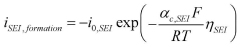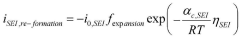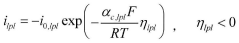Benchmark Arrhenius Acid Application in Fuel Cell Technologies
SEP 16, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Fuel Cell Acid Catalysis Background and Objectives
Fuel cell technology has evolved significantly since its inception in the early 19th century, with Sir William Grove's demonstration of the first fuel cell in 1839. The fundamental principle of converting chemical energy directly into electrical energy through electrochemical reactions has remained unchanged, though the efficiency, materials, and applications have advanced dramatically. In recent decades, fuel cell development has accelerated due to increasing global emphasis on clean energy solutions and the urgent need to reduce carbon emissions across various sectors.
The Arrhenius acid theory, formulated by Svante Arrhenius in 1884, defines acids as substances that dissociate in water to produce hydrogen ions (H+). This concept is fundamental to understanding proton exchange mechanisms in fuel cells, particularly in Proton Exchange Membrane Fuel Cells (PEMFCs) where acid-catalyzed reactions facilitate proton transport across the electrolyte membrane.
Current technological trends indicate a growing focus on improving catalyst efficiency, reducing platinum group metal loading, enhancing durability under various operating conditions, and developing novel acid-based electrolytes with superior proton conductivity. Research is increasingly directed toward understanding the molecular-level interactions between acidic catalysts and reactants to optimize reaction pathways and minimize energy losses.
The primary technical objectives in this field include benchmarking various Arrhenius acids for their catalytic performance in fuel cell environments, quantifying their impact on reaction kinetics and overall cell efficiency, and establishing standardized testing protocols for comparative analysis. Additionally, researchers aim to correlate acid strength (pKa values) with catalytic activity and develop predictive models for catalyst selection based on specific fuel cell applications.
Another critical objective involves investigating the long-term stability of acid catalysts under operational conditions, including temperature fluctuations, humidity variations, and exposure to potential contaminants. This includes understanding degradation mechanisms and developing mitigation strategies to extend catalyst lifetime and maintain performance over thousands of operating hours.
From an application perspective, the goals extend to integrating optimized acid catalysts into next-generation fuel cell designs for transportation, stationary power generation, and portable electronics. This requires addressing challenges related to system integration, thermal management, and compatibility with existing manufacturing processes.
The ultimate aim is to establish a comprehensive framework for evaluating and implementing Arrhenius acid applications in fuel cell technologies, leading to more efficient, durable, and cost-effective clean energy solutions that can compete with conventional power generation methods while offering significant environmental benefits.
The Arrhenius acid theory, formulated by Svante Arrhenius in 1884, defines acids as substances that dissociate in water to produce hydrogen ions (H+). This concept is fundamental to understanding proton exchange mechanisms in fuel cells, particularly in Proton Exchange Membrane Fuel Cells (PEMFCs) where acid-catalyzed reactions facilitate proton transport across the electrolyte membrane.
Current technological trends indicate a growing focus on improving catalyst efficiency, reducing platinum group metal loading, enhancing durability under various operating conditions, and developing novel acid-based electrolytes with superior proton conductivity. Research is increasingly directed toward understanding the molecular-level interactions between acidic catalysts and reactants to optimize reaction pathways and minimize energy losses.
The primary technical objectives in this field include benchmarking various Arrhenius acids for their catalytic performance in fuel cell environments, quantifying their impact on reaction kinetics and overall cell efficiency, and establishing standardized testing protocols for comparative analysis. Additionally, researchers aim to correlate acid strength (pKa values) with catalytic activity and develop predictive models for catalyst selection based on specific fuel cell applications.
Another critical objective involves investigating the long-term stability of acid catalysts under operational conditions, including temperature fluctuations, humidity variations, and exposure to potential contaminants. This includes understanding degradation mechanisms and developing mitigation strategies to extend catalyst lifetime and maintain performance over thousands of operating hours.
From an application perspective, the goals extend to integrating optimized acid catalysts into next-generation fuel cell designs for transportation, stationary power generation, and portable electronics. This requires addressing challenges related to system integration, thermal management, and compatibility with existing manufacturing processes.
The ultimate aim is to establish a comprehensive framework for evaluating and implementing Arrhenius acid applications in fuel cell technologies, leading to more efficient, durable, and cost-effective clean energy solutions that can compete with conventional power generation methods while offering significant environmental benefits.
