How to Choose Gas Mixtures for Temperature Programmed Reduction Experiments
MAR 7, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Gas Mixture Selection for TPR Background and Objectives
Temperature Programmed Reduction (TPR) has emerged as a fundamental characterization technique in catalysis research since its development in the 1960s. The method involves heating a sample in a reducing atmosphere while monitoring the consumption of reducing agents, providing crucial insights into the reducibility of metal oxides, supported catalysts, and complex oxide systems. The evolution of TPR methodology has been driven by the increasing demand for precise characterization of catalytic materials in petrochemical, environmental, and energy conversion applications.
The historical development of TPR techniques reveals a progressive refinement in gas mixture selection strategies. Early TPR experiments primarily utilized pure hydrogen as the reducing agent, but researchers quickly recognized that different gas compositions could reveal distinct reduction mechanisms and provide more comprehensive material characterization. The introduction of diluted hydrogen mixtures marked a significant advancement, enabling better control over reduction kinetics and improved detection sensitivity.
Current technological objectives in TPR gas mixture selection focus on achieving optimal balance between reduction efficiency and analytical precision. The primary goal involves selecting gas compositions that provide clear, interpretable reduction profiles while maintaining experimental reproducibility. Modern TPR applications demand gas mixtures that can differentiate between various reducible species within complex catalyst systems, particularly in multi-component materials where overlapping reduction peaks may obscure individual contributions.
The advancement of detection technologies has expanded the scope of achievable objectives in TPR experiments. Contemporary mass spectrometry and thermal conductivity detection systems enable the use of more sophisticated gas mixtures, including inert gas dilutions and alternative reducing agents beyond hydrogen. These technological improvements have established new targets for enhanced resolution of reduction events and improved quantitative analysis capabilities.
Strategic objectives in TPR gas mixture optimization encompass both fundamental understanding and practical applications. Researchers aim to develop standardized protocols that ensure consistent results across different laboratories while maintaining flexibility for specialized applications. The integration of computational modeling with experimental design has created opportunities for predictive gas mixture selection based on theoretical reduction thermodynamics.
The contemporary focus extends toward developing environmentally conscious TPR methodologies that minimize hazardous gas consumption while maintaining analytical quality. This objective aligns with broader sustainability goals in analytical chemistry and reflects the growing emphasis on green analytical practices in materials characterization research.
The historical development of TPR techniques reveals a progressive refinement in gas mixture selection strategies. Early TPR experiments primarily utilized pure hydrogen as the reducing agent, but researchers quickly recognized that different gas compositions could reveal distinct reduction mechanisms and provide more comprehensive material characterization. The introduction of diluted hydrogen mixtures marked a significant advancement, enabling better control over reduction kinetics and improved detection sensitivity.
Current technological objectives in TPR gas mixture selection focus on achieving optimal balance between reduction efficiency and analytical precision. The primary goal involves selecting gas compositions that provide clear, interpretable reduction profiles while maintaining experimental reproducibility. Modern TPR applications demand gas mixtures that can differentiate between various reducible species within complex catalyst systems, particularly in multi-component materials where overlapping reduction peaks may obscure individual contributions.
The advancement of detection technologies has expanded the scope of achievable objectives in TPR experiments. Contemporary mass spectrometry and thermal conductivity detection systems enable the use of more sophisticated gas mixtures, including inert gas dilutions and alternative reducing agents beyond hydrogen. These technological improvements have established new targets for enhanced resolution of reduction events and improved quantitative analysis capabilities.
Strategic objectives in TPR gas mixture optimization encompass both fundamental understanding and practical applications. Researchers aim to develop standardized protocols that ensure consistent results across different laboratories while maintaining flexibility for specialized applications. The integration of computational modeling with experimental design has created opportunities for predictive gas mixture selection based on theoretical reduction thermodynamics.
The contemporary focus extends toward developing environmentally conscious TPR methodologies that minimize hazardous gas consumption while maintaining analytical quality. This objective aligns with broader sustainability goals in analytical chemistry and reflects the growing emphasis on green analytical practices in materials characterization research.
Market Demand for Advanced TPR Gas Analysis Solutions
The global market for advanced Temperature Programmed Reduction (TPR) gas analysis solutions is experiencing significant growth driven by increasing demand for precise catalyst characterization across multiple industries. Pharmaceutical companies require sophisticated TPR systems to optimize drug synthesis processes, while petrochemical manufacturers depend on accurate catalyst analysis to improve refining efficiency and reduce operational costs. The automotive sector's push toward cleaner emissions has intensified the need for advanced catalyst development, creating substantial demand for TPR equipment capable of handling complex gas mixture configurations.
