How to Identify Optimal Solvent Ratios for Sonication Extraction
MAR 11, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Sonication Extraction Background and Optimization Goals
Sonication extraction has emerged as a pivotal technique in analytical chemistry and natural product isolation, leveraging ultrasonic waves to enhance mass transfer and accelerate extraction processes. This technology originated from the fundamental understanding that acoustic cavitation creates microscopic bubbles that collapse violently, generating localized high temperatures and pressures that facilitate the breakdown of cellular structures and improve solvent penetration. The evolution of sonication extraction spans several decades, beginning with basic ultrasonic baths in the 1960s and progressing to sophisticated probe-type sonicators and multi-frequency systems that offer precise control over extraction parameters.
The historical development of sonication extraction reveals a trajectory from empirical applications to scientifically optimized processes. Early implementations relied heavily on trial-and-error approaches, where researchers would test various solvent combinations without systematic understanding of the underlying mechanisms. The transition toward evidence-based optimization began in the 1990s when researchers started correlating acoustic parameters with extraction efficiency, leading to the recognition that solvent selection and ratio optimization represent critical determinants of extraction success.
Contemporary sonication extraction faces the fundamental challenge of balancing multiple competing factors to achieve optimal results. The primary technical objective centers on maximizing target compound recovery while minimizing extraction time, energy consumption, and solvent usage. This optimization challenge becomes particularly complex when dealing with multi-component systems where different target compounds exhibit varying solubilities in different solvent systems.
The identification of optimal solvent ratios represents a multidimensional optimization problem that must account for the physicochemical properties of target compounds, matrix characteristics, and acoustic propagation efficiency. Modern approaches aim to develop predictive models that can determine ideal solvent compositions based on compound polarity, molecular weight, and matrix compatibility, thereby reducing the need for extensive experimental screening.
Current technological goals emphasize the development of rapid screening methodologies that can systematically evaluate solvent ratio effects while incorporating real-time monitoring capabilities. Advanced systems integrate spectroscopic techniques with sonication equipment to provide immediate feedback on extraction progress, enabling dynamic adjustment of solvent ratios during the extraction process. These innovations represent a significant departure from traditional static approaches, moving toward adaptive extraction systems that can respond to changing conditions and optimize performance in real-time.
The historical development of sonication extraction reveals a trajectory from empirical applications to scientifically optimized processes. Early implementations relied heavily on trial-and-error approaches, where researchers would test various solvent combinations without systematic understanding of the underlying mechanisms. The transition toward evidence-based optimization began in the 1990s when researchers started correlating acoustic parameters with extraction efficiency, leading to the recognition that solvent selection and ratio optimization represent critical determinants of extraction success.
Contemporary sonication extraction faces the fundamental challenge of balancing multiple competing factors to achieve optimal results. The primary technical objective centers on maximizing target compound recovery while minimizing extraction time, energy consumption, and solvent usage. This optimization challenge becomes particularly complex when dealing with multi-component systems where different target compounds exhibit varying solubilities in different solvent systems.
The identification of optimal solvent ratios represents a multidimensional optimization problem that must account for the physicochemical properties of target compounds, matrix characteristics, and acoustic propagation efficiency. Modern approaches aim to develop predictive models that can determine ideal solvent compositions based on compound polarity, molecular weight, and matrix compatibility, thereby reducing the need for extensive experimental screening.
Current technological goals emphasize the development of rapid screening methodologies that can systematically evaluate solvent ratio effects while incorporating real-time monitoring capabilities. Advanced systems integrate spectroscopic techniques with sonication equipment to provide immediate feedback on extraction progress, enabling dynamic adjustment of solvent ratios during the extraction process. These innovations represent a significant departure from traditional static approaches, moving toward adaptive extraction systems that can respond to changing conditions and optimize performance in real-time.
