Optimize Ethyl Acetate Usage in Pharmaceutical Extractions
MAR 23, 20269 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
Ethyl Acetate Extraction Background and Optimization Goals
Ethyl acetate has emerged as a cornerstone solvent in pharmaceutical extraction processes due to its unique combination of moderate polarity, low toxicity, and favorable regulatory status. This organic solvent, classified as Class 3 by the International Council for Harmonisation (ICH), exhibits excellent selectivity for extracting active pharmaceutical ingredients (APIs) from complex matrices while maintaining compatibility with downstream purification processes. Its widespread adoption stems from the critical need to balance extraction efficiency with safety requirements in pharmaceutical manufacturing.
The pharmaceutical industry's reliance on ethyl acetate has intensified over the past two decades, driven by increasingly stringent regulatory frameworks and the growing emphasis on green chemistry principles. Traditional extraction solvents such as dichloromethane and chloroform face mounting restrictions due to environmental and health concerns, positioning ethyl acetate as a preferred alternative. Its biodegradable nature and relatively low environmental impact align with the industry's sustainability objectives while meeting the rigorous purity standards required for pharmaceutical applications.
Current extraction processes utilizing ethyl acetate face significant challenges in achieving optimal solvent utilization. Conventional methods often employ excessive solvent volumes to ensure complete extraction, resulting in substantial waste generation and increased operational costs. The lack of systematic optimization approaches has led to inefficient processes where solvent consumption can exceed theoretical requirements by 200-300%, directly impacting both economic viability and environmental footprint.
The primary optimization objectives center on developing methodologies to minimize ethyl acetate consumption while maintaining or enhancing extraction yields. This involves establishing precise solvent-to-feed ratios, optimizing contact times, and implementing advanced process control strategies. Secondary goals include reducing solvent losses through improved recovery systems and developing predictive models to determine optimal extraction parameters for different pharmaceutical compounds.
Technological advancement in this field aims to achieve a 40-60% reduction in ethyl acetate usage through process intensification techniques, real-time monitoring systems, and enhanced separation technologies. The integration of continuous extraction processes and automated solvent recovery systems represents key pathways toward achieving these ambitious targets while ensuring consistent product quality and regulatory compliance.
The pharmaceutical industry's reliance on ethyl acetate has intensified over the past two decades, driven by increasingly stringent regulatory frameworks and the growing emphasis on green chemistry principles. Traditional extraction solvents such as dichloromethane and chloroform face mounting restrictions due to environmental and health concerns, positioning ethyl acetate as a preferred alternative. Its biodegradable nature and relatively low environmental impact align with the industry's sustainability objectives while meeting the rigorous purity standards required for pharmaceutical applications.
Current extraction processes utilizing ethyl acetate face significant challenges in achieving optimal solvent utilization. Conventional methods often employ excessive solvent volumes to ensure complete extraction, resulting in substantial waste generation and increased operational costs. The lack of systematic optimization approaches has led to inefficient processes where solvent consumption can exceed theoretical requirements by 200-300%, directly impacting both economic viability and environmental footprint.
The primary optimization objectives center on developing methodologies to minimize ethyl acetate consumption while maintaining or enhancing extraction yields. This involves establishing precise solvent-to-feed ratios, optimizing contact times, and implementing advanced process control strategies. Secondary goals include reducing solvent losses through improved recovery systems and developing predictive models to determine optimal extraction parameters for different pharmaceutical compounds.
Technological advancement in this field aims to achieve a 40-60% reduction in ethyl acetate usage through process intensification techniques, real-time monitoring systems, and enhanced separation technologies. The integration of continuous extraction processes and automated solvent recovery systems represents key pathways toward achieving these ambitious targets while ensuring consistent product quality and regulatory compliance.
Market Demand for Efficient Pharmaceutical Extraction Processes
The pharmaceutical industry faces mounting pressure to enhance extraction efficiency while reducing environmental impact and operational costs. Traditional extraction processes often rely heavily on organic solvents like ethyl acetate, creating significant demand for optimization technologies that can minimize solvent consumption without compromising product quality or yield.
