UHMWPE's Impact on Cellular Frameworks for Tissue Engineering
AUG 6, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
UHMWPE in Tissue Engineering: Background and Objectives
Ultra-high-molecular-weight polyethylene (UHMWPE) has emerged as a promising material in the field of tissue engineering, particularly for its potential in cellular frameworks. The evolution of UHMWPE in biomedical applications can be traced back to its initial use in orthopedic implants, where its exceptional wear resistance and biocompatibility made it an ideal candidate for joint replacements. Over time, researchers began to explore its potential beyond traditional implant applications, recognizing its unique properties that could be harnessed for tissue engineering purposes.
The development of UHMWPE in tissue engineering is driven by the increasing demand for advanced biomaterials that can support cell growth, differentiation, and tissue regeneration. As the field of regenerative medicine continues to expand, there is a growing need for materials that can mimic the complex structures and mechanical properties of natural tissues. UHMWPE, with its high strength-to-weight ratio and ability to be processed into various forms, presents an attractive option for creating scaffolds and cellular frameworks.
The primary objective of incorporating UHMWPE into tissue engineering is to leverage its mechanical properties and biocompatibility to create robust, long-lasting cellular frameworks. These frameworks aim to provide a suitable environment for cell attachment, proliferation, and differentiation, ultimately leading to the formation of functional tissue constructs. Researchers are particularly interested in exploring how UHMWPE can be modified or combined with other materials to enhance its performance in tissue engineering applications.
One of the key technical goals in this field is to develop methods for surface modification of UHMWPE to improve its cell adhesion properties while maintaining its mechanical integrity. This involves investigating various techniques such as plasma treatment, chemical functionalization, and the incorporation of bioactive molecules. Another important objective is to optimize the porosity and interconnectivity of UHMWPE-based scaffolds to facilitate nutrient transport and cell migration throughout the structure.
The integration of UHMWPE with other biomaterials to create composite scaffolds is another area of focus. By combining UHMWPE with materials such as hydroxyapatite or bioactive glasses, researchers aim to create hybrid structures that can provide both mechanical support and bioactive cues for tissue regeneration. This approach is particularly relevant for applications in bone and cartilage tissue engineering, where the mechanical properties of UHMWPE can be complemented by the osteoinductive properties of other materials.
As research in this field progresses, there is a growing emphasis on understanding the long-term behavior of UHMWPE in biological environments. This includes studying its degradation characteristics, potential for wear particle generation, and interactions with surrounding tissues over extended periods. Such investigations are crucial for ensuring the safety and efficacy of UHMWPE-based tissue engineering constructs in clinical applications.
The development of UHMWPE in tissue engineering is driven by the increasing demand for advanced biomaterials that can support cell growth, differentiation, and tissue regeneration. As the field of regenerative medicine continues to expand, there is a growing need for materials that can mimic the complex structures and mechanical properties of natural tissues. UHMWPE, with its high strength-to-weight ratio and ability to be processed into various forms, presents an attractive option for creating scaffolds and cellular frameworks.
The primary objective of incorporating UHMWPE into tissue engineering is to leverage its mechanical properties and biocompatibility to create robust, long-lasting cellular frameworks. These frameworks aim to provide a suitable environment for cell attachment, proliferation, and differentiation, ultimately leading to the formation of functional tissue constructs. Researchers are particularly interested in exploring how UHMWPE can be modified or combined with other materials to enhance its performance in tissue engineering applications.
One of the key technical goals in this field is to develop methods for surface modification of UHMWPE to improve its cell adhesion properties while maintaining its mechanical integrity. This involves investigating various techniques such as plasma treatment, chemical functionalization, and the incorporation of bioactive molecules. Another important objective is to optimize the porosity and interconnectivity of UHMWPE-based scaffolds to facilitate nutrient transport and cell migration throughout the structure.
The integration of UHMWPE with other biomaterials to create composite scaffolds is another area of focus. By combining UHMWPE with materials such as hydroxyapatite or bioactive glasses, researchers aim to create hybrid structures that can provide both mechanical support and bioactive cues for tissue regeneration. This approach is particularly relevant for applications in bone and cartilage tissue engineering, where the mechanical properties of UHMWPE can be complemented by the osteoinductive properties of other materials.
