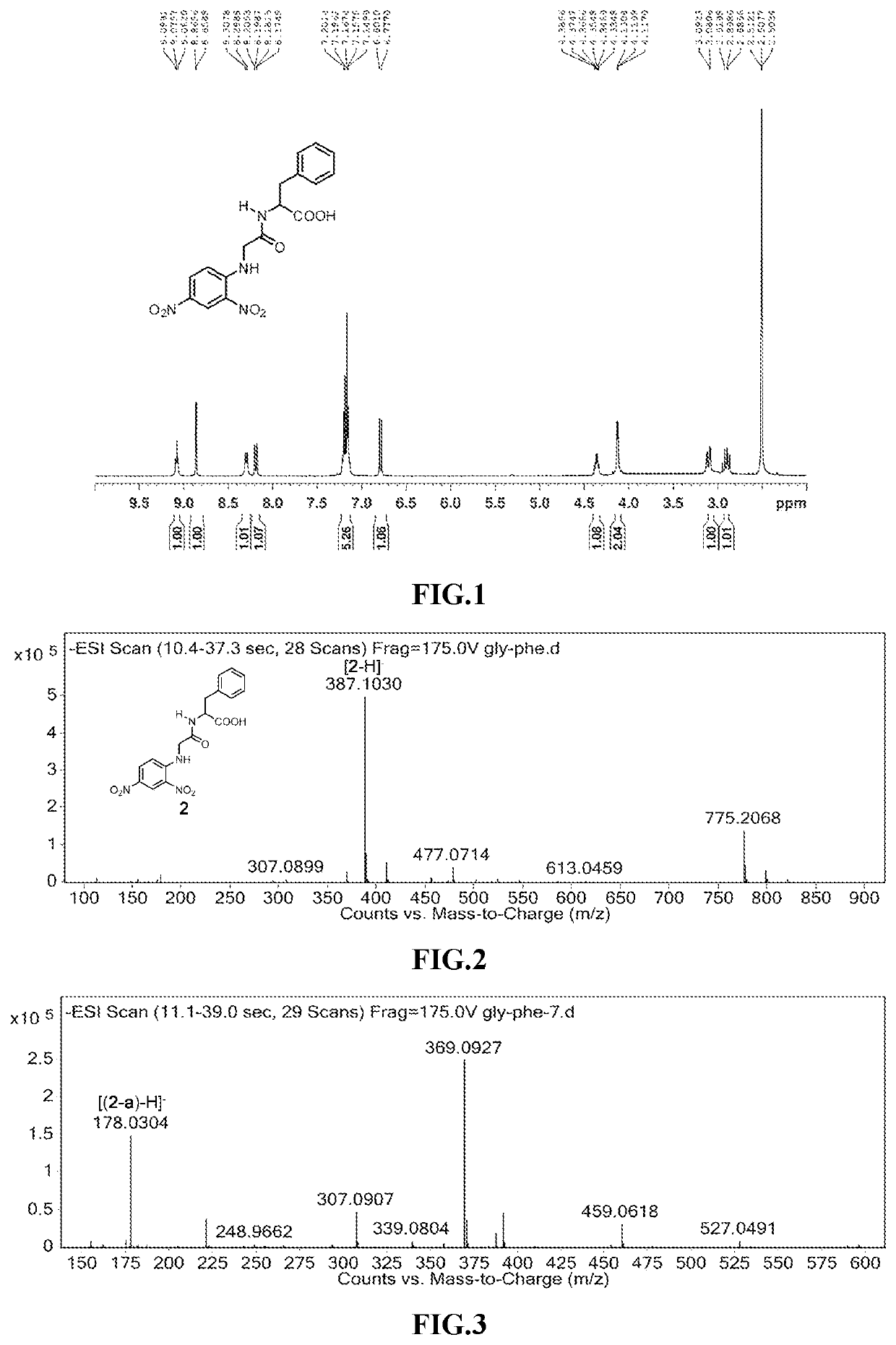
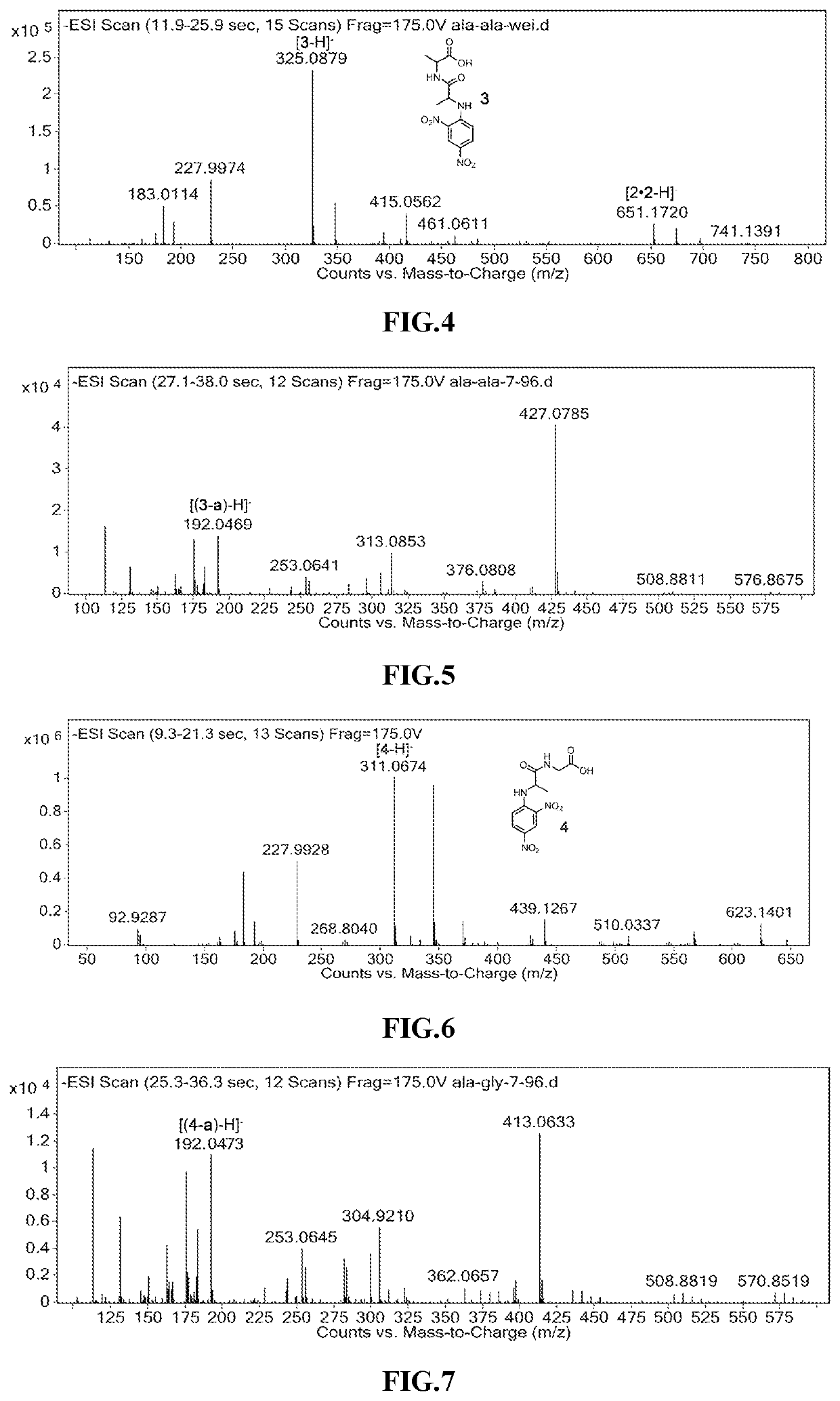
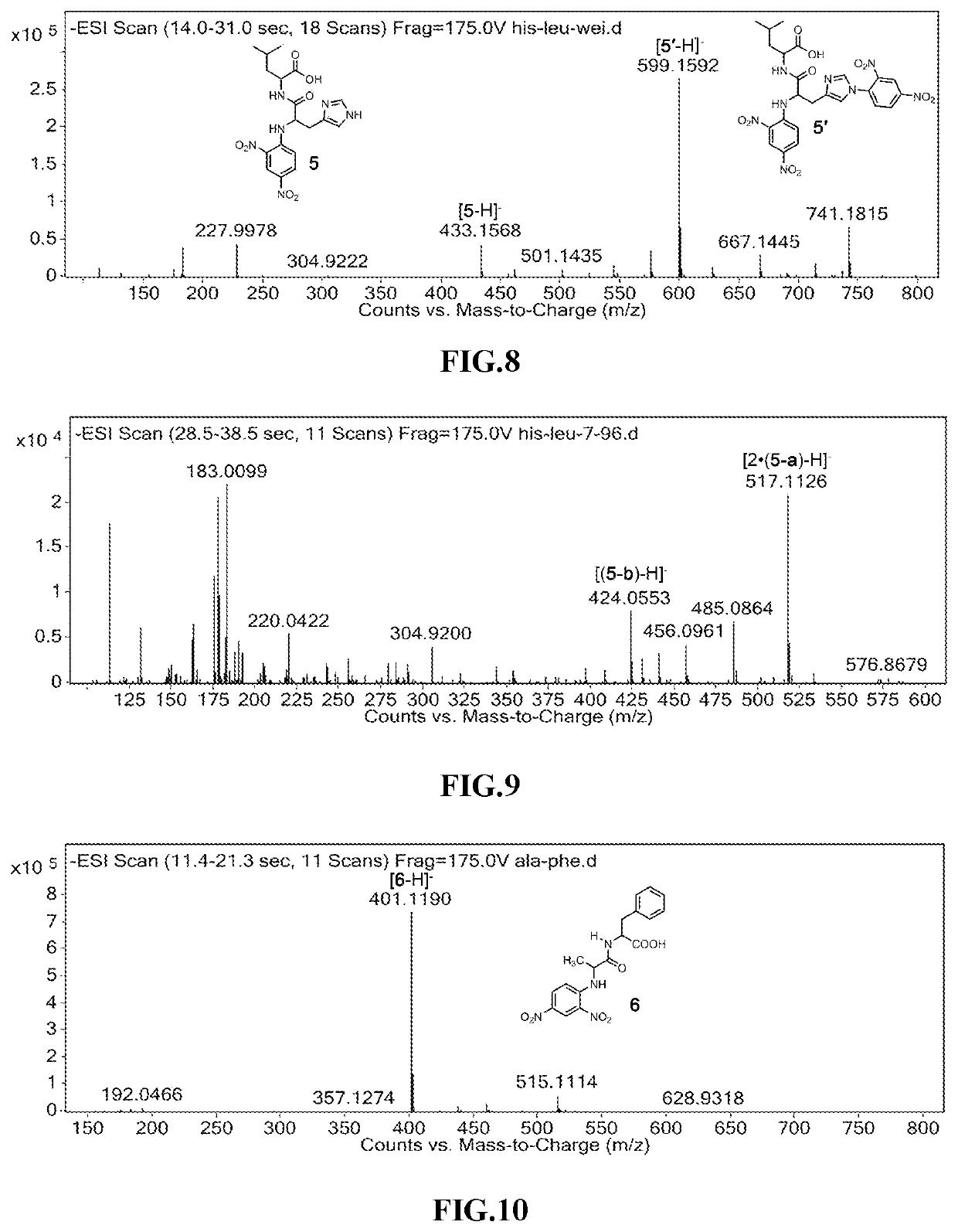
Amide Decomposition Patterns: Study Under Light Conditions
FEB 28, 20268 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Amide Photodegradation Background and Research Objectives
Amide compounds represent one of the most fundamental and ubiquitous chemical structures in both natural and synthetic systems, serving as the backbone of proteins, pharmaceuticals, polymers, and numerous industrial materials. The photochemical behavior of amides has garnered increasing attention due to their widespread environmental presence and potential degradation pathways under solar radiation. Understanding amide photodegradation mechanisms is crucial for predicting the fate of amide-containing compounds in natural environments, optimizing pharmaceutical stability, and developing sustainable chemical processes.
The historical development of amide photochemistry research can be traced back to early studies in the 1960s, when researchers first observed that certain amide bonds could undergo cleavage under ultraviolet irradiation. Initial investigations focused primarily on simple aliphatic and aromatic amides, revealing that photodegradation patterns were highly dependent on molecular structure, substitution patterns, and environmental conditions. Over subsequent decades, the field evolved to encompass more complex systems, including peptides, polyamides, and pharmaceutical compounds containing amide functionalities.
Recent advances in analytical techniques and computational chemistry have enabled more sophisticated investigations into amide photodegradation mechanisms. Modern research has revealed that amide photolysis can proceed through multiple pathways, including direct photolysis, sensitized reactions, and radical-mediated processes. The complexity of these mechanisms varies significantly based on factors such as wavelength, intensity, pH, temperature, and the presence of photosensitizers or quenchers.
The primary research objectives in studying amide decomposition patterns under light conditions encompass several critical areas. First, elucidating the fundamental photochemical mechanisms governing amide bond cleavage, including the identification of reactive intermediates and transition states. Second, establishing structure-activity relationships that can predict photodegradation susceptibility based on molecular characteristics. Third, quantifying the influence of environmental parameters on degradation kinetics and product formation.
