Quantifying Echogenicity: Best Practices in Endocrine Imaging
JAN 20, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Echogenicity Quantification Background and Objectives
Ultrasound imaging has become an indispensable diagnostic tool in endocrinology, particularly for evaluating thyroid, parathyroid, and adrenal glands. The echogenicity of tissue, which refers to the ability of tissue to reflect ultrasound waves, serves as a critical indicator for distinguishing between normal and pathological conditions. Traditionally, echogenicity assessment has relied heavily on subjective visual interpretation by radiologists, leading to significant inter-observer variability and inconsistent diagnostic outcomes. This subjective approach has created a pressing need for objective, reproducible quantification methods that can standardize diagnostic criteria and improve clinical decision-making.
The evolution of digital imaging technology and computational analysis has opened new avenues for transforming qualitative echogenicity assessment into quantitative measurements. Recent advances in image processing algorithms, machine learning techniques, and standardized imaging protocols have demonstrated the potential to extract numerical parameters from ultrasound images that correlate with tissue characteristics. These developments have sparked growing interest in establishing reliable quantification frameworks that can be integrated into routine clinical practice.
The primary objective of quantifying echogenicity in endocrine imaging is to establish objective, reproducible metrics that can accurately characterize tissue properties and differentiate between benign and malignant lesions. This involves developing standardized measurement protocols that account for technical variables such as equipment settings, probe frequency, and imaging depth. Additionally, the goal extends to creating reference databases of normal and pathological echogenicity values specific to different endocrine organs and patient populations.
Another critical objective is to enhance diagnostic accuracy and reduce dependency on operator expertise. By converting subjective visual assessments into quantifiable data, clinicians can achieve more consistent interpretations across different institutions and practitioners. Furthermore, quantitative echogenicity analysis aims to facilitate early detection of subtle tissue changes, monitor disease progression, and evaluate treatment responses with greater precision. Ultimately, these efforts seek to improve patient outcomes through more reliable, evidence-based diagnostic approaches in endocrine imaging.
The evolution of digital imaging technology and computational analysis has opened new avenues for transforming qualitative echogenicity assessment into quantitative measurements. Recent advances in image processing algorithms, machine learning techniques, and standardized imaging protocols have demonstrated the potential to extract numerical parameters from ultrasound images that correlate with tissue characteristics. These developments have sparked growing interest in establishing reliable quantification frameworks that can be integrated into routine clinical practice.
The primary objective of quantifying echogenicity in endocrine imaging is to establish objective, reproducible metrics that can accurately characterize tissue properties and differentiate between benign and malignant lesions. This involves developing standardized measurement protocols that account for technical variables such as equipment settings, probe frequency, and imaging depth. Additionally, the goal extends to creating reference databases of normal and pathological echogenicity values specific to different endocrine organs and patient populations.
Another critical objective is to enhance diagnostic accuracy and reduce dependency on operator expertise. By converting subjective visual assessments into quantifiable data, clinicians can achieve more consistent interpretations across different institutions and practitioners. Furthermore, quantitative echogenicity analysis aims to facilitate early detection of subtle tissue changes, monitor disease progression, and evaluate treatment responses with greater precision. Ultimately, these efforts seek to improve patient outcomes through more reliable, evidence-based diagnostic approaches in endocrine imaging.
Market Demand for Endocrine Imaging Solutions
The global endocrine imaging market is experiencing substantial growth driven by the rising prevalence of thyroid disorders, parathyroid diseases, and adrenal gland abnormalities. Thyroid nodules alone affect a significant portion of the adult population worldwide, with detection rates increasing due to improved screening protocols and heightened clinical awareness. The aging demographic in developed nations further amplifies demand, as endocrine disorders become more prevalent with age. Healthcare systems are increasingly prioritizing early detection and accurate diagnosis to reduce the burden of advanced-stage diseases and associated treatment costs.