Regulatory frameworks worldwide are tightening environmental standards for pharmaceutical manufacturing, driving companies to seek greener extraction alternatives. The European Medicines Agency and FDA have implemented stricter guidelines on residual solvent limits, particularly for Class 3 solvents like ethyl acetate. This regulatory landscape creates substantial market pull for technologies that can optimize solvent usage while maintaining compliance with pharmaceutical quality standards.
The global pharmaceutical extraction equipment market demonstrates robust growth, fueled by increasing drug development activities and the need for more efficient manufacturing processes. Generic drug manufacturers, facing intense price competition, are particularly motivated to adopt cost-effective extraction optimization technologies. These companies represent a significant market segment seeking solutions that can reduce raw material costs while maintaining product specifications.
Biopharmaceutical companies developing complex molecules and natural product extracts constitute another key market driver. These organizations require sophisticated extraction processes that can handle diverse chemical compounds while minimizing solvent waste. The growing trend toward personalized medicine and specialty pharmaceuticals further amplifies demand for flexible, efficient extraction systems.
Contract manufacturing organizations increasingly seek competitive advantages through process optimization capabilities. Their clients demand cost-effective manufacturing solutions, making ethyl acetate optimization technologies attractive investments. The outsourcing trend in pharmaceutical manufacturing expands the addressable market for extraction efficiency solutions.
Sustainability initiatives within pharmaceutical companies create additional market momentum. Corporate environmental responsibility programs and carbon footprint reduction goals align with ethyl acetate optimization objectives. Companies are investing in green chemistry initiatives that reduce solvent consumption and waste generation.
The market also responds to supply chain vulnerabilities exposed during recent global disruptions. Pharmaceutical manufacturers recognize that optimizing solvent usage reduces dependency on chemical suppliers and enhances operational resilience. This strategic consideration adds another dimension to market demand beyond immediate cost savings.
Emerging markets present significant growth opportunities as local pharmaceutical industries expand and modernize their manufacturing capabilities. These regions often prioritize cost-effective technologies that can improve competitiveness while meeting international quality standards.
Regulatory frameworks worldwide are tightening environmental standards for pharmaceutical manufacturing, driving companies to seek greener extraction alternatives. The European Medicines Agency and FDA have implemented stricter guidelines on residual solvent limits, particularly for Class 3 solvents like ethyl acetate. This regulatory landscape creates substantial market pull for technologies that can optimize solvent usage while maintaining compliance with pharmaceutical quality standards.
The global pharmaceutical extraction equipment market demonstrates robust growth, fueled by increasing drug development activities and the need for more efficient manufacturing processes. Generic drug manufacturers, facing intense price competition, are particularly motivated to adopt cost-effective extraction optimization technologies. These companies represent a significant market segment seeking solutions that can reduce raw material costs while maintaining product specifications.
Biopharmaceutical companies developing complex molecules and natural product extracts constitute another key market driver. These organizations require sophisticated extraction processes that can handle diverse chemical compounds while minimizing solvent waste. The growing trend toward personalized medicine and specialty pharmaceuticals further amplifies demand for flexible, efficient extraction systems.
Contract manufacturing organizations increasingly seek competitive advantages through process optimization capabilities. Their clients demand cost-effective manufacturing solutions, making ethyl acetate optimization technologies attractive investments. The outsourcing trend in pharmaceutical manufacturing expands the addressable market for extraction efficiency solutions.
Sustainability initiatives within pharmaceutical companies create additional market momentum. Corporate environmental responsibility programs and carbon footprint reduction goals align with ethyl acetate optimization objectives. Companies are investing in green chemistry initiatives that reduce solvent consumption and waste generation.
The market also responds to supply chain vulnerabilities exposed during recent global disruptions. Pharmaceutical manufacturers recognize that optimizing solvent usage reduces dependency on chemical suppliers and enhances operational resilience. This strategic consideration adds another dimension to market demand beyond immediate cost savings.