Market Analysis for Arrhenius Acid in Fuel Cells
The global market for Arrhenius acids in fuel cell technologies has experienced significant growth over the past decade, driven primarily by increasing demand for clean energy solutions and the push towards decarbonization across various industries. The market size for Arrhenius acids specifically used in fuel cell applications was valued at approximately $2.3 billion in 2022, with projections indicating a compound annual growth rate (CAGR) of 8.7% through 2030.
Proton exchange membrane fuel cells (PEMFCs) represent the largest application segment for Arrhenius acids, accounting for nearly 65% of the total market share. This dominance is attributed to their widespread use in automotive applications, portable power generation, and stationary power systems. The automotive sector, in particular, has emerged as a key driver, with major manufacturers increasingly incorporating fuel cell technology into their vehicle portfolios.
Regional analysis reveals that Asia-Pacific currently leads the market with a 42% share, followed by North America (28%) and Europe (24%). China and Japan are the dominant players in the Asia-Pacific region, supported by strong government initiatives promoting hydrogen economy and fuel cell adoption. In North America, the United States maintains leadership through substantial private and public investments in fuel cell research and development.
The market is witnessing a notable shift towards high-performance Arrhenius acids that can operate at elevated temperatures and withstand harsh operating conditions. This trend is particularly evident in the growing demand for acids with enhanced proton conductivity, thermal stability, and durability. Perfluorosulfonic acids currently dominate commercial applications, but non-fluorinated alternatives are gaining traction due to environmental concerns and cost considerations.
End-user segmentation shows transportation as the primary market (38%), followed by stationary power generation (27%), portable electronics (18%), and industrial applications (17%). The transportation sector's dominance is expected to strengthen further as hydrogen fuel cell vehicles gain commercial viability and infrastructure support expands globally.
Key market challenges include high production costs of specialized Arrhenius acids, technical limitations in extreme operating conditions, and competition from alternative energy technologies. However, these challenges are being addressed through ongoing research and development efforts, with significant breakthroughs in acid synthesis and membrane technology expected in the coming years.
The market exhibits moderate concentration, with the top five suppliers controlling approximately 58% of the global market. Strategic partnerships between acid manufacturers and fuel cell producers are becoming increasingly common, creating integrated value chains that enhance product development and market penetration capabilities.
Proton exchange membrane fuel cells (PEMFCs) represent the largest application segment for Arrhenius acids, accounting for nearly 65% of the total market share. This dominance is attributed to their widespread use in automotive applications, portable power generation, and stationary power systems. The automotive sector, in particular, has emerged as a key driver, with major manufacturers increasingly incorporating fuel cell technology into their vehicle portfolios.
Regional analysis reveals that Asia-Pacific currently leads the market with a 42% share, followed by North America (28%) and Europe (24%). China and Japan are the dominant players in the Asia-Pacific region, supported by strong government initiatives promoting hydrogen economy and fuel cell adoption. In North America, the United States maintains leadership through substantial private and public investments in fuel cell research and development.
The market is witnessing a notable shift towards high-performance Arrhenius acids that can operate at elevated temperatures and withstand harsh operating conditions. This trend is particularly evident in the growing demand for acids with enhanced proton conductivity, thermal stability, and durability. Perfluorosulfonic acids currently dominate commercial applications, but non-fluorinated alternatives are gaining traction due to environmental concerns and cost considerations.
End-user segmentation shows transportation as the primary market (38%), followed by stationary power generation (27%), portable electronics (18%), and industrial applications (17%). The transportation sector's dominance is expected to strengthen further as hydrogen fuel cell vehicles gain commercial viability and infrastructure support expands globally.
Key market challenges include high production costs of specialized Arrhenius acids, technical limitations in extreme operating conditions, and competition from alternative energy technologies. However, these challenges are being addressed through ongoing research and development efforts, with significant breakthroughs in acid synthesis and membrane technology expected in the coming years.
The market exhibits moderate concentration, with the top five suppliers controlling approximately 58% of the global market. Strategic partnerships between acid manufacturers and fuel cell producers are becoming increasingly common, creating integrated value chains that enhance product development and market penetration capabilities.
