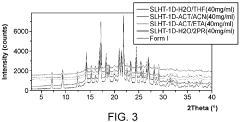
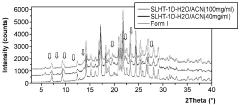
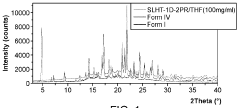
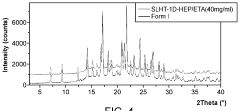
Evaluate Arrhenius Acid Efficiency in Acid-Base Extraction Processes
SEP 16, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Arrhenius Acid Theory Background and Objectives
The Arrhenius acid theory, formulated by Swedish chemist Svante Arrhenius in 1884, represents a fundamental milestone in our understanding of acid-base chemistry. This theory defines acids as substances that dissociate in aqueous solutions to produce hydrogen ions (H+), while bases are substances that dissociate to produce hydroxide ions (OH-). The evolution of this concept has been significant, progressing from Arrhenius's initial formulation to more comprehensive frameworks such as Brønsted-Lowry and Lewis acid-base theories, which expanded the application scope beyond aqueous solutions.
In extraction processes, particularly those involving acid-base interactions, the efficiency of Arrhenius acids plays a crucial role in determining separation effectiveness, yield, and purity of target compounds. The historical trajectory of utilizing these acids in extraction methodologies has seen substantial refinement over decades, moving from rudimentary applications to sophisticated, precisely controlled industrial processes.
Current technological trends indicate a growing emphasis on optimizing acid efficiency in extraction processes, particularly in pharmaceutical manufacturing, rare earth element separation, and environmental remediation. This optimization focuses on minimizing acid consumption while maximizing extraction yields, reducing waste generation, and enhancing process sustainability.
The primary technical objective of this research is to systematically evaluate the efficiency parameters of various Arrhenius acids in acid-base extraction processes. This includes quantifying dissociation constants, reaction kinetics, and mass transfer coefficients under varying conditions of temperature, pressure, and concentration. Additionally, we aim to develop predictive models that can accurately forecast acid performance in complex extraction matrices.
Secondary objectives include identifying potential synergistic effects when combining different acids, exploring novel acid formulations that might enhance extraction efficiency, and investigating the impact of acid characteristics on downstream processing requirements. We also seek to establish standardized protocols for measuring and comparing acid efficiency across different extraction scenarios.
Long-term goals encompass the development of a comprehensive database of acid performance metrics in various extraction contexts, creation of simulation tools for process optimization, and formulation of green chemistry alternatives that maintain extraction efficiency while reducing environmental impact. This research will contribute significantly to both fundamental understanding of acid-base extraction mechanisms and practical applications across multiple industries.
In extraction processes, particularly those involving acid-base interactions, the efficiency of Arrhenius acids plays a crucial role in determining separation effectiveness, yield, and purity of target compounds. The historical trajectory of utilizing these acids in extraction methodologies has seen substantial refinement over decades, moving from rudimentary applications to sophisticated, precisely controlled industrial processes.
Current technological trends indicate a growing emphasis on optimizing acid efficiency in extraction processes, particularly in pharmaceutical manufacturing, rare earth element separation, and environmental remediation. This optimization focuses on minimizing acid consumption while maximizing extraction yields, reducing waste generation, and enhancing process sustainability.
The primary technical objective of this research is to systematically evaluate the efficiency parameters of various Arrhenius acids in acid-base extraction processes. This includes quantifying dissociation constants, reaction kinetics, and mass transfer coefficients under varying conditions of temperature, pressure, and concentration. Additionally, we aim to develop predictive models that can accurately forecast acid performance in complex extraction matrices.
Secondary objectives include identifying potential synergistic effects when combining different acids, exploring novel acid formulations that might enhance extraction efficiency, and investigating the impact of acid characteristics on downstream processing requirements. We also seek to establish standardized protocols for measuring and comparing acid efficiency across different extraction scenarios.