As research in this field progresses, there is a growing emphasis on understanding the long-term behavior of UHMWPE in biological environments. This includes studying its degradation characteristics, potential for wear particle generation, and interactions with surrounding tissues over extended periods. Such investigations are crucial for ensuring the safety and efficacy of UHMWPE-based tissue engineering constructs in clinical applications.
Market Analysis for UHMWPE-based Cellular Scaffolds
The market for UHMWPE-based cellular scaffolds in tissue engineering is experiencing significant growth, driven by the increasing demand for advanced biomaterials in regenerative medicine. This market segment is part of the broader tissue engineering market, which is projected to reach substantial value in the coming years due to the rising prevalence of chronic diseases and the aging population.
UHMWPE-based scaffolds are gaining traction in various applications within tissue engineering, particularly in orthopedic and dental fields. The unique properties of UHMWPE, including its high strength-to-weight ratio, excellent wear resistance, and biocompatibility, make it an attractive material for cellular scaffolds. These characteristics address key market needs for durable and long-lasting implants that can support tissue regeneration.
The market demand for UHMWPE-based cellular scaffolds is primarily driven by the growing number of orthopedic surgeries, dental procedures, and the increasing adoption of regenerative medicine approaches. As healthcare systems worldwide focus on improving patient outcomes and reducing long-term healthcare costs, the demand for advanced biomaterials like UHMWPE scaffolds is expected to rise.
Geographically, North America and Europe currently dominate the market for UHMWPE-based cellular scaffolds, owing to their advanced healthcare infrastructure and higher adoption rates of innovative medical technologies. However, the Asia-Pacific region is emerging as a significant market, with rapid growth expected due to improving healthcare access and increasing investments in medical research and development.
The competitive landscape of the UHMWPE-based cellular scaffold market is characterized by a mix of established medical device companies and innovative startups. Key players are focusing on research and development to enhance the performance of UHMWPE scaffolds, particularly in terms of cell adhesion, proliferation, and differentiation. Collaborations between academic institutions and industry partners are also driving market growth through the development of novel scaffold designs and manufacturing techniques.
Despite the promising outlook, the market faces challenges such as stringent regulatory requirements and the high cost of development and production. These factors may impact market growth and adoption rates, particularly in emerging economies. However, ongoing advancements in manufacturing technologies, such as 3D printing and electrospinning, are expected to reduce production costs and improve scaffold customization, potentially expanding market opportunities.
In conclusion, the market for UHMWPE-based cellular scaffolds in tissue engineering shows strong growth potential, driven by technological advancements, increasing healthcare needs, and the material's unique properties. As research continues to demonstrate the efficacy of UHMWPE scaffolds in various tissue engineering applications, the market is poised for further expansion and innovation in the coming years.
UHMWPE-based scaffolds are gaining traction in various applications within tissue engineering, particularly in orthopedic and dental fields. The unique properties of UHMWPE, including its high strength-to-weight ratio, excellent wear resistance, and biocompatibility, make it an attractive material for cellular scaffolds. These characteristics address key market needs for durable and long-lasting implants that can support tissue regeneration.
The market demand for UHMWPE-based cellular scaffolds is primarily driven by the growing number of orthopedic surgeries, dental procedures, and the increasing adoption of regenerative medicine approaches. As healthcare systems worldwide focus on improving patient outcomes and reducing long-term healthcare costs, the demand for advanced biomaterials like UHMWPE scaffolds is expected to rise.
Geographically, North America and Europe currently dominate the market for UHMWPE-based cellular scaffolds, owing to their advanced healthcare infrastructure and higher adoption rates of innovative medical technologies. However, the Asia-Pacific region is emerging as a significant market, with rapid growth expected due to improving healthcare access and increasing investments in medical research and development.
The competitive landscape of the UHMWPE-based cellular scaffold market is characterized by a mix of established medical device companies and innovative startups. Key players are focusing on research and development to enhance the performance of UHMWPE scaffolds, particularly in terms of cell adhesion, proliferation, and differentiation. Collaborations between academic institutions and industry partners are also driving market growth through the development of novel scaffold designs and manufacturing techniques.