Market Demand for Efficient Extraction Technologies
The global extraction technology market is experiencing unprecedented growth driven by expanding applications across pharmaceutical, nutraceutical, food and beverage, and cosmetic industries. Sonication extraction, as an advanced green extraction technique, has emerged as a critical technology addressing the increasing demand for efficient, environmentally sustainable, and cost-effective extraction processes. The market's evolution reflects a fundamental shift from traditional extraction methods toward innovative approaches that maximize yield while minimizing environmental impact.
Pharmaceutical and biotechnology sectors represent the largest demand drivers for optimized sonication extraction technologies. The growing emphasis on natural product drug discovery and the need for precise extraction of bioactive compounds from plant materials have created substantial market opportunities. Companies are increasingly seeking standardized protocols for solvent ratio optimization to ensure consistent product quality and regulatory compliance. The ability to identify optimal solvent ratios directly impacts extraction efficiency, product purity, and overall manufacturing costs.
The nutraceutical industry's rapid expansion has further amplified demand for sophisticated extraction technologies. Consumer preferences for natural health products and functional foods have driven manufacturers to invest in advanced extraction capabilities. Optimal solvent ratio identification becomes crucial for maximizing the extraction of specific nutrients, antioxidants, and bioactive compounds while maintaining their biological activity and stability.
Environmental regulations and sustainability initiatives are reshaping market dynamics, creating strong demand for green extraction technologies. Sonication extraction's ability to reduce solvent consumption, minimize processing time, and eliminate toxic solvents aligns with industry sustainability goals. Companies are actively seeking technologies that can optimize solvent ratios to achieve maximum efficiency with minimal environmental footprint.
The food and beverage industry presents significant growth opportunities, particularly in flavor and fragrance extraction, essential oil production, and functional ingredient isolation. Market demand is driven by consumer preferences for natural ingredients and clean-label products. Manufacturers require precise solvent ratio optimization to maintain product consistency, enhance extraction yields, and meet stringent quality standards.
Emerging markets in Asia-Pacific and Latin America are experiencing accelerated adoption of advanced extraction technologies, driven by expanding pharmaceutical manufacturing capabilities and growing awareness of extraction efficiency benefits. These regions present substantial opportunities for companies offering comprehensive solutions for solvent ratio optimization in sonication extraction processes.
Pharmaceutical and biotechnology sectors represent the largest demand drivers for optimized sonication extraction technologies. The growing emphasis on natural product drug discovery and the need for precise extraction of bioactive compounds from plant materials have created substantial market opportunities. Companies are increasingly seeking standardized protocols for solvent ratio optimization to ensure consistent product quality and regulatory compliance. The ability to identify optimal solvent ratios directly impacts extraction efficiency, product purity, and overall manufacturing costs.
The nutraceutical industry's rapid expansion has further amplified demand for sophisticated extraction technologies. Consumer preferences for natural health products and functional foods have driven manufacturers to invest in advanced extraction capabilities. Optimal solvent ratio identification becomes crucial for maximizing the extraction of specific nutrients, antioxidants, and bioactive compounds while maintaining their biological activity and stability.
Environmental regulations and sustainability initiatives are reshaping market dynamics, creating strong demand for green extraction technologies. Sonication extraction's ability to reduce solvent consumption, minimize processing time, and eliminate toxic solvents aligns with industry sustainability goals. Companies are actively seeking technologies that can optimize solvent ratios to achieve maximum efficiency with minimal environmental footprint.
The food and beverage industry presents significant growth opportunities, particularly in flavor and fragrance extraction, essential oil production, and functional ingredient isolation. Market demand is driven by consumer preferences for natural ingredients and clean-label products. Manufacturers require precise solvent ratio optimization to maintain product consistency, enhance extraction yields, and meet stringent quality standards.
Emerging markets in Asia-Pacific and Latin America are experiencing accelerated adoption of advanced extraction technologies, driven by expanding pharmaceutical manufacturing capabilities and growing awareness of extraction efficiency benefits. These regions present substantial opportunities for companies offering comprehensive solutions for solvent ratio optimization in sonication extraction processes.