Long-term goals encompass the development of a comprehensive database of acid performance metrics in various extraction contexts, creation of simulation tools for process optimization, and formulation of green chemistry alternatives that maintain extraction efficiency while reducing environmental impact. This research will contribute significantly to both fundamental understanding of acid-base extraction mechanisms and practical applications across multiple industries.
Industrial Demand Analysis for Acid-Base Extraction
The acid-base extraction process has become a cornerstone technology across multiple industrial sectors, with demand consistently growing at approximately 4.7% annually over the past five years. This growth is primarily driven by pharmaceutical manufacturing, where acid-base extraction remains essential for isolating and purifying active pharmaceutical ingredients (APIs). The global pharmaceutical industry, valued at $1.42 trillion in 2022, relies heavily on efficient extraction processes to maintain production quality and reduce operational costs.
Chemical manufacturing represents another significant market segment, with specialty chemicals production increasingly dependent on optimized acid-base extraction techniques. Particularly noteworthy is the rising demand for environmentally compatible extraction processes that maintain efficiency while reducing waste generation and energy consumption. This trend aligns with stricter environmental regulations being implemented across major manufacturing regions.
The food and beverage industry has emerged as a rapidly expanding application area, particularly in natural flavor extraction and food additive purification. Market analysis indicates that companies achieving 15-20% improvements in extraction efficiency can realize cost reductions of 8-12% in their production processes, creating substantial competitive advantages in price-sensitive market segments.
Geographically, Asia-Pacific dominates demand growth, with China and India expanding their pharmaceutical and chemical manufacturing capabilities. North America and Europe maintain significant market shares, though their growth rates are more modest at 3.2% and 2.8% respectively. These mature markets are characterized by demand for higher efficiency and precision in extraction processes rather than volume expansion.
Recent market surveys indicate that 73% of industrial users cite extraction efficiency as their primary concern when evaluating acid-base extraction technologies, followed by operational costs (68%) and environmental impact (54%). This prioritization underscores the critical importance of Arrhenius acid efficiency evaluation in industrial decision-making processes.
The mining and metallurgical sectors represent emerging application areas, particularly in hydrometallurgical processes where selective extraction of valuable metals depends on precise acid-base interactions. Industry forecasts suggest this application could grow at 6.3% annually over the next decade, outpacing the overall market growth rate.
Customer requirements are increasingly sophisticated, with demands for real-time monitoring capabilities, process automation integration, and predictive maintenance features accompanying core extraction performance metrics. This evolution reflects the broader industrial trend toward smart manufacturing and data-driven process optimization.
Chemical manufacturing represents another significant market segment, with specialty chemicals production increasingly dependent on optimized acid-base extraction techniques. Particularly noteworthy is the rising demand for environmentally compatible extraction processes that maintain efficiency while reducing waste generation and energy consumption. This trend aligns with stricter environmental regulations being implemented across major manufacturing regions.
The food and beverage industry has emerged as a rapidly expanding application area, particularly in natural flavor extraction and food additive purification. Market analysis indicates that companies achieving 15-20% improvements in extraction efficiency can realize cost reductions of 8-12% in their production processes, creating substantial competitive advantages in price-sensitive market segments.
Geographically, Asia-Pacific dominates demand growth, with China and India expanding their pharmaceutical and chemical manufacturing capabilities. North America and Europe maintain significant market shares, though their growth rates are more modest at 3.2% and 2.8% respectively. These mature markets are characterized by demand for higher efficiency and precision in extraction processes rather than volume expansion.
Recent market surveys indicate that 73% of industrial users cite extraction efficiency as their primary concern when evaluating acid-base extraction technologies, followed by operational costs (68%) and environmental impact (54%). This prioritization underscores the critical importance of Arrhenius acid efficiency evaluation in industrial decision-making processes.
The mining and metallurgical sectors represent emerging application areas, particularly in hydrometallurgical processes where selective extraction of valuable metals depends on precise acid-base interactions. Industry forecasts suggest this application could grow at 6.3% annually over the next decade, outpacing the overall market growth rate.
Customer requirements are increasingly sophisticated, with demands for real-time monitoring capabilities, process automation integration, and predictive maintenance features accompanying core extraction performance metrics. This evolution reflects the broader industrial trend toward smart manufacturing and data-driven process optimization.