Research institutions and universities represent a substantial market segment, with growing investments in materials science and catalysis research programs. These academic facilities require versatile TPR systems that can accommodate diverse experimental protocols and gas mixture compositions. Government-funded research initiatives focused on clean energy technologies and environmental remediation have further expanded this market segment, particularly in regions with strong environmental regulations.
Industrial catalyst manufacturers constitute the largest market segment for advanced TPR solutions. These companies require high-throughput analysis capabilities to evaluate catalyst performance under various operating conditions. The ability to precisely control gas mixture compositions during TPR experiments has become critical for developing next-generation catalysts for hydrogen production, carbon capture, and renewable fuel synthesis applications.
The market demand is increasingly shifting toward automated TPR systems with integrated gas mixing capabilities. End users seek solutions that can automatically blend multiple gases with precise concentration control, reducing manual intervention and improving experimental reproducibility. This trend has created opportunities for suppliers offering comprehensive gas analysis platforms rather than standalone TPR instruments.
Emerging markets in Asia-Pacific regions show particularly strong growth potential, driven by expanding chemical processing industries and increasing research investments. Local manufacturers in these regions are seeking cost-effective TPR solutions that maintain analytical precision while offering flexible gas mixture configurations to support diverse application requirements.
The market also demonstrates growing demand for TPR systems capable of handling corrosive or hazardous gas mixtures safely. Industries working with sulfur-containing compounds or other reactive species require specialized equipment with enhanced safety features and corrosion-resistant components, creating a premium market segment for advanced TPR solutions.
Research institutions and universities represent a substantial market segment, with growing investments in materials science and catalysis research programs. These academic facilities require versatile TPR systems that can accommodate diverse experimental protocols and gas mixture compositions. Government-funded research initiatives focused on clean energy technologies and environmental remediation have further expanded this market segment, particularly in regions with strong environmental regulations.
Industrial catalyst manufacturers constitute the largest market segment for advanced TPR solutions. These companies require high-throughput analysis capabilities to evaluate catalyst performance under various operating conditions. The ability to precisely control gas mixture compositions during TPR experiments has become critical for developing next-generation catalysts for hydrogen production, carbon capture, and renewable fuel synthesis applications.
The market demand is increasingly shifting toward automated TPR systems with integrated gas mixing capabilities. End users seek solutions that can automatically blend multiple gases with precise concentration control, reducing manual intervention and improving experimental reproducibility. This trend has created opportunities for suppliers offering comprehensive gas analysis platforms rather than standalone TPR instruments.
Emerging markets in Asia-Pacific regions show particularly strong growth potential, driven by expanding chemical processing industries and increasing research investments. Local manufacturers in these regions are seeking cost-effective TPR solutions that maintain analytical precision while offering flexible gas mixture configurations to support diverse application requirements.
The market also demonstrates growing demand for TPR systems capable of handling corrosive or hazardous gas mixtures safely. Industries working with sulfur-containing compounds or other reactive species require specialized equipment with enhanced safety features and corrosion-resistant components, creating a premium market segment for advanced TPR solutions.
Current State and Challenges in TPR Gas Mixture Design
Temperature Programmed Reduction (TPR) experiments have become increasingly sophisticated in recent decades, yet the selection of appropriate gas mixtures remains a critical challenge that significantly impacts experimental outcomes. Current methodologies for gas mixture design in TPR applications often rely on empirical approaches rather than systematic optimization frameworks, leading to inconsistent results across different research groups and industrial applications.
The predominant approach in contemporary TPR gas mixture selection involves using hydrogen-containing reducing atmospheres, typically comprising 5-10% H2 in an inert carrier gas such as argon or nitrogen. However, this standardized approach fails to account for the diverse nature of materials being analyzed and the specific reduction mechanisms involved. Many researchers continue to employ generic gas compositions without considering the unique characteristics of their target materials, resulting in suboptimal experimental conditions.
One of the most significant challenges facing the field is the lack of standardized protocols for gas mixture optimization. Different laboratories often use varying concentrations of reducing agents, leading to difficulties in reproducing results and comparing data across studies. The absence of comprehensive databases correlating material properties with optimal gas compositions further complicates the selection process, forcing researchers to rely on trial-and-error methodologies.