Additionally, this research aims to develop predictive models for amide photostability assessment, which has direct applications in pharmaceutical development, environmental risk assessment, and materials science. Understanding these decomposition patterns is essential for designing more photostable amide-containing compounds and developing effective strategies for controlling unwanted photodegradation in various applications.
The historical development of amide photochemistry research can be traced back to early studies in the 1960s, when researchers first observed that certain amide bonds could undergo cleavage under ultraviolet irradiation. Initial investigations focused primarily on simple aliphatic and aromatic amides, revealing that photodegradation patterns were highly dependent on molecular structure, substitution patterns, and environmental conditions. Over subsequent decades, the field evolved to encompass more complex systems, including peptides, polyamides, and pharmaceutical compounds containing amide functionalities.
Recent advances in analytical techniques and computational chemistry have enabled more sophisticated investigations into amide photodegradation mechanisms. Modern research has revealed that amide photolysis can proceed through multiple pathways, including direct photolysis, sensitized reactions, and radical-mediated processes. The complexity of these mechanisms varies significantly based on factors such as wavelength, intensity, pH, temperature, and the presence of photosensitizers or quenchers.
The primary research objectives in studying amide decomposition patterns under light conditions encompass several critical areas. First, elucidating the fundamental photochemical mechanisms governing amide bond cleavage, including the identification of reactive intermediates and transition states. Second, establishing structure-activity relationships that can predict photodegradation susceptibility based on molecular characteristics. Third, quantifying the influence of environmental parameters on degradation kinetics and product formation.
Additionally, this research aims to develop predictive models for amide photostability assessment, which has direct applications in pharmaceutical development, environmental risk assessment, and materials science. Understanding these decomposition patterns is essential for designing more photostable amide-containing compounds and developing effective strategies for controlling unwanted photodegradation in various applications.
Market Demand for Photostable Amide Applications
The pharmaceutical industry represents the largest market segment for photostable amide applications, driven by the critical need to maintain drug efficacy throughout product lifecycles. Light-induced degradation of amide-containing pharmaceuticals poses significant challenges for drug manufacturers, particularly in the development of oral solid dosage forms, injectable formulations, and topical preparations. The global pharmaceutical market's emphasis on extended shelf life and stability under various storage conditions has created substantial demand for photostable amide compounds and protective formulation strategies.
Agricultural chemicals constitute another major market driver, where amide-based pesticides, herbicides, and fungicides require enhanced photostability to maintain field effectiveness. The increasing adoption of precision agriculture and integrated pest management systems has heightened the need for photostable formulations that can withstand prolonged sunlight exposure while maintaining biological activity. This demand is particularly pronounced in regions with intense solar radiation and extended growing seasons.
The cosmetics and personal care industry demonstrates growing market interest in photostable amide applications, especially in sunscreen formulations, anti-aging products, and color cosmetics. Consumer awareness regarding product stability and performance under light exposure has driven manufacturers to seek advanced amide compounds that resist photodegradation while maintaining desired functional properties.
Industrial applications present emerging market opportunities, particularly in polymer additives, coatings, and specialty chemicals where amide compounds serve as stabilizers, plasticizers, or functional modifiers. The automotive, aerospace, and construction industries increasingly demand materials with superior photostability to ensure long-term performance under outdoor conditions.
Market growth is further stimulated by regulatory requirements mandating comprehensive photostability testing for pharmaceutical products and increasing environmental concerns about chemical degradation products. The development of novel photostable amide derivatives and protective packaging solutions represents a rapidly expanding market segment, with particular emphasis on sustainable and environmentally friendly approaches to photostabilization.
Agricultural chemicals constitute another major market driver, where amide-based pesticides, herbicides, and fungicides require enhanced photostability to maintain field effectiveness. The increasing adoption of precision agriculture and integrated pest management systems has heightened the need for photostable formulations that can withstand prolonged sunlight exposure while maintaining biological activity. This demand is particularly pronounced in regions with intense solar radiation and extended growing seasons.
The cosmetics and personal care industry demonstrates growing market interest in photostable amide applications, especially in sunscreen formulations, anti-aging products, and color cosmetics. Consumer awareness regarding product stability and performance under light exposure has driven manufacturers to seek advanced amide compounds that resist photodegradation while maintaining desired functional properties.
Industrial applications present emerging market opportunities, particularly in polymer additives, coatings, and specialty chemicals where amide compounds serve as stabilizers, plasticizers, or functional modifiers. The automotive, aerospace, and construction industries increasingly demand materials with superior photostability to ensure long-term performance under outdoor conditions.
