How to Quantify Lithium Acetate Purity for Battery Use
SEP 10, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
Patsnap Eureka helps you evaluate technical feasibility & market potential.
Lithium Acetate Purity Quantification Background and Objectives
Lithium acetate has emerged as a critical component in the development of advanced battery technologies, particularly in the realm of lithium-ion batteries that power everything from portable electronics to electric vehicles. The evolution of this technology has been marked by continuous improvements in energy density, charging efficiency, and overall battery performance. Understanding the historical progression of lithium compounds in energy storage applications provides valuable context for current research efforts focused on purity quantification.
The importance of lithium acetate purity has grown exponentially as battery technologies have become more sophisticated. Impurities, even at trace levels, can significantly impact battery performance, safety, and longevity. Early battery technologies could tolerate higher impurity levels, but modern high-performance batteries require exceptionally pure materials to achieve desired specifications and reliability.
Current industry standards typically require lithium acetate purity levels exceeding 99.5%, with some advanced applications demanding 99.9% or higher. This trend toward increasingly stringent purity requirements has created a pressing need for more accurate, reliable, and efficient quantification methods. Traditional analytical techniques often lack the precision needed for today's demanding applications, creating a technological gap that must be addressed.
The primary objective of lithium acetate purity quantification research is to develop methodologies that can accurately detect and measure impurities at parts-per-million (ppm) or even parts-per-billion (ppb) levels. These methods must be not only highly sensitive but also reproducible, cost-effective, and suitable for implementation in both research and industrial settings.
Another critical goal is to establish standardized protocols that enable consistent quality control across the battery supply chain. The lack of universally accepted quantification methods has led to inconsistencies in reported purity levels, complicating material selection and quality assurance processes for manufacturers.
Recent technological advancements in analytical instrumentation, including high-resolution mass spectrometry, advanced chromatography techniques, and spectroscopic methods, have opened new possibilities for more precise purity assessments. These developments, coupled with computational approaches for data analysis, are creating promising pathways toward meeting the industry's evolving needs.
The ultimate aim of this technical research is to bridge the gap between current analytical capabilities and future battery requirements, ensuring that purity quantification methods keep pace with the rapid advancement of battery technologies. By achieving this objective, we can support the continued evolution of energy storage solutions that are essential for renewable energy integration and sustainable transportation systems.
The importance of lithium acetate purity has grown exponentially as battery technologies have become more sophisticated. Impurities, even at trace levels, can significantly impact battery performance, safety, and longevity. Early battery technologies could tolerate higher impurity levels, but modern high-performance batteries require exceptionally pure materials to achieve desired specifications and reliability.
Current industry standards typically require lithium acetate purity levels exceeding 99.5%, with some advanced applications demanding 99.9% or higher. This trend toward increasingly stringent purity requirements has created a pressing need for more accurate, reliable, and efficient quantification methods. Traditional analytical techniques often lack the precision needed for today's demanding applications, creating a technological gap that must be addressed.
The primary objective of lithium acetate purity quantification research is to develop methodologies that can accurately detect and measure impurities at parts-per-million (ppm) or even parts-per-billion (ppb) levels. These methods must be not only highly sensitive but also reproducible, cost-effective, and suitable for implementation in both research and industrial settings.
Another critical goal is to establish standardized protocols that enable consistent quality control across the battery supply chain. The lack of universally accepted quantification methods has led to inconsistencies in reported purity levels, complicating material selection and quality assurance processes for manufacturers.
Recent technological advancements in analytical instrumentation, including high-resolution mass spectrometry, advanced chromatography techniques, and spectroscopic methods, have opened new possibilities for more precise purity assessments. These developments, coupled with computational approaches for data analysis, are creating promising pathways toward meeting the industry's evolving needs.
The ultimate aim of this technical research is to bridge the gap between current analytical capabilities and future battery requirements, ensuring that purity quantification methods keep pace with the rapid advancement of battery technologies. By achieving this objective, we can support the continued evolution of energy storage solutions that are essential for renewable energy integration and sustainable transportation systems.