Current Solvent Selection Challenges in Sonication
Solvent selection in sonication extraction presents multifaceted challenges that significantly impact extraction efficiency and reproducibility. Traditional approaches often rely on empirical methods or literature precedents, which may not account for the specific physicochemical properties of target compounds or the unique conditions created during ultrasonic treatment. This conventional methodology frequently results in suboptimal extraction yields and inconsistent results across different laboratories and applications.
The complexity of solvent-analyte interactions under ultrasonic conditions creates additional layers of difficulty. Cavitation phenomena generate localized high-temperature and high-pressure zones that can alter solvent properties and molecular interactions in ways that differ substantially from conventional extraction methods. These dynamic conditions make it challenging to predict optimal solvent compositions based solely on standard physicochemical parameters such as polarity, boiling point, or viscosity.
Matrix effects pose another significant challenge in solvent selection for sonication extraction. Different sample matrices, ranging from plant tissues to synthetic materials, exhibit varying responses to ultrasonic treatment and solvent penetration. The heterogeneous nature of many samples means that a single solvent system may not effectively extract all target compounds, leading to incomplete recovery or selective extraction that compromises analytical accuracy.
Scale-up considerations further complicate solvent selection processes. Laboratory-scale optimization may not translate directly to larger-scale operations due to differences in ultrasonic power distribution, heat dissipation, and mass transfer characteristics. This scalability challenge often necessitates extensive re-optimization when transitioning from research to industrial applications.
Environmental and safety constraints increasingly influence solvent selection decisions. Regulatory pressures to minimize the use of hazardous solvents, coupled with sustainability concerns, limit the available solvent options. Green chemistry principles demand the consideration of biodegradability, toxicity, and environmental impact alongside extraction performance, creating a complex optimization problem that balances multiple competing objectives.
The lack of standardized methodologies for systematic solvent screening compounds these challenges. Without established protocols for evaluating solvent performance under sonication conditions, researchers often resort to trial-and-error approaches that are time-consuming and resource-intensive. This methodological gap hinders the development of predictive models and universal guidelines for solvent selection in ultrasonic extraction applications.
The complexity of solvent-analyte interactions under ultrasonic conditions creates additional layers of difficulty. Cavitation phenomena generate localized high-temperature and high-pressure zones that can alter solvent properties and molecular interactions in ways that differ substantially from conventional extraction methods. These dynamic conditions make it challenging to predict optimal solvent compositions based solely on standard physicochemical parameters such as polarity, boiling point, or viscosity.
Matrix effects pose another significant challenge in solvent selection for sonication extraction. Different sample matrices, ranging from plant tissues to synthetic materials, exhibit varying responses to ultrasonic treatment and solvent penetration. The heterogeneous nature of many samples means that a single solvent system may not effectively extract all target compounds, leading to incomplete recovery or selective extraction that compromises analytical accuracy.
Scale-up considerations further complicate solvent selection processes. Laboratory-scale optimization may not translate directly to larger-scale operations due to differences in ultrasonic power distribution, heat dissipation, and mass transfer characteristics. This scalability challenge often necessitates extensive re-optimization when transitioning from research to industrial applications.
Environmental and safety constraints increasingly influence solvent selection decisions. Regulatory pressures to minimize the use of hazardous solvents, coupled with sustainability concerns, limit the available solvent options. Green chemistry principles demand the consideration of biodegradability, toxicity, and environmental impact alongside extraction performance, creating a complex optimization problem that balances multiple competing objectives.
The lack of standardized methodologies for systematic solvent screening compounds these challenges. Without established protocols for evaluating solvent performance under sonication conditions, researchers often resort to trial-and-error approaches that are time-consuming and resource-intensive. This methodological gap hinders the development of predictive models and universal guidelines for solvent selection in ultrasonic extraction applications.