The complexity of multi-component systems presents another substantial challenge in current TPR gas mixture design. When analyzing complex materials containing multiple reducible species, determining the appropriate gas composition becomes increasingly difficult. Traditional single-component optimization approaches prove inadequate for these systems, as interactions between different reducible phases can significantly alter the required experimental conditions.
Current analytical capabilities also present limitations in real-time gas composition monitoring during TPR experiments. While mass spectrometry and thermal conductivity detection provide valuable insights, the dynamic nature of gas-solid interactions during temperature ramping creates challenges in maintaining optimal gas compositions throughout the experimental duration. This temporal variability in optimal conditions is poorly addressed by existing static gas mixture selection approaches.
The economic considerations of gas mixture selection represent an often-overlooked challenge in current practices. High-purity specialty gases and custom gas mixtures can significantly increase experimental costs, particularly for routine analytical applications. Balancing analytical performance with economic feasibility remains a persistent challenge, especially in industrial settings where cost-effectiveness is paramount.
Furthermore, safety considerations in gas mixture design have become increasingly important as TPR applications expand to include more reactive and potentially hazardous materials. Current guidelines for safe gas mixture selection are often inadequate for emerging applications, creating potential risks in laboratory environments and limiting the scope of materials that can be safely analyzed using conventional TPR methodologies.
The predominant approach in contemporary TPR gas mixture selection involves using hydrogen-containing reducing atmospheres, typically comprising 5-10% H2 in an inert carrier gas such as argon or nitrogen. However, this standardized approach fails to account for the diverse nature of materials being analyzed and the specific reduction mechanisms involved. Many researchers continue to employ generic gas compositions without considering the unique characteristics of their target materials, resulting in suboptimal experimental conditions.
One of the most significant challenges facing the field is the lack of standardized protocols for gas mixture optimization. Different laboratories often use varying concentrations of reducing agents, leading to difficulties in reproducing results and comparing data across studies. The absence of comprehensive databases correlating material properties with optimal gas compositions further complicates the selection process, forcing researchers to rely on trial-and-error methodologies.
The complexity of multi-component systems presents another substantial challenge in current TPR gas mixture design. When analyzing complex materials containing multiple reducible species, determining the appropriate gas composition becomes increasingly difficult. Traditional single-component optimization approaches prove inadequate for these systems, as interactions between different reducible phases can significantly alter the required experimental conditions.
Current analytical capabilities also present limitations in real-time gas composition monitoring during TPR experiments. While mass spectrometry and thermal conductivity detection provide valuable insights, the dynamic nature of gas-solid interactions during temperature ramping creates challenges in maintaining optimal gas compositions throughout the experimental duration. This temporal variability in optimal conditions is poorly addressed by existing static gas mixture selection approaches.
The economic considerations of gas mixture selection represent an often-overlooked challenge in current practices. High-purity specialty gases and custom gas mixtures can significantly increase experimental costs, particularly for routine analytical applications. Balancing analytical performance with economic feasibility remains a persistent challenge, especially in industrial settings where cost-effectiveness is paramount.
Furthermore, safety considerations in gas mixture design have become increasingly important as TPR applications expand to include more reactive and potentially hazardous materials. Current guidelines for safe gas mixture selection are often inadequate for emerging applications, creating potential risks in laboratory environments and limiting the scope of materials that can be safely analyzed using conventional TPR methodologies.
Existing Gas Mixture Strategies for TPR Applications
01 Gas mixture compositions for industrial applications
Gas mixtures can be formulated with specific compositions for various industrial applications. These mixtures typically combine different gases in precise ratios to achieve desired properties such as reactivity, stability, or specific chemical characteristics. The compositions may include inert gases, reactive gases, or combinations thereof to meet specific industrial requirements. The formulation process involves careful selection and blending of component gases to ensure optimal performance in target applications.- Gas mixture preparation and blending methods: Methods and apparatus for preparing gas mixtures involve precise blending techniques to achieve desired compositions. These methods include controlled mixing of multiple gas components, proportional blending systems, and automated mixing processes. The preparation techniques ensure accurate ratios and homogeneous distribution of gases in the final mixture. Various blending mechanisms and control systems are employed to maintain consistency and quality of the gas mixtures.