Emerging markets present significant growth opportunities as local pharmaceutical industries expand and modernize their manufacturing capabilities. These regions often prioritize cost-effective technologies that can improve competitiveness while meeting international quality standards.
Current State and Challenges in Ethyl Acetate Usage
Ethyl acetate currently serves as one of the most widely utilized organic solvents in pharmaceutical extraction processes, particularly valued for its moderate polarity, low toxicity profile, and favorable regulatory status. The solvent demonstrates exceptional performance in extracting active pharmaceutical ingredients from natural sources, synthetic intermediates, and complex matrices. Its relatively low boiling point of 77°C facilitates easy recovery through distillation, while its miscibility characteristics enable selective extraction of target compounds.
The pharmaceutical industry's reliance on ethyl acetate has intensified due to stringent environmental regulations that have restricted the use of more hazardous solvents such as dichloromethane and hexane. Current applications span across multiple pharmaceutical sectors, including natural product extraction, API purification, and formulation development. The solvent's compatibility with various extraction techniques, from traditional liquid-liquid extraction to modern supercritical fluid extraction, has established it as a versatile tool in pharmaceutical manufacturing.
Despite its widespread adoption, several critical challenges persist in optimizing ethyl acetate usage. Solvent consumption remains a primary concern, with many pharmaceutical processes exhibiting suboptimal solvent-to-feed ratios that significantly impact operational costs and environmental footprint. Traditional extraction protocols often employ excessive solvent volumes as a safety margin, leading to unnecessary waste generation and increased downstream processing requirements.
Recovery efficiency represents another substantial challenge, as conventional distillation methods typically achieve only 85-90% solvent recovery rates. The remaining 10-15% loss translates to significant economic and environmental costs, particularly in large-scale manufacturing operations. Additionally, the presence of water and other impurities in recovered ethyl acetate often necessitates extensive purification steps, further reducing overall process efficiency.
Process optimization faces technical limitations related to mass transfer kinetics and thermodynamic constraints. Many existing extraction systems operate under non-equilibrium conditions, resulting in incomplete extraction or requiring multiple extraction stages. The lack of real-time monitoring capabilities in most facilities prevents dynamic optimization based on actual process conditions, leading to conservative operating parameters that prioritize safety over efficiency.
Regulatory compliance adds another layer of complexity, as pharmaceutical manufacturers must balance solvent optimization with stringent quality requirements and residual solvent limits. The need to maintain consistent product quality while reducing solvent usage creates operational challenges that require sophisticated process control strategies and comprehensive validation protocols.
The pharmaceutical industry's reliance on ethyl acetate has intensified due to stringent environmental regulations that have restricted the use of more hazardous solvents such as dichloromethane and hexane. Current applications span across multiple pharmaceutical sectors, including natural product extraction, API purification, and formulation development. The solvent's compatibility with various extraction techniques, from traditional liquid-liquid extraction to modern supercritical fluid extraction, has established it as a versatile tool in pharmaceutical manufacturing.
Despite its widespread adoption, several critical challenges persist in optimizing ethyl acetate usage. Solvent consumption remains a primary concern, with many pharmaceutical processes exhibiting suboptimal solvent-to-feed ratios that significantly impact operational costs and environmental footprint. Traditional extraction protocols often employ excessive solvent volumes as a safety margin, leading to unnecessary waste generation and increased downstream processing requirements.
Recovery efficiency represents another substantial challenge, as conventional distillation methods typically achieve only 85-90% solvent recovery rates. The remaining 10-15% loss translates to significant economic and environmental costs, particularly in large-scale manufacturing operations. Additionally, the presence of water and other impurities in recovered ethyl acetate often necessitates extensive purification steps, further reducing overall process efficiency.
Process optimization faces technical limitations related to mass transfer kinetics and thermodynamic constraints. Many existing extraction systems operate under non-equilibrium conditions, resulting in incomplete extraction or requiring multiple extraction stages. The lack of real-time monitoring capabilities in most facilities prevents dynamic optimization based on actual process conditions, leading to conservative operating parameters that prioritize safety over efficiency.