Market growth is further stimulated by regulatory requirements mandating comprehensive photostability testing for pharmaceutical products and increasing environmental concerns about chemical degradation products. The development of novel photostable amide derivatives and protective packaging solutions represents a rapidly expanding market segment, with particular emphasis on sustainable and environmentally friendly approaches to photostabilization.
Current Challenges in Amide Photochemical Stability
Amide photochemical stability faces significant challenges that limit the practical application of amide-containing compounds in light-exposed environments. The primary constraint stems from the inherent susceptibility of amide bonds to photodegradation, particularly under UV radiation exposure. This vulnerability manifests through multiple degradation pathways, including direct photolysis and photosensitized oxidation reactions, leading to unpredictable decomposition patterns that compromise material integrity and performance.
The complexity of amide photodegradation mechanisms presents a fundamental challenge for researchers and industry practitioners. Unlike thermal degradation, which follows relatively predictable kinetic models, photochemical decomposition involves multiple competing reaction pathways that are highly dependent on environmental conditions such as wavelength, intensity, oxygen concentration, and the presence of photosensitizers. This multifactorial nature makes it extremely difficult to establish universal stability prediction models.
Current analytical methodologies face limitations in real-time monitoring of photodegradation processes. Traditional spectroscopic techniques often lack the sensitivity required to detect early-stage decomposition products, while accelerated aging tests may not accurately reflect real-world exposure conditions. The temporal resolution of existing monitoring systems is insufficient to capture rapid photochemical events, hindering comprehensive understanding of degradation kinetics.
Material design constraints represent another significant challenge in achieving enhanced photostability. Conventional stabilization approaches, such as UV absorbers and antioxidants, often provide limited protection and may introduce compatibility issues with the base amide matrix. The development of intrinsically photostable amide structures requires balancing molecular stability with functional properties, creating design trade-offs that are difficult to optimize.
Industrial implementation faces practical barriers related to cost-effectiveness and scalability. Advanced photostabilization technologies often involve expensive additives or complex processing conditions that may not be economically viable for large-scale production. Additionally, regulatory constraints on certain stabilizing agents limit the available options for enhancing amide photostability in consumer applications.
The lack of standardized testing protocols across different industries creates inconsistencies in photostability assessment. Varying exposure conditions, evaluation criteria, and performance metrics make it challenging to compare results across different research groups and establish industry-wide benchmarks for acceptable photostability levels.
The complexity of amide photodegradation mechanisms presents a fundamental challenge for researchers and industry practitioners. Unlike thermal degradation, which follows relatively predictable kinetic models, photochemical decomposition involves multiple competing reaction pathways that are highly dependent on environmental conditions such as wavelength, intensity, oxygen concentration, and the presence of photosensitizers. This multifactorial nature makes it extremely difficult to establish universal stability prediction models.
Current analytical methodologies face limitations in real-time monitoring of photodegradation processes. Traditional spectroscopic techniques often lack the sensitivity required to detect early-stage decomposition products, while accelerated aging tests may not accurately reflect real-world exposure conditions. The temporal resolution of existing monitoring systems is insufficient to capture rapid photochemical events, hindering comprehensive understanding of degradation kinetics.
Material design constraints represent another significant challenge in achieving enhanced photostability. Conventional stabilization approaches, such as UV absorbers and antioxidants, often provide limited protection and may introduce compatibility issues with the base amide matrix. The development of intrinsically photostable amide structures requires balancing molecular stability with functional properties, creating design trade-offs that are difficult to optimize.
Industrial implementation faces practical barriers related to cost-effectiveness and scalability. Advanced photostabilization technologies often involve expensive additives or complex processing conditions that may not be economically viable for large-scale production. Additionally, regulatory constraints on certain stabilizing agents limit the available options for enhancing amide photostability in consumer applications.
The lack of standardized testing protocols across different industries creates inconsistencies in photostability assessment. Varying exposure conditions, evaluation criteria, and performance metrics make it challenging to compare results across different research groups and establish industry-wide benchmarks for acceptable photostability levels.
Existing Photodegradation Mechanisms and Solutions
01 Thermal decomposition methods for amide compounds
Amide compounds can undergo thermal decomposition under controlled temperature conditions, typically involving heating to specific temperature ranges to break the amide bond. This process can be used for various applications including waste treatment, chemical synthesis, and material processing. The decomposition patterns are influenced by factors such as temperature, pressure, and the presence of catalysts or other chemical agents.- Thermal decomposition methods for amide compounds: Amide compounds can undergo thermal decomposition under controlled temperature conditions, typically involving heating to specific temperatures to break the amide bond. This process can be used for various applications including waste treatment, chemical synthesis, and material processing. The decomposition patterns are influenced by factors such as temperature, pressure, and the presence of catalysts or other chemical agents.