Ultrasound imaging remains the primary modality for endocrine gland evaluation due to its non-invasive nature, real-time visualization capabilities, and cost-effectiveness compared to CT or MRI. However, current qualitative assessment methods suffer from significant inter-observer variability, creating a critical need for standardized quantification tools. Clinicians face challenges in distinguishing benign from malignant nodules, leading to unnecessary biopsies and patient anxiety. The demand for objective, reproducible echogenicity quantification methods is particularly acute in thyroid nodule characterization, where subtle textural differences can indicate malignancy risk.
Healthcare providers and imaging equipment manufacturers are actively seeking solutions that integrate seamlessly into existing clinical workflows while providing quantitative metrics that enhance diagnostic confidence. The market shows strong interest in AI-powered analysis tools and standardized reporting systems that can reduce diagnostic uncertainty. Emerging markets in Asia-Pacific regions demonstrate accelerated adoption rates as healthcare infrastructure modernizes and access to advanced imaging technologies expands.
The shift toward personalized medicine and precision diagnostics further drives demand for quantitative imaging biomarkers in endocrine care. Regulatory bodies increasingly recognize the value of standardized imaging metrics for clinical decision-making, creating favorable conditions for innovative quantification technologies. The convergence of artificial intelligence, advanced image processing algorithms, and cloud-based diagnostic platforms presents significant commercial opportunities for solutions addressing echogenicity quantification challenges in endocrine imaging applications.
Ultrasound imaging remains the primary modality for endocrine gland evaluation due to its non-invasive nature, real-time visualization capabilities, and cost-effectiveness compared to CT or MRI. However, current qualitative assessment methods suffer from significant inter-observer variability, creating a critical need for standardized quantification tools. Clinicians face challenges in distinguishing benign from malignant nodules, leading to unnecessary biopsies and patient anxiety. The demand for objective, reproducible echogenicity quantification methods is particularly acute in thyroid nodule characterization, where subtle textural differences can indicate malignancy risk.
Healthcare providers and imaging equipment manufacturers are actively seeking solutions that integrate seamlessly into existing clinical workflows while providing quantitative metrics that enhance diagnostic confidence. The market shows strong interest in AI-powered analysis tools and standardized reporting systems that can reduce diagnostic uncertainty. Emerging markets in Asia-Pacific regions demonstrate accelerated adoption rates as healthcare infrastructure modernizes and access to advanced imaging technologies expands.
The shift toward personalized medicine and precision diagnostics further drives demand for quantitative imaging biomarkers in endocrine care. Regulatory bodies increasingly recognize the value of standardized imaging metrics for clinical decision-making, creating favorable conditions for innovative quantification technologies. The convergence of artificial intelligence, advanced image processing algorithms, and cloud-based diagnostic platforms presents significant commercial opportunities for solutions addressing echogenicity quantification challenges in endocrine imaging applications.
Current Challenges in Echogenicity Assessment
Echogenicity assessment in endocrine imaging faces significant technical and methodological challenges that hinder standardization and clinical reproducibility. The subjective nature of visual interpretation remains the primary obstacle, as radiologists rely heavily on personal experience and perception when evaluating tissue brightness patterns. This subjectivity introduces substantial inter-observer and intra-observer variability, making it difficult to establish consistent diagnostic criteria across different institutions and practitioners.
The lack of standardized reference frameworks compounds this problem. Current clinical practice often describes echogenicity using relative terms such as hypoechoic, isoechoic, or hyperechoic, comparing target tissues against adjacent structures like muscles or normal glandular tissue. However, these reference tissues themselves exhibit variable echogenicity depending on patient characteristics, equipment settings, and scanning techniques, leading to inconsistent baseline comparisons.
Technical factors related to ultrasound equipment present another layer of complexity. Different manufacturers employ proprietary image processing algorithms, gain settings, and frequency optimization strategies that significantly affect the displayed echogenicity. The absence of universal calibration standards means that identical tissues may appear differently across various ultrasound systems, limiting the transferability of diagnostic criteria and quantitative measurements between platforms.
Image acquisition parameters introduce additional variability that directly impacts echogenicity quantification. Factors such as probe frequency selection, focal zone positioning, time-gain compensation settings, and overall gain adjustments can dramatically alter the perceived brightness of tissues. Even subtle variations in probe pressure, scanning angle, or patient positioning can affect acoustic impedance matching and subsequent echo signal intensity, making reproducible measurements challenging.