Regulatory compliance adds another layer of complexity, as pharmaceutical manufacturers must balance solvent optimization with stringent quality requirements and residual solvent limits. The need to maintain consistent product quality while reducing solvent usage creates operational challenges that require sophisticated process control strategies and comprehensive validation protocols.
Existing Solutions for Ethyl Acetate Process Optimization
01 Ethyl acetate as solvent in pharmaceutical formulations
Ethyl acetate can be optimized as a solvent in pharmaceutical formulations for drug delivery systems, coating applications, and extraction processes. The optimization focuses on controlling solvent ratios, evaporation rates, and purity levels to enhance drug stability, bioavailability, and manufacturing efficiency. Process parameters such as temperature, pressure, and mixing conditions are adjusted to achieve optimal dissolution and particle size distribution.- Ethyl acetate as solvent in pharmaceutical formulations: Ethyl acetate can be optimized as a solvent in pharmaceutical formulations for drug delivery systems, coating applications, and extraction processes. The optimization focuses on controlling solvent ratios, evaporation rates, and purity levels to enhance drug stability, bioavailability, and manufacturing efficiency. Process parameters such as temperature, pressure, and mixing conditions are adjusted to achieve optimal dissolution and particle size distribution.
- Ethyl acetate recovery and recycling systems: Optimization of ethyl acetate usage through recovery and recycling systems involves implementing distillation, membrane separation, or adsorption technologies to reclaim and purify the solvent for reuse. These systems reduce waste, lower operational costs, and minimize environmental impact. Process optimization includes controlling recovery rates, energy consumption, and maintaining solvent quality throughout multiple cycles.
- Ethyl acetate in coating and film formation processes: Ethyl acetate serves as an optimized solvent in coating applications, including pharmaceutical coatings, food packaging films, and industrial surface treatments. Optimization strategies focus on controlling evaporation kinetics, film uniformity, adhesion properties, and coating thickness. Formulation adjustments involving viscosity modifiers, plasticizers, and co-solvents enhance the performance characteristics of the final coating.
- Ethyl acetate in extraction and purification processes: Optimization of ethyl acetate in extraction processes involves maximizing extraction efficiency, selectivity, and yield for target compounds from natural products, chemical intermediates, or waste streams. Parameters such as solvent-to-feed ratio, extraction time, temperature, and pH are optimized to improve separation performance. Multi-stage extraction and counter-current flow configurations further enhance process efficiency.
- Ethyl acetate usage in chemical synthesis and reaction optimization: Ethyl acetate functions as an optimized reaction medium or co-solvent in various chemical synthesis processes. Optimization involves selecting appropriate concentrations, reaction temperatures, and catalyst systems to maximize yield, selectivity, and reaction rates. The solvent properties of ethyl acetate, including polarity and miscibility, are leveraged to control reaction kinetics and product formation while minimizing side reactions and byproducts.
02 Ethyl acetate recovery and recycling systems
Optimization of ethyl acetate usage through recovery and recycling systems involves implementing distillation, membrane separation, or adsorption technologies to reclaim and purify the solvent for reuse. These systems reduce waste, lower operational costs, and minimize environmental impact. Process optimization includes controlling recovery rates, energy consumption, and maintaining solvent quality throughout multiple cycles.Expand Specific Solutions03 Ethyl acetate in coating and film formation processes
Ethyl acetate serves as an optimized solvent in coating applications, including film formation, surface treatment, and protective layer deposition. Optimization strategies focus on controlling evaporation kinetics, film thickness uniformity, and adhesion properties. Parameters such as concentration, application method, and drying conditions are adjusted to achieve desired coating characteristics and reduce solvent consumption.Expand Specific Solutions04 Ethyl acetate in extraction and purification processes
Optimization of ethyl acetate usage in extraction and purification involves enhancing selectivity, yield, and efficiency in separating target compounds from complex mixtures. This includes optimizing solvent-to-feed ratios, extraction time, temperature, and multi-stage extraction configurations. The approach improves product purity while minimizing solvent consumption and processing time in applications such as natural product extraction and chemical synthesis.Expand Specific Solutions05 Ethyl acetate in chemical synthesis and reaction optimization
