Liquid Nitrogen vs Nitrous Oxide: Thermal Expansion Differences
OCT 7, 202510 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Cryogenic Fluids Background and Research Objectives
Cryogenic fluids have been integral to numerous industrial and scientific applications since their discovery and commercial production in the late 19th century. Liquid nitrogen (LN2), with a boiling point of -196°C, emerged as one of the earliest widely used cryogenic fluids following air liquefaction processes developed by Carl von Linde in 1895. Nitrous oxide (N2O), though less commonly categorized as a true cryogenic fluid due to its higher boiling point of -88.5°C, shares many handling characteristics and applications with traditional cryogens.
The thermal expansion properties of these substances represent a critical area of study due to their significant impact on storage systems, transfer mechanisms, and safety protocols. Historically, the understanding of thermal expansion differences between liquid nitrogen and nitrous oxide has evolved from empirical observations to sophisticated thermodynamic modeling. This evolution has paralleled advancements in materials science, which has enabled the development of containment systems capable of withstanding extreme temperature gradients.
Current technological trends indicate growing precision requirements in cryogenic applications across multiple industries. In semiconductor manufacturing, nanometer-scale process control demands unprecedented understanding of thermal expansion behaviors. Similarly, in quantum computing, maintaining stable qubit states requires exquisite temperature management where even minor thermal fluctuations can compromise system integrity.
The medical field presents another frontier where these cryogenic fluids are increasingly utilized, from cryosurgery to biological sample preservation. Each application demands specific thermal expansion knowledge to ensure optimal performance and safety. The aerospace industry similarly relies on precise cryogenic fluid management for propulsion systems and satellite operations.
This research aims to establish a comprehensive comparative analysis of the thermal expansion characteristics of liquid nitrogen versus nitrous oxide across their entire usable temperature ranges. Specifically, we seek to quantify expansion coefficients under varying pressure conditions, develop predictive models for phase-change behaviors during rapid temperature transitions, and identify critical threshold points where expansion properties significantly diverge.
Additionally, this investigation will explore how these thermal expansion differences manifest in practical applications, particularly focusing on containment system design, transfer line engineering, and safety protocol development. By understanding these fundamental differences, we aim to establish design principles that can accommodate the unique behaviors of each fluid.
The ultimate objective is to develop a unified framework for cryogenic fluid management that accounts for the distinct thermal expansion properties of liquid nitrogen and nitrous oxide, enabling more efficient, safer, and more reliable cryogenic systems across industrial, medical, and scientific applications. This framework will serve as a foundation for next-generation cryogenic technologies that can precisely leverage the unique properties of each fluid.
The thermal expansion properties of these substances represent a critical area of study due to their significant impact on storage systems, transfer mechanisms, and safety protocols. Historically, the understanding of thermal expansion differences between liquid nitrogen and nitrous oxide has evolved from empirical observations to sophisticated thermodynamic modeling. This evolution has paralleled advancements in materials science, which has enabled the development of containment systems capable of withstanding extreme temperature gradients.
Current technological trends indicate growing precision requirements in cryogenic applications across multiple industries. In semiconductor manufacturing, nanometer-scale process control demands unprecedented understanding of thermal expansion behaviors. Similarly, in quantum computing, maintaining stable qubit states requires exquisite temperature management where even minor thermal fluctuations can compromise system integrity.
The medical field presents another frontier where these cryogenic fluids are increasingly utilized, from cryosurgery to biological sample preservation. Each application demands specific thermal expansion knowledge to ensure optimal performance and safety. The aerospace industry similarly relies on precise cryogenic fluid management for propulsion systems and satellite operations.
This research aims to establish a comprehensive comparative analysis of the thermal expansion characteristics of liquid nitrogen versus nitrous oxide across their entire usable temperature ranges. Specifically, we seek to quantify expansion coefficients under varying pressure conditions, develop predictive models for phase-change behaviors during rapid temperature transitions, and identify critical threshold points where expansion properties significantly diverge.
Additionally, this investigation will explore how these thermal expansion differences manifest in practical applications, particularly focusing on containment system design, transfer line engineering, and safety protocol development. By understanding these fundamental differences, we aim to establish design principles that can accommodate the unique behaviors of each fluid.
The ultimate objective is to develop a unified framework for cryogenic fluid management that accounts for the distinct thermal expansion properties of liquid nitrogen and nitrous oxide, enabling more efficient, safer, and more reliable cryogenic systems across industrial, medical, and scientific applications. This framework will serve as a foundation for next-generation cryogenic technologies that can precisely leverage the unique properties of each fluid.
