How to Evaluate Post-Sonication Turbidity for Sample Clarity
MAR 11, 202610 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Sonication Turbidity Assessment Background and Objectives
Sonication has emerged as a critical technique in various analytical and preparative processes, particularly in pharmaceutical, biotechnology, and materials science applications. The process involves applying ultrasonic energy to samples to achieve dispersion, dissolution, or extraction objectives. However, the effectiveness of sonication procedures is fundamentally dependent on achieving optimal sample clarity, which directly correlates with the intended analytical or preparative outcomes.
The evolution of sonication technology has progressed from basic ultrasonic baths to sophisticated probe sonicators with precise control parameters. Early applications focused primarily on simple dispersion tasks, but modern requirements demand more nuanced approaches to sample preparation where clarity assessment becomes paramount. This technological advancement has created an increasing need for standardized methods to evaluate post-sonication sample quality.
Turbidity measurement represents the most practical and widely adopted approach for assessing sample clarity following sonication procedures. The fundamental principle relies on quantifying light scattering caused by suspended particles or undissolved materials remaining in the sample matrix. This optical measurement technique provides immediate feedback on sonication effectiveness and enables process optimization through systematic evaluation protocols.
Current industry practices reveal significant variability in turbidity assessment methodologies, leading to inconsistent results and compromised reproducibility across different laboratories and applications. The absence of standardized evaluation protocols has created challenges in establishing reliable quality control measures and comparing results between different research groups or manufacturing facilities.
The primary objective of developing comprehensive turbidity evaluation methods centers on establishing quantitative criteria for sample clarity assessment. This involves defining acceptable turbidity thresholds for specific applications, implementing standardized measurement protocols, and creating decision-making frameworks for process optimization. These objectives aim to transform subjective visual assessments into objective, reproducible measurements that can support regulatory compliance and quality assurance requirements.
Secondary objectives include developing correlation models between turbidity measurements and downstream process performance, establishing time-dependent evaluation protocols to account for sample stability, and creating automated assessment systems that minimize operator variability. These goals collectively support the broader aim of enhancing sonication process reliability and ensuring consistent sample preparation outcomes across diverse applications and operational environments.
The evolution of sonication technology has progressed from basic ultrasonic baths to sophisticated probe sonicators with precise control parameters. Early applications focused primarily on simple dispersion tasks, but modern requirements demand more nuanced approaches to sample preparation where clarity assessment becomes paramount. This technological advancement has created an increasing need for standardized methods to evaluate post-sonication sample quality.
Turbidity measurement represents the most practical and widely adopted approach for assessing sample clarity following sonication procedures. The fundamental principle relies on quantifying light scattering caused by suspended particles or undissolved materials remaining in the sample matrix. This optical measurement technique provides immediate feedback on sonication effectiveness and enables process optimization through systematic evaluation protocols.
Current industry practices reveal significant variability in turbidity assessment methodologies, leading to inconsistent results and compromised reproducibility across different laboratories and applications. The absence of standardized evaluation protocols has created challenges in establishing reliable quality control measures and comparing results between different research groups or manufacturing facilities.
The primary objective of developing comprehensive turbidity evaluation methods centers on establishing quantitative criteria for sample clarity assessment. This involves defining acceptable turbidity thresholds for specific applications, implementing standardized measurement protocols, and creating decision-making frameworks for process optimization. These objectives aim to transform subjective visual assessments into objective, reproducible measurements that can support regulatory compliance and quality assurance requirements.
Secondary objectives include developing correlation models between turbidity measurements and downstream process performance, establishing time-dependent evaluation protocols to account for sample stability, and creating automated assessment systems that minimize operator variability. These goals collectively support the broader aim of enhancing sonication process reliability and ensuring consistent sample preparation outcomes across diverse applications and operational environments.
Market Demand for Post-Sonication Sample Analysis
The pharmaceutical and biotechnology industries represent the primary market drivers for post-sonication sample analysis technologies. These sectors require precise turbidity evaluation to ensure drug formulation consistency, particularly in the development of nanoparticle-based therapeutics and vaccine preparations. Regulatory compliance demands from agencies such as FDA and EMA necessitate robust analytical methods for demonstrating product quality and batch-to-batch consistency.
Laboratory automation trends are significantly expanding market opportunities for integrated turbidity measurement systems. High-throughput screening facilities and contract research organizations increasingly seek automated solutions that can process multiple samples simultaneously while maintaining measurement accuracy. This demand is particularly pronounced in pharmaceutical discovery phases where thousands of formulations require rapid clarity assessment.
The food and beverage industry presents substantial growth potential for post-sonication turbidity analysis applications. Quality control processes for emulsions, suspensions, and clarified products require reliable measurement techniques to ensure consumer safety and product consistency. Dairy processing, beverage clarification, and protein extraction operations specifically benefit from advanced turbidity evaluation methods.