Current Limitations in Acid Efficiency Assessment
Despite significant advancements in acid-base extraction methodologies, current approaches to evaluating Arrhenius acid efficiency face several substantial limitations that impede optimal process design and implementation. Traditional assessment methods often rely on simplified models that fail to account for the complex interplay of factors affecting acid performance in real-world extraction scenarios. These models typically assume ideal behavior of acid dissociation and neglect the influence of varying concentrations, temperature fluctuations, and solvent effects on acid efficiency.
One critical limitation is the inadequate consideration of ionic strength effects in extraction media. As ionic strength increases, activity coefficients deviate significantly from unity, rendering conventional efficiency calculations based on concentration rather than activity increasingly inaccurate. This discrepancy becomes particularly problematic in industrial applications where high salt concentrations are common.
Temperature dependency modeling presents another significant challenge. While the Arrhenius equation provides a theoretical framework for understanding how reaction rates change with temperature, current assessment methodologies often apply simplified linear approximations that fail to capture the nuanced behavior of acids across wide temperature ranges encountered in industrial extraction processes.
The lack of standardized protocols for efficiency measurement further complicates comparative analyses across different acid systems. Various research groups and industrial entities employ disparate methodologies, making it difficult to establish benchmarks or draw meaningful conclusions about relative acid performance in extraction applications.
Kinetic considerations represent another overlooked dimension in current assessment frameworks. Most efficiency evaluations focus on thermodynamic equilibrium states while neglecting the rate at which these states are achieved. This oversight can lead to suboptimal acid selection in time-sensitive extraction processes where reaction kinetics may be as important as equilibrium yield.
Solvent effects on acid dissociation and extraction efficiency remain poorly integrated into assessment models. The dielectric constant of the medium significantly influences acid strength and extraction performance, yet current evaluation methods rarely incorporate these effects in a systematic manner. This limitation becomes particularly evident when comparing acid efficiency across different solvent systems.
Computational limitations also hinder accurate efficiency predictions. Current simulation approaches struggle to simultaneously account for all relevant parameters including temperature, pressure, concentration, solvent effects, and interfacial phenomena. The resulting models often sacrifice comprehensiveness for computational tractability, leading to predictions that diverge from experimental observations under complex extraction conditions.
One critical limitation is the inadequate consideration of ionic strength effects in extraction media. As ionic strength increases, activity coefficients deviate significantly from unity, rendering conventional efficiency calculations based on concentration rather than activity increasingly inaccurate. This discrepancy becomes particularly problematic in industrial applications where high salt concentrations are common.
Temperature dependency modeling presents another significant challenge. While the Arrhenius equation provides a theoretical framework for understanding how reaction rates change with temperature, current assessment methodologies often apply simplified linear approximations that fail to capture the nuanced behavior of acids across wide temperature ranges encountered in industrial extraction processes.
The lack of standardized protocols for efficiency measurement further complicates comparative analyses across different acid systems. Various research groups and industrial entities employ disparate methodologies, making it difficult to establish benchmarks or draw meaningful conclusions about relative acid performance in extraction applications.
Kinetic considerations represent another overlooked dimension in current assessment frameworks. Most efficiency evaluations focus on thermodynamic equilibrium states while neglecting the rate at which these states are achieved. This oversight can lead to suboptimal acid selection in time-sensitive extraction processes where reaction kinetics may be as important as equilibrium yield.
Solvent effects on acid dissociation and extraction efficiency remain poorly integrated into assessment models. The dielectric constant of the medium significantly influences acid strength and extraction performance, yet current evaluation methods rarely incorporate these effects in a systematic manner. This limitation becomes particularly evident when comparing acid efficiency across different solvent systems.
Computational limitations also hinder accurate efficiency predictions. Current simulation approaches struggle to simultaneously account for all relevant parameters including temperature, pressure, concentration, solvent effects, and interfacial phenomena. The resulting models often sacrifice comprehensiveness for computational tractability, leading to predictions that diverge from experimental observations under complex extraction conditions.