The heterogeneous nature of endocrine pathologies further complicates standardized assessment. Thyroid nodules, for example, may exhibit mixed echogenicity patterns with calcifications, cystic components, or irregular internal architecture. Quantifying overall echogenicity in such heterogeneous lesions requires sophisticated analytical approaches that can account for spatial variations while providing clinically meaningful summary metrics.
Current quantification methods also struggle with limited dynamic range representation and inadequate correction for depth-dependent attenuation. As ultrasound waves penetrate deeper tissues, signal attenuation occurs, causing deeper structures to appear artificially hypoechoic. Existing compensation algorithms often fail to fully correct these physical phenomena, introducing systematic biases in echogenicity measurements that vary with lesion depth and tissue composition.
The lack of standardized reference frameworks compounds this problem. Current clinical practice often describes echogenicity using relative terms such as hypoechoic, isoechoic, or hyperechoic, comparing target tissues against adjacent structures like muscles or normal glandular tissue. However, these reference tissues themselves exhibit variable echogenicity depending on patient characteristics, equipment settings, and scanning techniques, leading to inconsistent baseline comparisons.
Technical factors related to ultrasound equipment present another layer of complexity. Different manufacturers employ proprietary image processing algorithms, gain settings, and frequency optimization strategies that significantly affect the displayed echogenicity. The absence of universal calibration standards means that identical tissues may appear differently across various ultrasound systems, limiting the transferability of diagnostic criteria and quantitative measurements between platforms.
Image acquisition parameters introduce additional variability that directly impacts echogenicity quantification. Factors such as probe frequency selection, focal zone positioning, time-gain compensation settings, and overall gain adjustments can dramatically alter the perceived brightness of tissues. Even subtle variations in probe pressure, scanning angle, or patient positioning can affect acoustic impedance matching and subsequent echo signal intensity, making reproducible measurements challenging.
The heterogeneous nature of endocrine pathologies further complicates standardized assessment. Thyroid nodules, for example, may exhibit mixed echogenicity patterns with calcifications, cystic components, or irregular internal architecture. Quantifying overall echogenicity in such heterogeneous lesions requires sophisticated analytical approaches that can account for spatial variations while providing clinically meaningful summary metrics.
Current quantification methods also struggle with limited dynamic range representation and inadequate correction for depth-dependent attenuation. As ultrasound waves penetrate deeper tissues, signal attenuation occurs, causing deeper structures to appear artificially hypoechoic. Existing compensation algorithms often fail to fully correct these physical phenomena, introducing systematic biases in echogenicity measurements that vary with lesion depth and tissue composition.
Existing Echogenicity Quantification Methods
01 Image processing methods for echogenicity quantification
Various image processing techniques are employed to quantify echogenicity in ultrasound images. These methods involve analyzing pixel intensity values, applying statistical algorithms, and utilizing computational models to measure and classify tissue echogenicity levels. Advanced signal processing approaches enable objective assessment of echo patterns and their distribution within regions of interest.- Image processing methods for echogenicity quantification: Various image processing techniques are employed to quantify echogenicity in ultrasound images. These methods involve analyzing pixel intensity distributions, calculating statistical parameters, and applying algorithms to measure the brightness and texture characteristics of tissue regions. Advanced computational approaches enable objective assessment of echogenic properties by converting visual information into numerical data that can be compared across different tissue types and pathological conditions.
- Contrast agent-based echogenicity enhancement and measurement: Contrast agents are utilized to enhance echogenicity for improved visualization and quantification in ultrasound imaging. These agents contain microbubbles or other materials that increase the acoustic reflectivity of blood vessels and tissues. Quantification methods measure the enhancement patterns, intensity changes, and temporal dynamics of contrast agent distribution to assess tissue perfusion, vascularity, and pathological features.
- Machine learning and artificial intelligence for echogenicity analysis: Machine learning algorithms and artificial intelligence systems are applied to automate and improve the accuracy of echogenicity quantification. These technologies can be trained on large datasets to recognize patterns, classify tissue types, and predict diagnostic outcomes based on echogenic characteristics. Neural networks and deep learning models process ultrasound images to extract features and provide objective measurements that reduce inter-observer variability.
