What Materials Improve CO₂ Capture Sorbent Efficiency
SEP 28, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
CO₂ Capture Materials Background and Objectives
Carbon dioxide capture and sequestration (CCS) has emerged as a critical technology in the global effort to mitigate climate change. The atmospheric concentration of CO₂ has increased dramatically since the industrial revolution, reaching levels unprecedented in human history. This technological domain has evolved from theoretical concepts in the 1970s to practical implementations in the 2000s, with significant acceleration in research and development over the past decade as climate concerns have intensified.
The evolution of CO₂ capture materials has progressed through several generations, beginning with conventional amine-based liquid sorbents, advancing to solid sorbents, and now exploring novel nanomaterials and hybrid systems. Each iteration has sought to address fundamental challenges of capture efficiency, energy requirements, and operational stability under various conditions.
Current technical objectives in this field focus on developing materials that can significantly improve the efficiency of CO₂ capture while reducing the associated energy penalty. Specifically, researchers aim to create sorbents with high CO₂ selectivity, rapid adsorption-desorption kinetics, and minimal regeneration energy requirements. The ideal material would maintain structural integrity over thousands of cycles while operating in the challenging environments typical of industrial emissions sources.
Another critical objective is scalability - developing materials that can be manufactured at industrial scale with consistent properties and reasonable costs. This includes considerations of raw material availability, synthesis complexity, and compatibility with existing infrastructure. The economic viability of any new capture material ultimately depends on achieving a balance between performance improvements and implementation costs.
Environmental sustainability represents an additional dimension of research goals. Next-generation materials must not only capture CO₂ effectively but do so with minimal environmental impact throughout their lifecycle. This includes considerations of toxicity, biodegradability, and the carbon footprint of the manufacturing process itself.
The technical trajectory points toward multifunctional materials that can simultaneously address multiple challenges. These include materials that can selectively capture CO₂ while rejecting other flue gas components, self-regenerate under mild conditions, or even convert captured CO₂ into valuable products through integrated catalytic functions.
Ultimately, the overarching goal is to develop materials that can enable economically viable carbon capture at the gigaton scale required to meaningfully impact climate change mitigation efforts. This represents not just an incremental improvement but a transformative advance in our technical capabilities for managing carbon emissions.
The evolution of CO₂ capture materials has progressed through several generations, beginning with conventional amine-based liquid sorbents, advancing to solid sorbents, and now exploring novel nanomaterials and hybrid systems. Each iteration has sought to address fundamental challenges of capture efficiency, energy requirements, and operational stability under various conditions.
Current technical objectives in this field focus on developing materials that can significantly improve the efficiency of CO₂ capture while reducing the associated energy penalty. Specifically, researchers aim to create sorbents with high CO₂ selectivity, rapid adsorption-desorption kinetics, and minimal regeneration energy requirements. The ideal material would maintain structural integrity over thousands of cycles while operating in the challenging environments typical of industrial emissions sources.
Another critical objective is scalability - developing materials that can be manufactured at industrial scale with consistent properties and reasonable costs. This includes considerations of raw material availability, synthesis complexity, and compatibility with existing infrastructure. The economic viability of any new capture material ultimately depends on achieving a balance between performance improvements and implementation costs.
Environmental sustainability represents an additional dimension of research goals. Next-generation materials must not only capture CO₂ effectively but do so with minimal environmental impact throughout their lifecycle. This includes considerations of toxicity, biodegradability, and the carbon footprint of the manufacturing process itself.
The technical trajectory points toward multifunctional materials that can simultaneously address multiple challenges. These include materials that can selectively capture CO₂ while rejecting other flue gas components, self-regenerate under mild conditions, or even convert captured CO₂ into valuable products through integrated catalytic functions.
Ultimately, the overarching goal is to develop materials that can enable economically viable carbon capture at the gigaton scale required to meaningfully impact climate change mitigation efforts. This represents not just an incremental improvement but a transformative advance in our technical capabilities for managing carbon emissions.
Market Analysis for Carbon Capture Technologies
The global carbon capture market is experiencing significant growth, driven by increasing environmental concerns and regulatory pressures to reduce greenhouse gas emissions. As of 2023, the carbon capture, utilization, and storage (CCUS) market was valued at approximately $4.2 billion, with projections indicating growth to reach $12.5 billion by 2030, representing a compound annual growth rate (CAGR) of 16.8%. This growth trajectory underscores the expanding commercial interest in technologies that enhance CO₂ capture efficiency.
Geographically, North America currently leads the market with about 38% share, followed by Europe at 32% and Asia-Pacific at 22%. The remaining 8% is distributed across other regions. This distribution reflects the concentration of industrial activities and regulatory frameworks supporting carbon capture initiatives in developed economies.