Market Applications and Demand Analysis for Cryogenic Fluids
The global cryogenic fluids market has been experiencing robust growth, driven primarily by expanding applications across multiple industries. The market value reached approximately $16.5 billion in 2022 and is projected to grow at a CAGR of 6.8% through 2030, with liquid nitrogen representing the largest segment at roughly 40% of the total market share. Nitrous oxide, while smaller in market volume, maintains significant presence in specialized applications, particularly in medical and food industries.
Healthcare sector remains the dominant consumer of cryogenic fluids, accounting for nearly 35% of global demand. Liquid nitrogen's extensive use in cryopreservation of biological samples, cryosurgery, and dermatological treatments has established it as the preferred cryogenic medium in medical applications. Meanwhile, nitrous oxide continues to serve as an important anesthetic agent, with global medical consumption exceeding 16,000 metric tons annually.
The food and beverage industry represents the second-largest market segment, where both substances find distinct applications. Liquid nitrogen's rapid freezing capabilities make it ideal for flash-freezing premium food products and creating novel culinary experiences, while nitrous oxide dominates the whipped cream dispensing market with annual consumption exceeding 8,000 metric tons in food applications alone.
Industrial manufacturing constitutes another significant demand driver, particularly for liquid nitrogen, which is extensively used in metal processing, electronics manufacturing, and as an industrial coolant. The semiconductor industry alone consumes approximately 12% of global liquid nitrogen production for cooling applications during manufacturing processes.
Regional analysis reveals North America and Europe as the largest markets for both cryogenic fluids, collectively accounting for over 60% of global consumption. However, the Asia-Pacific region demonstrates the highest growth rate at 8.2% annually, driven by rapid industrialization in China and India, and expanding healthcare infrastructure.
The differential thermal expansion properties between liquid nitrogen and nitrous oxide significantly influence their market applications. Liquid nitrogen's extremely low boiling point (-196°C) and minimal expansion during phase transition make it preferable for precision cooling applications in electronics and superconductivity research. Conversely, nitrous oxide's higher boiling point (-88.5°C) and greater thermal expansion coefficient create specific advantages in applications requiring controlled pressure generation, such as certain food processing techniques and automotive performance enhancement.
Market forecasts indicate specialized applications leveraging the unique thermal expansion characteristics of each fluid will drive premium segment growth, with projected value increases of 9.3% annually for specialized applications versus 5.7% for conventional uses through 2028.
Healthcare sector remains the dominant consumer of cryogenic fluids, accounting for nearly 35% of global demand. Liquid nitrogen's extensive use in cryopreservation of biological samples, cryosurgery, and dermatological treatments has established it as the preferred cryogenic medium in medical applications. Meanwhile, nitrous oxide continues to serve as an important anesthetic agent, with global medical consumption exceeding 16,000 metric tons annually.
The food and beverage industry represents the second-largest market segment, where both substances find distinct applications. Liquid nitrogen's rapid freezing capabilities make it ideal for flash-freezing premium food products and creating novel culinary experiences, while nitrous oxide dominates the whipped cream dispensing market with annual consumption exceeding 8,000 metric tons in food applications alone.
Industrial manufacturing constitutes another significant demand driver, particularly for liquid nitrogen, which is extensively used in metal processing, electronics manufacturing, and as an industrial coolant. The semiconductor industry alone consumes approximately 12% of global liquid nitrogen production for cooling applications during manufacturing processes.
Regional analysis reveals North America and Europe as the largest markets for both cryogenic fluids, collectively accounting for over 60% of global consumption. However, the Asia-Pacific region demonstrates the highest growth rate at 8.2% annually, driven by rapid industrialization in China and India, and expanding healthcare infrastructure.
The differential thermal expansion properties between liquid nitrogen and nitrous oxide significantly influence their market applications. Liquid nitrogen's extremely low boiling point (-196°C) and minimal expansion during phase transition make it preferable for precision cooling applications in electronics and superconductivity research. Conversely, nitrous oxide's higher boiling point (-88.5°C) and greater thermal expansion coefficient create specific advantages in applications requiring controlled pressure generation, such as certain food processing techniques and automotive performance enhancement.
Market forecasts indicate specialized applications leveraging the unique thermal expansion characteristics of each fluid will drive premium segment growth, with projected value increases of 9.3% annually for specialized applications versus 5.7% for conventional uses through 2028.