- Standardization and calibration techniques for echogenicity measurement: Standardization methods and calibration protocols are developed to ensure consistent and reproducible echogenicity measurements across different ultrasound systems and clinical settings. These techniques involve the use of reference phantoms, normalization procedures, and quality control measures to account for variations in equipment settings, operator technique, and patient factors. Calibration approaches enable reliable comparison of echogenicity values obtained from different examinations and institutions.
- Clinical applications of echogenicity quantification in disease diagnosis: Quantitative echogenicity assessment is applied in various clinical scenarios for disease diagnosis and monitoring. Applications include evaluation of liver steatosis, thyroid nodule characterization, breast lesion assessment, and kidney disease staging. Echogenicity measurements provide objective biomarkers that correlate with histological findings and disease severity, supporting clinical decision-making and treatment planning. These quantitative approaches complement traditional qualitative ultrasound interpretation.
02 Contrast agent-based echogenicity enhancement and measurement
Contrast agents are utilized to enhance echogenicity for improved visualization and quantification. These agents modify acoustic properties of tissues or blood flow, enabling better differentiation and measurement of echogenic characteristics. Quantification methods assess the enhancement patterns and intensity changes induced by contrast materials to provide diagnostic information.Expand Specific Solutions03 Automated classification and scoring systems for echogenicity
Automated systems employ machine learning algorithms and pattern recognition techniques to classify and score echogenicity levels. These systems analyze ultrasound data to categorize tissues based on their echogenic properties, providing standardized scoring metrics. The automation reduces observer variability and enables consistent quantitative assessment across different examinations.Expand Specific Solutions04 Three-dimensional echogenicity analysis and volumetric quantification
Three-dimensional imaging techniques enable comprehensive volumetric analysis of echogenicity. These approaches reconstruct spatial distribution of echogenic properties throughout tissue volumes, allowing for more accurate quantification compared to two-dimensional methods. Volumetric measurements provide enhanced diagnostic capabilities by capturing complete echogenic characteristics of anatomical structures.Expand Specific Solutions05 Real-time monitoring and dynamic echogenicity assessment
Real-time monitoring systems track temporal changes in echogenicity during procedures or physiological processes. These technologies enable dynamic assessment of echogenic properties, capturing variations over time. The continuous monitoring approach provides valuable information about tissue response, perfusion changes, and treatment effects through quantitative echogenicity measurements.Expand Specific Solutions
Key Players in Endocrine Imaging Systems
The field of quantifying echogenicity in endocrine imaging is experiencing significant growth as healthcare systems increasingly adopt AI-enhanced ultrasound technologies for thyroid and parathyroid diagnostics. The market demonstrates a mature competitive landscape with established medical device manufacturers like Shenzhen Mindray Bio-Medical Electronics, Siemens Healthcare Diagnostics, Koninklijke Philips, and Becton Dickinson dominating commercial deployment. Academic institutions including Xiamen University, Massachusetts Institute of Technology, and The University of Hong Kong are advancing fundamental research in quantitative ultrasound analysis. Specialized players such as AmCad BioMed are pioneering FDA-cleared computer-assisted detection systems, while diagnostic service providers like Laboratory Corporation of America Holdings integrate these technologies into clinical workflows. The technology has reached commercial maturity in breast and thyroid imaging applications, with ongoing innovation focused on AI-driven tissue characterization and automated diagnostic support systems.
Xiamen University
Technical Solution: Xiamen University has conducted extensive research on computer-aided diagnosis systems for thyroid nodule characterization using quantitative echogenicity analysis. Their research focuses on developing novel texture analysis algorithms that extract high-dimensional feature sets from ultrasound images, including first-order statistics, gray-level co-occurrence matrices, and wavelet-based features. The university has published multiple studies on standardization methodologies for echogenicity measurement, addressing challenges related to equipment variability and operator dependence. Their work includes development of deep learning models that integrate echogenicity patterns with clinical parameters for risk stratification of thyroid malignancy. Research teams have established large annotated datasets correlating quantitative echogenicity metrics with histopathological outcomes, contributing to evidence-based diagnostic criteria development.
