Emission Control Catalyst Efficiency Using Temperature Programmed Reduction
MAR 7, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Catalyst Efficiency Research Background and Objectives
Emission control catalysts have emerged as critical components in addressing global environmental challenges, particularly in reducing harmful pollutants from automotive and industrial sources. The increasing stringency of environmental regulations worldwide, including Euro VI standards in Europe and Tier 3 standards in the United States, has driven unprecedented demand for high-performance catalytic systems. These regulatory frameworks mandate significant reductions in nitrogen oxides, carbon monoxide, hydrocarbons, and particulate matter emissions, necessitating continuous advancement in catalyst technology.
The automotive industry's transition toward electrification presents both challenges and opportunities for catalyst development. While electric vehicle adoption is accelerating, internal combustion engines will remain prevalent for decades, particularly in commercial vehicles and emerging markets. This reality underscores the continued importance of optimizing catalyst efficiency to meet increasingly strict emission standards while maintaining cost-effectiveness and durability.
Temperature Programmed Reduction has gained prominence as a sophisticated analytical technique for characterizing catalyst performance and understanding fundamental reaction mechanisms. This methodology enables researchers to systematically evaluate how catalysts respond to controlled temperature variations, providing crucial insights into active site distribution, metal-support interactions, and reduction behavior under realistic operating conditions.
Current catalyst efficiency research faces several technical challenges, including the need to maintain performance across wider temperature ranges, improve low-temperature activity for cold-start conditions, and enhance durability under harsh operating environments. The complexity of modern exhaust aftertreatment systems, incorporating multiple catalyst types and advanced control strategies, demands comprehensive characterization methods that can provide detailed mechanistic understanding.
The primary objective of employing Temperature Programmed Reduction in catalyst efficiency research is to establish quantitative relationships between catalyst structure and performance parameters. This approach aims to identify optimal formulations, predict catalyst behavior under various operating conditions, and accelerate the development of next-generation emission control technologies. By leveraging TPR methodology, researchers seek to bridge the gap between fundamental catalyst science and practical automotive applications, ultimately contributing to cleaner transportation technologies and improved air quality.
The automotive industry's transition toward electrification presents both challenges and opportunities for catalyst development. While electric vehicle adoption is accelerating, internal combustion engines will remain prevalent for decades, particularly in commercial vehicles and emerging markets. This reality underscores the continued importance of optimizing catalyst efficiency to meet increasingly strict emission standards while maintaining cost-effectiveness and durability.
Temperature Programmed Reduction has gained prominence as a sophisticated analytical technique for characterizing catalyst performance and understanding fundamental reaction mechanisms. This methodology enables researchers to systematically evaluate how catalysts respond to controlled temperature variations, providing crucial insights into active site distribution, metal-support interactions, and reduction behavior under realistic operating conditions.
Current catalyst efficiency research faces several technical challenges, including the need to maintain performance across wider temperature ranges, improve low-temperature activity for cold-start conditions, and enhance durability under harsh operating environments. The complexity of modern exhaust aftertreatment systems, incorporating multiple catalyst types and advanced control strategies, demands comprehensive characterization methods that can provide detailed mechanistic understanding.
The primary objective of employing Temperature Programmed Reduction in catalyst efficiency research is to establish quantitative relationships between catalyst structure and performance parameters. This approach aims to identify optimal formulations, predict catalyst behavior under various operating conditions, and accelerate the development of next-generation emission control technologies. By leveraging TPR methodology, researchers seek to bridge the gap between fundamental catalyst science and practical automotive applications, ultimately contributing to cleaner transportation technologies and improved air quality.
Market Demand for Advanced Emission Control Technologies
The global automotive industry faces unprecedented pressure to reduce emissions, driving substantial market demand for advanced emission control technologies. Stringent environmental regulations across major markets, including Euro 7 standards in Europe, Tier 3 regulations in the United States, and China VI standards, mandate significant reductions in nitrogen oxides, particulate matter, and other harmful pollutants. These regulatory frameworks create a compelling market environment where automotive manufacturers must invest heavily in sophisticated catalyst systems to meet compliance requirements.
Heavy-duty vehicle segments represent particularly lucrative market opportunities for advanced emission control solutions. Commercial trucking, marine engines, and industrial equipment operators face increasing operational costs associated with emissions non-compliance, including substantial fines and operational restrictions in low-emission zones. This regulatory pressure translates into strong demand for high-efficiency catalytic systems that can maintain performance across diverse operating conditions and extended service intervals.
The passenger vehicle electrification trend paradoxically intensifies demand for advanced emission control technologies in remaining internal combustion engine applications. As automakers transition their portfolios, they require increasingly sophisticated catalyst systems to maximize the environmental performance of conventional powertrains during the transition period. Hybrid vehicle architectures present unique challenges, requiring catalysts that can efficiently handle intermittent engine operation and rapid temperature cycling.