- Gas mixture composition for specific applications: Specialized gas mixture compositions are formulated for particular industrial, medical, or scientific applications. These compositions include specific ratios of gases tailored to meet performance requirements. The mixtures may contain inert gases, reactive gases, or combinations thereof, designed to optimize functionality in their intended use. Formulations are developed to provide desired properties such as stability, reactivity, or specific physical characteristics.
- Gas mixture storage and containment systems: Systems and containers for storing gas mixtures are designed to maintain mixture integrity and prevent separation or degradation. These systems include specialized vessels, pressure containers, and storage tanks with features to ensure long-term stability. The containment solutions address challenges such as pressure management, temperature control, and prevention of contamination. Various materials and construction methods are employed to ensure safe and effective storage.
- Gas mixture delivery and dispensing apparatus: Apparatus and methods for delivering and dispensing gas mixtures provide controlled release and distribution of gases. These systems include regulators, flow control devices, and dispensing mechanisms that ensure accurate delivery of gas mixtures. The delivery systems are designed to maintain mixture composition during transfer and use. Various configurations allow for precise control of flow rates and pressure during dispensing operations.
- Gas mixture analysis and quality control: Methods and systems for analyzing gas mixtures ensure composition accuracy and quality standards. These techniques include monitoring systems, analytical methods, and testing procedures to verify mixture composition. Quality control processes involve measurement of component concentrations, detection of impurities, and validation of mixture properties. Various analytical instruments and procedures are employed to maintain consistency and meet specifications.
02 Methods for preparing and storing gas mixtures
Various methods and apparatus have been developed for the preparation, mixing, and storage of gas mixtures. These methods focus on achieving homogeneous distribution of component gases, maintaining stability during storage, and preventing separation or degradation. Techniques may include controlled mixing processes, specialized storage containers, and monitoring systems to ensure mixture integrity. The methods address challenges related to pressure management, temperature control, and prevention of contamination during preparation and storage phases.Expand Specific Solutions03 Gas mixtures for welding and cutting operations
Specialized gas mixtures have been developed for welding, cutting, and metal processing applications. These mixtures are designed to provide optimal shielding, heat transfer, and arc stability during metalworking operations. The compositions are tailored to specific metals and processes, incorporating gases that enhance weld quality, reduce oxidation, and improve operational efficiency. Such mixtures may include combinations of inert and active gases in proportions optimized for particular welding techniques and materials.Expand Specific Solutions04 Calibration and standard gas mixtures
Gas mixtures are formulated as calibration standards and reference materials for analytical instruments and detection systems. These mixtures contain precisely known concentrations of specific gases, enabling accurate calibration of measurement devices. The preparation involves stringent quality control measures to ensure accuracy and traceability of component concentrations. Such standard mixtures are essential for environmental monitoring, industrial process control, and laboratory analysis applications where precise gas detection and measurement are required.Expand Specific Solutions05 Gas mixtures for medical and respiratory applications
Therapeutic and medical gas mixtures have been developed for respiratory therapy, anesthesia, and other healthcare applications. These mixtures are formulated to meet strict medical standards and provide specific therapeutic benefits. The compositions are designed to support respiration, deliver anesthetic agents, or provide other medical treatments. Safety, purity, and precise composition control are critical factors in the formulation of medical gas mixtures to ensure patient safety and treatment efficacy.Expand Specific Solutions
Key Players in TPR Equipment and Gas Supply Industry
The temperature programmed reduction (TPR) gas mixture selection field represents a mature analytical technique within the broader catalyst characterization market, currently valued at several billion dollars globally. The industry has reached technological maturity with established protocols and standardized equipment, though innovation continues in automation and data analysis. Major players span diverse sectors: petrochemical giants like China Petroleum & Chemical Corp. and ExxonMobil Technology & Engineering Co. drive industrial applications, while specialty chemical companies including Air Liquide SA and Clariant International AG provide essential gas supplies and catalysts. Research institutions such as the Institute of Process Engineering, Chinese Academy of Sciences, and Delft University of Technology advance fundamental understanding, while technology companies like Siemens AG and Robert Bosch GmbH develop sophisticated instrumentation. This competitive landscape reflects TPR's critical role across petroleum refining, chemical manufacturing, and materials research, with established methodologies being enhanced through digital integration and automated systems.
China Petroleum & Chemical Corp.