Environmental monitoring sectors demonstrate increasing adoption of sonication-based sample preparation followed by turbidity analysis. Water treatment facilities, environmental consulting firms, and regulatory agencies utilize these techniques for assessing particulate contamination levels and treatment efficacy. The growing emphasis on water quality standards globally drives sustained demand for accurate turbidity measurement technologies.
Academic and research institutions constitute a stable market segment with consistent demand for advanced analytical capabilities. Universities, government research laboratories, and private research facilities require sophisticated turbidity evaluation systems for materials science, nanotechnology research, and fundamental studies of particle behavior in various media.
Emerging markets in Asia-Pacific regions show accelerated adoption rates driven by expanding pharmaceutical manufacturing capabilities and stricter quality control requirements. Local regulatory harmonization with international standards creates additional demand for validated analytical methods including post-sonication turbidity evaluation systems.
The market landscape indicates strong preference for integrated solutions that combine sonication capabilities with real-time turbidity monitoring. End users increasingly value systems offering automated data collection, statistical analysis capabilities, and regulatory compliance documentation features to streamline their analytical workflows and reduce operational complexity.
Laboratory automation trends are significantly expanding market opportunities for integrated turbidity measurement systems. High-throughput screening facilities and contract research organizations increasingly seek automated solutions that can process multiple samples simultaneously while maintaining measurement accuracy. This demand is particularly pronounced in pharmaceutical discovery phases where thousands of formulations require rapid clarity assessment.
The food and beverage industry presents substantial growth potential for post-sonication turbidity analysis applications. Quality control processes for emulsions, suspensions, and clarified products require reliable measurement techniques to ensure consumer safety and product consistency. Dairy processing, beverage clarification, and protein extraction operations specifically benefit from advanced turbidity evaluation methods.
Environmental monitoring sectors demonstrate increasing adoption of sonication-based sample preparation followed by turbidity analysis. Water treatment facilities, environmental consulting firms, and regulatory agencies utilize these techniques for assessing particulate contamination levels and treatment efficacy. The growing emphasis on water quality standards globally drives sustained demand for accurate turbidity measurement technologies.
Academic and research institutions constitute a stable market segment with consistent demand for advanced analytical capabilities. Universities, government research laboratories, and private research facilities require sophisticated turbidity evaluation systems for materials science, nanotechnology research, and fundamental studies of particle behavior in various media.
Emerging markets in Asia-Pacific regions show accelerated adoption rates driven by expanding pharmaceutical manufacturing capabilities and stricter quality control requirements. Local regulatory harmonization with international standards creates additional demand for validated analytical methods including post-sonication turbidity evaluation systems.
The market landscape indicates strong preference for integrated solutions that combine sonication capabilities with real-time turbidity monitoring. End users increasingly value systems offering automated data collection, statistical analysis capabilities, and regulatory compliance documentation features to streamline their analytical workflows and reduce operational complexity.
Current Turbidity Measurement Challenges and Limitations
Traditional turbidity measurement methods face significant limitations when applied to post-sonication sample evaluation. Conventional nephelometric turbidimeters, while effective for standard water quality assessments, struggle with the complex particle size distributions and dynamic settling behaviors characteristic of sonicated samples. These instruments typically operate at fixed wavelengths and detection angles, which may not capture the full spectrum of light scattering phenomena occurring in samples containing freshly dispersed nanoparticles and microparticles.
The temporal instability of post-sonication samples presents a critical measurement challenge. Particle aggregation and settling occur rapidly after sonication cessation, creating a narrow time window for accurate turbidity assessment. Standard measurement protocols often require sample equilibration periods that are incompatible with the dynamic nature of sonicated dispersions. This temporal constraint forces operators to choose between measurement precision and sample representativeness.
Particle size heterogeneity in sonicated samples introduces substantial measurement artifacts. Different particle populations scatter light with varying intensities and angular distributions, leading to non-linear relationships between actual particle concentration and measured turbidity values. Submicron particles exhibit Rayleigh scattering behavior, while larger particles follow Mie scattering principles, creating complex interference patterns that conventional turbidimeters cannot adequately resolve.
Sample matrix effects significantly complicate turbidity interpretation in post-sonication applications. The presence of dissolved salts, surfactants, or organic compounds can alter particle surface properties and aggregation kinetics, directly impacting light scattering characteristics. These matrix interactions are particularly pronounced in biological and pharmaceutical samples where protein adsorption and ionic strength variations create unpredictable measurement responses.
Calibration standardization remains problematic across different sample types and sonication conditions. Traditional formazin standards poorly represent the optical properties of sonicated dispersions, leading to systematic measurement errors. The lack of universally accepted reference materials for post-sonication turbidity assessment hampers inter-laboratory comparability and method validation efforts.