Market Demand Analysis for High-Purity Battery Materials
The global market for high-purity battery materials has experienced exponential growth in recent years, primarily driven by the rapid expansion of electric vehicle (EV) production and renewable energy storage systems. Lithium-based batteries, particularly lithium-ion batteries, have emerged as the dominant technology in these applications, creating substantial demand for high-purity lithium compounds such as lithium acetate.
Market research indicates that the global lithium-ion battery market was valued at approximately $41.1 billion in 2021 and is projected to reach $116.6 billion by 2030, representing a compound annual growth rate (CAGR) of 12.3%. This growth directly translates to increased demand for battery-grade lithium compounds, including lithium acetate, which must meet stringent purity requirements.
The automotive sector represents the largest consumer of high-purity lithium materials, accounting for over 60% of the total market share. Major automotive manufacturers have announced ambitious electrification targets, with several planning to phase out internal combustion engines entirely within the next decade. This transition is expected to further accelerate demand for high-purity battery materials.
Energy storage systems (ESS) constitute another rapidly growing market segment, driven by the increasing integration of renewable energy sources into power grids. The intermittent nature of renewable energy generation necessitates efficient storage solutions, creating additional demand for high-capacity, high-performance batteries that require ultra-pure lithium compounds.
Consumer electronics continue to represent a significant market for lithium batteries, though their share is gradually decreasing relative to automotive and energy storage applications. Nevertheless, the trend toward more powerful, longer-lasting portable devices maintains steady demand for high-purity battery materials in this sector.
Geographically, Asia-Pacific dominates the market, with China, Japan, and South Korea collectively accounting for over 70% of global lithium battery production. However, significant investments in battery manufacturing capacity are underway in Europe and North America, driven by concerns about supply chain security and government incentives for domestic production.
Industry analysts highlight that battery manufacturers are increasingly focused on material purity as a key factor in enhancing battery performance, safety, and longevity. Impurities in lithium compounds can significantly impact electrochemical performance, accelerate capacity degradation, and potentially compromise safety. Consequently, there is growing willingness among manufacturers to pay premium prices for materials with verified high purity levels.
The market trend clearly indicates that quantification methods for lithium acetate purity are not merely technical considerations but critical factors in determining commercial viability and market acceptance. Battery manufacturers are implementing increasingly stringent quality control measures, creating market pressure for advanced analytical techniques that can reliably verify material purity.
Market research indicates that the global lithium-ion battery market was valued at approximately $41.1 billion in 2021 and is projected to reach $116.6 billion by 2030, representing a compound annual growth rate (CAGR) of 12.3%. This growth directly translates to increased demand for battery-grade lithium compounds, including lithium acetate, which must meet stringent purity requirements.
The automotive sector represents the largest consumer of high-purity lithium materials, accounting for over 60% of the total market share. Major automotive manufacturers have announced ambitious electrification targets, with several planning to phase out internal combustion engines entirely within the next decade. This transition is expected to further accelerate demand for high-purity battery materials.
Energy storage systems (ESS) constitute another rapidly growing market segment, driven by the increasing integration of renewable energy sources into power grids. The intermittent nature of renewable energy generation necessitates efficient storage solutions, creating additional demand for high-capacity, high-performance batteries that require ultra-pure lithium compounds.
Consumer electronics continue to represent a significant market for lithium batteries, though their share is gradually decreasing relative to automotive and energy storage applications. Nevertheless, the trend toward more powerful, longer-lasting portable devices maintains steady demand for high-purity battery materials in this sector.
Geographically, Asia-Pacific dominates the market, with China, Japan, and South Korea collectively accounting for over 70% of global lithium battery production. However, significant investments in battery manufacturing capacity are underway in Europe and North America, driven by concerns about supply chain security and government incentives for domestic production.
Industry analysts highlight that battery manufacturers are increasingly focused on material purity as a key factor in enhancing battery performance, safety, and longevity. Impurities in lithium compounds can significantly impact electrochemical performance, accelerate capacity degradation, and potentially compromise safety. Consequently, there is growing willingness among manufacturers to pay premium prices for materials with verified high purity levels.
The market trend clearly indicates that quantification methods for lithium acetate purity are not merely technical considerations but critical factors in determining commercial viability and market acceptance. Battery manufacturers are implementing increasingly stringent quality control measures, creating market pressure for advanced analytical techniques that can reliably verify material purity.