Ethyl acetate is optimized as a reaction medium or reagent in chemical synthesis processes. Optimization focuses on reaction kinetics, selectivity, and yield by controlling solvent polarity, reaction temperature, catalyst concentration, and residence time. The optimization also addresses solvent compatibility with reactants and products, minimizing side reactions, and facilitating product isolation and purification steps.Expand Specific Solutions
Key Players in Pharmaceutical Extraction Equipment Industry
The pharmaceutical extraction industry utilizing ethyl acetate is experiencing steady growth driven by increasing demand for natural product isolation and purification processes. The market demonstrates moderate maturity with established players across chemical manufacturing and pharmaceutical sectors. Technology maturity varies significantly among key participants, with specialty chemical manufacturers like Rhodia Operations SASU, Celanese International Corp., and Eastman Chemical Co. leading in advanced solvent production and optimization technologies. Viridis Chemical LLC represents emerging innovation with proprietary catalytic processes for renewable ethyl acetate production. Pharmaceutical companies including Astellas Pharma, Wockhardt Ltd., and Janssen Biotech demonstrate mature extraction applications, while research institutions like Beijing University of Traditional Chinese Medicine and National Taiwan University contribute to process optimization research. The competitive landscape shows consolidation around sustainable production methods and enhanced extraction efficiency, with companies like Altus Formulation focusing on safer delivery technologies and K.D. Pharma Bexbach specializing in purification processes for pharmaceutical applications.
Celanese International Corp.
Technical Solution: Celanese has developed advanced ethyl acetate production and purification technologies for pharmaceutical applications. Their approach focuses on high-purity ethyl acetate manufacturing using optimized esterification processes with enhanced catalyst systems. The company implements continuous distillation columns with improved separation efficiency, achieving over 99.8% purity levels suitable for pharmaceutical extractions. Their technology includes solvent recovery systems that can reclaim up to 95% of ethyl acetate from extraction processes, significantly reducing waste and operational costs. Additionally, they have developed specialized grade ethyl acetate with reduced impurity profiles specifically designed for sensitive pharmaceutical compound extractions, ensuring minimal interference with active pharmaceutical ingredients.
Strengths: Leading chemical manufacturer with extensive solvent production expertise and established pharmaceutical supply chains. Weaknesses: Limited focus on extraction process optimization compared to solvent production.
Tianjin Institute of Pharmaceutical Research Co., Ltd.
Technical Solution: Tianjin Institute has developed comprehensive ethyl acetate optimization protocols for traditional Chinese medicine and modern pharmaceutical extractions. Their approach combines traditional extraction wisdom with modern process engineering, utilizing ethyl acetate in multi-stage extraction systems that maximize active compound recovery. The institute has created standardized procedures for ethyl acetate usage optimization that include pre-treatment methods to enhance extraction efficiency, reducing overall solvent requirements by 30-50%. Their technology incorporates temperature-controlled extraction systems with ethyl acetate recycling capabilities, achieving both economic and environmental benefits. The institute also provides extensive research on ethyl acetate selectivity for different pharmaceutical compounds, offering guidance on optimal extraction conditions including solvent-to-material ratios, extraction duration, and post-extraction purification steps to maximize pharmaceutical compound purity and yield.
Strengths: Strong research foundation with expertise in both traditional and modern pharmaceutical extraction methods. Weaknesses: Limited commercial scale implementation and technology transfer capabilities compared to multinational corporations.
Core Innovations in Solvent Recovery and Recycling
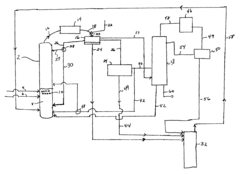
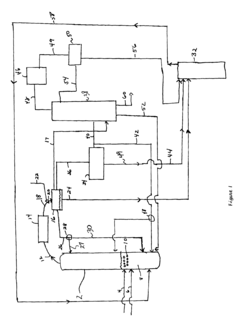
Process for separating ethyl acetate from liquid mixtures by extractive distillation
PatentInactiveGB765334A
Innovation
- The process employs extractive distillation in a fractionating column with an auxiliary liquid that forms an azeotropic mixture with ethyl acetate, allowing it to be separated from other components, while the auxiliary liquid distills overhead with water, enabling efficient separation of ethyl acetate from complex mixtures using a suitable washing liquid chosen based on Raoult's law and hydrogen bonding criteria.