- Catalytic decomposition of amide structures: Catalytic processes can facilitate the decomposition of amide bonds through the use of metal catalysts, enzymes, or other catalytic agents. These methods often operate under milder conditions compared to thermal decomposition and can provide more selective breakdown patterns. The catalytic approach allows for better control over the decomposition products and reaction pathways.
- Hydrolytic decomposition mechanisms: Amides can be decomposed through hydrolysis reactions, where water molecules break the amide bond to form carboxylic acids and amines. This process can occur under acidic or basic conditions and may be accelerated by temperature or enzymatic catalysis. The hydrolytic decomposition pattern is particularly relevant in biological systems and environmental degradation processes.
- Oxidative decomposition pathways: Oxidative decomposition of amides involves the use of oxidizing agents or conditions to break down the amide structure. This can result in various oxidation products depending on the specific conditions and oxidants used. The process is relevant in environmental remediation, chemical synthesis, and analytical applications where complete mineralization or specific product formation is desired.
- Photochemical and radiation-induced decomposition: Amide compounds can undergo decomposition when exposed to ultraviolet light, gamma radiation, or other forms of electromagnetic radiation. This photochemical or radiation-induced decomposition follows specific patterns that depend on the wavelength, intensity, and duration of exposure. Such processes are utilized in sterilization, polymer degradation studies, and advanced oxidation processes.
02 Catalytic decomposition of amide structures
Catalytic processes can facilitate the decomposition of amide bonds through the use of metal catalysts, acid catalysts, or base catalysts. These methods allow for more controlled decomposition at lower temperatures compared to purely thermal methods. The catalytic approach can selectively break amide bonds while preserving other functional groups in complex molecules, making it valuable for pharmaceutical and chemical manufacturing applications.Expand Specific Solutions03 Hydrolytic decomposition pathways
Amides can be decomposed through hydrolysis reactions in the presence of water, acids, or bases. This decomposition pathway involves the cleavage of the amide bond to form carboxylic acids and amines. The rate and mechanism of hydrolytic decomposition depend on pH conditions, temperature, and the structural characteristics of the amide compound. This method is commonly employed in environmental remediation and biochemical processes.Expand Specific Solutions04 Oxidative decomposition mechanisms
Oxidative decomposition of amides involves the use of oxidizing agents or oxidative conditions to break down the amide structure. This process can lead to the formation of various oxidation products including carboxylic acids, aldehydes, and nitrogen-containing compounds. The oxidative pathway is particularly relevant in environmental chemistry, waste treatment, and the degradation of amide-containing pollutants.Expand Specific Solutions05 Photochemical and radiation-induced decomposition
Amide compounds can undergo decomposition when exposed to ultraviolet light, radiation, or other forms of electromagnetic energy. This photochemical decomposition follows specific patterns depending on the wavelength of light, intensity of radiation, and molecular structure of the amide. The process can result in homolytic or heterolytic cleavage of the amide bond and is important in photochemistry, material degradation studies, and sterilization applications.Expand Specific Solutions
Key Players in Photochemistry and Amide Research
The study of amide decomposition patterns under light conditions represents an emerging research area in the early development stage, with significant potential across pharmaceutical, materials science, and photochemical applications. The market remains nascent but shows promise given the involvement of major industrial players alongside academic institutions. Technology maturity varies considerably across participants, with established corporations like FUJIFILM Corp., Canon Inc., and Toyota Motor Corp. bringing advanced photochemical and materials expertise, while semiconductor leaders such as Taiwan Semiconductor Manufacturing Co. and SK hynix contribute precision manufacturing capabilities. Academic institutions including Zhejiang University of Technology, Soochow University, and Hokkaido University drive fundamental research innovation. Chemical specialists like Shin-Etsu Chemical and Eastman Chemical provide materials science foundations, while technology giants IBM and 3M Innovative Properties offer computational and analytical solutions, creating a diverse ecosystem spanning from basic research to potential commercial applications.
FUJIFILM Corp.