Current Analytical Methods and Technical Limitations
The quantification of lithium acetate purity for battery applications currently relies on several analytical methods, each with specific capabilities and limitations. Inductively Coupled Plasma Mass Spectrometry (ICP-MS) and Inductively Coupled Plasma Optical Emission Spectroscopy (ICP-OES) represent the gold standards for elemental analysis, offering detection limits in the parts-per-billion range. These techniques excel at identifying metallic impurities that can significantly impact battery performance but require sophisticated equipment and specialized training.
X-ray Diffraction (XRD) provides critical information about the crystalline structure of lithium acetate, helping identify phase impurities and structural anomalies. However, XRD struggles with amorphous impurities and requires relatively large sample sizes, limiting its application in production environments where material conservation is essential.
Fourier Transform Infrared Spectroscopy (FTIR) and Nuclear Magnetic Resonance (NMR) spectroscopy offer insights into molecular structure and organic impurities. While powerful for identifying functional groups and molecular arrangements, these methods often lack the sensitivity required for trace contaminant detection crucial in high-performance battery applications.
Thermal analysis techniques, including Differential Scanning Calorimetry (DSC) and Thermogravimetric Analysis (TGA), provide valuable data on thermal stability and decomposition patterns but offer limited information about specific impurities. These methods serve better as complementary techniques rather than primary purity assessment tools.
A significant technical limitation across all methods is the lack of standardized protocols specifically developed for battery-grade lithium acetate. The battery industry has historically borrowed analytical approaches from pharmaceutical or general chemical analysis, which may not address the unique requirements of electrochemical applications.
Sample preparation represents another critical challenge, as lithium compounds are hygroscopic and reactive with atmospheric carbon dioxide. Exposure during handling can introduce artifacts that compromise analytical accuracy. Additionally, the high sensitivity of modern batteries to even trace impurities pushes detection requirements below what many conventional methods can reliably achieve.
Interlaboratory reproducibility remains problematic, with different facilities often reporting varying purity levels for identical samples. This inconsistency stems from equipment variations, analyst expertise differences, and the absence of universally accepted reference materials specifically for battery-grade lithium compounds.
Cost and throughput limitations also constrain comprehensive purity analysis in production environments. High-precision analytical methods typically require expensive equipment and considerable analysis time, creating bottlenecks in manufacturing processes where rapid quality control decisions are essential.
X-ray Diffraction (XRD) provides critical information about the crystalline structure of lithium acetate, helping identify phase impurities and structural anomalies. However, XRD struggles with amorphous impurities and requires relatively large sample sizes, limiting its application in production environments where material conservation is essential.
Fourier Transform Infrared Spectroscopy (FTIR) and Nuclear Magnetic Resonance (NMR) spectroscopy offer insights into molecular structure and organic impurities. While powerful for identifying functional groups and molecular arrangements, these methods often lack the sensitivity required for trace contaminant detection crucial in high-performance battery applications.
Thermal analysis techniques, including Differential Scanning Calorimetry (DSC) and Thermogravimetric Analysis (TGA), provide valuable data on thermal stability and decomposition patterns but offer limited information about specific impurities. These methods serve better as complementary techniques rather than primary purity assessment tools.
A significant technical limitation across all methods is the lack of standardized protocols specifically developed for battery-grade lithium acetate. The battery industry has historically borrowed analytical approaches from pharmaceutical or general chemical analysis, which may not address the unique requirements of electrochemical applications.
Sample preparation represents another critical challenge, as lithium compounds are hygroscopic and reactive with atmospheric carbon dioxide. Exposure during handling can introduce artifacts that compromise analytical accuracy. Additionally, the high sensitivity of modern batteries to even trace impurities pushes detection requirements below what many conventional methods can reliably achieve.
Interlaboratory reproducibility remains problematic, with different facilities often reporting varying purity levels for identical samples. This inconsistency stems from equipment variations, analyst expertise differences, and the absence of universally accepted reference materials specifically for battery-grade lithium compounds.
Cost and throughput limitations also constrain comprehensive purity analysis in production environments. High-precision analytical methods typically require expensive equipment and considerable analysis time, creating bottlenecks in manufacturing processes where rapid quality control decisions are essential.