Despite the promising outlook, the market faces challenges such as stringent regulatory requirements and the high cost of development and production. These factors may impact market growth and adoption rates, particularly in emerging economies. However, ongoing advancements in manufacturing technologies, such as 3D printing and electrospinning, are expected to reduce production costs and improve scaffold customization, potentially expanding market opportunities.
In conclusion, the market for UHMWPE-based cellular scaffolds in tissue engineering shows strong growth potential, driven by technological advancements, increasing healthcare needs, and the material's unique properties. As research continues to demonstrate the efficacy of UHMWPE scaffolds in various tissue engineering applications, the market is poised for further expansion and innovation in the coming years.
Current Challenges in UHMWPE Cellular Frameworks
The integration of Ultra-High Molecular Weight Polyethylene (UHMWPE) into cellular frameworks for tissue engineering presents several significant challenges that researchers and engineers must address. One of the primary obstacles is the inherent hydrophobicity of UHMWPE, which can hinder cell adhesion and proliferation. This characteristic makes it difficult to create a suitable microenvironment for cell growth and tissue formation, potentially limiting the effectiveness of UHMWPE-based scaffolds in tissue engineering applications.
Another challenge lies in the mechanical properties of UHMWPE when used in cellular frameworks. While UHMWPE is known for its excellent wear resistance and durability, achieving the right balance between strength and flexibility in cellular structures remains complex. The material's high stiffness can sometimes lead to a mismatch with the mechanical properties of natural tissues, potentially causing stress shielding effects or inadequate load transfer to developing tissues.
Surface modification of UHMWPE presents yet another hurdle in its application to cellular frameworks. Enhancing the biocompatibility and cell-material interactions often requires altering the surface properties of UHMWPE. However, the material's chemical inertness makes it resistant to many conventional surface modification techniques, necessitating the development of novel approaches to improve its biological performance without compromising its bulk properties.
The processing and fabrication of UHMWPE into complex, porous structures suitable for tissue engineering also pose significant challenges. Traditional manufacturing methods may struggle to create the intricate architectures required for optimal cell growth and nutrient diffusion. Advanced fabrication techniques, such as 3D printing or electrospinning, face difficulties in processing UHMWPE due to its high molecular weight and viscosity in the melt state.
Biodegradability and integration with host tissues represent additional concerns in UHMWPE-based cellular frameworks. The material's bioinert nature and resistance to degradation can impede the gradual replacement of the scaffold with newly formed tissue, a crucial aspect of many tissue engineering strategies. This characteristic necessitates careful consideration of the long-term implications of UHMWPE implants and their interaction with surrounding tissues.
Lastly, ensuring uniform cell distribution and vascularization within UHMWPE cellular frameworks remains a significant challenge. The material's properties can affect cell migration and the formation of blood vessels, potentially leading to uneven tissue growth or inadequate nutrient supply to cells in the scaffold's interior. Overcoming these limitations requires innovative approaches to scaffold design and surface functionalization to promote cellular infiltration and angiogenesis.
Another challenge lies in the mechanical properties of UHMWPE when used in cellular frameworks. While UHMWPE is known for its excellent wear resistance and durability, achieving the right balance between strength and flexibility in cellular structures remains complex. The material's high stiffness can sometimes lead to a mismatch with the mechanical properties of natural tissues, potentially causing stress shielding effects or inadequate load transfer to developing tissues.
Surface modification of UHMWPE presents yet another hurdle in its application to cellular frameworks. Enhancing the biocompatibility and cell-material interactions often requires altering the surface properties of UHMWPE. However, the material's chemical inertness makes it resistant to many conventional surface modification techniques, necessitating the development of novel approaches to improve its biological performance without compromising its bulk properties.
The processing and fabrication of UHMWPE into complex, porous structures suitable for tissue engineering also pose significant challenges. Traditional manufacturing methods may struggle to create the intricate architectures required for optimal cell growth and nutrient diffusion. Advanced fabrication techniques, such as 3D printing or electrospinning, face difficulties in processing UHMWPE due to its high molecular weight and viscosity in the melt state.