Current Technical Challenges in Thermal Expansion Management
Managing thermal expansion differences between liquid nitrogen and nitrous oxide presents significant technical challenges due to their distinct physical properties. Liquid nitrogen, with a boiling point of -196°C, exhibits minimal thermal expansion until it approaches its phase change, while nitrous oxide, boiling at -88.5°C, demonstrates more pronounced expansion characteristics across its usable temperature range. This fundamental difference creates complexities in containment system design and material selection.
The primary challenge lies in developing containment vessels that can accommodate these different expansion behaviors without compromising structural integrity. Materials used must maintain their mechanical properties at cryogenic temperatures while withstanding the stress induced by thermal cycling. Traditional metals like stainless steel become brittle at extreme low temperatures, while specialized alloys add significant cost to system design.
Sealing systems represent another critical challenge area. The substantial temperature differentials between ambient conditions and operating temperatures create significant contraction in sealing materials, potentially leading to leakage. Current elastomeric seals often fail to maintain their flexibility and sealing properties across the wide temperature ranges required, particularly with liquid nitrogen applications.
Thermal insulation systems must balance competing requirements of minimizing heat transfer while accommodating differential expansion. Vacuum-jacketed vessels with multi-layer insulation remain the gold standard but present manufacturing complexities and increased costs. The insulation efficiency directly impacts boil-off rates, which differ significantly between the two cryogens due to their different latent heats of vaporization.
Pressure management systems face unique challenges with each cryogen. Liquid nitrogen's extremely low temperature creates rapid pressure buildup during warming, while nitrous oxide's higher vapor pressure at equivalent temperatures requires more robust pressure relief systems. Current pressure relief technologies often struggle to provide consistent performance across the temperature ranges experienced during normal operation and potential fault conditions.
Instrumentation and monitoring systems face reliability issues in cryogenic environments. Sensors must function accurately at extreme temperatures while accommodating the physical movement caused by thermal expansion and contraction. Current sensor technologies often demonstrate drift or complete failure when exposed to rapid temperature changes, particularly in liquid nitrogen applications.
Transfer systems for both cryogens must address the challenges of thermal shock and expansion during cool-down operations. Rapid cooling of warm transfer lines can induce significant thermal stress, leading to premature component failure. Current quick-connect couplings often leak during thermal cycling, creating both safety hazards and product loss.
The primary challenge lies in developing containment vessels that can accommodate these different expansion behaviors without compromising structural integrity. Materials used must maintain their mechanical properties at cryogenic temperatures while withstanding the stress induced by thermal cycling. Traditional metals like stainless steel become brittle at extreme low temperatures, while specialized alloys add significant cost to system design.
Sealing systems represent another critical challenge area. The substantial temperature differentials between ambient conditions and operating temperatures create significant contraction in sealing materials, potentially leading to leakage. Current elastomeric seals often fail to maintain their flexibility and sealing properties across the wide temperature ranges required, particularly with liquid nitrogen applications.
Thermal insulation systems must balance competing requirements of minimizing heat transfer while accommodating differential expansion. Vacuum-jacketed vessels with multi-layer insulation remain the gold standard but present manufacturing complexities and increased costs. The insulation efficiency directly impacts boil-off rates, which differ significantly between the two cryogens due to their different latent heats of vaporization.
Pressure management systems face unique challenges with each cryogen. Liquid nitrogen's extremely low temperature creates rapid pressure buildup during warming, while nitrous oxide's higher vapor pressure at equivalent temperatures requires more robust pressure relief systems. Current pressure relief technologies often struggle to provide consistent performance across the temperature ranges experienced during normal operation and potential fault conditions.
Instrumentation and monitoring systems face reliability issues in cryogenic environments. Sensors must function accurately at extreme temperatures while accommodating the physical movement caused by thermal expansion and contraction. Current sensor technologies often demonstrate drift or complete failure when exposed to rapid temperature changes, particularly in liquid nitrogen applications.
Transfer systems for both cryogens must address the challenges of thermal shock and expansion during cool-down operations. Rapid cooling of warm transfer lines can induce significant thermal stress, leading to premature component failure. Current quick-connect couplings often leak during thermal cycling, creating both safety hazards and product loss.