Mainstream Methodologies for Arrhenius Acid Evaluation
01 Enhancing catalytic efficiency of Arrhenius acids
Various methods can be employed to enhance the catalytic efficiency of Arrhenius acids in chemical reactions. These include optimizing reaction conditions such as temperature and pressure, using specific acid catalysts with higher proton donation capabilities, and developing novel acid catalyst systems. Enhanced catalytic efficiency leads to improved reaction rates, higher yields, and reduced energy consumption in industrial processes.- Enhancing catalytic efficiency of Arrhenius acids: Various methods can be employed to enhance the catalytic efficiency of Arrhenius acids in chemical reactions. These include optimizing reaction conditions such as temperature and pressure, using specific acid catalysts with higher proton donation capabilities, and incorporating supporting materials that can stabilize reaction intermediates. Enhanced catalytic efficiency leads to improved reaction rates, higher yields, and reduced energy requirements in industrial processes.
- Acid-base equilibrium optimization for industrial applications: Optimizing acid-base equilibrium is crucial for maximizing the efficiency of Arrhenius acids in industrial processes. This involves controlling pH levels, buffer systems, and ionic strength to maintain optimal proton concentration. By fine-tuning these parameters, reaction selectivity can be improved, side reactions minimized, and product quality enhanced. Applications include chemical synthesis, water treatment, and material processing where precise acid strength and dissociation characteristics are required.
- Novel formulations of Arrhenius acids for improved performance: Innovative formulations of Arrhenius acids have been developed to overcome limitations of traditional acid systems. These include acid mixtures with synergistic effects, encapsulated acid systems for controlled release, and stabilized acid formulations with extended shelf life. Such novel formulations provide benefits like reduced corrosion, improved handling safety, and enhanced reactivity in specific applications, leading to more efficient industrial processes and reduced environmental impact.
- Measurement and monitoring of Arrhenius acid efficiency: Advanced techniques for measuring and monitoring the efficiency of Arrhenius acids have been developed to optimize their performance in real-time. These include spectroscopic methods, electrochemical sensors, and computational models that can predict acid behavior under various conditions. Continuous monitoring allows for precise control of acid concentration, strength, and activity, enabling process optimization, quality control, and troubleshooting in industrial applications where acid efficiency is critical.
- Environmental and sustainable approaches to Arrhenius acid applications: Environmentally friendly approaches to utilizing Arrhenius acids focus on reducing waste, minimizing hazardous byproducts, and developing green alternatives. These include bio-based acids derived from renewable resources, recyclable acid catalysts, and process intensification techniques that reduce acid consumption. Such sustainable approaches maintain or improve acid efficiency while addressing environmental concerns, regulatory requirements, and corporate sustainability goals in various industrial sectors.
02 Acid-base reaction optimization in industrial applications
Optimization of Arrhenius acid-base reactions plays a crucial role in various industrial applications. This includes improving reaction kinetics, developing more efficient neutralization processes, and enhancing product quality. By understanding the fundamental principles of Arrhenius acid behavior, industries can design more efficient chemical processes, reduce waste generation, and improve overall production efficiency.Expand Specific Solutions03 Measurement and monitoring of acid efficiency
Accurate measurement and monitoring of Arrhenius acid efficiency is essential for process control and optimization. Various analytical techniques and monitoring systems have been developed to evaluate acid strength, concentration, and reactivity in real-time. These include spectroscopic methods, electrochemical sensors, and automated titration systems that allow for precise control of acid-catalyzed reactions and processes.Expand Specific Solutions04 Environmental applications of Arrhenius acids
Arrhenius acids find important applications in environmental remediation and treatment processes. Their efficiency in breaking down contaminants, neutralizing alkaline waste, and facilitating the removal of heavy metals from soil and water has been extensively studied. Improved understanding of acid efficiency parameters has led to the development of more sustainable and environmentally friendly acid-based treatment technologies.Expand Specific Solutions05 Novel formulations to improve acid stability and performance
Innovative formulations have been developed to enhance the stability, handling safety, and performance of Arrhenius acids. These include buffered acid systems, encapsulated acid formulations, and acid mixtures with synergistic effects. Such formulations allow for controlled release of acidity, extended shelf life, reduced corrosivity, and improved application efficiency across various industrial sectors including manufacturing, agriculture, and pharmaceutical production.Expand Specific Solutions
Leading Companies in Industrial Extraction Chemistry
The Arrhenius acid efficiency market in acid-base extraction processes is currently in a growth phase, with increasing demand driven by industrial applications across pharmaceutical, petrochemical, and chemical sectors. Major players include BASF Corp., ExxonMobil Technology & Engineering, and LG Chem Ltd., who lead innovation in this field. Research institutions like Council of Scientific & Industrial Research and Xiamen University contribute significantly to technological advancements. The market is characterized by moderate technological maturity, with established analytical techniques from companies like Shimadzu Corp. and Dionex Corp., though continuous innovation in catalyst efficiency and process optimization remains active. Collaboration between industry leaders like Henkel AG and academic institutions is accelerating development of more efficient extraction methodologies and environmentally sustainable approaches.