Industrial applications beyond automotive sectors demonstrate growing market appetite for advanced emission control solutions. Power generation facilities, chemical processing plants, and manufacturing operations face tightening environmental standards that necessitate deployment of high-performance catalytic systems. These applications often require customized solutions capable of handling specific pollutant profiles and operating conditions.
Emerging markets present significant growth opportunities as developing economies implement stricter environmental standards. Countries across Asia-Pacific, Latin America, and Africa are progressively adopting more stringent emission regulations, creating substantial demand for proven emission control technologies. Local manufacturing requirements and technology transfer expectations in these markets drive demand for adaptable, cost-effective catalyst solutions.
The market increasingly values catalyst systems that demonstrate measurable efficiency improvements through advanced characterization techniques. End users seek technologies backed by comprehensive performance data, including temperature programmed reduction analysis, to ensure optimal return on investment and regulatory compliance certainty.
Heavy-duty vehicle segments represent particularly lucrative market opportunities for advanced emission control solutions. Commercial trucking, marine engines, and industrial equipment operators face increasing operational costs associated with emissions non-compliance, including substantial fines and operational restrictions in low-emission zones. This regulatory pressure translates into strong demand for high-efficiency catalytic systems that can maintain performance across diverse operating conditions and extended service intervals.
The passenger vehicle electrification trend paradoxically intensifies demand for advanced emission control technologies in remaining internal combustion engine applications. As automakers transition their portfolios, they require increasingly sophisticated catalyst systems to maximize the environmental performance of conventional powertrains during the transition period. Hybrid vehicle architectures present unique challenges, requiring catalysts that can efficiently handle intermittent engine operation and rapid temperature cycling.
Industrial applications beyond automotive sectors demonstrate growing market appetite for advanced emission control solutions. Power generation facilities, chemical processing plants, and manufacturing operations face tightening environmental standards that necessitate deployment of high-performance catalytic systems. These applications often require customized solutions capable of handling specific pollutant profiles and operating conditions.
Emerging markets present significant growth opportunities as developing economies implement stricter environmental standards. Countries across Asia-Pacific, Latin America, and Africa are progressively adopting more stringent emission regulations, creating substantial demand for proven emission control technologies. Local manufacturing requirements and technology transfer expectations in these markets drive demand for adaptable, cost-effective catalyst solutions.
The market increasingly values catalyst systems that demonstrate measurable efficiency improvements through advanced characterization techniques. End users seek technologies backed by comprehensive performance data, including temperature programmed reduction analysis, to ensure optimal return on investment and regulatory compliance certainty.
Current TPR Analysis Limitations in Catalyst Research
Temperature Programmed Reduction (TPR) has emerged as a fundamental characterization technique for evaluating emission control catalysts, yet several inherent limitations constrain its effectiveness in comprehensive catalyst research. The conventional TPR methodology faces significant challenges in providing complete insights into the complex redox behaviors that govern catalyst performance in real-world automotive applications.
One primary limitation lies in the oversimplified experimental conditions employed in standard TPR analysis. Traditional TPR experiments utilize pure hydrogen as the reducing agent under controlled laboratory conditions, which fails to replicate the complex gas mixtures and dynamic operating environments encountered in actual exhaust systems. This disconnect between laboratory conditions and real-world applications creates substantial gaps in understanding catalyst behavior under practical operating scenarios.
The temporal resolution of conventional TPR systems presents another critical constraint. Most commercial TPR instruments operate with relatively slow heating rates and limited data acquisition frequencies, making it challenging to capture rapid redox transitions that occur during catalyst operation. This limitation becomes particularly problematic when studying modern three-way catalysts that must respond rapidly to fluctuating air-fuel ratios in engine exhaust streams.
Quantitative analysis represents a significant challenge in current TPR methodologies. While TPR provides valuable qualitative information about reduction temperatures and peak characteristics, converting these signals into meaningful quantitative data regarding active site concentrations, dispersion, and accessibility remains complex and often unreliable. The overlapping reduction peaks from different metal species and support interactions further complicate accurate deconvolution and quantification efforts.
Sample preparation and handling procedures introduce additional uncertainties in TPR analysis. Catalyst samples often undergo pretreatment steps that may not accurately reflect their actual state during operation. Furthermore, the static nature of TPR measurements fails to account for the dynamic restructuring and sintering processes that occur under real operating conditions, particularly at elevated temperatures typical of exhaust aftertreatment systems.
The interpretation of TPR data becomes increasingly complex when dealing with multi-component catalyst systems commonly used in modern emission control applications. The interactions between different active phases, promoters, and support materials create intricate reduction profiles that are difficult to deconvolute using conventional analysis approaches. This complexity often leads to oversimplified interpretations that may not accurately reflect the true catalyst behavior.
Current TPR instrumentation also faces limitations in terms of detection sensitivity and selectivity. While hydrogen consumption can be monitored effectively, the simultaneous analysis of product formation and intermediate species remains challenging with standard equipment configurations. This limitation restricts the ability to fully understand the mechanistic pathways involved in catalyst reduction and subsequent catalytic processes.