Established Protocols for Lithium Acetate Purity Assessment
01 Purification methods for lithium acetate
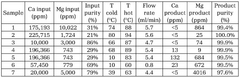
Various purification methods are employed to achieve high-purity lithium acetate, including recrystallization, filtration, and solvent extraction techniques. These methods effectively remove impurities such as metal ions, organic contaminants, and insoluble particles. Advanced purification processes can achieve pharmaceutical or battery-grade lithium acetate with purity levels exceeding 99.9%.- Purification methods for lithium acetate: Various methods are employed to purify lithium acetate to achieve high purity levels. These methods include recrystallization, filtration, and chemical treatments to remove impurities. The purification process is critical for applications requiring high-purity lithium acetate, such as in battery technology and pharmaceutical manufacturing. Advanced purification techniques can achieve purity levels exceeding 99.9%, which is essential for sensitive applications.
- Analytical techniques for determining lithium acetate purity: Various analytical methods are used to determine the purity of lithium acetate, including spectroscopic techniques, chromatography, and titration. These methods allow for the quantification of impurities and confirmation of the lithium acetate content. Advanced analytical techniques can detect impurities at parts-per-million levels, ensuring that the lithium acetate meets the required specifications for various applications. Quality control protocols typically involve multiple analytical methods to comprehensively assess purity.
- Applications requiring high-purity lithium acetate: High-purity lithium acetate is essential for various applications, including lithium-ion battery production, pharmaceutical formulations, and specialty chemical synthesis. The purity requirements vary depending on the specific application, with some requiring ultra-high purity levels. In battery applications, impurities can significantly impact performance and longevity. In pharmaceutical applications, high purity is necessary to meet regulatory standards and ensure product safety and efficacy.
- Impact of impurities on lithium acetate performance: Impurities in lithium acetate can significantly affect its performance in various applications. Common impurities include other metal ions, organic compounds, and moisture. These impurities can interfere with chemical reactions, reduce effectiveness in specific applications, and cause unwanted side reactions. Understanding the impact of specific impurities is crucial for establishing appropriate purity specifications and ensuring consistent performance in end-use applications.
- Industrial production of high-purity lithium acetate: Industrial processes for producing high-purity lithium acetate involve careful control of reaction conditions, raw material selection, and post-production purification steps. Manufacturers employ various techniques to minimize contamination during production and handling. Scaling up laboratory purification methods presents challenges that must be addressed to maintain purity levels in industrial production. Advanced manufacturing technologies and clean room facilities may be utilized to achieve the highest purity grades required for specialized applications.
02 Analytical techniques for purity determination
Several analytical techniques are used to determine the purity of lithium acetate, including inductively coupled plasma mass spectrometry (ICP-MS), atomic absorption spectroscopy, X-ray diffraction, and high-performance liquid chromatography (HPLC). These methods enable precise quantification of trace impurities and verification of lithium acetate purity for various applications.Expand Specific Solutions03 High-purity lithium acetate for battery applications
High-purity lithium acetate is crucial for lithium-ion battery manufacturing, where impurities can significantly impact battery performance and lifespan. Purification processes specifically designed for battery applications focus on removing transition metal contaminants and achieving consistent quality. Ultra-high purity lithium acetate serves as a precursor for cathode materials and electrolyte components.Expand Specific Solutions04 Pharmaceutical and biomedical grade purity standards
Pharmaceutical and biomedical applications require lithium acetate with stringent purity specifications to ensure safety and efficacy. Purification processes for these applications focus on removing toxic impurities and achieving compliance with pharmacopeia standards. High-purity lithium acetate is used in various medical formulations and as a reagent in molecular biology applications.Expand Specific Solutions05 Industrial scale production of high-purity lithium acetate
Industrial manufacturing processes have been developed to produce high-purity lithium acetate at commercial scale. These processes incorporate continuous flow systems, quality control measures, and specialized equipment to maintain purity throughout production. Innovations in manufacturing technology have enabled cost-effective production of high-purity lithium acetate while minimizing environmental impact.Expand Specific Solutions
Key Industry Players in Battery Materials Analysis
The lithium acetate purity quantification market for battery applications is in a growth phase, with increasing demand driven by the expanding lithium-ion battery sector. The market size is projected to grow significantly as electric vehicle adoption accelerates globally. Technologically, the field shows moderate maturity with established analytical methods, but innovation continues. Key players represent diverse specializations: GS Yuasa, Hefei Guoxuan, and Pure Battery Technologies focus on battery manufacturing; Honeywell and Resonac provide analytical instrumentation; while Ascend Elements and Cirba Solutions specialize in battery recycling technologies. Academic institutions like University of Maryland and Chinese Academy of Sciences contribute fundamental research, creating a competitive landscape balanced between established corporations and emerging specialized firms developing advanced purity assessment methodologies.