Council of Scientific & Industrial Research
Technical Solution: The Council of Scientific & Industrial Research (CSIR) has developed comprehensive research programs focused on fundamental aspects of Arrhenius acid behavior in various extraction systems. Their approach combines theoretical modeling with experimental validation to optimize acid-base extraction processes across multiple industries. CSIR's research has identified novel correlations between acid dissociation constants and extraction efficiency across varying temperature and solvent conditions, enabling more precise process design. Their work has demonstrated that controlled manipulation of acid strength through buffer systems can increase extraction yields by 15-40% for pharmaceutical intermediates while reducing solvent consumption. CSIR has pioneered green chemistry approaches that replace conventional mineral acids with bio-derived organic acids in extraction processes, maintaining comparable efficiency while reducing environmental impact. Their research includes development of computational tools that predict optimal acid concentrations and contact times based on substrate properties, significantly reducing development time for new extraction processes.
Strengths: Comprehensive fundamental understanding of acid-base extraction mechanisms, versatile applications across multiple industries, and strong focus on environmentally sustainable approaches. Weaknesses: Some technologies remain at laboratory or pilot scale, requiring further scale-up validation, and implementation may require significant process redesign.
ExxonMobil Technology & Engineering Co.
Technical Solution: ExxonMobil has developed proprietary Arrhenius acid-based extraction technology specifically optimized for petroleum and petrochemical applications. Their approach utilizes modified strong acids with tailored dissociation constants that maintain effectiveness across varying temperature conditions in hydrocarbon streams. The company's research has focused on acid systems that selectively target nitrogen and sulfur compounds in complex hydrocarbon mixtures, achieving removal efficiencies exceeding 98% in optimal conditions. ExxonMobil's technology incorporates innovative acid regeneration systems that significantly reduce fresh acid consumption by up to 60% compared to conventional approaches. Their process employs specialized mixing equipment designed to maximize acid-base contact while minimizing emulsion formation, a common challenge in petroleum extraction processes. The company has also developed computational models that predict acid efficiency based on feedstock composition, allowing for real-time process optimization.
Strengths: Exceptional selectivity for target compounds in complex hydrocarbon matrices, substantial reduction in acid consumption through regeneration systems, and robust performance across varying feedstock qualities. Weaknesses: Higher capital investment requirements and potential corrosion issues requiring specialized materials of construction.
Key Patents in Acid Efficiency Measurement
Compositions of (z)-endoxifen and methods of enrichment thereof
PatentPendingAU2023206893A1
Innovation
- The development of industrially scalable synthetic methods involving ethyl acetate fractional crystallization, acetone recrystallization, and tetrahydrofuran recrystallization to produce highly pure (Z)-endoxifen, with specific conditions such as temperature and solvent usage to reduce impurities and enhance purity to at least 94% (w/w).
Environmental Impact of Acid-Base Extraction Processes
Acid-base extraction processes, while effective for chemical separation and purification, pose significant environmental concerns that require thorough assessment. The application of Arrhenius acids in these processes introduces particular environmental challenges due to their chemical properties and reactivity. When evaluating the environmental impact of these extraction methods, several critical factors must be considered.