Existing Solvent Ratio Optimization Approaches
01 Optimization of solvent-to-material ratios in ultrasonic extraction
The ratio of extraction solvent to raw material is a critical parameter in sonication extraction processes. Optimal ratios typically range from 5:1 to 30:1 (v/w) depending on the material being extracted. Higher ratios generally improve extraction efficiency by providing sufficient solvent contact with the material, but excessive ratios may dilute the extract and increase processing costs. The optimal ratio must be determined experimentally for each specific application to balance extraction yield and economic considerations.- Optimization of solvent-to-material ratios in ultrasonic extraction: The ratio of extraction solvent to raw material is a critical parameter in sonication extraction processes. Optimal ratios typically range from 5:1 to 30:1 (v/w) depending on the material being extracted. Higher ratios generally improve extraction efficiency by providing sufficient solvent contact with the material, but excessive ratios may dilute the extract and increase processing costs. The optimal ratio must be determined experimentally for each specific application to balance extraction yield, concentration, and economic considerations.
- Multi-stage extraction with varying solvent ratios: Sequential extraction processes employing different solvent-to-material ratios at each stage can enhance overall extraction efficiency. Initial stages may use lower ratios for concentrated extraction of readily accessible compounds, followed by subsequent stages with higher ratios to extract remaining components. This approach maximizes yield while minimizing total solvent consumption and allows for selective extraction of different compound classes based on their solubility characteristics.
- Temperature-dependent solvent ratio adjustments: The optimal solvent-to-material ratio in sonication extraction varies with temperature due to changes in solvent properties and solubility. At elevated temperatures, lower solvent ratios may be sufficient due to increased extraction kinetics and solubility. Conversely, room temperature extractions typically require higher ratios to achieve comparable yields. Integration of temperature control with ratio optimization enables more efficient extraction protocols.
- Solvent ratio optimization for specific compound classes: Different bioactive compounds require specific solvent-to-material ratios for optimal extraction under sonication. Polar compounds may require higher ratios of polar solvents, while lipophilic substances can be efficiently extracted with lower ratios of non-polar solvents. The ratio must be tailored to the target compounds' physicochemical properties, including molecular weight, polarity, and matrix interactions, to maximize selective extraction and minimize co-extraction of unwanted materials.
- Scale-up considerations for solvent ratio in industrial sonication: Translating laboratory-scale solvent ratios to industrial-scale sonication extraction requires consideration of factors such as ultrasonic power distribution, vessel geometry, and mass transfer limitations. Industrial processes may require adjusted ratios compared to laboratory conditions to maintain extraction efficiency. Pilot-scale studies are essential to determine appropriate scaling factors and ensure that solvent ratios remain economically viable while maintaining product quality and yield at commercial production volumes.
02 Multi-stage extraction with varying solvent ratios
Sequential extraction processes employing different solvent-to-material ratios at each stage can enhance overall extraction efficiency. The first extraction stage typically uses a lower solvent ratio to obtain concentrated extracts, followed by subsequent stages with higher ratios to maximize yield. This approach allows for better recovery of target compounds while maintaining extract quality and reducing solvent consumption compared to single-stage extraction methods.Expand Specific Solutions03 Solvent ratio adjustment based on particle size and material properties
The optimal solvent-to-material ratio in sonication extraction varies depending on the physical characteristics of the raw material, including particle size, moisture content, and density. Finely ground materials generally require lower solvent ratios due to increased surface area, while coarser materials need higher ratios to ensure adequate penetration. Material porosity and cellular structure also influence the required solvent volume for effective extraction under ultrasonic conditions.Expand Specific Solutions04 Temperature-dependent solvent ratio optimization in ultrasonic extraction
The relationship between extraction temperature and solvent ratio is significant in sonication processes. At elevated temperatures, lower solvent ratios may be sufficient due to increased solubility and mass transfer rates. Conversely, room temperature extractions often require higher solvent ratios to achieve comparable yields. The synergistic effect of ultrasonic cavitation and temperature on solvent penetration allows for optimization of both parameters simultaneously to improve extraction efficiency.Expand Specific Solutions05 Solvent mixture ratios and composition in ultrasonic extraction