Biodegradability and integration with host tissues represent additional concerns in UHMWPE-based cellular frameworks. The material's bioinert nature and resistance to degradation can impede the gradual replacement of the scaffold with newly formed tissue, a crucial aspect of many tissue engineering strategies. This characteristic necessitates careful consideration of the long-term implications of UHMWPE implants and their interaction with surrounding tissues.
Lastly, ensuring uniform cell distribution and vascularization within UHMWPE cellular frameworks remains a significant challenge. The material's properties can affect cell migration and the formation of blood vessels, potentially leading to uneven tissue growth or inadequate nutrient supply to cells in the scaffold's interior. Overcoming these limitations requires innovative approaches to scaffold design and surface functionalization to promote cellular infiltration and angiogenesis.
Existing UHMWPE Cellular Framework Solutions
01 Fabrication methods for UHMWPE cellular frameworks
Various techniques are employed to create UHMWPE cellular frameworks, including gel spinning, electrospinning, and 3D printing. These methods allow for the production of porous structures with controlled architecture, enhancing the material's properties for specific applications such as tissue engineering scaffolds or lightweight structural components.- Manufacturing methods for UHMWPE cellular frameworks: Various techniques are employed to create UHMWPE cellular frameworks, including compression molding, extrusion, and 3D printing. These methods allow for the production of lightweight, high-strength structures with controlled porosity and cell size. The manufacturing processes can be optimized to achieve specific mechanical properties and density requirements for different applications.
- Composite materials incorporating UHMWPE cellular frameworks: UHMWPE cellular frameworks are used as reinforcing components in composite materials. These composites combine the high strength and low weight of UHMWPE with other materials to create structures with enhanced mechanical properties. Applications include aerospace, automotive, and protective equipment industries, where the composites offer improved impact resistance and energy absorption.
- Surface modification of UHMWPE cellular frameworks: Surface treatments and modifications are applied to UHMWPE cellular frameworks to enhance their properties and compatibility with other materials. These modifications can improve adhesion in composite structures, increase wear resistance, or add functionality such as antimicrobial properties. Techniques may include plasma treatment, chemical etching, or coating applications.
- Biomedical applications of UHMWPE cellular frameworks: UHMWPE cellular frameworks find extensive use in biomedical applications due to their biocompatibility and mechanical properties. They are utilized in orthopedic implants, tissue engineering scaffolds, and drug delivery systems. The porous structure allows for tissue ingrowth and controlled release of therapeutic agents, while the material's durability ensures long-term performance in the body.
- Characterization and testing of UHMWPE cellular frameworks: Various methods are employed to characterize and test UHMWPE cellular frameworks, including mechanical testing, microscopy, and thermal analysis. These techniques help evaluate properties such as compressive strength, fatigue resistance, cell size distribution, and thermal stability. The data obtained from these tests is crucial for optimizing the frameworks for specific applications and ensuring quality control in manufacturing processes.
02 Modification of UHMWPE for improved properties
UHMWPE can be modified through various processes to enhance its mechanical, thermal, and chemical properties. These modifications may include crosslinking, blending with other polymers, or incorporating nanoparticles. Such improvements can lead to better wear resistance, increased strength, and enhanced biocompatibility for medical applications.Expand Specific Solutions03 Applications of UHMWPE cellular frameworks in medical devices
UHMWPE cellular frameworks find extensive use in medical devices, particularly in orthopedic implants and tissue engineering scaffolds. The porous structure allows for bone ingrowth and improved integration with surrounding tissues, while the material's biocompatibility and mechanical properties make it suitable for long-term implantation.Expand Specific Solutions04 UHMWPE cellular frameworks in industrial applications
The unique properties of UHMWPE cellular frameworks make them suitable for various industrial applications. These include filtration systems, impact-resistant structures, and lightweight components for aerospace and automotive industries. The material's high strength-to-weight ratio and chemical resistance contribute to its versatility in these fields.Expand Specific Solutions05 Characterization and testing of UHMWPE cellular frameworks
Various methods are employed to characterize and test UHMWPE cellular frameworks, including mechanical testing, microscopy, and spectroscopy techniques. These analyses help in understanding the material's structure-property relationships, optimizing manufacturing processes, and ensuring quality control for specific applications.Expand Specific Solutions
Key Players in UHMWPE Tissue Engineering Research
The field of UHMWPE's impact on cellular frameworks for tissue engineering is in a nascent stage of development, with significant potential for growth. The market size is expanding as research progresses, driven by the increasing demand for advanced biomaterials in regenerative medicine. Technologically, the field is still evolving, with varying levels of maturity across different applications. Key players like The General Hospital Corp., Massachusetts Institute of Technology, and DSM IP Assets BV are at the forefront of research and development, leveraging their expertise in materials science and biomedical engineering. Universities such as Zhejiang University and the University of Southern California are contributing to the knowledge base, while companies like Smith & Nephew Orthopaedics GmbH and DePuy Synthes Products, Inc. are exploring commercial applications. The competitive landscape is characterized by a mix of academic institutions, research organizations, and medical device companies, indicating a collaborative yet competitive environment for innovation in this emerging field.