Instrument sensitivity limitations become apparent when evaluating highly clarified samples or detecting subtle changes in particle distributions. Many commercial turbidimeters lack sufficient resolution to distinguish between different degrees of sample clarity achieved through optimized sonication protocols, limiting their utility for process optimization and quality control applications.
The temporal instability of post-sonication samples presents a critical measurement challenge. Particle aggregation and settling occur rapidly after sonication cessation, creating a narrow time window for accurate turbidity assessment. Standard measurement protocols often require sample equilibration periods that are incompatible with the dynamic nature of sonicated dispersions. This temporal constraint forces operators to choose between measurement precision and sample representativeness.
Particle size heterogeneity in sonicated samples introduces substantial measurement artifacts. Different particle populations scatter light with varying intensities and angular distributions, leading to non-linear relationships between actual particle concentration and measured turbidity values. Submicron particles exhibit Rayleigh scattering behavior, while larger particles follow Mie scattering principles, creating complex interference patterns that conventional turbidimeters cannot adequately resolve.
Sample matrix effects significantly complicate turbidity interpretation in post-sonication applications. The presence of dissolved salts, surfactants, or organic compounds can alter particle surface properties and aggregation kinetics, directly impacting light scattering characteristics. These matrix interactions are particularly pronounced in biological and pharmaceutical samples where protein adsorption and ionic strength variations create unpredictable measurement responses.
Calibration standardization remains problematic across different sample types and sonication conditions. Traditional formazin standards poorly represent the optical properties of sonicated dispersions, leading to systematic measurement errors. The lack of universally accepted reference materials for post-sonication turbidity assessment hampers inter-laboratory comparability and method validation efforts.
Instrument sensitivity limitations become apparent when evaluating highly clarified samples or detecting subtle changes in particle distributions. Many commercial turbidimeters lack sufficient resolution to distinguish between different degrees of sample clarity achieved through optimized sonication protocols, limiting their utility for process optimization and quality control applications.
Existing Turbidity Measurement Solutions Post-Sonication
01 Sonication methods for particle size reduction and dispersion
Sonication techniques are employed to reduce particle size and improve dispersion of suspended materials in liquid samples. The ultrasonic energy breaks down aggregates and creates more uniform particle distributions, which directly impacts the turbidity and clarity of samples. Various sonication parameters such as frequency, amplitude, and duration can be optimized to achieve desired clarity levels in different sample types.- Sonication methods for improving sample clarity: Various sonication techniques and parameters can be employed to enhance the clarity of turbid samples. The application of ultrasonic energy helps break down particles and aggregates that cause turbidity, resulting in clearer samples. Different sonication durations, frequencies, and power levels can be optimized depending on the sample type to achieve maximum clarity improvement.
- Turbidity measurement and monitoring systems: Advanced measurement systems and devices are used to assess sample clarity before and after sonication treatment. These systems employ optical sensors, light scattering techniques, and automated monitoring to quantify turbidity levels. Real-time monitoring capabilities allow for precise control of the sonication process to achieve desired clarity levels.
- Filtration and separation techniques post-sonication: Following sonication treatment, various filtration and separation methods can be applied to further enhance sample clarity. These techniques include membrane filtration, centrifugation, and sedimentation processes that remove residual particles and debris. The combination of sonication with subsequent filtration steps provides superior clarity compared to either method alone.
- Chemical additives and treatment agents for clarity enhancement: Specific chemical agents and additives can be introduced to samples in conjunction with sonication to improve clarity outcomes. These substances may include dispersants, surfactants, or clarifying agents that work synergistically with ultrasonic treatment. The chemical composition and concentration of these additives are optimized to maximize particle dispersion and prevent reaggregation.
- Application-specific sonication protocols for different sample types: Customized sonication protocols have been developed for various sample matrices including biological fluids, pharmaceutical formulations, and industrial suspensions. Each application requires specific parameter adjustments to address unique turbidity challenges while preserving sample integrity. These protocols consider factors such as sample volume, viscosity, and the nature of suspended particles.