Strengths: Strong academic research foundation with numerous peer-reviewed publications, innovative algorithm development with focus on clinical validation, collaborative networks with medical institutions. Weaknesses: Limited commercial product development and market deployment, research prototypes may lack regulatory approval for clinical use, scalability challenges for widespread implementation.
Shenzhen Mindray Bio-Medical Electronics Co., Ltd.
Technical Solution: Mindray has developed advanced ultrasound imaging systems with integrated quantitative echogenicity analysis capabilities for endocrine applications. Their solutions incorporate automated texture analysis algorithms that measure grayscale distribution patterns in thyroid and parathyroid tissues. The technology utilizes histogram-based statistical parameters including mean echogenicity, standard deviation, and skewness to characterize tissue properties. Their systems feature real-time processing capabilities with standardized reference phantoms for calibration, enabling reproducible measurements across different scanning sessions and operators. The platform integrates machine learning algorithms to correlate echogenicity patterns with pathological findings, supporting differential diagnosis of thyroid nodules and parathyroid adenomas.
Strengths: Comprehensive product portfolio with strong market presence in medical ultrasound, robust calibration methodology ensuring measurement consistency. Weaknesses: Limited published clinical validation data compared to research institutions, proprietary algorithms may lack transparency for academic research applications.
Core Innovations in Texture Analysis Algorithms
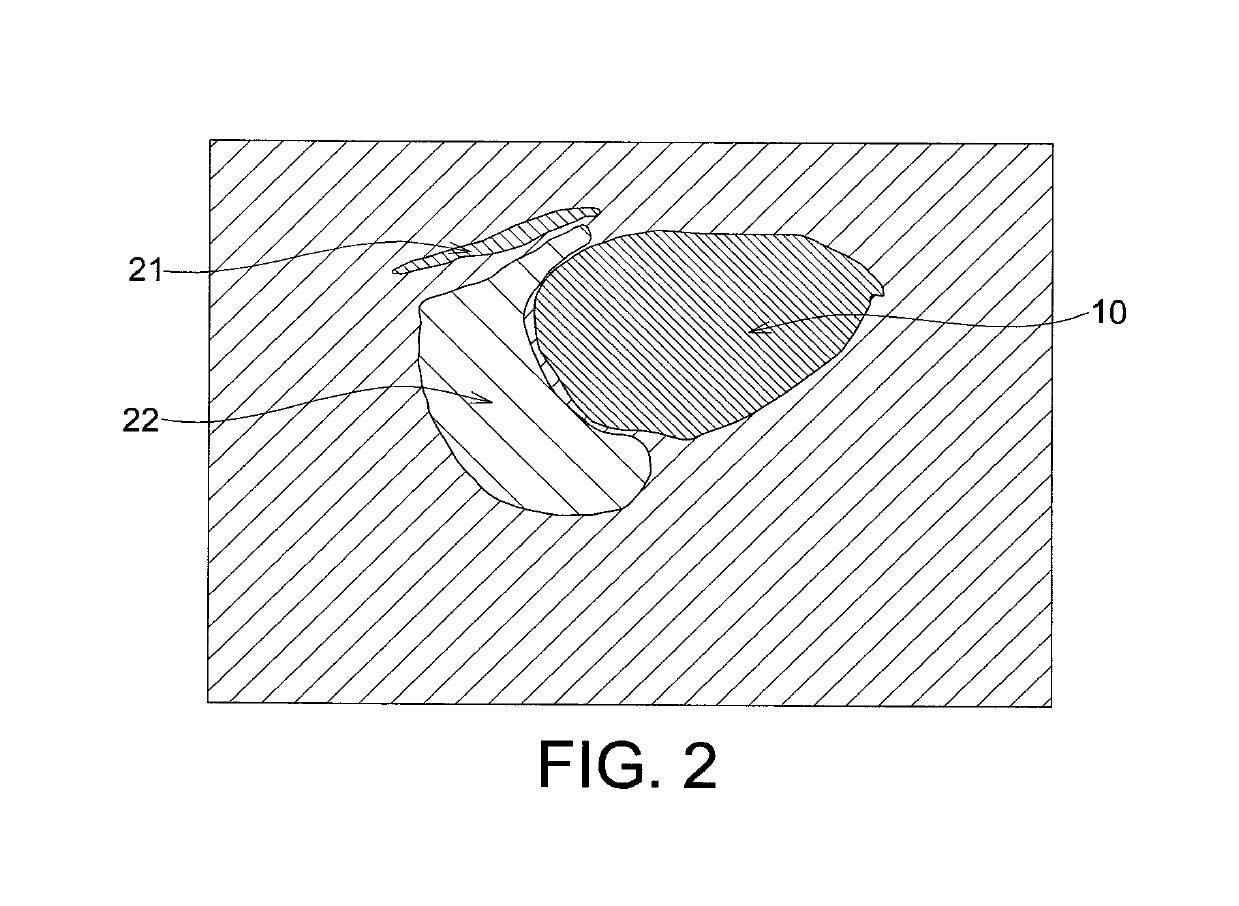
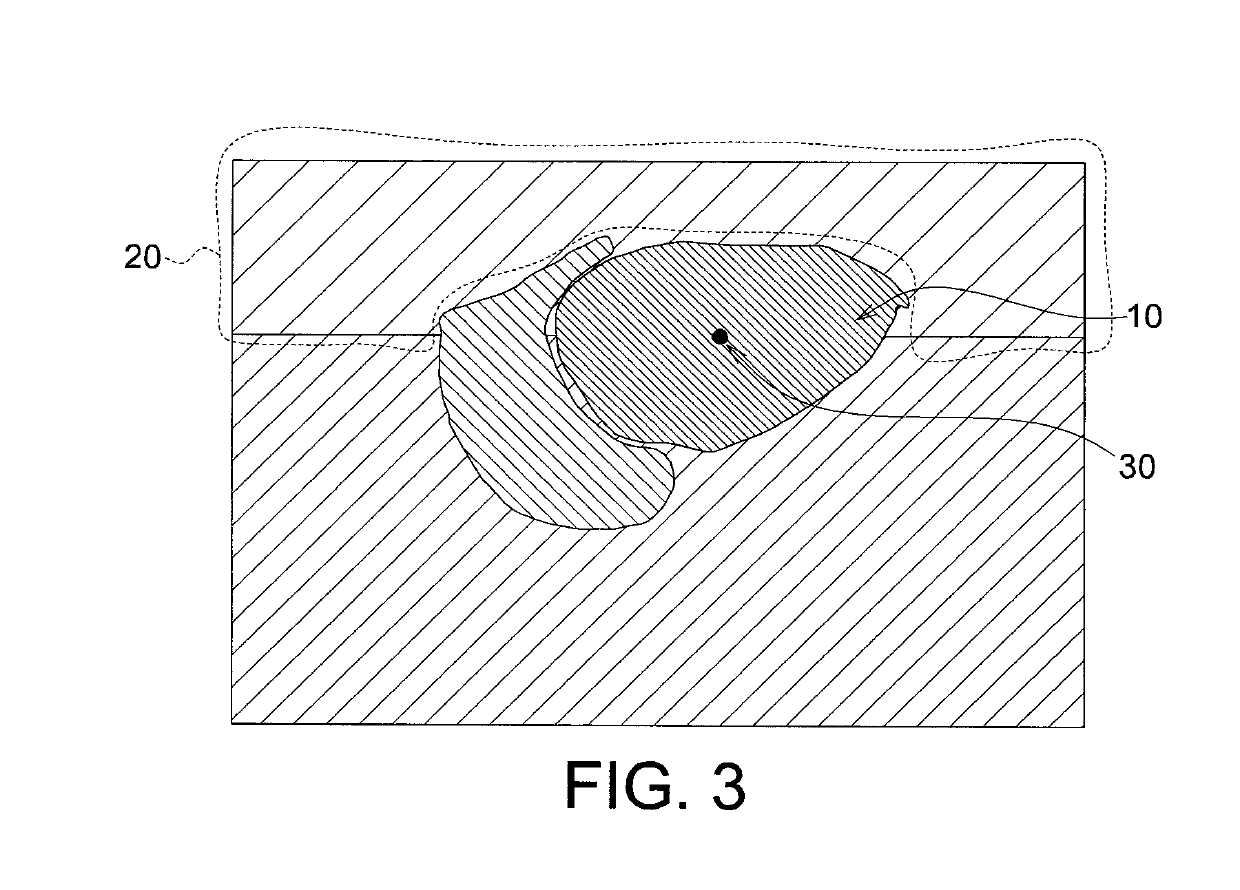
Echogenicity quantification method and calibration method for ultrasonic device using echogenicity index
PatentActiveEP3029634A1
Innovation
- An echogenicity quantification method that calculates an echogenicity index by averaging and normalizing grayscale values within a Region Of Interest (ROI) and a reference region, excluding outliers, to provide an objective and consistent measure across different ultrasonic devices.