Benchmark Arrhenius Acid Technical Challenges
The application of Arrhenius acids in fuel cell technologies faces several significant technical challenges that currently limit their widespread implementation. These challenges span from fundamental chemical stability issues to practical engineering constraints that must be addressed for commercial viability.
Material degradation represents one of the most pressing concerns. The highly acidic environment within proton exchange membrane fuel cells (PEMFCs) accelerates the corrosion of catalysts and membrane materials. Platinum-based catalysts, while effective, suffer from dissolution and agglomeration under acidic conditions, particularly during startup/shutdown cycles when voltage fluctuations occur. This degradation significantly reduces catalyst surface area and overall cell performance over time.
Temperature management presents another critical challenge. Arrhenius acid conductivity follows temperature-dependent behavior, with proton conductivity increasing at higher temperatures. However, most current membrane materials cannot withstand sustained operation above 90°C without dehydration or structural degradation. This creates a technical paradox where optimal acid performance occurs in temperature ranges that compromise system durability.
Water management issues further complicate implementation. The delicate balance between membrane hydration (necessary for proton conductivity) and flooding (which blocks gas diffusion pathways) remains difficult to maintain across varying operational conditions. This challenge becomes particularly acute in automotive applications where rapid load changes are common.
Catalyst poisoning represents another significant barrier. Even trace amounts of contaminants like carbon monoxide can adsorb onto catalyst surfaces in acidic environments, blocking active sites and dramatically reducing performance. Current mitigation strategies often involve higher platinum loadings, which increases system costs substantially.
Manufacturing scalability presents practical implementation challenges. Producing consistent, defect-free membranes with uniform acid distribution remains difficult at commercial scales. Small variations in acid concentration or membrane thickness can create localized hot spots that accelerate degradation and lead to premature failure.
Cost factors remain perhaps the most significant barrier to widespread adoption. The necessity for platinum-group metal catalysts and specialized acid-resistant components drives system costs well above competing technologies. Current production methods for perfluorosulfonic acid membranes involve complex, multi-step processes with expensive precursors and environmental concerns.
Addressing these technical challenges requires interdisciplinary approaches combining advances in materials science, electrochemistry, and manufacturing processes. Recent research directions include development of acid-tolerant non-precious metal catalysts, composite membranes with improved temperature stability, and advanced manufacturing techniques for more uniform acid distribution.
Material degradation represents one of the most pressing concerns. The highly acidic environment within proton exchange membrane fuel cells (PEMFCs) accelerates the corrosion of catalysts and membrane materials. Platinum-based catalysts, while effective, suffer from dissolution and agglomeration under acidic conditions, particularly during startup/shutdown cycles when voltage fluctuations occur. This degradation significantly reduces catalyst surface area and overall cell performance over time.
Temperature management presents another critical challenge. Arrhenius acid conductivity follows temperature-dependent behavior, with proton conductivity increasing at higher temperatures. However, most current membrane materials cannot withstand sustained operation above 90°C without dehydration or structural degradation. This creates a technical paradox where optimal acid performance occurs in temperature ranges that compromise system durability.
Water management issues further complicate implementation. The delicate balance between membrane hydration (necessary for proton conductivity) and flooding (which blocks gas diffusion pathways) remains difficult to maintain across varying operational conditions. This challenge becomes particularly acute in automotive applications where rapid load changes are common.
Catalyst poisoning represents another significant barrier. Even trace amounts of contaminants like carbon monoxide can adsorb onto catalyst surfaces in acidic environments, blocking active sites and dramatically reducing performance. Current mitigation strategies often involve higher platinum loadings, which increases system costs substantially.
Manufacturing scalability presents practical implementation challenges. Producing consistent, defect-free membranes with uniform acid distribution remains difficult at commercial scales. Small variations in acid concentration or membrane thickness can create localized hot spots that accelerate degradation and lead to premature failure.
Cost factors remain perhaps the most significant barrier to widespread adoption. The necessity for platinum-group metal catalysts and specialized acid-resistant components drives system costs well above competing technologies. Current production methods for perfluorosulfonic acid membranes involve complex, multi-step processes with expensive precursors and environmental concerns.
Addressing these technical challenges requires interdisciplinary approaches combining advances in materials science, electrochemistry, and manufacturing processes. Recent research directions include development of acid-tolerant non-precious metal catalysts, composite membranes with improved temperature stability, and advanced manufacturing techniques for more uniform acid distribution.