02 Turbidity measurement and monitoring systems
Advanced turbidity measurement systems utilize optical sensors and detection methods to assess sample clarity after sonication treatment. These systems can provide real-time monitoring of turbidity changes and quantify the effectiveness of sonication processes. Measurement techniques include nephelometric methods, light scattering analysis, and spectrophotometric approaches to evaluate particle concentration and sample transparency.Expand Specific Solutions03 Filtration and clarification post-treatment methods
Following sonication, additional filtration and clarification techniques can be applied to further improve sample clarity. These methods include membrane filtration, centrifugation, and settling processes that remove residual particles and aggregates. The combination of sonication with subsequent clarification steps enhances overall sample quality and reduces turbidity to acceptable levels for various analytical applications.Expand Specific Solutions04 Chemical additives and stabilizers for clarity enhancement
Chemical agents and stabilizing compounds can be incorporated into samples to maintain clarity after sonication treatment. These additives prevent re-aggregation of particles, modify surface properties, and improve long-term stability of dispersions. Various formulations including surfactants, dispersants, and pH modifiers work synergistically with sonication to achieve and maintain optimal sample clarity.Expand Specific Solutions05 Quality control and validation protocols for clarity assessment
Standardized protocols and validation methods are essential for ensuring consistent sample clarity results after sonication. These include establishing baseline turbidity measurements, defining acceptance criteria, and implementing quality control procedures. Documentation of sonication parameters, measurement conditions, and sample handling procedures ensures reproducibility and reliability of clarity assessments across different batches and applications.Expand Specific Solutions
Key Players in Sonication Equipment and Turbidity Analysis
The post-sonication turbidity evaluation market represents an emerging niche within the broader analytical instrumentation sector, currently in its early development stage with limited market size but growing demand across pharmaceutical, biotechnology, and materials science applications. The competitive landscape is fragmented, featuring diverse players ranging from established pharmaceutical giants like Takeda Pharmaceutical and Samsung Electronics to specialized research institutions including The Rockefeller University, Purdue Research Foundation, and Ocean University of China. Technology maturity varies significantly across participants, with companies like Boehringer Mannheim and Process Instruments demonstrating advanced analytical capabilities, while newer entrants such as Kashiv Biosciences and Jiangsu Gaiya Environmental Science & Technology are developing innovative approaches. The field benefits from strong academic-industry collaboration, evidenced by contributions from Colorado School of Mines and Naval Research Laboratory, suggesting robust foundational research supporting commercial applications. Overall, the market shows promising growth potential despite its nascent stage.
Dow Global Technologies LLC
Technical Solution: Dow has developed advanced turbidity measurement systems that utilize multi-angle light scattering technology for post-sonication sample analysis. Their approach combines nephelometric and turbidimetric principles to provide accurate turbidity readings even in challenging sample matrices. The system incorporates automated sample handling with real-time monitoring capabilities, allowing for continuous assessment of sample clarity changes following ultrasonic treatment. Their proprietary algorithms compensate for particle size distribution variations and optical interference, ensuring reliable measurements across different sample types and concentrations.
Strengths: Robust measurement technology with high accuracy and automated processing capabilities. Weaknesses: High equipment costs and complexity may limit accessibility for smaller laboratories.
Process Instruments (UK) Ltd.
Technical Solution: Process Instruments specializes in online turbidity monitoring solutions specifically designed for post-sonication applications. Their technology employs dual-beam optical systems with infrared light sources to minimize color interference and provide stable measurements. The instruments feature self-cleaning mechanisms and automatic calibration protocols to maintain measurement accuracy over extended periods. Their systems integrate seamlessly with laboratory information management systems (LIMS) and provide real-time data logging with statistical analysis capabilities for trend monitoring and quality control purposes.
Strengths: Specialized expertise in turbidity measurement with reliable online monitoring capabilities. Weaknesses: Limited to specific application ranges and may require frequent maintenance in harsh environments.
Core Innovations in Turbidity Assessment Technologies
Device for determining end point of sample pretreatment through real-time turbidity measurement
PatentWO2025206884A1
Innovation
- A device that measures turbidity in real-time during sample pretreatment, using a control panel to determine the end point based on turbidity change rates, ensuring the pretreatment process is optimized for accurate TOC measurement by terminating when the turbidity change rate is within a certain level, typically 30%, and includes features for ultrasonic irradiation and alkaline elution to enhance sample preparation.