Echogenicity quantification method and calibration method for ultrasonic device using echogenicity index
PatentActiveUS10249037B2
Innovation
- An echogenicity quantification method that calculates an echogenicity index by averaging and normalizing grayscale values within a Region Of Interest (ROI) and a reference region, excluding outliers, to provide an objective and consistent measure across different ultrasonic devices and operators.
Clinical Validation and Standardization Requirements
Clinical validation represents a critical gateway for translating quantitative echogenicity measurements from research concepts into reliable diagnostic tools for endocrine imaging. Rigorous validation protocols must demonstrate that quantitative metrics consistently correlate with histopathological findings, clinical outcomes, and established diagnostic criteria across diverse patient populations. Multi-center prospective studies are essential to establish diagnostic thresholds, sensitivity, and specificity values that can be confidently applied in routine clinical practice. These validation efforts must account for variations in patient demographics, disease stages, and comorbidities to ensure broad applicability and minimize bias in clinical decision-making.
Standardization requirements encompass multiple technical and operational dimensions that directly impact measurement reproducibility. Equipment calibration protocols must be established to ensure consistent acoustic output and image acquisition parameters across different ultrasound platforms and manufacturers. Reference phantoms with known echogenic properties should be developed specifically for endocrine applications, enabling periodic quality assurance checks and inter-institutional comparisons. Imaging protocols require detailed specification of transducer selection, frequency settings, gain adjustments, and region-of-interest placement to minimize operator-dependent variability.
Data reporting standards must define unified terminology, measurement units, and statistical methods for expressing echogenicity values. International consensus guidelines should establish minimum dataset requirements, including metadata documentation of acquisition parameters and quality indicators. Interoperability standards are necessary to facilitate seamless integration of quantitative echogenicity data into electronic health records and picture archiving systems, enabling longitudinal tracking and multi-site data aggregation.
Regulatory pathways for clinical adoption require comprehensive documentation demonstrating analytical validity, clinical validity, and clinical utility. Certification processes must address software validation, algorithm transparency, and performance monitoring in real-world settings. Continuous post-market surveillance mechanisms should track measurement accuracy degradation and identify emerging technical limitations. Training and certification programs for clinicians are equally important to ensure proper technique execution and appropriate interpretation of quantitative results within clinical contexts.
Standardization requirements encompass multiple technical and operational dimensions that directly impact measurement reproducibility. Equipment calibration protocols must be established to ensure consistent acoustic output and image acquisition parameters across different ultrasound platforms and manufacturers. Reference phantoms with known echogenic properties should be developed specifically for endocrine applications, enabling periodic quality assurance checks and inter-institutional comparisons. Imaging protocols require detailed specification of transducer selection, frequency settings, gain adjustments, and region-of-interest placement to minimize operator-dependent variability.
Data reporting standards must define unified terminology, measurement units, and statistical methods for expressing echogenicity values. International consensus guidelines should establish minimum dataset requirements, including metadata documentation of acquisition parameters and quality indicators. Interoperability standards are necessary to facilitate seamless integration of quantitative echogenicity data into electronic health records and picture archiving systems, enabling longitudinal tracking and multi-site data aggregation.