By application sector, power generation represents the largest market segment at 41%, followed by industrial processes (34%), natural gas processing (15%), and other applications (10%). The dominance of power generation highlights the critical need for carbon capture solutions in electricity production, particularly in coal and natural gas power plants.
The market for CO₂ capture sorbent materials specifically is witnessing rapid innovation and commercialization. Traditional amine-based sorbents currently hold approximately 65% of the material market share, while emerging materials such as metal-organic frameworks (MOFs), zeolites, and carbon-based sorbents collectively account for about 30%, with other novel materials comprising the remaining 5%.
Key market drivers include strengthening climate policies, carbon pricing mechanisms, and government incentives for clean energy technologies. The Inflation Reduction Act in the United States, which increased the 45Q tax credit for carbon sequestration to $85 per ton, has significantly boosted market interest. Similarly, the European Union's Emissions Trading System (EU ETS) and Carbon Border Adjustment Mechanism (CBAM) are creating financial incentives for carbon capture implementation.
Customer segments for improved CO₂ capture sorbents include power utilities, cement manufacturers, steel producers, chemical companies, and oil and gas operators. These industries face increasing pressure to decarbonize their operations while maintaining economic viability, creating demand for more efficient and cost-effective capture solutions.
Market barriers include high capital costs, energy penalties associated with capture processes, and limited infrastructure for CO₂ transport and storage. The levelized cost of carbon capture currently ranges from $40-120 per ton of CO₂, depending on the industry and technology employed, highlighting the need for more efficient sorbent materials to reduce operational costs.
Geographically, North America currently leads the market with about 38% share, followed by Europe at 32% and Asia-Pacific at 22%. The remaining 8% is distributed across other regions. This distribution reflects the concentration of industrial activities and regulatory frameworks supporting carbon capture initiatives in developed economies.
By application sector, power generation represents the largest market segment at 41%, followed by industrial processes (34%), natural gas processing (15%), and other applications (10%). The dominance of power generation highlights the critical need for carbon capture solutions in electricity production, particularly in coal and natural gas power plants.
The market for CO₂ capture sorbent materials specifically is witnessing rapid innovation and commercialization. Traditional amine-based sorbents currently hold approximately 65% of the material market share, while emerging materials such as metal-organic frameworks (MOFs), zeolites, and carbon-based sorbents collectively account for about 30%, with other novel materials comprising the remaining 5%.
Key market drivers include strengthening climate policies, carbon pricing mechanisms, and government incentives for clean energy technologies. The Inflation Reduction Act in the United States, which increased the 45Q tax credit for carbon sequestration to $85 per ton, has significantly boosted market interest. Similarly, the European Union's Emissions Trading System (EU ETS) and Carbon Border Adjustment Mechanism (CBAM) are creating financial incentives for carbon capture implementation.
Customer segments for improved CO₂ capture sorbents include power utilities, cement manufacturers, steel producers, chemical companies, and oil and gas operators. These industries face increasing pressure to decarbonize their operations while maintaining economic viability, creating demand for more efficient and cost-effective capture solutions.
Market barriers include high capital costs, energy penalties associated with capture processes, and limited infrastructure for CO₂ transport and storage. The levelized cost of carbon capture currently ranges from $40-120 per ton of CO₂, depending on the industry and technology employed, highlighting the need for more efficient sorbent materials to reduce operational costs.
Current Sorbent Technologies and Limitations
Carbon dioxide capture technologies have evolved significantly over the past decades, with various sorbent materials being developed and optimized. Currently, the most widely deployed CO₂ capture technologies utilize liquid amine-based sorbents, particularly monoethanolamine (MEA) solutions. These systems typically achieve 85-95% capture efficiency in industrial settings but suffer from high regeneration energy requirements (3.5-4.2 GJ/ton CO₂), equipment corrosion issues, and solvent degradation over time. The operational costs associated with these limitations have hindered widespread commercial adoption beyond niche applications.
Solid sorbents represent another major category, including activated carbons, zeolites, and metal-organic frameworks (MOFs). Activated carbons offer moderate CO₂ adsorption capacity (2-3 mmol/g) with the advantages of low cost and high stability, but demonstrate poor selectivity in the presence of water vapor. Zeolites exhibit higher adsorption capacities (up to 5 mmol/g) and excellent selectivity at ambient temperatures, though their performance deteriorates significantly in humid conditions and they require substantial energy for regeneration.