Technical Solution: FUJIFILM has developed advanced photochemical analysis systems for studying molecular decomposition under controlled light exposure. Their technology utilizes proprietary spectroscopic methods combined with high-resolution imaging to monitor amide bond cleavage in real-time. The company's approach integrates UV-Vis spectrophotometry with mass spectrometry detection, enabling precise tracking of decomposition intermediates and end products. Their systems feature automated light intensity control and wavelength selection capabilities, allowing researchers to study photodegradation patterns across different spectral ranges. The technology includes specialized sample chambers with inert atmosphere control to isolate light-induced effects from oxidative degradation.
Strengths: Established expertise in photographic chemistry and light-sensitive materials, comprehensive analytical instrumentation portfolio. Weaknesses: Limited focus on pharmaceutical applications, higher equipment costs compared to academic solutions.
Canon, Inc.
Technical Solution: Canon has developed optical measurement systems specifically designed for photodegradation studies of organic compounds including amides. Their technology employs high-precision LED arrays with narrow bandwidth control for selective wavelength exposure studies. The system integrates advanced CCD sensors for real-time monitoring of sample changes during light exposure. Canon's approach includes proprietary algorithms for analyzing spectral shifts and molecular structural changes. Their equipment features temperature-controlled sample stages and automated data acquisition systems that can track decomposition kinetics over extended periods. The technology supports both continuous and pulsed light exposure protocols for comprehensive photostability testing.
Strengths: Superior optical precision and imaging technology, robust automated systems for long-term studies. Weaknesses: Primarily hardware-focused with limited chemical analysis software, expensive initial investment.
Core Patents in Amide Photostability Enhancement
Method for photocleavage of amide bonds
PatentActiveUS20220289665A1
Innovation
- A method involving the reaction of 2,4-dinitrofluorobenzene with an α-amino acid to form a compound that undergoes photocleavage under light irradiation, using mild conditions like weak acids or bases, and a range of organic solvents, allowing for the cleavage of amide bonds.
Method and device for decomposing amine-based compound
PatentInactiveJP2006239478A
Innovation
- Applying ultraviolet rays with energy equal to or higher than the dissociation energy of N-H bonds to a solution containing an amine-based compound while in contact with a photocatalyst to facilitate rapid decomposition.
Environmental Impact of Amide Photodegradation
The environmental implications of amide photodegradation represent a critical intersection between chemical stability and ecological sustainability. When amide compounds undergo photolytic decomposition under natural or artificial light conditions, they generate various byproducts that can significantly impact environmental systems. These degradation products often include smaller molecular fragments, free radicals, and potentially toxic intermediates that may persist in environmental matrices longer than the parent compounds.
Aquatic ecosystems face particular vulnerability to amide photodegradation products. The breakdown of pharmaceutical amides, agricultural chemicals, and industrial compounds in water bodies can lead to the formation of metabolites with altered bioavailability and toxicity profiles. These transformation products may exhibit enhanced mobility through soil and groundwater systems, potentially reaching drinking water sources and affecting aquatic organisms at concentrations previously considered safe for the parent compounds.
Atmospheric photodegradation of volatile amide compounds contributes to secondary organic aerosol formation and can influence regional air quality. The photochemical reactions involving amides in the troposphere generate nitrogen-containing organic compounds that participate in complex atmospheric chemistry cycles. These processes can affect the formation of ground-level ozone and contribute to the deposition of reactive nitrogen species in terrestrial ecosystems.
Soil environments experience long-term consequences from amide photodegradation, particularly in agricultural settings where amide-based pesticides and fertilizers are commonly applied. The photolytic breakdown products can alter soil microbial communities, affect nutrient cycling processes, and potentially accumulate in food chains. The persistence and mobility of these degradation products vary significantly depending on soil composition, pH levels, and organic matter content.
The bioaccumulation potential of amide photodegradation products presents ongoing environmental concerns. While parent amide compounds may have well-characterized environmental fate profiles, their photolytic products often lack comprehensive toxicological assessment. This knowledge gap creates uncertainty in environmental risk assessment and regulatory frameworks, highlighting the need for systematic evaluation of photodegradation pathways and their ecological consequences across different environmental compartments.
Aquatic ecosystems face particular vulnerability to amide photodegradation products. The breakdown of pharmaceutical amides, agricultural chemicals, and industrial compounds in water bodies can lead to the formation of metabolites with altered bioavailability and toxicity profiles. These transformation products may exhibit enhanced mobility through soil and groundwater systems, potentially reaching drinking water sources and affecting aquatic organisms at concentrations previously considered safe for the parent compounds.