Comparative Analysis of LN2 and N2O Thermal Properties
01 Thermal expansion properties of cryogenic fluids
Liquid nitrogen and nitrous oxide exhibit unique thermal expansion characteristics when transitioning from liquid to gas states. These cryogenic fluids undergo significant volume expansion when heated, which is crucial for various industrial applications. The thermal expansion coefficients differ between these substances, with liquid nitrogen expanding approximately 700 times its volume when vaporized, while nitrous oxide has different expansion properties that affect its handling and storage requirements.- Thermal expansion properties of cryogenic fluids: The thermal expansion characteristics of liquid nitrogen and nitrous oxide are critical in various applications. These cryogenic fluids exhibit significant volume changes during phase transitions from liquid to gas. Understanding these expansion properties is essential for designing storage systems, transfer equipment, and safety mechanisms. The thermal expansion coefficients differ substantially between liquid nitrogen (-196°C) and nitrous oxide (-88.5°C), affecting their behavior in pressurized systems.
- Pressure management systems for cryogenic fluids: Specialized pressure management systems are required to handle the thermal expansion of liquid nitrogen and nitrous oxide. These systems include pressure relief valves, expansion chambers, and controlled venting mechanisms to prevent dangerous pressure buildup. The significant volume expansion ratio when these cryogenic liquids convert to gas (approximately 1:700 for liquid nitrogen) necessitates careful engineering of containment vessels and transfer lines to accommodate pressure fluctuations during temperature changes.
- Applications utilizing thermal expansion properties: The thermal expansion properties of liquid nitrogen and nitrous oxide are leveraged in various industrial and medical applications. These include cryogenic cooling systems, food freezing processes, medical tissue preservation, and specialized manufacturing techniques. The controlled expansion of these cryogenic fluids enables precise temperature regulation in sensitive processes. Additionally, the rapid expansion properties are utilized in certain propulsion systems, pressure-driven mechanisms, and specialized extraction techniques.
- Safety considerations for handling expanding cryogenic fluids: Safety protocols are essential when working with liquid nitrogen and nitrous oxide due to their significant thermal expansion properties. Rapid expansion can create oxygen-deficient atmospheres, cause pressure vessel failures, or result in cryogenic burns. Engineering controls include proper insulation, pressure relief systems, oxygen monitoring, and specialized containment vessels designed to withstand the stresses of thermal cycling. Personnel training and protective equipment requirements are specified to mitigate risks associated with the unpredictable expansion behavior of these cryogenic fluids.
- Measurement and control systems for thermal expansion: Advanced measurement and control systems are employed to monitor and manage the thermal expansion of liquid nitrogen and nitrous oxide. These systems incorporate temperature sensors, pressure transducers, flow meters, and computerized control algorithms to maintain safe operating conditions. Real-time monitoring allows for predictive management of expansion events, while automated safety systems can initiate pressure relief or emergency shutdown procedures when expansion parameters exceed predetermined thresholds. These control systems are critical in applications where precise management of the cryogenic fluid state is required.
02 Pressure management systems for cryogenic fluids
Specialized pressure management systems are essential for handling the thermal expansion of liquid nitrogen and nitrous oxide. These systems include pressure relief valves, expansion chambers, and controlled venting mechanisms to prevent dangerous pressure buildup during temperature changes. The design of these systems must account for the rapid expansion rates of these cryogenic fluids to ensure safe operation in various applications, from medical devices to industrial processes.Expand Specific Solutions03 Temperature control mechanisms for expansion management
Advanced temperature control mechanisms are employed to manage the thermal expansion of liquid nitrogen and nitrous oxide. These systems utilize precise temperature sensors, insulation techniques, and heat exchange methods to maintain optimal conditions and control expansion rates. By regulating temperature gradients and heat transfer, these mechanisms prevent uncontrolled expansion and ensure efficient operation in applications ranging from cryotherapy to food freezing processes.Expand Specific Solutions04 Container and storage vessel design for expansion accommodation
Specialized containers and storage vessels are designed to accommodate the thermal expansion properties of liquid nitrogen and nitrous oxide. These designs incorporate expansion spaces, flexible components, and reinforced structures to withstand pressure changes during temperature fluctuations. The materials used must maintain structural integrity at extremely low temperatures while providing sufficient safety margins for the substantial volume changes that occur during phase transitions.Expand Specific Solutions05 Applications utilizing controlled thermal expansion
Various applications leverage the controlled thermal expansion properties of liquid nitrogen and nitrous oxide. These include cryogenic cooling systems, medical devices, food processing equipment, and specialized industrial processes. By harnessing the predictable expansion characteristics of these fluids, engineers develop efficient systems for rapid cooling, pressure generation, and mechanical actuation. The significant volume change during phase transition provides energy that can be captured and utilized in numerous innovative applications.Expand Specific Solutions
Leading Companies and Research Institutions in Cryogenics
The thermal expansion differences between liquid nitrogen and nitrous oxide represent a critical area of study in cryogenic applications, currently in a growth phase with an estimated market size of $20-25 billion. The technology is approaching maturity with significant advancements from key players. Air Liquide and Linde GmbH lead industrial gas applications, while semiconductor manufacturers like Samsung Electronics, SK Hynix, and KIOXIA are driving innovation in electronics cooling. NITROcrete has pioneered specialized concrete applications, while Air Products & Chemicals and Hangzhou Oxygen Plant Group are advancing equipment design. Research institutions including Technical University of Denmark and Maastricht University contribute fundamental thermal expansion studies, creating a competitive landscape balanced between established industrial giants and specialized niche innovators.