Massachusetts Institute of Technology
Technical Solution: MIT has developed a novel approach to incorporate UHMWPE into cellular frameworks for tissue engineering. They have created a hybrid scaffold combining UHMWPE fibers with biodegradable polymers to enhance mechanical properties while maintaining biocompatibility[1]. The UHMWPE fibers are aligned within the scaffold to mimic natural tissue structures, providing directional strength and support for cell growth. MIT researchers have also explored surface modification techniques to improve cell adhesion to the UHMWPE components, addressing one of the material's key limitations in tissue engineering applications[2]. Additionally, they have investigated the use of UHMWPE-based nanocomposites to further enhance the scaffold's properties and cell interactions[3].
Strengths: Superior mechanical properties, customizable scaffold architecture, and potential for long-term implant stability. Weaknesses: Potential challenges in biodegradation and complete tissue integration.
DSM IP Assets BV
Technical Solution: DSM has developed a proprietary UHMWPE fiber technology called Dyneema®, which they have adapted for tissue engineering applications. Their approach involves creating highly oriented UHMWPE fiber networks that can be integrated into cellular frameworks[4]. DSM has focused on optimizing the surface properties of these fibers to enhance cell attachment and proliferation, using techniques such as plasma treatment and biofunctionalization[5]. They have also explored the use of UHMWPE in combination with other biocompatible materials to create composite scaffolds with tailored mechanical and biological properties. DSM's research has shown promising results in applications such as ligament and tendon reconstruction, where the high strength and fatigue resistance of UHMWPE are particularly beneficial[6].
Strengths: Excellent mechanical properties, potential for long-term implant durability, and versatility in scaffold design. Weaknesses: Limited biodegradability and potential challenges in achieving full tissue integration.
Innovations in UHMWPE Scaffold Design
Uhmwpe paste extruded tubes
PatentPendingUS20240066777A1
Innovation
- The development of UHMWPE tubes with an average wall thickness of less than 0.1 mm, produced by a paste extrusion method with high machine direction orientation, which are sterilizable by radiation and exhibit high tensile strength, lubricity, and abrasion resistance, using eco-friendly solvents like d-limonene.
Ultra-high molecular weight polyethylene for joint surface
PatentActiveJP2017201037A
Innovation
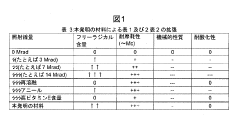
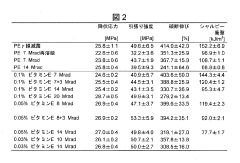
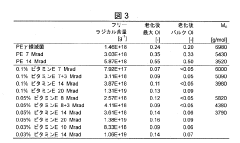
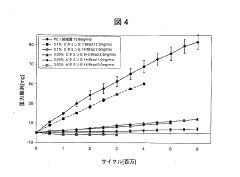
- A method involving the addition of trace amounts (0.02 to 0.12 wt%) of vitamin E to UHMWPE powder before molding, followed by gamma irradiation at doses between 5 and 20 Mrad, without subsequent heat treatment, to create a composition with improved wear resistance, oxidative stability, and mechanical properties.