The use of mixed solvent systems in sonication extraction requires careful consideration of both the solvent-to-material ratio and the ratio of different solvents in the mixture. Binary or ternary solvent systems can enhance selectivity and extraction efficiency for specific compounds. The proportion of polar to non-polar solvents, along with the total solvent volume relative to material mass, must be optimized to maximize target compound recovery while minimizing co-extraction of unwanted substances.Expand Specific Solutions
Key Players in Sonication Equipment Industry
The sonication extraction technology for optimal solvent ratio identification represents a mature field experiencing steady growth, driven by increasing demand for efficient extraction processes across pharmaceutical, food, and chemical industries. The market demonstrates moderate expansion with established applications in natural product extraction and analytical chemistry. Technology maturity varies significantly among key players, with pharmaceutical companies like F. Hoffmann-La Roche Ltd., Shanghai Hutchison Pharmaceuticals, and Shijiazhuang Yiling Pharmaceutical leading in commercial applications, while research institutions including MIT, University of Bonn, and Anhui University drive fundamental innovations. Specialized technology companies such as Sonichem Technologies Ltd. and Nano Green Technology represent advanced implementation capabilities. The competitive landscape shows convergence between traditional pharmaceutical manufacturers adopting sonication methods and emerging clean-tech companies developing novel ultrasonic processes, indicating a transitioning industry moving toward more sophisticated, environmentally sustainable extraction technologies with optimized solvent utilization.
Council of Scientific & Industrial Research
Technical Solution: CSIR has developed systematic methodologies for optimizing solvent ratios in sonication extraction through their various research institutes. Their approach combines traditional experimental design with modern analytical techniques to identify optimal extraction parameters. CSIR researchers utilize factorial design experiments and response surface methodology to systematically evaluate the effects of different solvent ratios on extraction efficiency. The organization has developed standardized protocols for screening solvent combinations across different natural product extraction applications. Their methodology incorporates cost-effectiveness analysis and environmental impact assessment when selecting optimal solvent systems. CSIR's approach emphasizes the use of green solvents and sustainable extraction practices. The research teams have developed databases of optimal solvent ratios for various plant materials and bioactive compounds, providing valuable reference data for industrial applications.
Strengths: Extensive research network, focus on sustainable practices, comprehensive databases. Weaknesses: Limited commercial partnerships, slower technology transfer processes.
F. Hoffmann-La Roche Ltd.
Technical Solution: Roche has developed comprehensive methodologies for optimizing solvent ratios in sonication extraction as part of their pharmaceutical manufacturing processes. Their approach integrates Design of Experiment (DoE) methodologies with advanced analytical techniques to systematically evaluate solvent combinations. The company employs high-throughput screening platforms that can simultaneously test multiple solvent ratio combinations under controlled sonication conditions. Their process development teams utilize statistical modeling and response surface methodology to identify optimal extraction parameters. Roche's technology incorporates real-time process analytical technology (PAT) to monitor extraction efficiency and adjust solvent ratios dynamically. The system considers regulatory compliance requirements and environmental impact factors when selecting optimal solvent combinations for pharmaceutical applications.
Strengths: Extensive pharmaceutical expertise, robust regulatory compliance framework, advanced analytical capabilities. Weaknesses: Focus primarily on pharmaceutical applications, high development costs.
Core Innovations in Solvent System Design
Solvent selection process
PatentInactiveUS20140262961A1
Innovation
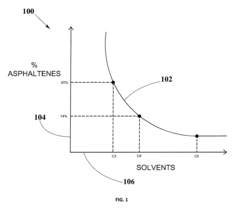
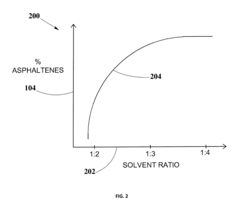
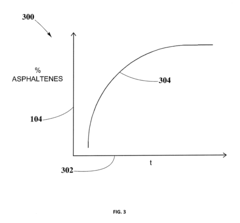
- A method for solvent selection using a computer-based analysis integrating market demands, oil characteristics, equipment capabilities, and solvent properties, including charts and algorithms to optimize solvent choice for asphaltenes separation, reducing solvent usage and equipment requirements.