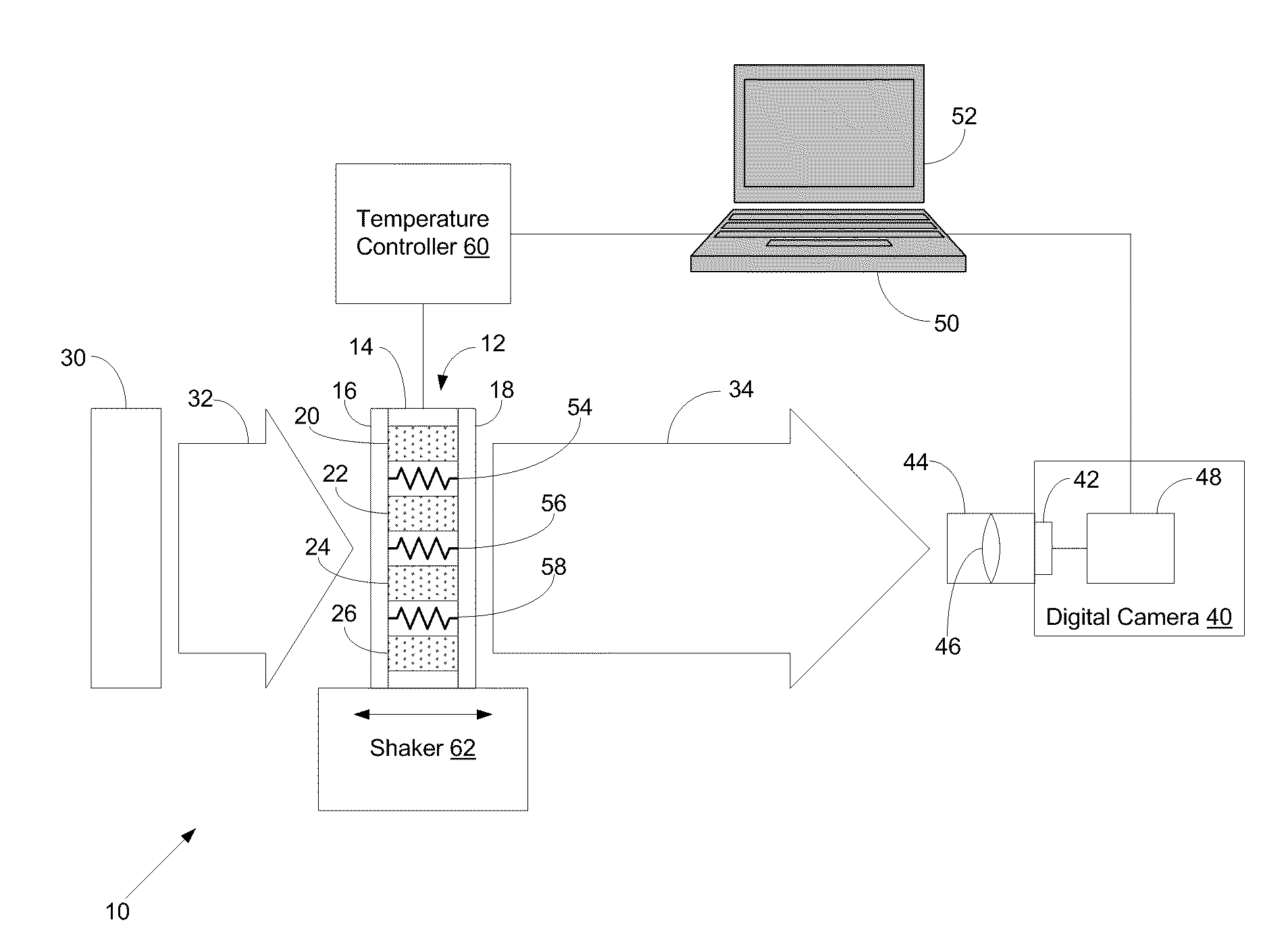
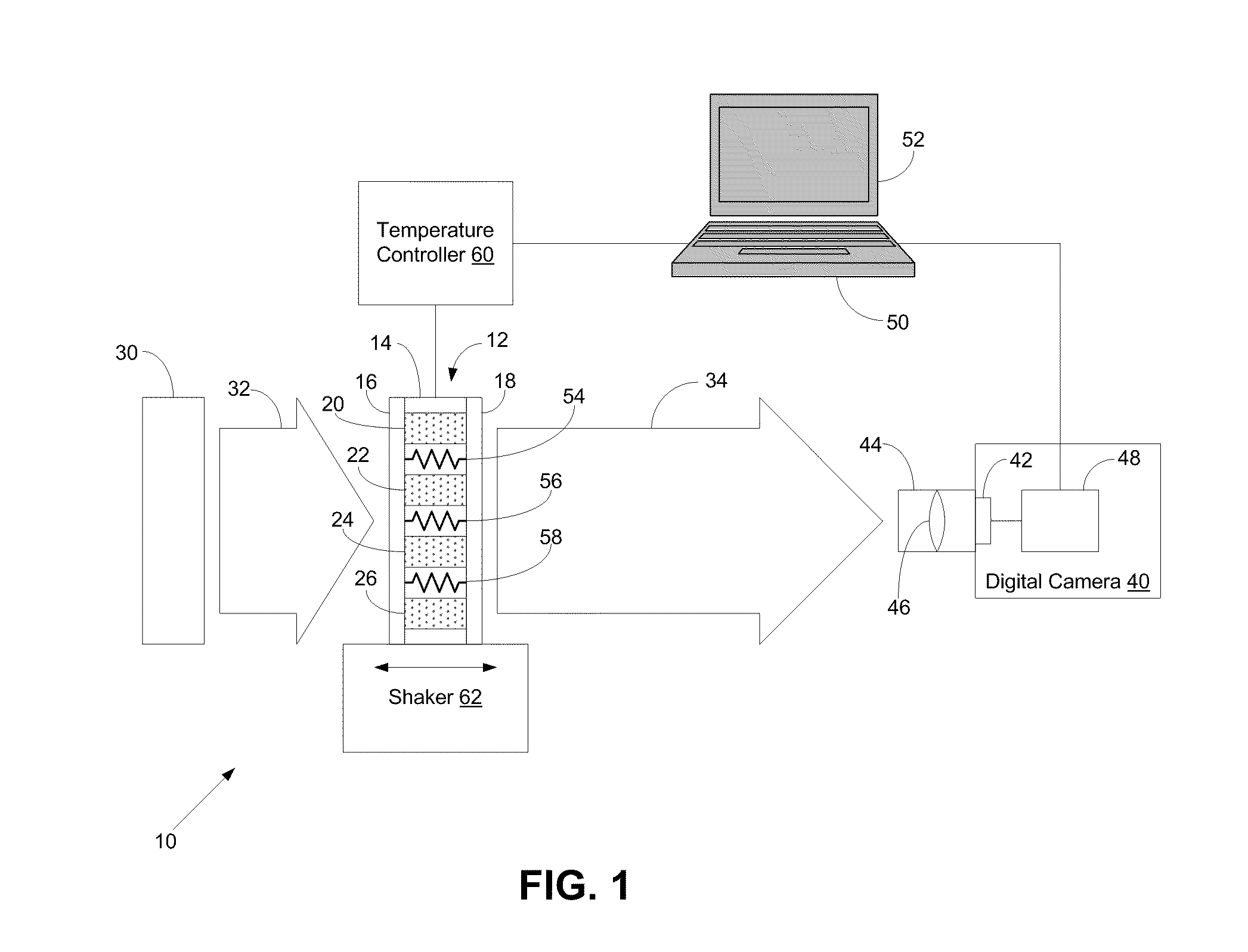
Systems and Methods for High-Throughput Turbidity Measurements
PatentInactiveUS20100110220A1
Innovation
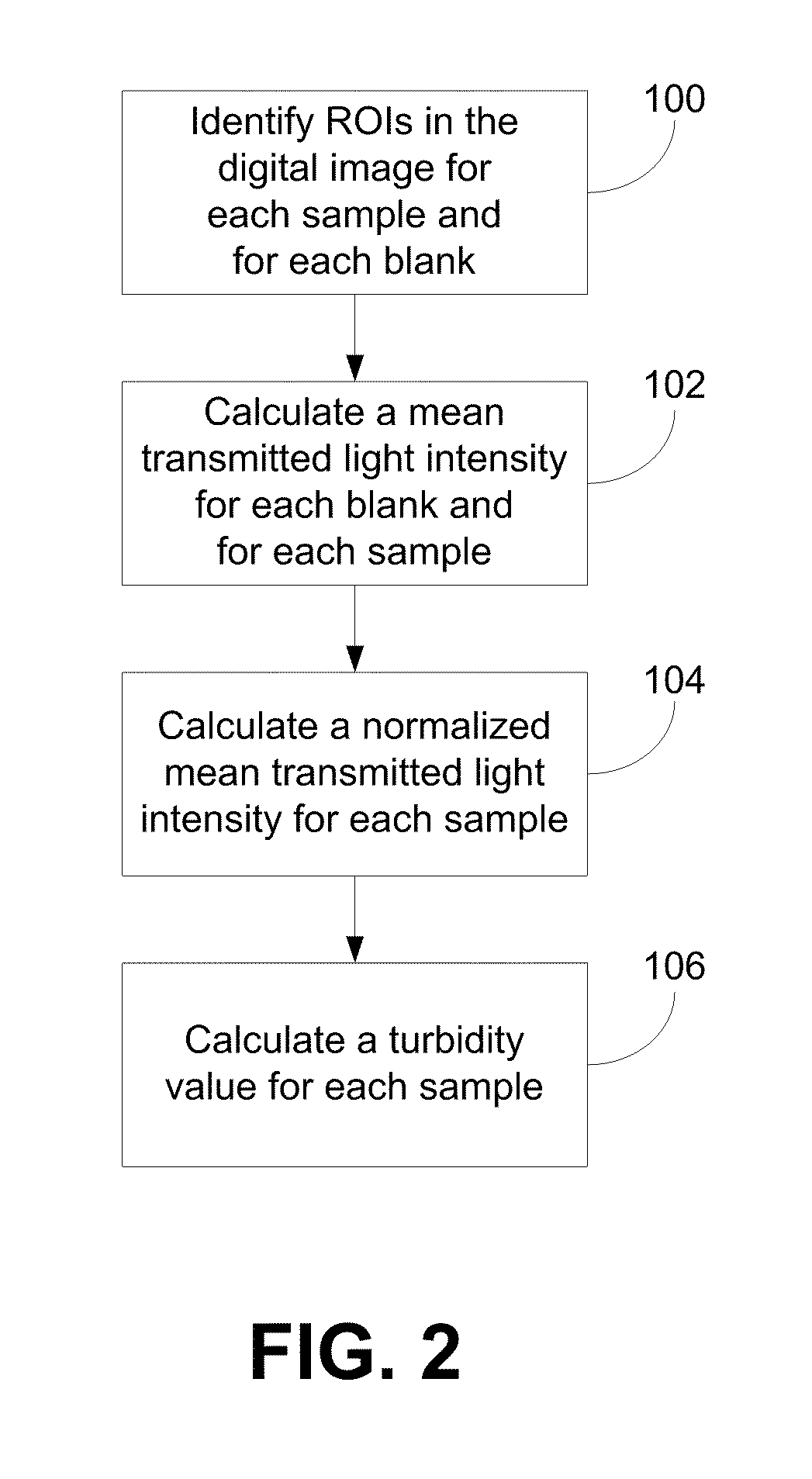
- A system comprising a sample assembly, a light source, a digital camera, and a temperature controller that allows for simultaneous turbidity measurements across multiple samples and blanks, with temperature control and image analysis to determine turbidity values and other parameters in a high-throughput manner.