GS Yuasa International Ltd.
Technical Solution: GS Yuasa has developed a specialized electrochemical impedance spectroscopy (EIS) method for quantifying lithium acetate purity specifically for battery applications. Their approach measures the electrochemical response of lithium acetate samples under controlled potential conditions across a frequency spectrum, generating characteristic impedance patterns that correlate with purity levels. The system employs a reference electrode setup with platinum working electrodes and proprietary electrolyte formulations optimized for lithium acetate analysis. GS Yuasa's method can detect impurities affecting ionic conductivity at concentrations below 0.05%, which is particularly relevant for battery performance. Their analytical protocol includes temperature-controlled measurements at 25°C, 45°C, and 60°C to evaluate thermal stability of the material. The resulting impedance data is processed through their proprietary algorithm that generates a "Battery Suitability Index" (BSI) that correlates strongly with cycle life performance in actual battery testing. The system is complemented by X-ray diffraction analysis to verify crystalline structure integrity and identify any crystalline impurities[4][6].
Strengths: The electrochemical approach directly measures properties relevant to battery performance rather than just chemical composition. The multi-temperature testing provides insight into thermal stability that other methods miss. Weaknesses: The method requires specialized electrochemical equipment and expertise to operate correctly. The impedance patterns can be affected by sample preparation variables, requiring strict standardization of testing protocols.
Robert Bosch GmbH
Technical Solution: Bosch has pioneered a dual-verification lithium acetate purity quantification system specifically designed for battery applications. Their approach combines traditional titration methods with advanced spectroscopic techniques. The first verification layer employs potentiometric titration with standardized reagents to determine acetate content with precision of ±0.1%. The second layer utilizes Fourier Transform Infrared Spectroscopy (FTIR) with attenuated total reflection (ATR) to identify and quantify specific molecular impurities through characteristic absorption patterns. Bosch's system incorporates machine learning algorithms that analyze spectral data against a proprietary database of known impurity signatures, enabling detection of contaminants at concentrations as low as 0.01%. Their method also includes Karl Fischer titration for precise water content determination, as moisture significantly impacts lithium acetate's performance in battery applications. The entire process is automated through Bosch's Manufacturing Analytics Platform, which provides real-time quality monitoring and trend analysis[2][5].
Strengths: The dual-verification approach significantly reduces false positives/negatives compared to single-method systems. Integration with manufacturing analytics enables continuous process improvement and early detection of quality drift. Weaknesses: The system requires regular calibration and validation against reference standards to maintain accuracy. The proprietary machine learning models require substantial training data specific to each manufacturing environment to achieve optimal performance.
Critical Analytical Techniques and Instrumentation Review
Lithium purification process
PatentWO2025120543A1
Innovation
- A simplified and cost-effective lithium purification process involving a two-vessel continuous-loop crystallization method that achieves high-purity lithium carbonate (99.9 wt%) from crude lithium carbonate in a single step, reducing the need for pre-processing and reagents.
Detection method of solid electrolyte lithium aluminum titanium phosphate (LATP)
PatentActiveCN110554078A
Innovation
- By reasonably setting the proportion of solid electrolyte homogenate and the surface quality of the electrodes, the solid electrolyte LATP electrodes and separators, and metal lithium assembled button half cells were prepared. The gradient rate discharge test method was used to determine the purity and rate performance of the solid electrolyte LATP.
Quality Control Standards and Certification Requirements
The quantification of lithium acetate purity for battery applications necessitates adherence to stringent quality control standards and certification requirements. International organizations such as the International Organization for Standardization (ISO) have established comprehensive frameworks, with ISO 9001 serving as the foundational quality management system standard applicable to lithium acetate production processes. More specifically, ISO/IEC 17025 provides guidelines for testing laboratories involved in purity analysis, ensuring measurement accuracy and reliability.