Biocompatibility and Safety Considerations
The biocompatibility and safety considerations of Ultra-High Molecular Weight Polyethylene (UHMWPE) in cellular frameworks for tissue engineering are crucial aspects that require thorough examination. UHMWPE has shown promising potential in this field due to its excellent mechanical properties and wear resistance, but its interaction with biological systems must be carefully evaluated.
One of the primary concerns is the material's ability to integrate with host tissues without eliciting adverse reactions. Studies have demonstrated that UHMWPE generally exhibits good biocompatibility, with minimal inflammatory responses observed in various in vitro and in vivo models. However, the surface properties of UHMWPE can significantly influence cell adhesion and proliferation, which are essential for successful tissue engineering applications.
The potential release of wear particles from UHMWPE-based scaffolds is another critical safety consideration. While UHMWPE is known for its low wear rate, the generation of microscopic particles over time cannot be entirely eliminated. These particles may trigger local inflammatory responses or potentially migrate to other parts of the body, raising concerns about long-term safety.
To address these issues, various surface modification techniques have been explored to enhance the biocompatibility of UHMWPE. These include plasma treatment, chemical functionalization, and coating with bioactive molecules. Such modifications aim to improve cell attachment, promote tissue integration, and reduce the risk of particle generation.
The degradation behavior of UHMWPE in physiological environments is another important factor to consider. Although UHMWPE is generally considered biologically inert, long-term exposure to bodily fluids and mechanical stresses may lead to subtle changes in its structure or surface properties. These changes could potentially affect the material's performance and safety profile over time.
Sterilization methods for UHMWPE-based cellular frameworks must also be carefully selected to ensure both effective microbial elimination and preservation of the material's properties. Conventional sterilization techniques, such as gamma irradiation, may induce oxidative degradation in UHMWPE, potentially compromising its mechanical integrity and biocompatibility.
Regulatory considerations play a significant role in the development and approval of UHMWPE-based tissue engineering products. Comprehensive preclinical and clinical studies are necessary to demonstrate the long-term safety and efficacy of these materials in specific applications. This includes evaluating potential systemic effects, genotoxicity, and carcinogenicity risks associated with UHMWPE implants.
One of the primary concerns is the material's ability to integrate with host tissues without eliciting adverse reactions. Studies have demonstrated that UHMWPE generally exhibits good biocompatibility, with minimal inflammatory responses observed in various in vitro and in vivo models. However, the surface properties of UHMWPE can significantly influence cell adhesion and proliferation, which are essential for successful tissue engineering applications.
The potential release of wear particles from UHMWPE-based scaffolds is another critical safety consideration. While UHMWPE is known for its low wear rate, the generation of microscopic particles over time cannot be entirely eliminated. These particles may trigger local inflammatory responses or potentially migrate to other parts of the body, raising concerns about long-term safety.
To address these issues, various surface modification techniques have been explored to enhance the biocompatibility of UHMWPE. These include plasma treatment, chemical functionalization, and coating with bioactive molecules. Such modifications aim to improve cell attachment, promote tissue integration, and reduce the risk of particle generation.
The degradation behavior of UHMWPE in physiological environments is another important factor to consider. Although UHMWPE is generally considered biologically inert, long-term exposure to bodily fluids and mechanical stresses may lead to subtle changes in its structure or surface properties. These changes could potentially affect the material's performance and safety profile over time.
Sterilization methods for UHMWPE-based cellular frameworks must also be carefully selected to ensure both effective microbial elimination and preservation of the material's properties. Conventional sterilization techniques, such as gamma irradiation, may induce oxidative degradation in UHMWPE, potentially compromising its mechanical integrity and biocompatibility.
Regulatory considerations play a significant role in the development and approval of UHMWPE-based tissue engineering products. Comprehensive preclinical and clinical studies are necessary to demonstrate the long-term safety and efficacy of these materials in specific applications. This includes evaluating potential systemic effects, genotoxicity, and carcinogenicity risks associated with UHMWPE implants.