One primary limitation lies in the oversimplified experimental conditions employed in standard TPR analysis. Traditional TPR experiments utilize pure hydrogen as the reducing agent under controlled laboratory conditions, which fails to replicate the complex gas mixtures and dynamic operating environments encountered in actual exhaust systems. This disconnect between laboratory conditions and real-world applications creates substantial gaps in understanding catalyst behavior under practical operating scenarios.
The temporal resolution of conventional TPR systems presents another critical constraint. Most commercial TPR instruments operate with relatively slow heating rates and limited data acquisition frequencies, making it challenging to capture rapid redox transitions that occur during catalyst operation. This limitation becomes particularly problematic when studying modern three-way catalysts that must respond rapidly to fluctuating air-fuel ratios in engine exhaust streams.
Quantitative analysis represents a significant challenge in current TPR methodologies. While TPR provides valuable qualitative information about reduction temperatures and peak characteristics, converting these signals into meaningful quantitative data regarding active site concentrations, dispersion, and accessibility remains complex and often unreliable. The overlapping reduction peaks from different metal species and support interactions further complicate accurate deconvolution and quantification efforts.
Sample preparation and handling procedures introduce additional uncertainties in TPR analysis. Catalyst samples often undergo pretreatment steps that may not accurately reflect their actual state during operation. Furthermore, the static nature of TPR measurements fails to account for the dynamic restructuring and sintering processes that occur under real operating conditions, particularly at elevated temperatures typical of exhaust aftertreatment systems.
The interpretation of TPR data becomes increasingly complex when dealing with multi-component catalyst systems commonly used in modern emission control applications. The interactions between different active phases, promoters, and support materials create intricate reduction profiles that are difficult to deconvolute using conventional analysis approaches. This complexity often leads to oversimplified interpretations that may not accurately reflect the true catalyst behavior.
Current TPR instrumentation also faces limitations in terms of detection sensitivity and selectivity. While hydrogen consumption can be monitored effectively, the simultaneous analysis of product formation and intermediate species remains challenging with standard equipment configurations. This limitation restricts the ability to fully understand the mechanistic pathways involved in catalyst reduction and subsequent catalytic processes.
Existing TPR Solutions for Catalyst Efficiency Analysis
01 Catalyst composition and precious metal loading optimization
Improving emission control catalyst efficiency through optimized catalyst compositions, including the selection and ratio of precious metals such as platinum, palladium, and rhodium. The loading amount, dispersion, and particle size of these active metals on support materials significantly affect catalytic activity and durability. Advanced formulations focus on maximizing metal utilization while minimizing costs and maintaining high conversion efficiency for pollutants.- Use of precious metal catalysts for emission control: Precious metals such as platinum, palladium, and rhodium are widely used as active catalytic materials in emission control systems. These metals demonstrate high catalytic activity for converting harmful pollutants including carbon monoxide, hydrocarbons, and nitrogen oxides into less harmful substances. The efficiency of these catalysts can be enhanced through optimized dispersion on support materials and controlled particle size distribution to maximize the active surface area available for catalytic reactions.
- Catalyst support materials and structures: The selection and design of catalyst support materials play a crucial role in determining overall catalyst efficiency. Various support structures including ceramic honeycomb substrates, zeolites, and metal oxide carriers provide high surface area and thermal stability. The support material's properties such as porosity, pore size distribution, and thermal resistance directly influence the dispersion of active catalytic components and the durability of the catalyst system under high-temperature exhaust conditions.
- Oxygen storage capacity enhancement: Incorporating oxygen storage components such as cerium oxide and cerium-zirconium mixed oxides significantly improves catalyst performance under fluctuating air-fuel ratio conditions. These materials can store oxygen during lean conditions and release it during rich conditions, thereby maintaining optimal catalytic activity across varying exhaust compositions. The oxygen storage capacity helps to buffer transient changes in exhaust gas composition and extends the operational window for efficient pollutant conversion.
- Catalyst aging resistance and thermal stability: Improving the thermal stability and resistance to aging is essential for maintaining long-term catalyst efficiency. Various approaches include the use of stabilizing additives, protective coatings, and advanced preparation methods that prevent sintering of active metal particles and degradation of support materials at high temperatures. Enhanced thermal stability ensures that the catalyst maintains its structural integrity and catalytic activity throughout its operational lifetime, even under severe thermal cycling conditions typical in automotive applications.
- Multi-layer and zoned catalyst configurations: Advanced catalyst systems employ multi-layer or zoned configurations to optimize the conversion of different pollutants at various temperature ranges and positions within the exhaust system. These designs may include separate layers for oxidation and reduction reactions, or zones with different catalyst compositions tailored to specific exhaust conditions. Such configurations allow for improved light-off performance at low temperatures while maintaining high conversion efficiency at elevated temperatures, thereby enhancing overall system performance across the entire operating range.