Process improvement for continuous ethyl acetate production
PatentInactiveUS6768021B2
Innovation
- The process involves using a membrane separation unit to remove water from the condensed reaction stream, recycling the dried stream back into the production process, and employing an additional distillation zone to produce purified ethyl acetate with minimal acid content, thereby optimizing water management and increasing process capacity.
Environmental Regulations for Pharmaceutical Solvents
The pharmaceutical industry faces increasingly stringent environmental regulations governing solvent usage, with ethyl acetate being subject to comprehensive oversight due to its widespread application in extraction processes. Regulatory frameworks across major pharmaceutical markets, including the United States, European Union, and Asia-Pacific regions, have established specific guidelines for volatile organic compound emissions, waste management, and worker exposure limits.
The U.S. Environmental Protection Agency classifies ethyl acetate under the Clean Air Act's National Emission Standards for Hazardous Air Pollutants, requiring pharmaceutical manufacturers to implement maximum achievable control technology for emission reduction. The regulation mandates continuous monitoring of ethyl acetate concentrations in workplace environments, with permissible exposure limits set at 400 parts per million over an eight-hour time-weighted average.
European regulations under the Industrial Emissions Directive impose strict requirements on pharmaceutical facilities using ethyl acetate in extraction processes. The REACH regulation requires comprehensive registration of ethyl acetate usage quantities exceeding one ton annually, including detailed safety data sheets and risk assessment documentation. Additionally, the Seveso III Directive establishes threshold quantities for ethyl acetate storage, triggering enhanced safety management requirements for facilities exceeding 5,000 tons.
Waste disposal regulations present significant compliance challenges for pharmaceutical extraction operations. The Resource Conservation and Recovery Act in the United States classifies ethyl acetate-containing waste streams as hazardous materials, requiring specialized treatment and disposal protocols. Similar regulations in the European Union mandate waste minimization strategies and recovery programs, with increasing emphasis on solvent recycling and reuse technologies.
Recent regulatory trends indicate a shift toward more restrictive emission standards and enhanced reporting requirements. The International Council for Harmonisation guidelines increasingly emphasize green chemistry principles, encouraging pharmaceutical manufacturers to optimize solvent usage through process intensification and alternative extraction methodologies. These evolving regulations create both compliance pressures and innovation opportunities for ethyl acetate optimization in pharmaceutical extraction applications.
The U.S. Environmental Protection Agency classifies ethyl acetate under the Clean Air Act's National Emission Standards for Hazardous Air Pollutants, requiring pharmaceutical manufacturers to implement maximum achievable control technology for emission reduction. The regulation mandates continuous monitoring of ethyl acetate concentrations in workplace environments, with permissible exposure limits set at 400 parts per million over an eight-hour time-weighted average.
European regulations under the Industrial Emissions Directive impose strict requirements on pharmaceutical facilities using ethyl acetate in extraction processes. The REACH regulation requires comprehensive registration of ethyl acetate usage quantities exceeding one ton annually, including detailed safety data sheets and risk assessment documentation. Additionally, the Seveso III Directive establishes threshold quantities for ethyl acetate storage, triggering enhanced safety management requirements for facilities exceeding 5,000 tons.
Waste disposal regulations present significant compliance challenges for pharmaceutical extraction operations. The Resource Conservation and Recovery Act in the United States classifies ethyl acetate-containing waste streams as hazardous materials, requiring specialized treatment and disposal protocols. Similar regulations in the European Union mandate waste minimization strategies and recovery programs, with increasing emphasis on solvent recycling and reuse technologies.