Technical Solution: Develops comprehensive gas mixture selection protocols for TPR experiments focusing on petrochemical catalyst characterization. Their approach involves systematic evaluation of H2/Ar ratios ranging from 5-20% for optimal reduction profiles, with specialized attention to sulfur-resistant catalysts used in refining processes. The company employs multi-stage gas switching techniques, starting with inert purging using high-purity nitrogen, followed by controlled H2/N2 mixtures for initial reduction phases, and concluding with pure hydrogen for complete reduction. Their methodology incorporates real-time mass spectrometry monitoring to track water formation and consumption patterns, enabling precise determination of metal dispersion and support interactions in industrial catalysts.
Strengths: Extensive industrial catalyst testing experience, robust analytical capabilities. Weaknesses: Limited focus on novel catalyst materials, primarily petroleum-oriented applications.
DuPont de Nemours, Inc.
Technical Solution: Implements advanced gas mixture optimization strategies for TPR analysis of specialty chemical catalysts, particularly focusing on precious metal and mixed oxide systems. Their protocol emphasizes the use of dilute hydrogen concentrations (2-10% H2 in Ar) to prevent sintering and ensure controlled reduction kinetics. DuPont's approach includes pre-treatment with oxygen-containing mixtures to establish consistent oxidation states, followed by systematic reduction using calibrated gas blends. They utilize sophisticated flow control systems with mass flow controllers to maintain precise gas composition throughout temperature ramping cycles, typically from ambient to 900°C. The methodology incorporates baseline correction techniques and employs reference materials for quantitative hydrogen consumption measurements.
Strengths: High precision analytical methods, extensive materials science expertise. Weaknesses: Higher operational costs, complex instrumentation requirements.
Core Innovations in TPR Gas Composition Optimization
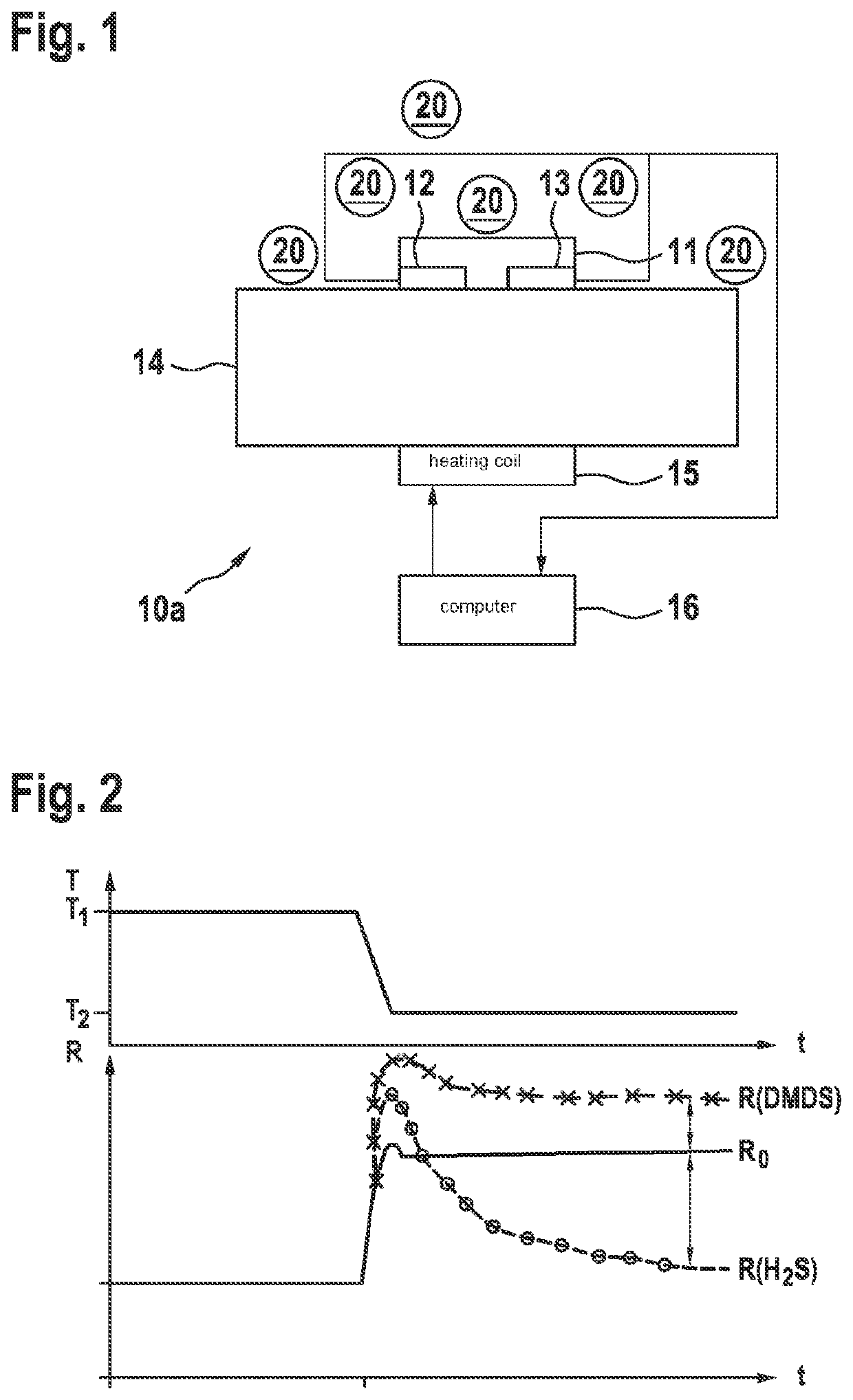
Methods for analyzing a gas mixture and gas sensor
PatentActiveUS20220034835A1
Innovation
- A method involving a metal oxide layer, preferably with tin oxide and doped with palladium or platinum, where electrical resistance measurements are taken at multiple temperatures, including temperature pulses, to differentiate and quantify components in a gas mixture, using a binary or dual temperature pulse approach to avoid compensation effects.
Method and apparatus for reducing a nitrogen oxide
PatentInactiveUS20040126286A1
Innovation
- A method and apparatus utilizing gas analyzers to determine the compositional content of nitrogen oxide-containing gas mixtures, allowing for precise calculation and control of reducing agent injection, including the use of catalysts for selective catalytic reduction, to minimize unreacted reducing agent release.