Quality Standards for Laboratory Sample Processing
Quality standards for laboratory sample processing in post-sonication turbidity evaluation represent a critical framework ensuring reproducible and accurate measurements of sample clarity. These standards encompass multiple dimensions including measurement protocols, equipment calibration requirements, environmental controls, and documentation procedures that collectively guarantee the reliability of turbidity assessments following ultrasonic treatment.
The foundation of quality standards begins with establishing standardized measurement conditions. Temperature control within ±2°C of the specified measurement temperature is essential, as thermal variations significantly impact particle behavior and light scattering properties. Sample containers must meet specific optical requirements, including uniform wall thickness and absence of scratches or contaminants that could interfere with turbidity readings. The measurement environment requires controlled lighting conditions to prevent external light interference with nephelometric or spectrophotometric measurements.
Calibration protocols form another cornerstone of quality standards. Primary turbidity standards, typically formazin-based solutions with certified turbidity values, must be prepared according to established procedures and verified against secondary standards. Calibration verification should occur at minimum daily, with full calibration curves generated weekly or following any significant equipment maintenance. The acceptable deviation from certified values typically ranges within ±2% for high-precision applications.
Sample handling procedures within quality standards address critical factors affecting measurement accuracy. Post-sonication samples require standardized settling times, typically 2-5 minutes, to allow transient cavitation effects to dissipate while preventing significant particle sedimentation. Sample volume requirements, mixing protocols, and measurement timing must be strictly controlled to ensure consistency across different operators and measurement sessions.
Documentation and traceability requirements establish comprehensive record-keeping systems tracking sample identification, processing parameters, measurement conditions, and results. Quality control charts monitoring blank measurements, standard recoveries, and duplicate sample precision provide ongoing verification of system performance. Acceptance criteria for measurement precision, typically requiring relative standard deviations below 5% for replicate measurements, ensure data quality meets analytical requirements.
Regular proficiency testing and method validation components verify that laboratory procedures consistently meet established performance criteria. These standards also define corrective action protocols when measurements fall outside acceptable ranges, including equipment troubleshooting procedures, recalibration requirements, and sample reprocessing criteria. Integration with broader laboratory quality management systems ensures that post-sonication turbidity evaluation maintains the highest standards of analytical reliability and regulatory compliance.
The foundation of quality standards begins with establishing standardized measurement conditions. Temperature control within ±2°C of the specified measurement temperature is essential, as thermal variations significantly impact particle behavior and light scattering properties. Sample containers must meet specific optical requirements, including uniform wall thickness and absence of scratches or contaminants that could interfere with turbidity readings. The measurement environment requires controlled lighting conditions to prevent external light interference with nephelometric or spectrophotometric measurements.
Calibration protocols form another cornerstone of quality standards. Primary turbidity standards, typically formazin-based solutions with certified turbidity values, must be prepared according to established procedures and verified against secondary standards. Calibration verification should occur at minimum daily, with full calibration curves generated weekly or following any significant equipment maintenance. The acceptable deviation from certified values typically ranges within ±2% for high-precision applications.
Sample handling procedures within quality standards address critical factors affecting measurement accuracy. Post-sonication samples require standardized settling times, typically 2-5 minutes, to allow transient cavitation effects to dissipate while preventing significant particle sedimentation. Sample volume requirements, mixing protocols, and measurement timing must be strictly controlled to ensure consistency across different operators and measurement sessions.