Air Liquide SA
Technical Solution: Air Liquide has developed comprehensive thermal expansion management systems specifically addressing the differences between liquid nitrogen (LN2) and nitrous oxide (N2O). Their technology utilizes proprietary cryogenic vessel designs with multi-layered insulation systems that accommodate the significantly different thermal expansion coefficients - LN2 expanding approximately 696 times when converting from liquid to gas at room temperature, while N2O expands about 666 times. The company's Dual-Phase Expansion Control (DPEC) technology incorporates variable-geometry pressure relief systems calibrated to the specific expansion characteristics of each cryogen, preventing pressure buildup during phase transitions. Their advanced monitoring systems track temperature gradients across storage vessels in real-time, automatically adjusting pressure release mechanisms based on the specific thermal expansion properties of the contained cryogen.
Strengths: Industry-leading expertise in cryogenic fluid management with proprietary vessel designs specifically engineered for different thermal expansion behaviors. Weaknesses: Their solutions typically require significant infrastructure investment and specialized maintenance protocols, limiting accessibility for smaller operations.
Air Products & Chemicals, Inc.
Technical Solution: Air Products has pioneered differential thermal expansion compensation technology for cryogenic applications, specifically addressing the unique challenges posed by liquid nitrogen and nitrous oxide. Their CryoExpand™ system incorporates specialized materials with carefully calculated expansion joints that accommodate the 696:1 volume expansion ratio of liquid nitrogen compared to the approximately 666:1 ratio of nitrous oxide when transitioning from liquid to gas phase at standard temperature. The company's approach includes precision-engineered storage and delivery systems with variable-geometry internal components that automatically adjust to the different expansion behaviors of these cryogens. Their technology incorporates real-time monitoring of thermal gradients and expansion rates, with predictive algorithms that anticipate pressure changes based on the specific thermodynamic properties of each substance, enabling safe and efficient handling across diverse industrial applications.
Strengths: Comprehensive engineering approach that addresses both material selection and system design to manage different thermal expansion behaviors. Weaknesses: Solutions tend to be highly specialized for specific applications, potentially limiting cross-industry adaptability.
Key Scientific Principles Behind Thermal Expansion Differences
Flow meter used for metering the energy applied to a method employing a cryogenic fluid
PatentActiveUS20180340810A1
Innovation
- Separate the gas and liquid phases of the cryogenic fluid before injection, measure the mass flow rate of each phase, and calculate the associated energy flow rates considering variations in enthalpy and temperature, then recombine them for precise energy delivery to the downstream process.
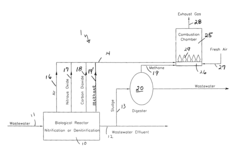
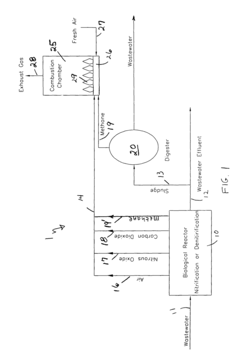
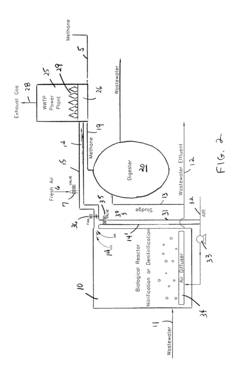
Nitrous oxide reduction from biological wastewater treatment processes
PatentInactiveUS8157993B1
Innovation
- Adapting nitrous oxide technology from high-performance racing engines by breaking down nitrous oxide into nitrogen and oxygen, which is then recirculated and used in the combustion chamber of methane gas generators at wastewater treatment plants, reducing thermal NOx production and eliminating atmospheric emissions.