02 Support material and washcoat technology
Enhancement of catalyst efficiency through advanced support materials and washcoat formulations. The support structure, surface area, pore size distribution, and thermal stability of materials like alumina, ceria-zirconia, and zeolites play crucial roles in catalyst performance. Washcoat technology involves optimizing the coating process, layer thickness, and adhesion properties to maximize active site accessibility and pollutant conversion.Expand Specific Solutions03 Catalyst aging resistance and thermal durability
Technologies focused on improving catalyst longevity and maintaining efficiency under high-temperature operating conditions. This includes the development of thermally stable materials, sintering-resistant formulations, and protective additives that prevent catalyst deactivation. Methods to enhance resistance to poisoning from fuel impurities and maintain catalytic activity over extended service life are critical for long-term emission control performance.Expand Specific Solutions04 Three-way catalyst and oxygen storage capacity
Optimization of three-way catalysts for simultaneous reduction of nitrogen oxides, carbon monoxide, and hydrocarbons. Enhancement of oxygen storage capacity through ceria-based materials and mixed oxides enables efficient operation across varying air-fuel ratios. The dynamic oxygen storage and release capability is essential for maintaining high conversion efficiency during transient engine operating conditions and cold start scenarios.Expand Specific Solutions05 Catalyst structure and substrate design
Improvements in catalyst efficiency through optimized substrate geometries, cell densities, and flow-through designs. Honeycomb structures, wall-flow filters, and advanced substrate configurations enhance mass transfer, reduce pressure drop, and increase contact between exhaust gases and catalytic surfaces. Innovative designs balance mechanical strength, thermal shock resistance, and catalytic performance to achieve superior emission control across various operating conditions.Expand Specific Solutions
Key Players in Emission Control Catalyst Industry
The emission control catalyst efficiency research field represents a mature technology sector within the automotive industry's transition phase toward stricter environmental regulations. The market demonstrates substantial scale driven by global emission standards like Euro 6 and EPA Tier 3, with established automotive manufacturers including Ford Global Technologies LLC, Toyota Motor Corp., Honda Motor Co., BMW AG, Mercedes-Benz Group AG, and Volkswagen AG leading development efforts. Technology maturity varies significantly across the competitive landscape, where traditional catalyst specialists like Johnson Matthey Plc and Cataler Corp. possess advanced temperature programmed reduction expertise, while automotive OEMs such as GM Global Technology Operations LLC and Hyundai Motor Co. focus on integration optimization. Chinese players including China Petroleum & Chemical Corp. and SINOPEC Beijing Research Institute are rapidly advancing their catalyst technologies. Component suppliers like DENSO Corp., Robert Bosch GmbH, and Continental Emitec demonstrate sophisticated substrate and system integration capabilities, indicating a highly competitive environment with established technological foundations but ongoing innovation in efficiency optimization methodologies.
Toyota Motor Corp.
Technical Solution: Toyota utilizes temperature programmed reduction techniques to enhance the efficiency of their three-way catalysts and lean NOx trap systems. Their TPR research focuses on understanding the reducibility characteristics of cerium-zirconium mixed oxides and precious metal dispersion effects on catalyst performance. The company employs TPR analysis to optimize catalyst preparation methods, particularly for achieving optimal metal-support interactions that enhance oxygen storage capacity and catalytic activity. Toyota's TPR methodology includes systematic studies of reduction peak shifts corresponding to different catalyst aging states, enabling development of more durable emission control systems. Their research integrates TPR data with engine dynamometer testing to validate catalyst performance improvements and ensure compliance with stringent emission standards across various driving conditions.
Strengths: Extensive automotive expertise with strong focus on durability and real-world performance validation through comprehensive testing. Weaknesses: Conservative approach to new technologies may slow adoption of breakthrough catalyst innovations.
Cataler Corp.
Technical Solution: Cataler Corporation develops emission control catalysts using TPR analysis to optimize catalyst formulations for automotive and industrial applications. Their TPR methodology focuses on characterizing the reduction behavior of multi-component catalyst systems, particularly for three-way catalysts and diesel aftertreatment systems. The company employs TPR techniques to understand metal-support interactions and optimize precious metal loading for maximum catalytic efficiency. Their research includes systematic TPR studies to correlate reduction temperatures with catalyst activity profiles, enabling development of cost-effective catalyst formulations with reduced precious metal content while maintaining performance standards. Cataler integrates TPR data with accelerated aging tests to ensure catalyst durability and compliance with emission regulations across various market segments.
Strengths: Focused catalyst manufacturer with deep expertise in TPR analysis and cost-effective catalyst development for diverse applications. Weaknesses: Smaller scale compared to major automotive suppliers may limit research resources and global market penetration capabilities.