Metal-organic frameworks have emerged as promising candidates with record-setting CO₂ adsorption capacities exceeding 7 mmol/g under optimal conditions. Notable examples include Mg-MOF-74 and HKUST-1. However, MOFs face challenges including high synthesis costs, limited stability in industrial conditions, and scalability issues that have restricted their commercial deployment despite laboratory success.
Amine-functionalized solid sorbents combine the high selectivity of amines with the structural advantages of solid supports. Materials such as amine-grafted silicas and amine-impregnated porous polymers demonstrate CO₂ capacities of 3-6 mmol/g with improved stability compared to liquid amines. Nevertheless, these materials still face challenges including amine leaching during cycling and gradual capacity degradation.
Calcium looping technology, utilizing limestone (CaCO₃) as a sorbent, offers very high theoretical capacity but suffers from rapid performance degradation due to sintering during multiple carbonation-calcination cycles. Current research focuses on developing sintering-resistant materials through various doping strategies.
A critical limitation across all current sorbent technologies is the trade-off between adsorption capacity and regeneration energy. Materials with high binding affinity for CO₂ typically require more energy for regeneration, creating an efficiency bottleneck. Additionally, most advanced sorbents demonstrate significant performance gaps between laboratory conditions and real-world industrial environments containing contaminants like SOx, NOx, and water vapor.
The economic viability threshold for widespread adoption of carbon capture technologies is generally considered to be below $40/ton CO₂, a target that current sorbent technologies struggle to achieve when accounting for capital costs, operational expenses, and regeneration energy requirements.
Solid sorbents represent another major category, including activated carbons, zeolites, and metal-organic frameworks (MOFs). Activated carbons offer moderate CO₂ adsorption capacity (2-3 mmol/g) with the advantages of low cost and high stability, but demonstrate poor selectivity in the presence of water vapor. Zeolites exhibit higher adsorption capacities (up to 5 mmol/g) and excellent selectivity at ambient temperatures, though their performance deteriorates significantly in humid conditions and they require substantial energy for regeneration.
Metal-organic frameworks have emerged as promising candidates with record-setting CO₂ adsorption capacities exceeding 7 mmol/g under optimal conditions. Notable examples include Mg-MOF-74 and HKUST-1. However, MOFs face challenges including high synthesis costs, limited stability in industrial conditions, and scalability issues that have restricted their commercial deployment despite laboratory success.
Amine-functionalized solid sorbents combine the high selectivity of amines with the structural advantages of solid supports. Materials such as amine-grafted silicas and amine-impregnated porous polymers demonstrate CO₂ capacities of 3-6 mmol/g with improved stability compared to liquid amines. Nevertheless, these materials still face challenges including amine leaching during cycling and gradual capacity degradation.
Calcium looping technology, utilizing limestone (CaCO₃) as a sorbent, offers very high theoretical capacity but suffers from rapid performance degradation due to sintering during multiple carbonation-calcination cycles. Current research focuses on developing sintering-resistant materials through various doping strategies.
A critical limitation across all current sorbent technologies is the trade-off between adsorption capacity and regeneration energy. Materials with high binding affinity for CO₂ typically require more energy for regeneration, creating an efficiency bottleneck. Additionally, most advanced sorbents demonstrate significant performance gaps between laboratory conditions and real-world industrial environments containing contaminants like SOx, NOx, and water vapor.
The economic viability threshold for widespread adoption of carbon capture technologies is generally considered to be below $40/ton CO₂, a target that current sorbent technologies struggle to achieve when accounting for capital costs, operational expenses, and regeneration energy requirements.
State-of-the-Art Sorbent Enhancement Methods
01 Metal-organic frameworks (MOFs) for CO₂ capture
Metal-organic frameworks are advanced porous materials with high surface area and tunable pore structures that demonstrate exceptional CO₂ adsorption capacity. These materials can be engineered with specific metal centers and organic linkers to enhance selectivity and efficiency for carbon dioxide capture. MOFs offer advantages such as low regeneration energy requirements and stability over multiple adsorption-desorption cycles, making them promising candidates for industrial-scale carbon capture applications.- Metal-organic frameworks (MOFs) for CO₂ capture: Metal-organic frameworks (MOFs) are highly porous crystalline materials that can be engineered for efficient CO₂ capture. These materials offer high surface area, tunable pore sizes, and customizable chemical functionality that can selectively adsorb CO₂. The efficiency of MOFs can be enhanced by incorporating specific metal centers and organic linkers that increase CO₂ binding affinity while maintaining regeneration capabilities under mild conditions.