Battery-grade lithium acetate must comply with industry-specific standards such as those set by the International Electrotechnical Commission (IEC) and ASTM International. The IEC 62660 series addresses lithium-ion cells for electric vehicle applications, while ASTM E1613 outlines standard test methods for determination of metals in reagents by atomic absorption spectroscopy, applicable to lithium compound purity assessment.
Regional certification bodies impose additional requirements that manufacturers must satisfy. In North America, Underwriters Laboratories (UL) certification is often mandatory, with UL 1642 specifically addressing lithium batteries. The European Union requires CE marking, indicating compliance with the EU Battery Directive (2006/66/EC) and REACH regulations governing chemical substances. Asian markets, particularly Japan and South Korea, have established their own certification systems through organizations like JIS (Japanese Industrial Standards) and KS (Korean Standards).
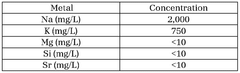
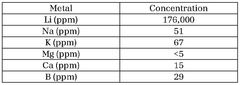
Quality control protocols for lithium acetate typically mandate regular testing for moisture content (≤0.5%), heavy metal impurities (≤10 ppm for most metals), and particle size distribution. Analytical techniques must be validated according to USP <1225> or equivalent standards, with defined acceptance criteria for method precision, accuracy, and detection limits. Documentation requirements include certificates of analysis (CoA) for each production batch, detailing test results against specifications.
Traceability represents another critical aspect of quality assurance, with manufacturers required to maintain comprehensive records of raw material sources, production processes, and analytical test results. Many certification schemes now incorporate sustainability criteria, requiring documentation of environmental management systems (ISO 14001) and responsible sourcing practices, particularly for lithium as a critical battery material.
Emerging trends in certification requirements include the development of specialized standards for battery recycling and circular economy principles, with organizations like the Global Battery Alliance promoting responsible value chains. As battery technologies evolve, certification requirements are increasingly focusing on performance characteristics such as cycle life and safety under extreme conditions, necessitating more sophisticated purity testing methodologies.
Battery-grade lithium acetate must comply with industry-specific standards such as those set by the International Electrotechnical Commission (IEC) and ASTM International. The IEC 62660 series addresses lithium-ion cells for electric vehicle applications, while ASTM E1613 outlines standard test methods for determination of metals in reagents by atomic absorption spectroscopy, applicable to lithium compound purity assessment.
Regional certification bodies impose additional requirements that manufacturers must satisfy. In North America, Underwriters Laboratories (UL) certification is often mandatory, with UL 1642 specifically addressing lithium batteries. The European Union requires CE marking, indicating compliance with the EU Battery Directive (2006/66/EC) and REACH regulations governing chemical substances. Asian markets, particularly Japan and South Korea, have established their own certification systems through organizations like JIS (Japanese Industrial Standards) and KS (Korean Standards).
Quality control protocols for lithium acetate typically mandate regular testing for moisture content (≤0.5%), heavy metal impurities (≤10 ppm for most metals), and particle size distribution. Analytical techniques must be validated according to USP <1225> or equivalent standards, with defined acceptance criteria for method precision, accuracy, and detection limits. Documentation requirements include certificates of analysis (CoA) for each production batch, detailing test results against specifications.
Traceability represents another critical aspect of quality assurance, with manufacturers required to maintain comprehensive records of raw material sources, production processes, and analytical test results. Many certification schemes now incorporate sustainability criteria, requiring documentation of environmental management systems (ISO 14001) and responsible sourcing practices, particularly for lithium as a critical battery material.
Emerging trends in certification requirements include the development of specialized standards for battery recycling and circular economy principles, with organizations like the Global Battery Alliance promoting responsible value chains. As battery technologies evolve, certification requirements are increasingly focusing on performance characteristics such as cycle life and safety under extreme conditions, necessitating more sophisticated purity testing methodologies.