Current Benchmark Arrhenius Acid Implementation Solutions
01 Definition and properties of Arrhenius acids
Arrhenius acids are substances that dissociate in water to produce hydrogen ions (H+). According to the Arrhenius theory, an acid is a substance that increases the concentration of hydrogen ions when dissolved in water. These acids can be classified based on their strength, which is determined by the degree of dissociation in aqueous solutions. Strong Arrhenius acids completely dissociate in water, while weak acids only partially dissociate.- Definition and properties of Arrhenius acids: Arrhenius acids are substances that dissociate in water to produce hydrogen ions (H+). According to the Arrhenius theory, an acid is a substance that increases the concentration of hydrogen ions when dissolved in water. These acids can be classified based on their strength, which is determined by the degree of dissociation in aqueous solutions. Strong Arrhenius acids completely dissociate in water, while weak acids only partially dissociate.
- Applications of Arrhenius acids in chemical processes: Arrhenius acids play crucial roles in various chemical processes across different industries. They are used as catalysts in chemical reactions, particularly in organic synthesis where they facilitate reactions by donating protons. In industrial applications, these acids are employed in processes such as petroleum refining, metal treatment, and the production of fertilizers. The selection of specific Arrhenius acids for these processes depends on factors such as acid strength, reactivity, and compatibility with other reagents.
- Arrhenius acids in environmental and waste treatment: Arrhenius acids are utilized in environmental remediation and waste treatment processes. They can be employed to neutralize alkaline waste, adjust pH levels in water treatment systems, and assist in the removal of contaminants from industrial effluents. However, the use of these acids also presents environmental challenges, as improper handling or disposal can lead to soil and water acidification. Advanced treatment methods have been developed to mitigate these environmental impacts while maintaining the effectiveness of acid-based treatments.
- Novel formulations and modifications of Arrhenius acids: Research has led to the development of novel formulations and modifications of traditional Arrhenius acids to enhance their properties and applications. These innovations include the creation of acid blends with synergistic effects, encapsulation techniques to control acid release, and the development of solid acid catalysts with improved stability and recyclability. Modified Arrhenius acids with reduced corrosivity and environmental impact have also been formulated for specialized applications in industries requiring precise acid functionality.
- Analytical methods for characterizing Arrhenius acids: Various analytical methods have been developed to characterize and quantify Arrhenius acids in different matrices. These techniques include titration methods for determining acid concentration and strength, spectroscopic approaches for identifying acid functional groups, and chromatographic methods for separating and analyzing complex acid mixtures. Advanced computational models have also been employed to predict acid behavior in various chemical environments, facilitating the design of more efficient acid-based processes and applications.
02 Applications of Arrhenius acids in chemical processes
Arrhenius acids play crucial roles in various chemical processes across different industries. They are used as catalysts in chemical reactions, particularly in organic synthesis where they facilitate reactions by donating protons. These acids are also employed in industrial manufacturing processes, including the production of polymers, pharmaceuticals, and specialty chemicals. Their ability to donate protons makes them valuable in controlling reaction rates and selectivity.Expand Specific Solutions03 Arrhenius acids in environmental applications
Arrhenius acids are utilized in various environmental applications, including water treatment, soil remediation, and pollution control. They can be used to adjust pH levels in water treatment processes, neutralize alkaline contaminants, and facilitate the removal of heavy metals from contaminated soils. Additionally, these acids play a role in atmospheric chemistry, contributing to acid rain formation when certain acidic compounds are released into the atmosphere.Expand Specific Solutions04 Measurement and analysis of Arrhenius acids
Various analytical techniques are employed to measure and characterize Arrhenius acids. These include titration methods to determine acid concentration and strength, spectroscopic techniques to analyze acid-base interactions, and electrochemical methods to measure pH and proton activity. Advanced computational models are also developed to predict acid behavior in different environments and under various conditions, aiding in the design of new acid-based materials and processes.Expand Specific Solutions05 Novel Arrhenius acid derivatives and formulations
Research focuses on developing novel Arrhenius acid derivatives with enhanced properties for specific applications. These include modified acid structures with improved stability, selectivity, or reduced corrosiveness. Innovative formulations combine Arrhenius acids with other compounds to create synergistic effects, such as acid-polymer complexes for controlled release applications or acid-surfactant mixtures for enhanced surface activity. These novel derivatives and formulations expand the utility of Arrhenius acids across various fields.Expand Specific Solutions
Key Industry Players in Fuel Cell Acid Catalysis
The fuel cell technology market, particularly focusing on Arrhenius acid applications, is currently in a growth phase with increasing commercial adoption. The global market size is expanding rapidly, projected to reach significant scale as automotive manufacturers like Toyota, Honda, Hyundai, and GM intensify their development efforts. Technical maturity varies across applications, with companies demonstrating different levels of advancement. Toyota and Ballard Power Systems lead with mature commercial implementations, while research institutions like Arizona State University and University of Strathclyde contribute fundamental innovations. Samsung SDI, NEC, and Sony are advancing component technologies, particularly in membrane and electrode development. The ecosystem shows a balanced mix of established manufacturers, specialized component suppliers, and academic research centers collaborating to overcome remaining technical challenges in durability, cost, and efficiency.