- Amine-functionalized sorbents: Amine-functionalized materials represent a significant class of CO₂ capture sorbents with high efficiency. These materials work through chemical adsorption (chemisorption) where amine groups react with CO₂ to form carbamates or bicarbonates. Various supports including silica, polymers, and porous carbons can be functionalized with amines to enhance CO₂ capture capacity and selectivity. The efficiency of these sorbents depends on amine loading, accessibility, and stability during multiple adsorption-desorption cycles.
- Solid oxide and alkali metal-based sorbents: Solid oxide and alkali metal-based materials offer promising CO₂ capture efficiency at elevated temperatures. These include calcium oxide (CaO), magnesium oxide (MgO), and lithium-based sorbents that capture CO₂ through carbonation reactions. The efficiency of these materials is determined by their cyclic stability, resistance to sintering, and regeneration energy requirements. Various dopants and support structures can be incorporated to enhance the performance and longevity of these high-temperature sorbents.
- Novel composite and hybrid sorbent materials: Composite and hybrid materials combine the advantages of different sorbent types to achieve enhanced CO₂ capture efficiency. These materials may integrate organic and inorganic components, creating synergistic effects that improve adsorption capacity, selectivity, and regeneration properties. Examples include polymer-inorganic composites, layered double hydroxides, and hierarchical porous structures. The efficiency of these hybrid materials can be optimized by controlling the composition, morphology, and interfacial properties of the constituent components.
- Process optimization and efficiency metrics for CO₂ capture: Improving the overall efficiency of CO₂ capture systems involves optimizing process parameters and developing standardized metrics for performance evaluation. Key factors include energy consumption for regeneration, working capacity under practical conditions, selectivity in mixed gas streams, and long-term stability. Advanced process designs such as temperature/pressure swing adsorption, vacuum swing adsorption, and integrated capture systems can significantly enhance the efficiency of CO₂ capture operations while reducing the overall energy penalty and operational costs.
02 Amine-functionalized sorbents
Amine-functionalized materials represent a significant advancement in CO₂ capture technology. These sorbents incorporate various amine groups that chemically bind with CO₂ through carbamate formation. The efficiency of these materials can be enhanced by optimizing the amine loading, pore structure, and support materials. Solid supports such as silica, polymers, or carbon-based materials can be modified with amines to create high-capacity, selective CO₂ capture systems with improved regeneration properties and reduced energy consumption.Expand Specific Solutions03 Zeolite and molecular sieve-based CO₂ adsorbents
Zeolites and molecular sieves are crystalline aluminosilicate materials with well-defined pore structures that enable selective adsorption of CO₂. These materials utilize physical adsorption mechanisms based on molecular size exclusion and can be modified to enhance their CO₂ capture efficiency. The performance of zeolite-based sorbents can be improved through ion exchange, framework modification, and optimization of the Si/Al ratio. These materials offer advantages including high thermal stability, mechanical strength, and relatively low cost for large-scale carbon capture applications.Expand Specific Solutions04 Novel composite and hybrid sorbent materials
Composite and hybrid materials combine the advantages of different types of sorbents to achieve enhanced CO₂ capture performance. These materials may integrate organic and inorganic components, such as polymer-inorganic hybrids, mixed matrix materials, or layered structures. By synergistically combining multiple capture mechanisms, these composites can achieve higher adsorption capacities, improved selectivity, and better stability. Advanced manufacturing techniques like 3D printing and controlled synthesis methods are being employed to optimize the structure and performance of these hybrid sorbent systems.Expand Specific Solutions05 Process optimization and efficiency enhancement techniques
Various techniques and methodologies have been developed to enhance the overall efficiency of CO₂ capture processes using sorbent materials. These include temperature and pressure swing adsorption processes, vacuum swing adsorption, and hybrid capture systems. Innovations in process design, such as fluidized bed reactors, moving bed systems, and integrated capture-regeneration cycles, can significantly improve the energy efficiency and economics of carbon capture. Additionally, advanced monitoring and control systems, heat integration strategies, and optimization algorithms contribute to maximizing the performance of CO₂ capture sorbents in practical applications.Expand Specific Solutions
Leading Organizations in Carbon Capture Research
The CO₂ capture sorbent efficiency market is in a growth phase, with increasing global focus on carbon reduction technologies driving expansion. The market is projected to reach significant scale as carbon capture becomes essential for climate goals. Technologically, the field shows varying maturity levels across different approaches. Leading players represent diverse sectors: commercial entities like Climeworks AG and Global Thermostat are pioneering direct air capture technologies, while established industrial companies such as Wacker Chemie and China Petroleum & Chemical Corp. are leveraging their chemical expertise. Academic institutions (Arizona State University, Georgia Tech, University of Queensland) contribute fundamental research, creating a competitive landscape where collaboration between industry and academia is accelerating innovation in sorbent materials with improved efficiency, selectivity, and regeneration capabilities.