Safety Standards for TPR Gas Handling and Operations
Temperature Programmed Reduction experiments involve handling potentially hazardous gas mixtures, making adherence to comprehensive safety standards paramount for laboratory operations. The primary safety concern stems from the use of hydrogen-containing reducing gases, which present significant fire and explosion risks when improperly managed. International safety standards, including OSHA regulations and NFPA codes, mandate specific protocols for hydrogen concentrations exceeding 4% by volume in air, as this approaches the lower explosive limit.
Gas handling infrastructure must comply with rigorous engineering standards to prevent catastrophic failures. All gas delivery systems require pressure relief valves, flame arrestors, and emergency shut-off mechanisms positioned at strategic locations throughout the experimental setup. Stainless steel tubing with appropriate pressure ratings must be employed, with regular leak detection protocols using hydrogen-sensitive detectors capable of detecting concentrations as low as 25 ppm.
Ventilation systems represent a critical safety component, requiring minimum air exchange rates of 6-12 changes per hour in TPR laboratories. Emergency ventilation systems must activate automatically upon detection of gas leaks, with backup power systems ensuring continuous operation during power failures. Gas detection systems should incorporate multiple sensor types, including catalytic bead sensors for hydrogen and infrared sensors for carbon monoxide monitoring.
Personnel safety protocols mandate comprehensive training programs covering gas properties, emergency procedures, and proper equipment operation. Personal protective equipment requirements include flame-resistant laboratory coats, safety glasses, and emergency breathing apparatus readily accessible within the laboratory environment. Regular safety audits and equipment calibration schedules ensure ongoing compliance with evolving safety standards.
Storage and handling procedures for gas cylinders must follow DOT regulations for compressed gas transportation and storage. Cylinder securing systems, proper labeling protocols, and segregation requirements for incompatible gases form essential components of comprehensive safety management. Emergency response procedures must include clear evacuation routes, communication protocols, and coordination with local emergency services familiar with laboratory-specific hazards.
Gas handling infrastructure must comply with rigorous engineering standards to prevent catastrophic failures. All gas delivery systems require pressure relief valves, flame arrestors, and emergency shut-off mechanisms positioned at strategic locations throughout the experimental setup. Stainless steel tubing with appropriate pressure ratings must be employed, with regular leak detection protocols using hydrogen-sensitive detectors capable of detecting concentrations as low as 25 ppm.