Core TPR Innovations for Emission Control Applications
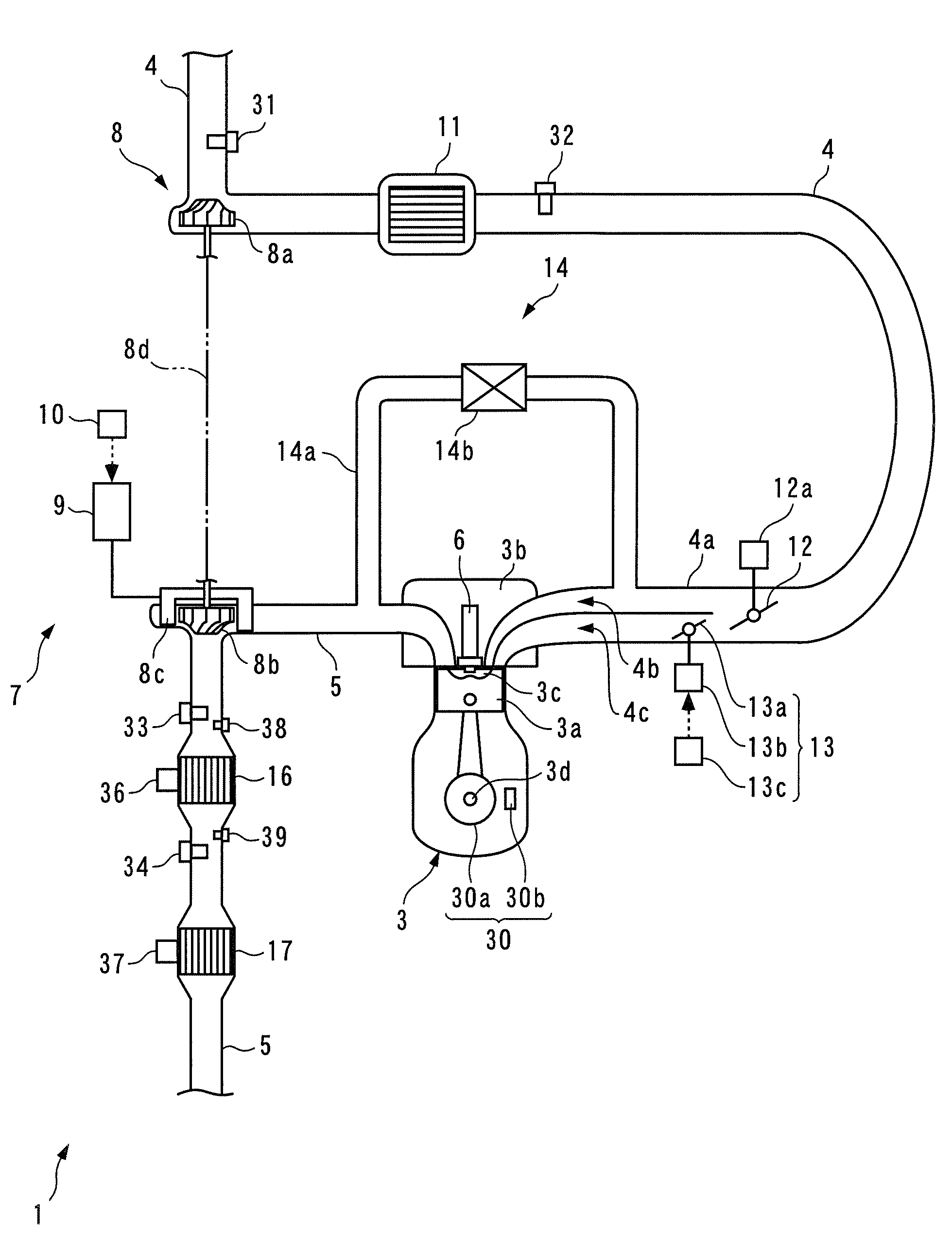
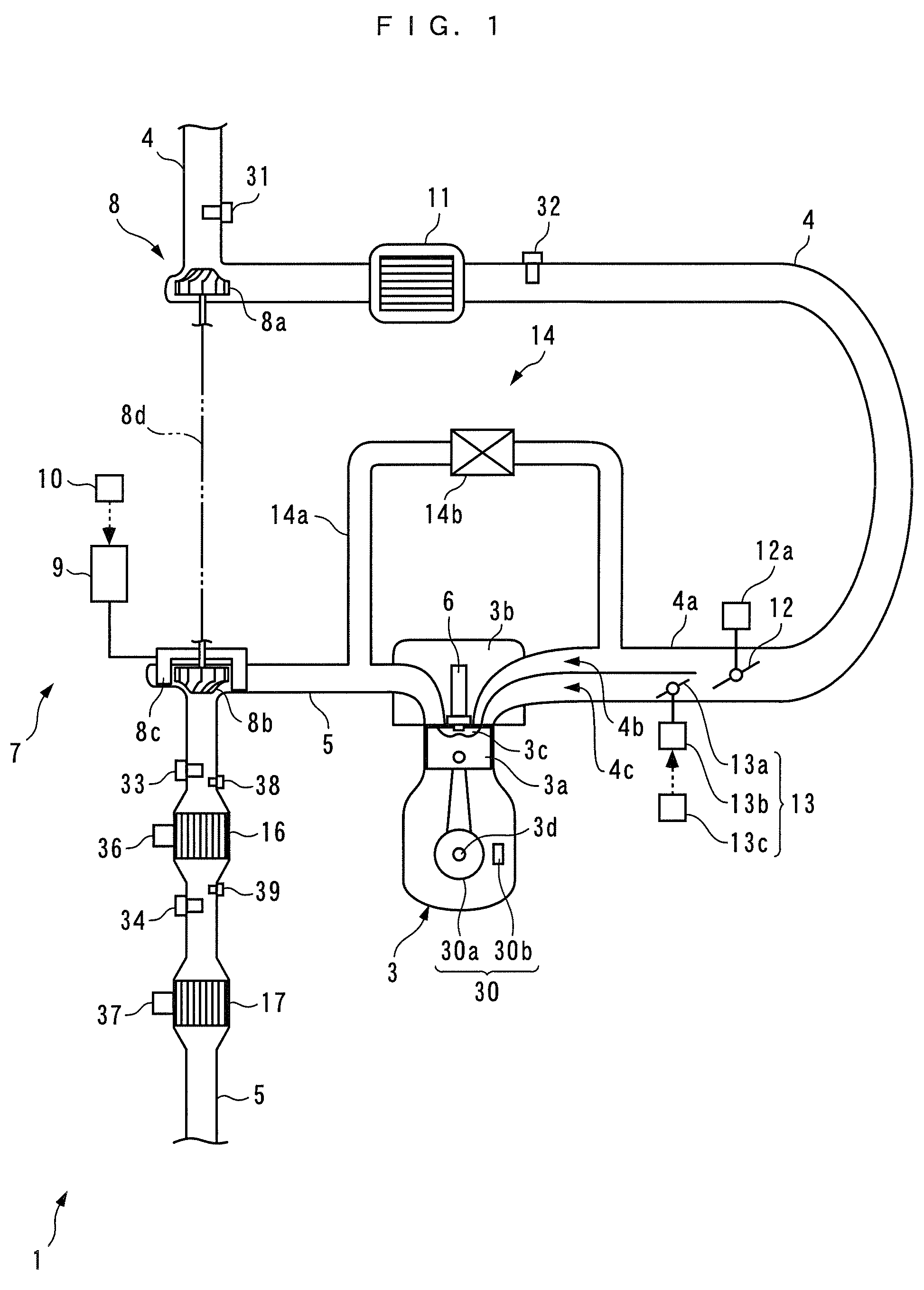
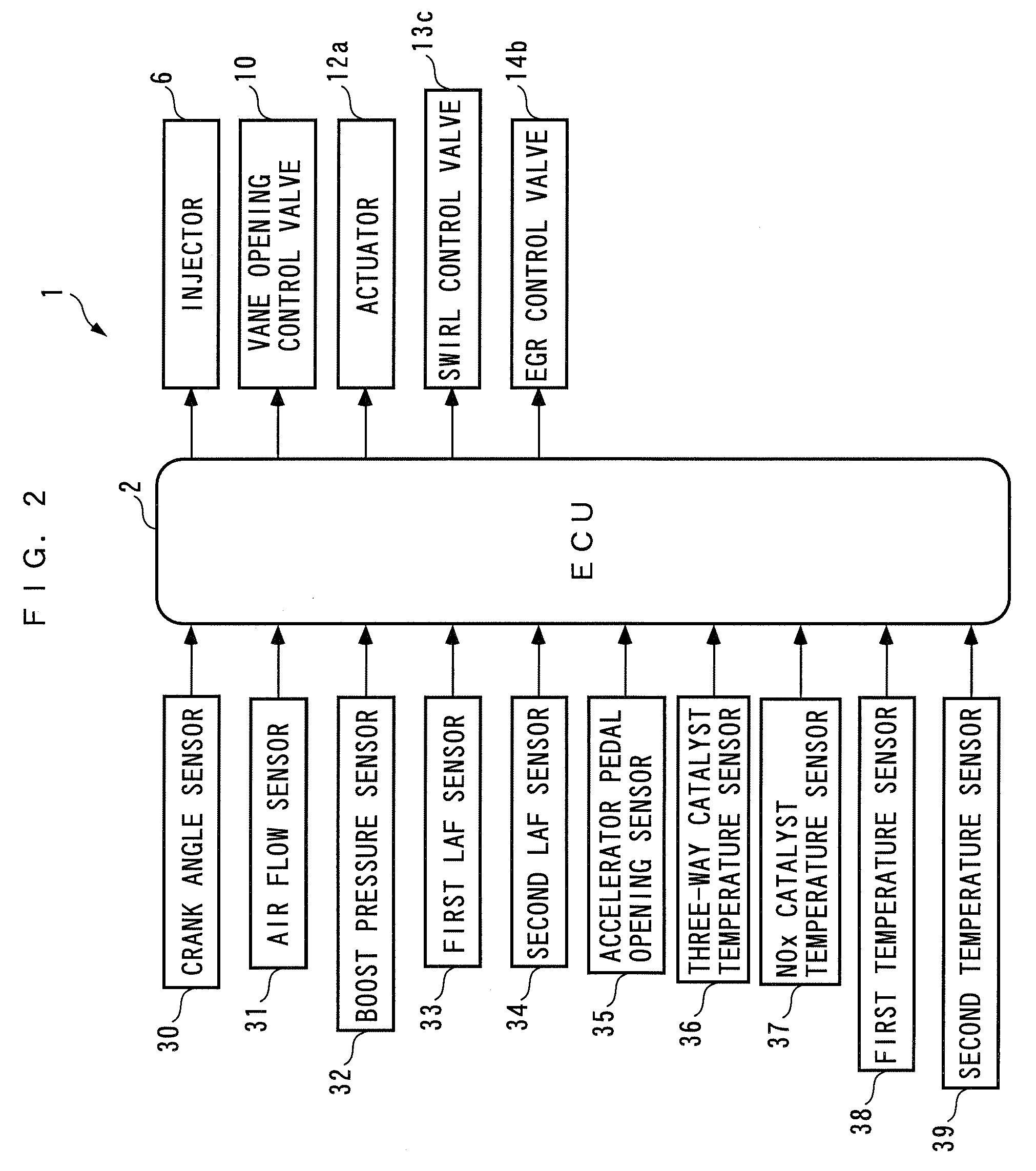
Exhaust emission control device and method for internal combustion engine, and engine control unit
PatentInactiveUS8117829B2
Innovation
- An exhaust emission control device that estimates the degradation of the upstream catalyst and adjusts the reduction control time period to ensure a just enough reducing agent is supplied to the NOx catalyst, considering the consumption variance based on catalyst degradation, thereby optimizing NOx reduction and fuel economy.
Internal combustion engine emission control apparatus and method
PatentInactiveEP1355049A3
Innovation
- The system employs split passages with different pipe lengths and exhaust choke valves to distribute exhaust gas flow proportions, ensuring that at least one emission control catalyst operates within a predetermined temperature condition, thereby maintaining high purification rates across a broader temperature range.
Environmental Regulations for Automotive Emissions
The automotive industry operates under an increasingly stringent framework of environmental regulations designed to minimize vehicular emissions and their impact on air quality and public health. These regulations have evolved significantly over the past five decades, establishing progressively tighter limits on pollutant emissions and driving technological innovation in emission control systems.