Regulatory Pathway for UHMWPE Tissue Engineered Products
The regulatory pathway for UHMWPE tissue engineered products is complex and multifaceted, requiring careful navigation through various regulatory bodies and compliance standards. In the United States, the Food and Drug Administration (FDA) plays a pivotal role in overseeing the development and commercialization of these innovative medical devices.
The classification of UHMWPE tissue engineered products typically falls under Class III medical devices, which are subject to the most stringent regulatory controls due to their potential risks and novel applications. This classification necessitates a premarket approval (PMA) application, a process that demands substantial clinical data to demonstrate safety and efficacy.
Key steps in the regulatory pathway include preclinical testing, which involves in vitro and in vivo studies to assess biocompatibility, mechanical properties, and potential toxicity. These studies are crucial for establishing a solid foundation for subsequent clinical trials and regulatory submissions.
Clinical trials for UHMWPE tissue engineered products generally follow a phased approach. Phase I trials focus on safety and feasibility in a small cohort of patients. Phase II trials expand to larger patient populations and assess preliminary efficacy. Phase III trials are pivotal, large-scale studies designed to provide definitive evidence of safety and effectiveness.
Throughout the development process, manufacturers must adhere to Good Manufacturing Practices (GMP) and implement robust quality management systems. This ensures consistency in product quality and facilitates compliance with regulatory requirements.
The regulatory submission process involves compiling comprehensive documentation, including detailed descriptions of the manufacturing process, quality control measures, and all preclinical and clinical data. The FDA review process is rigorous and may involve advisory committee meetings to evaluate the product's risk-benefit profile.
Post-market surveillance is a critical component of the regulatory pathway, requiring manufacturers to monitor and report adverse events, conduct post-approval studies if mandated, and maintain ongoing compliance with regulatory standards.
Internationally, regulatory pathways may vary, but many countries align with or reference FDA standards. In the European Union, for instance, UHMWPE tissue engineered products would likely be regulated under the Medical Device Regulation (MDR), requiring CE marking through a notified body assessment.
Navigating these regulatory pathways demands a multidisciplinary approach, involving expertise in regulatory affairs, clinical research, quality assurance, and engineering. Successful commercialization of UHMWPE tissue engineered products hinges on a thorough understanding and meticulous execution of these regulatory requirements.
The classification of UHMWPE tissue engineered products typically falls under Class III medical devices, which are subject to the most stringent regulatory controls due to their potential risks and novel applications. This classification necessitates a premarket approval (PMA) application, a process that demands substantial clinical data to demonstrate safety and efficacy.
Key steps in the regulatory pathway include preclinical testing, which involves in vitro and in vivo studies to assess biocompatibility, mechanical properties, and potential toxicity. These studies are crucial for establishing a solid foundation for subsequent clinical trials and regulatory submissions.
Clinical trials for UHMWPE tissue engineered products generally follow a phased approach. Phase I trials focus on safety and feasibility in a small cohort of patients. Phase II trials expand to larger patient populations and assess preliminary efficacy. Phase III trials are pivotal, large-scale studies designed to provide definitive evidence of safety and effectiveness.
Throughout the development process, manufacturers must adhere to Good Manufacturing Practices (GMP) and implement robust quality management systems. This ensures consistency in product quality and facilitates compliance with regulatory requirements.
The regulatory submission process involves compiling comprehensive documentation, including detailed descriptions of the manufacturing process, quality control measures, and all preclinical and clinical data. The FDA review process is rigorous and may involve advisory committee meetings to evaluate the product's risk-benefit profile.
Post-market surveillance is a critical component of the regulatory pathway, requiring manufacturers to monitor and report adverse events, conduct post-approval studies if mandated, and maintain ongoing compliance with regulatory standards.
Internationally, regulatory pathways may vary, but many countries align with or reference FDA standards. In the European Union, for instance, UHMWPE tissue engineered products would likely be regulated under the Medical Device Regulation (MDR), requiring CE marking through a notified body assessment.
Navigating these regulatory pathways demands a multidisciplinary approach, involving expertise in regulatory affairs, clinical research, quality assurance, and engineering. Successful commercialization of UHMWPE tissue engineered products hinges on a thorough understanding and meticulous execution of these regulatory requirements.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!