Recent regulatory trends indicate a shift toward more restrictive emission standards and enhanced reporting requirements. The International Council for Harmonisation guidelines increasingly emphasize green chemistry principles, encouraging pharmaceutical manufacturers to optimize solvent usage through process intensification and alternative extraction methodologies. These evolving regulations create both compliance pressures and innovation opportunities for ethyl acetate optimization in pharmaceutical extraction applications.
Process Safety Considerations in Ethyl Acetate Handling
Ethyl acetate handling in pharmaceutical extraction processes requires comprehensive safety protocols due to its inherent hazardous properties. As a volatile organic compound with a flash point of -4°C, ethyl acetate poses significant fire and explosion risks. The compound exhibits moderate toxicity through inhalation and skin contact, necessitating strict exposure controls and personal protective equipment requirements. Its relatively low boiling point of 77°C contributes to rapid vapor formation, creating potential atmospheric hazards in enclosed spaces.
Fire prevention measures constitute the primary safety concern in ethyl acetate operations. All electrical equipment must meet explosion-proof standards, and static electricity control systems are essential during transfer operations. Hot work permits and flame-free zones must be strictly enforced within designated areas. Emergency fire suppression systems should utilize foam or carbon dioxide rather than water, which can spread the solvent and exacerbate fire conditions.
Ventilation system design plays a critical role in maintaining safe working environments. Local exhaust ventilation at transfer points and process equipment prevents vapor accumulation, while general dilution ventilation maintains ambient concentrations below permissible exposure limits. Vapor detection systems with automatic alarms provide early warning of potentially dangerous atmospheric conditions, enabling prompt corrective actions.
Personal protective equipment protocols must address both acute and chronic exposure risks. Respiratory protection using organic vapor cartridges is mandatory in areas where engineering controls cannot maintain safe exposure levels. Chemical-resistant gloves and protective clothing prevent dermal absorption, while eye protection guards against splash hazards during manual operations.
Storage and handling procedures require specialized considerations for ethyl acetate's reactive properties. Containers must be grounded during transfer operations to prevent static discharge. Temperature control systems prevent excessive vapor generation, while secondary containment systems manage potential spill scenarios. Regular inspection protocols for storage vessels, piping systems, and safety equipment ensure continued operational integrity.
Emergency response planning encompasses spill containment, fire suppression, and medical treatment protocols. Personnel training programs must cover recognition of exposure symptoms, proper use of safety equipment, and emergency evacuation procedures. Regular safety drills and equipment testing maintain organizational preparedness for potential incidents involving ethyl acetate handling operations.
Fire prevention measures constitute the primary safety concern in ethyl acetate operations. All electrical equipment must meet explosion-proof standards, and static electricity control systems are essential during transfer operations. Hot work permits and flame-free zones must be strictly enforced within designated areas. Emergency fire suppression systems should utilize foam or carbon dioxide rather than water, which can spread the solvent and exacerbate fire conditions.
Ventilation system design plays a critical role in maintaining safe working environments. Local exhaust ventilation at transfer points and process equipment prevents vapor accumulation, while general dilution ventilation maintains ambient concentrations below permissible exposure limits. Vapor detection systems with automatic alarms provide early warning of potentially dangerous atmospheric conditions, enabling prompt corrective actions.
Personal protective equipment protocols must address both acute and chronic exposure risks. Respiratory protection using organic vapor cartridges is mandatory in areas where engineering controls cannot maintain safe exposure levels. Chemical-resistant gloves and protective clothing prevent dermal absorption, while eye protection guards against splash hazards during manual operations.
Storage and handling procedures require specialized considerations for ethyl acetate's reactive properties. Containers must be grounded during transfer operations to prevent static discharge. Temperature control systems prevent excessive vapor generation, while secondary containment systems manage potential spill scenarios. Regular inspection protocols for storage vessels, piping systems, and safety equipment ensure continued operational integrity.
Emergency response planning encompasses spill containment, fire suppression, and medical treatment protocols. Personnel training programs must cover recognition of exposure symptoms, proper use of safety equipment, and emergency evacuation procedures. Regular safety drills and equipment testing maintain organizational preparedness for potential incidents involving ethyl acetate handling operations.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!