Atmospheric photodegradation of volatile amide compounds contributes to secondary organic aerosol formation and can influence regional air quality. The photochemical reactions involving amides in the troposphere generate nitrogen-containing organic compounds that participate in complex atmospheric chemistry cycles. These processes can affect the formation of ground-level ozone and contribute to the deposition of reactive nitrogen species in terrestrial ecosystems.
Soil environments experience long-term consequences from amide photodegradation, particularly in agricultural settings where amide-based pesticides and fertilizers are commonly applied. The photolytic breakdown products can alter soil microbial communities, affect nutrient cycling processes, and potentially accumulate in food chains. The persistence and mobility of these degradation products vary significantly depending on soil composition, pH levels, and organic matter content.
The bioaccumulation potential of amide photodegradation products presents ongoing environmental concerns. While parent amide compounds may have well-characterized environmental fate profiles, their photolytic products often lack comprehensive toxicological assessment. This knowledge gap creates uncertainty in environmental risk assessment and regulatory frameworks, highlighting the need for systematic evaluation of photodegradation pathways and their ecological consequences across different environmental compartments.
Safety Regulations for Photosensitive Amide Compounds
The handling of photosensitive amide compounds requires comprehensive safety protocols due to their inherent instability under light exposure and potential for hazardous decomposition reactions. These compounds present unique challenges in laboratory and industrial settings, necessitating specialized regulatory frameworks that address both photochemical reactivity and decomposition byproduct toxicity.
Primary safety regulations mandate strict light control measures for all photosensitive amide compounds. Storage facilities must maintain complete darkness or utilize amber-filtered lighting systems with wavelengths above 550 nanometers to minimize photochemical activation. Temperature-controlled environments below 25°C are required to reduce thermal acceleration of light-induced decomposition processes. All containers must be constructed from UV-blocking materials, typically amber glass or specialized polymer compositions that filter harmful radiation spectra.
Personnel handling protocols establish mandatory protective equipment requirements including UV-protective eyewear, chemical-resistant gloves, and full-coverage laboratory coats. Respiratory protection becomes critical when working with volatile amide compounds, as photodecomposition can generate toxic nitrogen-containing vapors and reactive intermediates. Training programs must emphasize recognition of photodegradation indicators such as color changes, gas evolution, or unusual odors that signal active decomposition processes.
Waste management regulations for photosensitive amides require immediate neutralization and disposal procedures to prevent accumulation of unstable materials. Decomposed amide residues often contain carcinogenic aromatic amines or reactive carbonyl compounds that demand specialized treatment protocols. Emergency response procedures must account for rapid decomposition scenarios, including fire suppression systems compatible with nitrogen-containing organic compounds and evacuation protocols for toxic vapor release incidents.
Transportation regulations classify photosensitive amides under specialized hazardous material categories, requiring light-proof packaging and temperature monitoring throughout the supply chain. Documentation must include photostability data, decomposition kinetics, and emergency contact information for specialized hazmat response teams. Regular safety audits ensure compliance with evolving regulatory standards as new decomposition pathways and hazards are identified through ongoing research.
Primary safety regulations mandate strict light control measures for all photosensitive amide compounds. Storage facilities must maintain complete darkness or utilize amber-filtered lighting systems with wavelengths above 550 nanometers to minimize photochemical activation. Temperature-controlled environments below 25°C are required to reduce thermal acceleration of light-induced decomposition processes. All containers must be constructed from UV-blocking materials, typically amber glass or specialized polymer compositions that filter harmful radiation spectra.
Personnel handling protocols establish mandatory protective equipment requirements including UV-protective eyewear, chemical-resistant gloves, and full-coverage laboratory coats. Respiratory protection becomes critical when working with volatile amide compounds, as photodecomposition can generate toxic nitrogen-containing vapors and reactive intermediates. Training programs must emphasize recognition of photodegradation indicators such as color changes, gas evolution, or unusual odors that signal active decomposition processes.
Waste management regulations for photosensitive amides require immediate neutralization and disposal procedures to prevent accumulation of unstable materials. Decomposed amide residues often contain carcinogenic aromatic amines or reactive carbonyl compounds that demand specialized treatment protocols. Emergency response procedures must account for rapid decomposition scenarios, including fire suppression systems compatible with nitrogen-containing organic compounds and evacuation protocols for toxic vapor release incidents.
Transportation regulations classify photosensitive amides under specialized hazardous material categories, requiring light-proof packaging and temperature monitoring throughout the supply chain. Documentation must include photostability data, decomposition kinetics, and emergency contact information for specialized hazmat response teams. Regular safety audits ensure compliance with evolving regulatory standards as new decomposition pathways and hazards are identified through ongoing research.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!