Regulatory pathways for clinical adoption require comprehensive documentation demonstrating analytical validity, clinical validity, and clinical utility. Certification processes must address software validation, algorithm transparency, and performance monitoring in real-world settings. Continuous post-market surveillance mechanisms should track measurement accuracy degradation and identify emerging technical limitations. Training and certification programs for clinicians are equally important to ensure proper technique execution and appropriate interpretation of quantitative results within clinical contexts.
Integration with PACS and Diagnostic Workflows
The seamless integration of echogenicity quantification tools with Picture Archiving and Communication Systems (PACS) represents a critical enabler for clinical adoption in endocrine imaging. Modern PACS infrastructure must accommodate advanced quantitative analysis modules that can process ultrasound images while maintaining compatibility with existing DICOM standards. This integration ensures that echogenicity measurements are automatically embedded within patient imaging records, eliminating manual data entry and reducing potential transcription errors. The technical implementation requires robust application programming interfaces that facilitate bidirectional communication between quantification software and institutional PACS servers, enabling real-time data exchange and archival storage.
Workflow optimization demands that echogenicity analysis tools operate within the natural clinical examination sequence without disrupting radiologist efficiency. Ideally, quantification algorithms should execute as background processes during image acquisition or immediately post-capture, presenting results within seconds to support immediate clinical decision-making. The user interface must be intuitive, allowing clinicians to review quantitative metrics alongside conventional B-mode images through unified viewing platforms. Structured reporting templates that incorporate standardized echogenicity parameters facilitate consistent documentation and enable downstream data mining for quality assurance and research purposes.
Interoperability challenges arise from the heterogeneous nature of healthcare IT ecosystems, where multiple vendor systems must communicate effectively. Adherence to HL7 FHIR standards and IHE profiles becomes essential for ensuring that echogenicity data can be shared across departmental boundaries and integrated with electronic health records. Cloud-based solutions are emerging as viable alternatives, offering scalable computational resources for complex quantification algorithms while maintaining secure access to imaging data through encrypted connections and compliance with healthcare privacy regulations.
The diagnostic workflow must also accommodate quality control mechanisms that validate quantification accuracy before clinical interpretation. Automated alerts for suboptimal image quality or measurement inconsistencies help maintain diagnostic reliability. Furthermore, integration with clinical decision support systems can leverage echogenicity data to trigger protocol-specific recommendations, such as suggesting biopsy for thyroid nodules exhibiting markedly hypoechoic characteristics. This comprehensive integration transforms echogenicity quantification from an isolated analytical tool into an integral component of evidence-based endocrine diagnostics.
Workflow optimization demands that echogenicity analysis tools operate within the natural clinical examination sequence without disrupting radiologist efficiency. Ideally, quantification algorithms should execute as background processes during image acquisition or immediately post-capture, presenting results within seconds to support immediate clinical decision-making. The user interface must be intuitive, allowing clinicians to review quantitative metrics alongside conventional B-mode images through unified viewing platforms. Structured reporting templates that incorporate standardized echogenicity parameters facilitate consistent documentation and enable downstream data mining for quality assurance and research purposes.
Interoperability challenges arise from the heterogeneous nature of healthcare IT ecosystems, where multiple vendor systems must communicate effectively. Adherence to HL7 FHIR standards and IHE profiles becomes essential for ensuring that echogenicity data can be shared across departmental boundaries and integrated with electronic health records. Cloud-based solutions are emerging as viable alternatives, offering scalable computational resources for complex quantification algorithms while maintaining secure access to imaging data through encrypted connections and compliance with healthcare privacy regulations.
The diagnostic workflow must also accommodate quality control mechanisms that validate quantification accuracy before clinical interpretation. Automated alerts for suboptimal image quality or measurement inconsistencies help maintain diagnostic reliability. Furthermore, integration with clinical decision support systems can leverage echogenicity data to trigger protocol-specific recommendations, such as suggesting biopsy for thyroid nodules exhibiting markedly hypoechoic characteristics. This comprehensive integration transforms echogenicity quantification from an isolated analytical tool into an integral component of evidence-based endocrine diagnostics.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!