Ventilation systems represent a critical safety component, requiring minimum air exchange rates of 6-12 changes per hour in TPR laboratories. Emergency ventilation systems must activate automatically upon detection of gas leaks, with backup power systems ensuring continuous operation during power failures. Gas detection systems should incorporate multiple sensor types, including catalytic bead sensors for hydrogen and infrared sensors for carbon monoxide monitoring.
Personnel safety protocols mandate comprehensive training programs covering gas properties, emergency procedures, and proper equipment operation. Personal protective equipment requirements include flame-resistant laboratory coats, safety glasses, and emergency breathing apparatus readily accessible within the laboratory environment. Regular safety audits and equipment calibration schedules ensure ongoing compliance with evolving safety standards.
Storage and handling procedures for gas cylinders must follow DOT regulations for compressed gas transportation and storage. Cylinder securing systems, proper labeling protocols, and segregation requirements for incompatible gases form essential components of comprehensive safety management. Emergency response procedures must include clear evacuation routes, communication protocols, and coordination with local emergency services familiar with laboratory-specific hazards.
Environmental Impact of TPR Gas Mixture Disposal
The environmental implications of TPR gas mixture disposal represent a critical consideration in laboratory operations and industrial applications. Traditional TPR experiments typically utilize hydrogen-containing gas mixtures, which upon completion generate waste streams containing unreacted hydrogen, water vapor, and various reduction byproducts. The disposal of these gaseous effluents requires careful environmental assessment, as improper handling can contribute to atmospheric emissions and potential safety hazards.
Hydrogen-rich waste streams pose unique environmental challenges due to their high flammability and potential for atmospheric release. When vented directly to atmosphere, these gases can contribute to local air quality degradation and represent inefficient resource utilization. The environmental impact varies significantly depending on the carrier gas composition, with helium-based mixtures presenting resource depletion concerns due to helium's non-renewable nature, while nitrogen-based systems offer more sustainable alternatives.
Water vapor generation during TPR processes, while seemingly benign, can contribute to localized humidity increases in laboratory environments and may carry trace contaminants from the reduction process. The condensation and subsequent disposal of this water requires consideration of potential dissolved species that may have environmental implications if released into wastewater systems without proper treatment.
Industrial-scale TPR applications amplify these environmental concerns significantly. Large-volume gas consumption and waste generation necessitate comprehensive disposal strategies that often involve gas recovery systems, catalytic combustion units, or specialized waste treatment facilities. The carbon footprint associated with gas production, transportation, and disposal becomes a substantial factor in overall process sustainability assessment.
Emerging disposal technologies focus on gas recovery and recycling systems that can capture and purify unreacted hydrogen for reuse, significantly reducing both environmental impact and operational costs. Advanced scrubbing systems and catalytic treatment methods are being developed to neutralize harmful byproducts before atmospheric release. These innovations represent a shift toward circular economy principles in TPR methodology, where waste minimization and resource recovery become integral components of experimental design and industrial implementation strategies.
Hydrogen-rich waste streams pose unique environmental challenges due to their high flammability and potential for atmospheric release. When vented directly to atmosphere, these gases can contribute to local air quality degradation and represent inefficient resource utilization. The environmental impact varies significantly depending on the carrier gas composition, with helium-based mixtures presenting resource depletion concerns due to helium's non-renewable nature, while nitrogen-based systems offer more sustainable alternatives.
Water vapor generation during TPR processes, while seemingly benign, can contribute to localized humidity increases in laboratory environments and may carry trace contaminants from the reduction process. The condensation and subsequent disposal of this water requires consideration of potential dissolved species that may have environmental implications if released into wastewater systems without proper treatment.
Industrial-scale TPR applications amplify these environmental concerns significantly. Large-volume gas consumption and waste generation necessitate comprehensive disposal strategies that often involve gas recovery systems, catalytic combustion units, or specialized waste treatment facilities. The carbon footprint associated with gas production, transportation, and disposal becomes a substantial factor in overall process sustainability assessment.
Emerging disposal technologies focus on gas recovery and recycling systems that can capture and purify unreacted hydrogen for reuse, significantly reducing both environmental impact and operational costs. Advanced scrubbing systems and catalytic treatment methods are being developed to neutralize harmful byproducts before atmospheric release. These innovations represent a shift toward circular economy principles in TPR methodology, where waste minimization and resource recovery become integral components of experimental design and industrial implementation strategies.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!