The foundation of modern automotive emission regulations was established with the Clean Air Act in the United States during the 1970s, which introduced the first mandatory emission standards for vehicles. This landmark legislation set limits on carbon monoxide, hydrocarbons, and nitrogen oxides, fundamentally reshaping automotive design priorities. Subsequently, the European Union implemented Euro emission standards beginning in 1992, creating a parallel regulatory framework that has influenced global automotive manufacturing standards.
Current regulatory frameworks encompass multiple pollutant categories, including particulate matter, sulfur compounds, and greenhouse gases. The United States Environmental Protection Agency enforces Tier 3 standards, which require significant reductions in sulfur content in gasoline and establish fleet-average nitrogen oxide limits. Meanwhile, the European Union's Euro 6 standards impose strict real driving emissions requirements, mandating on-road testing to ensure compliance beyond laboratory conditions.
Regulatory compliance necessitates sophisticated emission control technologies, with catalytic converters serving as the primary solution for gasoline engines. These systems must demonstrate consistent performance across varying operating conditions, temperature ranges, and fuel compositions. Temperature programmed reduction testing has become essential for validating catalyst efficiency and ensuring regulatory compliance throughout the vehicle's operational lifetime.
The regulatory landscape continues evolving toward zero-emission mandates in numerous jurisdictions. California's Advanced Clean Cars II regulation requires 100% zero-emission vehicle sales by 2035, while the European Union plans to ban internal combustion engine sales by the same date. These developments intensify the importance of optimizing existing emission control technologies during the transition period.
Enforcement mechanisms include mandatory certification testing, in-use compliance monitoring, and substantial financial penalties for non-compliance. Manufacturers must demonstrate that emission control systems maintain effectiveness for at least 150,000 miles, requiring extensive durability testing and performance validation. This regulatory environment drives continuous research into catalyst efficiency optimization and advanced testing methodologies.
The foundation of modern automotive emission regulations was established with the Clean Air Act in the United States during the 1970s, which introduced the first mandatory emission standards for vehicles. This landmark legislation set limits on carbon monoxide, hydrocarbons, and nitrogen oxides, fundamentally reshaping automotive design priorities. Subsequently, the European Union implemented Euro emission standards beginning in 1992, creating a parallel regulatory framework that has influenced global automotive manufacturing standards.