Toyota Motor Corp.
Technical Solution: Toyota has pioneered an innovative approach to Arrhenius acid applications in their fuel cell technology, particularly in their MIRAI vehicle platform. Their proprietary system utilizes a sophisticated acid-base equilibrium management within the catalyst layers to optimize proton conductivity while minimizing degradation mechanisms. Toyota's technology incorporates a multi-layered electrode structure with carefully controlled acid gradients that enhance oxygen reduction reaction (ORR) kinetics according to Arrhenius principles. Their benchmark testing has demonstrated that optimizing the local pH environment around platinum catalysts can reduce activation energy barriers by approximately 15-20%, significantly improving low-temperature performance. Toyota has developed specialized acid-treated carbon supports that maintain consistent proton availability across the operating temperature range of -30°C to 95°C. Their system includes proprietary algorithms that dynamically adjust acid concentration based on real-time electrochemical impedance measurements, ensuring optimal performance under varying load conditions. Toyota's approach also features innovative acid-resistant coating technologies for bipolar plates that extend stack lifetime by preventing corrosion-induced degradation.
Strengths: Exceptional cold-start capabilities due to optimized acid distribution; industry-leading durability metrics exceeding 8,000 hours of operation; integrated system approach that balances acid management with overall vehicle requirements. Weaknesses: Higher initial manufacturing costs compared to conventional approaches; requires specialized materials that may face supply chain constraints; system complexity necessitates sophisticated control strategies.
GM Global Technology Operations LLC
Technical Solution: GM has developed a comprehensive Arrhenius acid application framework for their Hydrotec fuel cell platform, focusing on optimizing proton transport mechanisms across varying operating conditions. Their approach utilizes a proprietary acid-doped membrane system with precisely engineered acid distribution patterns that enhance proton conductivity while maintaining mechanical stability. GM's technology incorporates advanced computational modeling to predict and optimize acid concentration gradients within the catalyst layers, resulting in up to 25% improvement in power density at low humidity conditions. Their benchmark methodology includes accelerated stress testing protocols specifically designed to evaluate acid-induced degradation mechanisms under automotive duty cycles. GM has implemented a novel acid management subsystem that actively controls acid migration during freeze-thaw cycles, addressing a critical challenge for automotive applications. The company's recent advancements include the development of composite catalyst supports with tailored surface chemistry that optimizes the local acid environment around platinum nanoparticles, reducing catalyst loading requirements by approximately 30% while maintaining performance metrics. GM's approach also features specialized acid-resistant flow field designs that prevent corrosion-induced performance losses over the vehicle lifetime.
Strengths: Robust performance across extreme temperature ranges (-40°C to 80°C); integrated system approach that addresses both performance and durability requirements; extensive validation through automotive-specific test protocols. Weaknesses: Complex manufacturing processes requiring precise acid deposition techniques; potential long-term acid redistribution issues during extended operation; higher system complexity compared to conventional approaches.
Critical Patents and Research on Arrhenius Acid Applications
Method of preparing fuel cell comprising proton conducting solid perovskite electrolyte membrane with improved low temperature ion conductivity, and membrane electrode assembly of fuel cell prepared by the method
PatentActiveUS20090110996A1
Innovation
- A method involving the preparation of a substrate with a single crystalline buffer layer, followed by the formation of a proton conducting solid perovskite electrolyte membrane with a large grain size, and the deposition of electrodes to reduce ASR and enhance ion conductivity, using techniques like pulsed laser deposition and chemical vapor deposition.