Safety Protocols and Risk Management for Cryogenic Applications
Working with cryogenic substances requires comprehensive safety protocols due to their extreme temperatures and unique physical properties. When comparing liquid nitrogen (-196°C) and nitrous oxide (-88.5°C), their different thermal expansion characteristics create distinct safety challenges. Liquid nitrogen expands approximately 700 times when transitioning from liquid to gas, while nitrous oxide expands about 300 times, necessitating different containment strategies and pressure relief mechanisms.
Risk assessment for cryogenic applications must account for these expansion differences. Facilities handling liquid nitrogen require more robust pressure relief systems and larger-capacity venting mechanisms to accommodate its greater expansion potential. Conversely, nitrous oxide systems, while requiring less expansion capacity, present additional risks due to its oxidizing properties that can intensify combustion processes.
Personal protective equipment requirements vary slightly between these substances. Both require insulated gloves, face shields, and appropriate footwear, but the extreme cold of liquid nitrogen demands materials rated for lower temperatures. Oxygen monitoring becomes critical in enclosed spaces for both substances, as they can displace oxygen and create asphyxiation hazards, though liquid nitrogen presents a greater risk due to its higher expansion ratio.
Emergency response protocols must be tailored to each substance's properties. Liquid nitrogen spills require immediate evacuation of larger areas due to the rapid volume expansion and extreme cold. Nitrous oxide incidents necessitate additional precautions regarding potential fire acceleration. First aid procedures for cryogenic burns remain similar, though liquid nitrogen exposure typically results in more severe tissue damage due to its lower temperature.
Storage and handling guidelines reflect these differences as well. Liquid nitrogen containers require more frequent pressure checks and more substantial insulation. The Dewar vessels used must accommodate greater pressure buildup and have appropriate venting mechanisms. Nitrous oxide storage, while still requiring specialized equipment, can utilize systems with lower pressure ratings but must incorporate safeguards against oxidizer-related hazards.
Training programs for personnel working with these cryogens should emphasize their distinct properties. Staff must understand the specific expansion characteristics and associated risks of each substance. Simulation exercises should include scenarios reflecting the different emergency response requirements for liquid nitrogen's extreme cold and greater expansion versus nitrous oxide's moderate expansion and oxidizing properties.
Regulatory compliance frameworks differ slightly between these substances, with liquid nitrogen often subject to stricter pressure vessel regulations due to its greater expansion potential, while nitrous oxide may face additional scrutiny under regulations governing oxidizing agents and controlled substances in certain jurisdictions.
Risk assessment for cryogenic applications must account for these expansion differences. Facilities handling liquid nitrogen require more robust pressure relief systems and larger-capacity venting mechanisms to accommodate its greater expansion potential. Conversely, nitrous oxide systems, while requiring less expansion capacity, present additional risks due to its oxidizing properties that can intensify combustion processes.
Personal protective equipment requirements vary slightly between these substances. Both require insulated gloves, face shields, and appropriate footwear, but the extreme cold of liquid nitrogen demands materials rated for lower temperatures. Oxygen monitoring becomes critical in enclosed spaces for both substances, as they can displace oxygen and create asphyxiation hazards, though liquid nitrogen presents a greater risk due to its higher expansion ratio.
Emergency response protocols must be tailored to each substance's properties. Liquid nitrogen spills require immediate evacuation of larger areas due to the rapid volume expansion and extreme cold. Nitrous oxide incidents necessitate additional precautions regarding potential fire acceleration. First aid procedures for cryogenic burns remain similar, though liquid nitrogen exposure typically results in more severe tissue damage due to its lower temperature.
Storage and handling guidelines reflect these differences as well. Liquid nitrogen containers require more frequent pressure checks and more substantial insulation. The Dewar vessels used must accommodate greater pressure buildup and have appropriate venting mechanisms. Nitrous oxide storage, while still requiring specialized equipment, can utilize systems with lower pressure ratings but must incorporate safeguards against oxidizer-related hazards.