Current regulatory frameworks encompass multiple pollutant categories, including particulate matter, sulfur compounds, and greenhouse gases. The United States Environmental Protection Agency enforces Tier 3 standards, which require significant reductions in sulfur content in gasoline and establish fleet-average nitrogen oxide limits. Meanwhile, the European Union's Euro 6 standards impose strict real driving emissions requirements, mandating on-road testing to ensure compliance beyond laboratory conditions.
Regulatory compliance necessitates sophisticated emission control technologies, with catalytic converters serving as the primary solution for gasoline engines. These systems must demonstrate consistent performance across varying operating conditions, temperature ranges, and fuel compositions. Temperature programmed reduction testing has become essential for validating catalyst efficiency and ensuring regulatory compliance throughout the vehicle's operational lifetime.
The regulatory landscape continues evolving toward zero-emission mandates in numerous jurisdictions. California's Advanced Clean Cars II regulation requires 100% zero-emission vehicle sales by 2035, while the European Union plans to ban internal combustion engine sales by the same date. These developments intensify the importance of optimizing existing emission control technologies during the transition period.
Enforcement mechanisms include mandatory certification testing, in-use compliance monitoring, and substantial financial penalties for non-compliance. Manufacturers must demonstrate that emission control systems maintain effectiveness for at least 150,000 miles, requiring extensive durability testing and performance validation. This regulatory environment drives continuous research into catalyst efficiency optimization and advanced testing methodologies.
Catalyst Lifecycle Assessment and Sustainability
The lifecycle assessment of emission control catalysts represents a critical framework for evaluating environmental sustainability throughout the entire catalyst operational period. This comprehensive approach encompasses raw material extraction, manufacturing processes, operational performance, and end-of-life management. Temperature programmed reduction studies provide essential data for understanding catalyst degradation patterns and optimizing replacement schedules, directly impacting the overall environmental footprint of emission control systems.
Sustainability considerations in catalyst development focus on minimizing the use of precious metals such as platinum, palladium, and rhodium. These materials, while highly effective in catalytic applications, present significant supply chain vulnerabilities and environmental extraction impacts. Research efforts increasingly target the development of alternative formulations using abundant earth elements or reduced precious metal loadings without compromising catalytic efficiency. Temperature programmed reduction analysis enables precise characterization of these alternative materials, supporting the transition toward more sustainable catalyst compositions.
The operational lifecycle phase represents the most significant environmental impact period for emission control catalysts. During this phase, catalyst efficiency directly correlates with pollutant reduction effectiveness and fuel consumption optimization. Temperature programmed reduction monitoring provides real-time insights into catalyst performance degradation, enabling predictive maintenance strategies that extend operational lifespans and reduce replacement frequency.
End-of-life catalyst management presents both challenges and opportunities for sustainability enhancement. Spent catalysts contain recoverable precious metals that can be recycled through specialized processing techniques. However, the recycling process itself requires energy-intensive procedures and generates secondary waste streams. Lifecycle assessment methodologies help quantify the net environmental benefits of recycling versus disposal, informing optimal end-of-life strategies.
Emerging sustainability metrics for catalyst systems include carbon footprint analysis, resource depletion potential, and circular economy indicators. These comprehensive assessment frameworks integrate temperature programmed reduction data with broader environmental impact categories, enabling holistic optimization of catalyst systems for both performance and sustainability objectives.
Sustainability considerations in catalyst development focus on minimizing the use of precious metals such as platinum, palladium, and rhodium. These materials, while highly effective in catalytic applications, present significant supply chain vulnerabilities and environmental extraction impacts. Research efforts increasingly target the development of alternative formulations using abundant earth elements or reduced precious metal loadings without compromising catalytic efficiency. Temperature programmed reduction analysis enables precise characterization of these alternative materials, supporting the transition toward more sustainable catalyst compositions.
The operational lifecycle phase represents the most significant environmental impact period for emission control catalysts. During this phase, catalyst efficiency directly correlates with pollutant reduction effectiveness and fuel consumption optimization. Temperature programmed reduction monitoring provides real-time insights into catalyst performance degradation, enabling predictive maintenance strategies that extend operational lifespans and reduce replacement frequency.
End-of-life catalyst management presents both challenges and opportunities for sustainability enhancement. Spent catalysts contain recoverable precious metals that can be recycled through specialized processing techniques. However, the recycling process itself requires energy-intensive procedures and generates secondary waste streams. Lifecycle assessment methodologies help quantify the net environmental benefits of recycling versus disposal, informing optimal end-of-life strategies.
Emerging sustainability metrics for catalyst systems include carbon footprint analysis, resource depletion potential, and circular economy indicators. These comprehensive assessment frameworks integrate temperature programmed reduction data with broader environmental impact categories, enabling holistic optimization of catalyst systems for both performance and sustainability objectives.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!