Solvent extraction process
PatentInactiveGB821998A
Innovation
- A mixed ether glycol solvent with a general formula HO(C2H4O)x(CH3O)yH, containing both oxypropylene and oxyethylene units, is used for selective extraction, offering a balance between selectivity and solubility, stability, and constant vapor pressure, allowing for continuous reuse and efficient separation of aromatic hydrocarbons from other hydrocarbons.
Environmental Impact of Solvent Usage
The environmental implications of solvent selection and usage in sonication extraction processes represent a critical consideration that extends beyond immediate operational efficiency. Traditional organic solvents commonly employed in extraction procedures, including hexane, chloroform, and methanol, pose significant environmental risks through their production, application, and disposal phases. These conventional solvents often exhibit high volatility, contributing to atmospheric pollution and greenhouse gas emissions, while their manufacturing processes typically involve energy-intensive petrochemical pathways that generate substantial carbon footprints.
Water contamination emerges as a primary environmental concern when optimizing solvent ratios for sonication extraction. Improper disposal of spent solvent mixtures can lead to groundwater contamination, affecting local ecosystems and potentially entering drinking water supplies. The persistence of certain organic solvents in environmental matrices means that even small-scale laboratory operations can contribute to long-term ecological damage if waste management protocols are inadequate.
The growing emphasis on green chemistry principles has catalyzed the development of environmentally benign solvent alternatives for sonication extraction applications. Supercritical carbon dioxide, ionic liquids, and deep eutectic solvents represent promising alternatives that can significantly reduce environmental impact while maintaining extraction efficiency. These green solvents often demonstrate superior biodegradability, reduced toxicity profiles, and lower atmospheric ozone depletion potential compared to conventional organic solvents.
Lifecycle assessment considerations play an increasingly important role in solvent ratio optimization strategies. The environmental impact extends beyond direct solvent usage to encompass energy consumption during sonication processes, solvent recovery and recycling operations, and waste treatment requirements. Optimal solvent ratios must therefore balance extraction efficiency with minimized environmental burden, considering factors such as solvent recyclability, energy requirements for separation processes, and overall waste generation.
Regulatory frameworks worldwide are increasingly stringent regarding solvent emissions and waste disposal, driving innovation toward more sustainable extraction methodologies. The implementation of volatile organic compound regulations and hazardous waste classification systems necessitates careful consideration of environmental impact during solvent ratio optimization, influencing both operational costs and regulatory compliance requirements for industrial-scale sonication extraction processes.
Water contamination emerges as a primary environmental concern when optimizing solvent ratios for sonication extraction. Improper disposal of spent solvent mixtures can lead to groundwater contamination, affecting local ecosystems and potentially entering drinking water supplies. The persistence of certain organic solvents in environmental matrices means that even small-scale laboratory operations can contribute to long-term ecological damage if waste management protocols are inadequate.
The growing emphasis on green chemistry principles has catalyzed the development of environmentally benign solvent alternatives for sonication extraction applications. Supercritical carbon dioxide, ionic liquids, and deep eutectic solvents represent promising alternatives that can significantly reduce environmental impact while maintaining extraction efficiency. These green solvents often demonstrate superior biodegradability, reduced toxicity profiles, and lower atmospheric ozone depletion potential compared to conventional organic solvents.
Lifecycle assessment considerations play an increasingly important role in solvent ratio optimization strategies. The environmental impact extends beyond direct solvent usage to encompass energy consumption during sonication processes, solvent recovery and recycling operations, and waste treatment requirements. Optimal solvent ratios must therefore balance extraction efficiency with minimized environmental burden, considering factors such as solvent recyclability, energy requirements for separation processes, and overall waste generation.