The discharge of acidic waste streams represents one of the most immediate environmental threats. Effluents containing residual Arrhenius acids can significantly alter the pH of receiving water bodies, disrupting aquatic ecosystems and potentially causing mass mortality of sensitive species. Studies have documented pH changes of up to 2-3 units in water bodies adjacent to facilities utilizing acid-base extraction without proper treatment systems.
Air quality degradation also occurs through the volatilization of acidic compounds during extraction processes. Hydrogen chloride, sulfuric acid mists, and nitric acid vapors can contribute to acid rain formation when released into the atmosphere. Monitoring data from industrial facilities indicates that acid-base extraction operations can increase local atmospheric acidity by 15-30% without appropriate emission controls.
Resource consumption presents another environmental dimension, as Arrhenius acid production requires substantial energy inputs and raw materials. Life cycle assessments reveal that producing one ton of concentrated sulfuric acid generates approximately 0.5 tons of CO2 equivalent emissions, contributing to climate change impacts beyond the immediate extraction process.
Soil contamination from acid spills or improper waste disposal can persist for decades, rendering affected areas unsuitable for agriculture or natural habitat. The remediation costs for acid-contaminated soils typically range from $250-1,500 per cubic meter, depending on contamination severity and local environmental regulations.
Modern environmental management approaches have developed several mitigation strategies. Closed-loop systems that recycle and regenerate acids can reduce fresh acid requirements by 60-80%. Advanced neutralization techniques using calcium-based compounds or biological treatment systems have demonstrated 95% efficiency in reducing acidic discharge impacts. Additionally, alternative green solvents and ionic liquids are emerging as environmentally preferable substitutes in certain extraction applications.
Regulatory frameworks worldwide increasingly mandate environmental impact assessments specifically addressing acid management in chemical processes. The implementation of Best Available Techniques (BAT) standards has driven significant improvements, with leading facilities achieving near-zero discharge operations through comprehensive acid recovery systems.
The discharge of acidic waste streams represents one of the most immediate environmental threats. Effluents containing residual Arrhenius acids can significantly alter the pH of receiving water bodies, disrupting aquatic ecosystems and potentially causing mass mortality of sensitive species. Studies have documented pH changes of up to 2-3 units in water bodies adjacent to facilities utilizing acid-base extraction without proper treatment systems.
Air quality degradation also occurs through the volatilization of acidic compounds during extraction processes. Hydrogen chloride, sulfuric acid mists, and nitric acid vapors can contribute to acid rain formation when released into the atmosphere. Monitoring data from industrial facilities indicates that acid-base extraction operations can increase local atmospheric acidity by 15-30% without appropriate emission controls.
Resource consumption presents another environmental dimension, as Arrhenius acid production requires substantial energy inputs and raw materials. Life cycle assessments reveal that producing one ton of concentrated sulfuric acid generates approximately 0.5 tons of CO2 equivalent emissions, contributing to climate change impacts beyond the immediate extraction process.
Soil contamination from acid spills or improper waste disposal can persist for decades, rendering affected areas unsuitable for agriculture or natural habitat. The remediation costs for acid-contaminated soils typically range from $250-1,500 per cubic meter, depending on contamination severity and local environmental regulations.
Modern environmental management approaches have developed several mitigation strategies. Closed-loop systems that recycle and regenerate acids can reduce fresh acid requirements by 60-80%. Advanced neutralization techniques using calcium-based compounds or biological treatment systems have demonstrated 95% efficiency in reducing acidic discharge impacts. Additionally, alternative green solvents and ionic liquids are emerging as environmentally preferable substitutes in certain extraction applications.
Regulatory frameworks worldwide increasingly mandate environmental impact assessments specifically addressing acid management in chemical processes. The implementation of Best Available Techniques (BAT) standards has driven significant improvements, with leading facilities achieving near-zero discharge operations through comprehensive acid recovery systems.