Environmental Impact of Analytical Processes
The analytical processes used to quantify lithium acetate purity for battery applications carry significant environmental implications that warrant careful consideration. Traditional analytical methods such as atomic absorption spectroscopy (AAS), inductively coupled plasma mass spectrometry (ICP-MS), and high-performance liquid chromatography (HPLC) consume substantial amounts of energy and generate chemical waste that can harm ecosystems if improperly managed.
Solvent usage represents a major environmental concern in these analytical processes. Many purity assessment techniques require organic solvents like acetonitrile, methanol, and hexane, which are derived from non-renewable petroleum resources. These solvents often possess high volatility, contributing to air pollution and potential ozone depletion when released into the atmosphere. Additionally, their production and disposal generate significant carbon footprints.
Water consumption in analytical laboratories presents another environmental challenge. Techniques such as ion chromatography and titration methods require large volumes of ultrapure water, which necessitates energy-intensive purification processes. The resulting wastewater frequently contains trace amounts of lithium compounds and other chemicals that conventional treatment facilities may not effectively remove before discharge.
Energy utilization across the analytical lifecycle constitutes a substantial environmental factor. Instruments like mass spectrometers and spectrophotometers demand continuous power during operation and often require climate-controlled environments, further increasing energy consumption. This energy demand translates to greenhouse gas emissions when derived from fossil fuel sources, contributing to climate change impacts.
Chemical reagents employed in lithium acetate analysis, including acids, bases, and indicators, present toxicity concerns for aquatic ecosystems. Improper disposal of these substances can lead to bioaccumulation in organisms and disrupt ecological balances. Particularly concerning are heavy metal standards used for calibration, which pose long-term environmental persistence issues.
Recent advancements in green analytical chemistry offer promising alternatives with reduced environmental footprints. Miniaturization through microfluidic devices significantly decreases reagent consumption and waste generation. Solvent-free techniques and the replacement of hazardous chemicals with environmentally benign alternatives represent important progress toward sustainable analytical practices.
Life cycle assessment (LCA) studies indicate that implementing recycling systems for analytical waste can reduce the environmental impact by up to 40%. Furthermore, transitioning to renewable energy sources for laboratory operations could decrease the carbon footprint of lithium acetate purity analysis by approximately 60%, highlighting the importance of holistic approaches to environmental impact mitigation in analytical processes.
Solvent usage represents a major environmental concern in these analytical processes. Many purity assessment techniques require organic solvents like acetonitrile, methanol, and hexane, which are derived from non-renewable petroleum resources. These solvents often possess high volatility, contributing to air pollution and potential ozone depletion when released into the atmosphere. Additionally, their production and disposal generate significant carbon footprints.
Water consumption in analytical laboratories presents another environmental challenge. Techniques such as ion chromatography and titration methods require large volumes of ultrapure water, which necessitates energy-intensive purification processes. The resulting wastewater frequently contains trace amounts of lithium compounds and other chemicals that conventional treatment facilities may not effectively remove before discharge.
Energy utilization across the analytical lifecycle constitutes a substantial environmental factor. Instruments like mass spectrometers and spectrophotometers demand continuous power during operation and often require climate-controlled environments, further increasing energy consumption. This energy demand translates to greenhouse gas emissions when derived from fossil fuel sources, contributing to climate change impacts.
Chemical reagents employed in lithium acetate analysis, including acids, bases, and indicators, present toxicity concerns for aquatic ecosystems. Improper disposal of these substances can lead to bioaccumulation in organisms and disrupt ecological balances. Particularly concerning are heavy metal standards used for calibration, which pose long-term environmental persistence issues.
Recent advancements in green analytical chemistry offer promising alternatives with reduced environmental footprints. Miniaturization through microfluidic devices significantly decreases reagent consumption and waste generation. Solvent-free techniques and the replacement of hazardous chemicals with environmentally benign alternatives represent important progress toward sustainable analytical practices.
Life cycle assessment (LCA) studies indicate that implementing recycling systems for analytical waste can reduce the environmental impact by up to 40%. Furthermore, transitioning to renewable energy sources for laboratory operations could decrease the carbon footprint of lithium acetate purity analysis by approximately 60%, highlighting the importance of holistic approaches to environmental impact mitigation in analytical processes.
Unlock deeper insights with Patsnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with Patsnap Eureka AI Agent Platform!