Training programs for personnel working with these cryogens should emphasize their distinct properties. Staff must understand the specific expansion characteristics and associated risks of each substance. Simulation exercises should include scenarios reflecting the different emergency response requirements for liquid nitrogen's extreme cold and greater expansion versus nitrous oxide's moderate expansion and oxidizing properties.
Regulatory compliance frameworks differ slightly between these substances, with liquid nitrogen often subject to stricter pressure vessel regulations due to its greater expansion potential, while nitrous oxide may face additional scrutiny under regulations governing oxidizing agents and controlled substances in certain jurisdictions.
Environmental Impact and Sustainability Considerations
The environmental impact of cryogenic fluids like liquid nitrogen and nitrous oxide extends far beyond their immediate applications, encompassing production, transportation, usage, and disposal phases. Liquid nitrogen, primarily produced through air separation processes, requires significant energy input but leaves minimal direct environmental footprint when released, as nitrogen constitutes approximately 78% of Earth's atmosphere. Its production carbon footprint varies between 0.4-1.8 kg CO2e per kilogram, depending on energy sources used in the separation process.
Nitrous oxide presents more complex environmental considerations. While also produced industrially, N2O is a potent greenhouse gas with a global warming potential approximately 298 times that of carbon dioxide over a 100-year period. Accidental releases during handling or intentional venting contribute to atmospheric concentrations, with N2O remaining in the atmosphere for an average of 114 years. Additionally, nitrous oxide contributes to stratospheric ozone depletion, further complicating its environmental profile.
The thermal expansion differences between these substances influence their sustainability profiles. Liquid nitrogen's higher expansion ratio (1:694) compared to nitrous oxide (1:437) means more gaseous product per unit of liquid, potentially reducing transportation emissions. However, this same property necessitates more robust containment systems, requiring additional materials and energy in infrastructure development.
Energy efficiency considerations favor liquid nitrogen in many applications. Its lower boiling point (-196°C vs -88.5°C for N2O) makes it more energy-intensive to produce initially, but its inert nature eliminates many environmental risks associated with nitrous oxide. Modern liquid nitrogen production facilities have implemented significant efficiency improvements, reducing energy consumption by approximately 30% over the past two decades.
Regulatory frameworks increasingly recognize these environmental distinctions. The Kigali Amendment to the Montreal Protocol does not directly regulate nitrogen but places restrictions on nitrous oxide due to its ozone-depleting potential. Similarly, carbon pricing mechanisms in various jurisdictions may affect the economic viability of nitrous oxide applications where alternatives exist.
Lifecycle assessment studies indicate that applications leveraging the thermal expansion properties of these substances should consider not only immediate performance characteristics but also broader environmental impacts. For temperature-sensitive applications where either substance might be suitable, liquid nitrogen increasingly represents the more sustainable choice despite higher initial production energy requirements.
Nitrous oxide presents more complex environmental considerations. While also produced industrially, N2O is a potent greenhouse gas with a global warming potential approximately 298 times that of carbon dioxide over a 100-year period. Accidental releases during handling or intentional venting contribute to atmospheric concentrations, with N2O remaining in the atmosphere for an average of 114 years. Additionally, nitrous oxide contributes to stratospheric ozone depletion, further complicating its environmental profile.
The thermal expansion differences between these substances influence their sustainability profiles. Liquid nitrogen's higher expansion ratio (1:694) compared to nitrous oxide (1:437) means more gaseous product per unit of liquid, potentially reducing transportation emissions. However, this same property necessitates more robust containment systems, requiring additional materials and energy in infrastructure development.
Energy efficiency considerations favor liquid nitrogen in many applications. Its lower boiling point (-196°C vs -88.5°C for N2O) makes it more energy-intensive to produce initially, but its inert nature eliminates many environmental risks associated with nitrous oxide. Modern liquid nitrogen production facilities have implemented significant efficiency improvements, reducing energy consumption by approximately 30% over the past two decades.
Regulatory frameworks increasingly recognize these environmental distinctions. The Kigali Amendment to the Montreal Protocol does not directly regulate nitrogen but places restrictions on nitrous oxide due to its ozone-depleting potential. Similarly, carbon pricing mechanisms in various jurisdictions may affect the economic viability of nitrous oxide applications where alternatives exist.
Lifecycle assessment studies indicate that applications leveraging the thermal expansion properties of these substances should consider not only immediate performance characteristics but also broader environmental impacts. For temperature-sensitive applications where either substance might be suitable, liquid nitrogen increasingly represents the more sustainable choice despite higher initial production energy requirements.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!