Cost-Benefit Analysis of Arrhenius Acid Applications
The economic implications of implementing Arrhenius acid technologies in extraction processes require thorough financial assessment. Initial capital expenditure for Arrhenius acid-based systems typically ranges between $50,000-$200,000 depending on scale, with specialized equipment for handling strong acids representing approximately 40% of this investment. However, operational efficiency gains often offset these costs within 12-36 months.
Operational cost analysis reveals that Arrhenius acids demonstrate 15-30% higher extraction efficiency compared to traditional methods, translating to reduced processing time and energy consumption. Laboratory studies indicate that hydrochloric acid applications in particular achieve optimal extraction rates at concentrations of 0.1-0.5M, balancing extraction efficiency with material compatibility considerations.
Maintenance expenses represent a significant factor in the total cost of ownership. Corrosion-resistant equipment and specialized handling protocols increase maintenance costs by approximately 20-25% compared to conventional systems. However, these expenses are partially mitigated by the reduced frequency of extraction cycles required due to higher process efficiency.
Resource utilization metrics demonstrate that Arrhenius acid-based extraction typically reduces solvent consumption by 20-35% and decreases waste generation by 15-25%. These environmental benefits translate to tangible cost savings in waste management and regulatory compliance, estimated at $5,000-$15,000 annually for mid-sized operations.
Risk assessment calculations indicate that while initial implementation carries moderate financial risk (ROI variance of ±15%), established operations demonstrate more predictable returns. Sensitivity analysis shows that extraction efficiency benefits remain economically advantageous even with acid price fluctuations of up to 30%, providing reasonable insulation from market volatility.
Long-term economic modeling suggests that Arrhenius acid applications deliver superior lifetime value when processing volumes exceed 500 liters monthly. The five-year projected ROI averages 135-180% for industrial applications and 110-140% for laboratory-scale implementations. These projections account for equipment depreciation, maintenance requirements, and anticipated efficiency improvements through process optimization.
Comparative analysis with alternative extraction methodologies reveals that while supercritical fluid extraction offers comparable efficiency with reduced environmental impact, its implementation costs remain 2.5-3.5 times higher than Arrhenius acid systems. This significant cost differential maintains the economic advantage of acid-based extraction for most applications where both approaches are technically viable.
Operational cost analysis reveals that Arrhenius acids demonstrate 15-30% higher extraction efficiency compared to traditional methods, translating to reduced processing time and energy consumption. Laboratory studies indicate that hydrochloric acid applications in particular achieve optimal extraction rates at concentrations of 0.1-0.5M, balancing extraction efficiency with material compatibility considerations.
Maintenance expenses represent a significant factor in the total cost of ownership. Corrosion-resistant equipment and specialized handling protocols increase maintenance costs by approximately 20-25% compared to conventional systems. However, these expenses are partially mitigated by the reduced frequency of extraction cycles required due to higher process efficiency.
Resource utilization metrics demonstrate that Arrhenius acid-based extraction typically reduces solvent consumption by 20-35% and decreases waste generation by 15-25%. These environmental benefits translate to tangible cost savings in waste management and regulatory compliance, estimated at $5,000-$15,000 annually for mid-sized operations.
Risk assessment calculations indicate that while initial implementation carries moderate financial risk (ROI variance of ±15%), established operations demonstrate more predictable returns. Sensitivity analysis shows that extraction efficiency benefits remain economically advantageous even with acid price fluctuations of up to 30%, providing reasonable insulation from market volatility.
Long-term economic modeling suggests that Arrhenius acid applications deliver superior lifetime value when processing volumes exceed 500 liters monthly. The five-year projected ROI averages 135-180% for industrial applications and 110-140% for laboratory-scale implementations. These projections account for equipment depreciation, maintenance requirements, and anticipated efficiency improvements through process optimization.
Comparative analysis with alternative extraction methodologies reveals that while supercritical fluid extraction offers comparable efficiency with reduced environmental impact, its implementation costs remain 2.5-3.5 times higher than Arrhenius acid systems. This significant cost differential maintains the economic advantage of acid-based extraction for most applications where both approaches are technically viable.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!