Regulatory frameworks worldwide are increasingly stringent regarding solvent emissions and waste disposal, driving innovation toward more sustainable extraction methodologies. The implementation of volatile organic compound regulations and hazardous waste classification systems necessitates careful consideration of environmental impact during solvent ratio optimization, influencing both operational costs and regulatory compliance requirements for industrial-scale sonication extraction processes.
Quality Standards for Extraction Processes
Quality standards for sonication extraction processes encompass multiple critical parameters that ensure reproducible and effective extraction outcomes. These standards establish benchmarks for extraction efficiency, purity levels, and consistency across different operational conditions. The development of comprehensive quality frameworks has become essential as sonication extraction gains wider adoption in pharmaceutical, nutraceutical, and analytical chemistry applications.
Extraction efficiency standards typically require minimum yield thresholds ranging from 85-95% for target compounds, depending on the specific application and matrix complexity. These benchmarks are established through validated analytical methods including HPLC, GC-MS, or spectrophotometric techniques. Purity specifications often mandate that extracted compounds meet pharmaceutical-grade requirements, with impurity levels below 0.1% for critical applications.
Reproducibility standards demand that extraction processes demonstrate coefficient of variation values below 5% across multiple batches and operators. This requirement necessitates strict control over solvent ratios, temperature fluctuations, and sonication parameters. Documentation protocols must include detailed batch records, equipment calibration certificates, and environmental monitoring data to ensure traceability and compliance.
Solvent quality specifications form a cornerstone of extraction standards, requiring reagent-grade or higher purity solvents with documented water content, residual impurities, and stability profiles. Solvent ratio tolerances are typically maintained within ±2% of target values, with continuous monitoring systems employed to detect deviations. Storage and handling protocols must prevent contamination and degradation that could compromise extraction performance.
Safety and environmental standards mandate proper ventilation systems, waste disposal procedures, and operator protection protocols. Regulatory compliance requirements vary by jurisdiction but commonly include adherence to ICH guidelines, FDA regulations, or equivalent international standards. Quality management systems must incorporate risk assessment methodologies, change control procedures, and continuous improvement processes.
Validation protocols require comprehensive testing across the operational range, including worst-case scenarios and stress conditions. Statistical analysis methods must demonstrate process capability and control limits, with ongoing monitoring systems to detect trends and deviations. These standards collectively ensure that sonication extraction processes deliver consistent, high-quality results while maintaining safety and regulatory compliance.
Extraction efficiency standards typically require minimum yield thresholds ranging from 85-95% for target compounds, depending on the specific application and matrix complexity. These benchmarks are established through validated analytical methods including HPLC, GC-MS, or spectrophotometric techniques. Purity specifications often mandate that extracted compounds meet pharmaceutical-grade requirements, with impurity levels below 0.1% for critical applications.
Reproducibility standards demand that extraction processes demonstrate coefficient of variation values below 5% across multiple batches and operators. This requirement necessitates strict control over solvent ratios, temperature fluctuations, and sonication parameters. Documentation protocols must include detailed batch records, equipment calibration certificates, and environmental monitoring data to ensure traceability and compliance.
Solvent quality specifications form a cornerstone of extraction standards, requiring reagent-grade or higher purity solvents with documented water content, residual impurities, and stability profiles. Solvent ratio tolerances are typically maintained within ±2% of target values, with continuous monitoring systems employed to detect deviations. Storage and handling protocols must prevent contamination and degradation that could compromise extraction performance.
Safety and environmental standards mandate proper ventilation systems, waste disposal procedures, and operator protection protocols. Regulatory compliance requirements vary by jurisdiction but commonly include adherence to ICH guidelines, FDA regulations, or equivalent international standards. Quality management systems must incorporate risk assessment methodologies, change control procedures, and continuous improvement processes.
Validation protocols require comprehensive testing across the operational range, including worst-case scenarios and stress conditions. Statistical analysis methods must demonstrate process capability and control limits, with ongoing monitoring systems to detect trends and deviations. These standards collectively ensure that sonication extraction processes deliver consistent, high-quality results while maintaining safety and regulatory compliance.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!