology in high-volume settings. After initial implementation, marginal costs decrease substantially with increased testing volume, creating economies of scale that outperform traditional diagnostics after approximately 10,000 tests annually. This makes Raman particularly cost-effective for large hospitals and reference laboratories.
Infrastructure considerations include specialized laboratory space with controlled temperature and vibration isolation, adding 15-20% to implementation costs compared to conventional diagnostic platforms. However, Raman's minimal waste generation reduces disposal costs by approximately 60% compared to chemical-intensive methods.
The total cost of ownership analysis reveals that while Raman spectroscopy requires higher initial investment, its 10-year operational costs typically fall 25-30% below comparable conventional diagnostic platforms when accounting for reagents, maintenance, and personnel expenses. This long-term economic advantage must be balanced against immediate budgetary constraints and institutional capacity for technology adoption.
Operational costs must also be evaluated comprehensively. While Raman systems have minimal reagent requirements, reducing recurring expenses by approximately 30-40% compared to chemical-based diagnostics, they demand specialized maintenance. Annual maintenance contracts typically cost 8-12% of the initial investment, and specialized technicians command higher salaries than conventional laboratory personnel.
Training represents another substantial cost factor. Laboratory staff require extensive training (40-60 hours initially) to operate Raman equipment effectively and interpret spectral data accurately. This training investment exceeds that of conventional diagnostic methods by approximately 200%, though this gap narrows with time as expertise develops.
The return on investment timeline differs significantly from other diagnostic approaches. Raman diagnostics typically achieve financial break-even in 3-5 years, compared to 1-2 years for conventional methods. However, long-term economic benefits emerge through reduced per-test costs (approximately $2-5 per Raman test versus $8-15 for comparable molecular tests) and minimal consumable requirements.
Scalability economics favor Raman technology in high-volume settings. After initial implementation, marginal costs decrease substantially with increased testing volume, creating economies of scale that outperform traditional diagnostics after approximately 10,000 tests annually. This makes Raman particularly cost-effective for large hospitals and reference laboratories.
Infrastructure considerations include specialized laboratory space with controlled temperature and vibration isolation, adding 15-20% to implementation costs compared to conventional diagnostic platforms. However, Raman's minimal waste generation reduces disposal costs by approximately 60% compared to chemical-intensive methods.
The total cost of ownership analysis reveals that while Raman spectroscopy requires higher initial investment, its 10-year operational costs typically fall 25-30% below comparable conventional diagnostic platforms when accounting for reagents, maintenance, and personnel expenses. This long-term economic advantage must be balanced against immediate budgetary constraints and institutional capacity for technology adoption.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!