Documentation and traceability requirements establish comprehensive record-keeping systems tracking sample identification, processing parameters, measurement conditions, and results. Quality control charts monitoring blank measurements, standard recoveries, and duplicate sample precision provide ongoing verification of system performance. Acceptance criteria for measurement precision, typically requiring relative standard deviations below 5% for replicate measurements, ensure data quality meets analytical requirements.
Regular proficiency testing and method validation components verify that laboratory procedures consistently meet established performance criteria. These standards also define corrective action protocols when measurements fall outside acceptable ranges, including equipment troubleshooting procedures, recalibration requirements, and sample reprocessing criteria. Integration with broader laboratory quality management systems ensures that post-sonication turbidity evaluation maintains the highest standards of analytical reliability and regulatory compliance.
Automation Trends in Sample Preparation and Analysis
The automation landscape in sample preparation and analysis has undergone significant transformation, particularly in turbidity measurement applications following sonication processes. Modern laboratory environments increasingly demand streamlined workflows that minimize manual intervention while maximizing measurement accuracy and reproducibility. This shift toward automation has been driven by the need to handle larger sample volumes, reduce human error, and ensure consistent analytical results across different operators and time periods.
Robotic liquid handling systems have emerged as cornerstone technologies in automated sample preparation workflows. These systems integrate sophisticated pipetting mechanisms with turbidity measurement capabilities, enabling seamless transition from sonication to clarity assessment. Advanced platforms now incorporate multi-channel dispensing units that can simultaneously process dozens of samples, significantly reducing processing time compared to traditional manual methods. The integration of automated sample tracking through barcode or RFID systems ensures complete traceability throughout the analytical process.
Inline turbidity monitoring represents a major advancement in real-time sample analysis. Modern systems employ continuous flow cells equipped with nephelometric sensors that provide instantaneous turbidity readings as samples pass through the measurement chamber. These systems eliminate the need for discrete sampling and manual measurement, reducing contamination risks and improving measurement precision. The integration of automated dilution systems allows for optimal measurement range adjustment without operator intervention.
Machine learning algorithms are increasingly being implemented to optimize measurement protocols and predict optimal sonication parameters. These intelligent systems analyze historical data patterns to automatically adjust measurement timing, sample positioning, and calibration intervals. Predictive maintenance capabilities help prevent instrument drift and ensure consistent performance over extended operational periods.
Cloud-based data management platforms have revolutionized how turbidity data is collected, processed, and analyzed. These systems enable remote monitoring of multiple instruments, automated report generation, and seamless integration with laboratory information management systems. Real-time alerts and automated quality control checks ensure immediate detection of measurement anomalies or system malfunctions.
The convergence of Internet of Things technologies with traditional analytical instruments has created smart laboratory ecosystems where turbidity measurements are automatically triggered based on sonication completion signals. This level of integration represents the future direction of automated sample analysis, where entire analytical workflows operate with minimal human oversight while maintaining the highest standards of analytical quality and regulatory compliance.
Robotic liquid handling systems have emerged as cornerstone technologies in automated sample preparation workflows. These systems integrate sophisticated pipetting mechanisms with turbidity measurement capabilities, enabling seamless transition from sonication to clarity assessment. Advanced platforms now incorporate multi-channel dispensing units that can simultaneously process dozens of samples, significantly reducing processing time compared to traditional manual methods. The integration of automated sample tracking through barcode or RFID systems ensures complete traceability throughout the analytical process.
Inline turbidity monitoring represents a major advancement in real-time sample analysis. Modern systems employ continuous flow cells equipped with nephelometric sensors that provide instantaneous turbidity readings as samples pass through the measurement chamber. These systems eliminate the need for discrete sampling and manual measurement, reducing contamination risks and improving measurement precision. The integration of automated dilution systems allows for optimal measurement range adjustment without operator intervention.
Machine learning algorithms are increasingly being implemented to optimize measurement protocols and predict optimal sonication parameters. These intelligent systems analyze historical data patterns to automatically adjust measurement timing, sample positioning, and calibration intervals. Predictive maintenance capabilities help prevent instrument drift and ensure consistent performance over extended operational periods.
Cloud-based data management platforms have revolutionized how turbidity data is collected, processed, and analyzed. These systems enable remote monitoring of multiple instruments, automated report generation, and seamless integration with laboratory information management systems. Real-time alerts and automated quality control checks ensure immediate detection of measurement anomalies or system malfunctions.
The convergence of Internet of Things technologies with traditional analytical instruments has created smart laboratory ecosystems where turbidity measurements are automatically triggered based on sonication completion signals. This level of integration represents the future direction of automated sample analysis, where entire analytical workflows operate with minimal human oversight while maintaining the highest standards of analytical quality and regulatory compliance.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!