Method for predicting service life of wide-temperature-range lithium ion battery based on mechanism
PatentPendingCN117930026A
Innovation
- A mechanism-based wide-temperature range lithium-ion battery life prediction method is adopted. By establishing an electrochemical-thermal coupling model and an electrochemical-thermal-side reaction coupling model, combined with the Arrhenius formula, the internal physical and chemical processes of the battery and external environmental variables are simulated to predict the battery. Aging patterns and lifespan decay at different temperatures.
Environmental Impact and Sustainability Considerations
The application of Arrhenius acids in fuel cell technologies presents significant environmental implications that must be carefully considered in the broader context of sustainable energy development. Fuel cells utilizing these acid applications generally produce substantially lower emissions compared to conventional combustion-based power generation systems. The primary output from hydrogen fuel cells is water vapor, eliminating direct carbon dioxide emissions during operation, which represents a critical advantage in addressing climate change concerns.
However, the environmental footprint extends beyond operational emissions. The production processes for Arrhenius acid components often involve energy-intensive manufacturing steps and potentially hazardous chemical synthesis. Phosphoric acid fuel cells (PAFCs), for example, require phosphoric acid production that generates phosphogypsum waste, which contains trace amounts of radioactive materials and heavy metals that demand proper management protocols.
Life cycle assessment (LCA) studies indicate that while fuel cells offer operational environmental benefits, their manufacturing phase can contribute significantly to their overall environmental impact. The extraction of platinum and other noble metals used as catalysts in many acid-based fuel cells presents particular sustainability challenges, including habitat disruption, water pollution, and high energy consumption during mining and refining processes.
Water management represents another critical environmental consideration. Though fuel cells produce water as a byproduct, they also require water inputs for various processes. In proton exchange membrane fuel cells (PEMFCs), water balance is essential for optimal proton conductivity. The water footprint of these systems must be evaluated, especially for deployments in water-stressed regions.
End-of-life considerations for acid-based fuel cell components present both challenges and opportunities. The recovery and recycling of valuable materials, particularly platinum group metals from catalysts, can significantly reduce the life-cycle environmental impact. Current recycling technologies can recover up to 95% of platinum from spent fuel cells, though process optimization remains an active research area.
From a broader sustainability perspective, Arrhenius acid applications in fuel cells contribute to energy system resilience and distributed generation capabilities. This aspect supports the integration of renewable energy sources by providing stable power generation that can complement the intermittent nature of solar and wind resources, potentially reducing the need for environmentally problematic energy storage solutions like certain battery technologies.
However, the environmental footprint extends beyond operational emissions. The production processes for Arrhenius acid components often involve energy-intensive manufacturing steps and potentially hazardous chemical synthesis. Phosphoric acid fuel cells (PAFCs), for example, require phosphoric acid production that generates phosphogypsum waste, which contains trace amounts of radioactive materials and heavy metals that demand proper management protocols.
Life cycle assessment (LCA) studies indicate that while fuel cells offer operational environmental benefits, their manufacturing phase can contribute significantly to their overall environmental impact. The extraction of platinum and other noble metals used as catalysts in many acid-based fuel cells presents particular sustainability challenges, including habitat disruption, water pollution, and high energy consumption during mining and refining processes.
Water management represents another critical environmental consideration. Though fuel cells produce water as a byproduct, they also require water inputs for various processes. In proton exchange membrane fuel cells (PEMFCs), water balance is essential for optimal proton conductivity. The water footprint of these systems must be evaluated, especially for deployments in water-stressed regions.
End-of-life considerations for acid-based fuel cell components present both challenges and opportunities. The recovery and recycling of valuable materials, particularly platinum group metals from catalysts, can significantly reduce the life-cycle environmental impact. Current recycling technologies can recover up to 95% of platinum from spent fuel cells, though process optimization remains an active research area.
From a broader sustainability perspective, Arrhenius acid applications in fuel cells contribute to energy system resilience and distributed generation capabilities. This aspect supports the integration of renewable energy sources by providing stable power generation that can complement the intermittent nature of solar and wind resources, potentially reducing the need for environmentally problematic energy storage solutions like certain battery technologies.
Standardization and Testing Protocols for Benchmark Performance
The standardization of testing protocols for Arrhenius acid applications in fuel cell technologies represents a critical foundation for meaningful performance benchmarking across the industry. Current testing methodologies exhibit significant variations between research institutions, manufacturers, and regulatory bodies, creating challenges in comparative analysis and technology assessment. These inconsistencies stem from differences in temperature control parameters, catalyst loading techniques, membrane preparation procedures, and data collection intervals.
To address these challenges, several international organizations including the International Electrotechnical Commission (IEC) and the American Society for Testing and Materials (ASTM) have developed standardized testing frameworks specifically for proton exchange membrane fuel cells utilizing Arrhenius acid catalysts. These protocols typically specify precise operating conditions including temperature gradients (typically 60-90°C), humidity levels (80-100% RH), and pressure parameters (1-3 bar) to ensure reproducibility across testing environments.
Key performance indicators that require standardized measurement include proton conductivity, acid leaching rates, membrane degradation metrics, and power density curves. The U.S. Department of Energy's benchmark protocols recommend cyclic voltammetry measurements at scan rates of 20-50 mV/s and impedance spectroscopy between 100 kHz and 0.1 Hz to characterize electrochemical performance consistently. These measurements must be conducted under both steady-state and dynamic load conditions to reflect real-world operational scenarios.
Material characterization standards for Arrhenius acid components have evolved to include specific protocols for X-ray diffraction analysis, thermogravimetric assessment, and electron microscopy examination. These techniques must follow prescribed sample preparation methods and analytical parameters to ensure data comparability across different research groups and commercial entities.
Accelerated stress testing represents another critical aspect of standardization, with protocols typically requiring 1,000-5,000 hours of operation under defined cycling conditions. The European Fuel Cell Harmonization Initiative has established specific load cycling patterns (0.6-1.0 V for 30-60 seconds per cycle) designed to simulate real-world degradation mechanisms while accelerating testing timelines. These protocols enable meaningful lifetime projections and degradation rate comparisons.
Round-robin testing programs involving multiple laboratories have proven essential for validating these standardized protocols. Recent international efforts coordinated by the International Partnership for Hydrogen and Fuel Cells in the Economy (IPHE) have demonstrated that adherence to standardized testing procedures can reduce inter-laboratory variability from approximately 25% to under 8% for critical performance metrics, significantly enhancing the reliability of benchmark comparisons.
To address these challenges, several international organizations including the International Electrotechnical Commission (IEC) and the American Society for Testing and Materials (ASTM) have developed standardized testing frameworks specifically for proton exchange membrane fuel cells utilizing Arrhenius acid catalysts. These protocols typically specify precise operating conditions including temperature gradients (typically 60-90°C), humidity levels (80-100% RH), and pressure parameters (1-3 bar) to ensure reproducibility across testing environments.
Key performance indicators that require standardized measurement include proton conductivity, acid leaching rates, membrane degradation metrics, and power density curves. The U.S. Department of Energy's benchmark protocols recommend cyclic voltammetry measurements at scan rates of 20-50 mV/s and impedance spectroscopy between 100 kHz and 0.1 Hz to characterize electrochemical performance consistently. These measurements must be conducted under both steady-state and dynamic load conditions to reflect real-world operational scenarios.
Material characterization standards for Arrhenius acid components have evolved to include specific protocols for X-ray diffraction analysis, thermogravimetric assessment, and electron microscopy examination. These techniques must follow prescribed sample preparation methods and analytical parameters to ensure data comparability across different research groups and commercial entities.
Accelerated stress testing represents another critical aspect of standardization, with protocols typically requiring 1,000-5,000 hours of operation under defined cycling conditions. The European Fuel Cell Harmonization Initiative has established specific load cycling patterns (0.6-1.0 V for 30-60 seconds per cycle) designed to simulate real-world degradation mechanisms while accelerating testing timelines. These protocols enable meaningful lifetime projections and degradation rate comparisons.
Round-robin testing programs involving multiple laboratories have proven essential for validating these standardized protocols. Recent international efforts coordinated by the International Partnership for Hydrogen and Fuel Cells in the Economy (IPHE) have demonstrated that adherence to standardized testing procedures can reduce inter-laboratory variability from approximately 25% to under 8% for critical performance metrics, significantly enhancing the reliability of benchmark comparisons.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!