Climeworks AG
Technical Solution: Climeworks has developed a direct air capture (DAC) technology using solid sorbent materials arranged in modular collectors. Their proprietary amine-functionalized filter material selectively captures CO₂ when air passes through it. The process involves a two-step temperature swing adsorption cycle where ambient air flows through the collector, binding CO₂ to the sorbent surface. Once saturated, the collector is heated to 80-100°C, releasing concentrated CO₂ that can be collected for storage or utilization. Their latest generation technology has achieved significant efficiency improvements, reducing energy requirements by approximately 80% compared to their first-generation systems, with current energy consumption around 2-2.5 MWh per ton of CO₂ captured. Climeworks has deployed commercial plants in Switzerland and Iceland, with their Orca plant in Iceland capturing 4,000 tons of CO₂ annually and their Mammoth plant aiming to scale to 36,000 tons.
Strengths: Modular design allows for scalable deployment; integration with geothermal energy in Iceland provides renewable heat source for regeneration; permanent storage solution through mineralization in basalt rock. Weaknesses: Still relatively high energy requirements compared to theoretical minimums; current cost estimated at $600-800 per ton of CO₂, requiring further cost reduction for widespread adoption; limited capture capacity compared to global emissions.
Shanghai Advanced Research Institute
Technical Solution: Shanghai Advanced Research Institute (SARI) has developed advanced metal-organic framework (MOF) materials for CO₂ capture with exceptional performance characteristics. Their research focuses on creating hierarchically porous structures with tailored chemical functionalities that maximize CO₂ selectivity and adsorption capacity. SARI's innovative MOFs incorporate nitrogen-rich functional groups and open metal sites that create strong binding interactions with CO₂ molecules while maintaining relatively low regeneration energy requirements. One of their breakthrough materials demonstrated a CO₂ uptake capacity exceeding 4.5 mmol/g under ambient conditions with a CO₂/N₂ selectivity factor above 80. Their materials engineering approach includes post-synthetic modification techniques to enhance stability in humid conditions—a common challenge for many sorbent materials. SARI has also developed composite materials combining MOFs with graphene oxide to improve heat transfer properties during the adsorption-desorption cycle, addressing one of the key efficiency bottlenecks in temperature swing processes. Recent work has focused on scalable synthesis methods to transition these materials from laboratory to industrial applications.
Strengths: Exceptionally high CO₂ selectivity and capacity compared to conventional materials; tunable pore structures allowing optimization for different operating conditions; relatively good stability under repeated cycling. Weaknesses: Manufacturing costs remain high for large-scale production; some materials show performance degradation in the presence of moisture or contaminants; energy requirements for regeneration still present challenges for commercial viability.
Key Innovations in High-Efficiency CO₂ Sorbents
Sorbents, systems including sorbents, and methods using the sorbents
PatentPendingUS20240335784A1
Innovation
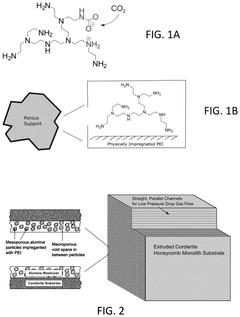
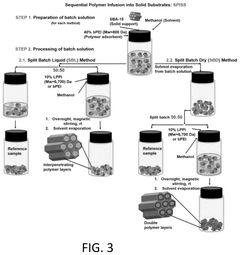
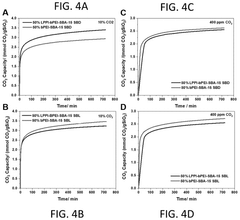
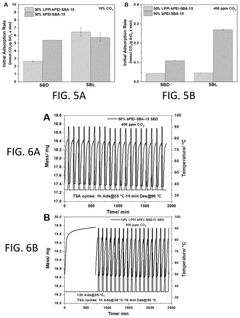
- Development of sorbents comprising a CO2-philic phase with a combination of polypropylenimine and polyethylenimine, which provides improved oxidative stability and hydrophilicity, allowing for efficient CO2 capture and regeneration, and are integrated into a structured support for enhanced performance.
Quality control method for co2 capture material
PatentWO2025084925A1
Innovation
- A simplified quality control method involving the use of an indicator dye that adsorbs onto the sorbent, followed by image capture and analysis to indirectly quantify the CO2 capture capacity based on colour intensity, eliminating the need for expensive laboratory equipment.
Environmental Impact Assessment of Capture Materials
The environmental implications of CO₂ capture materials extend far beyond their primary function of reducing greenhouse gas emissions. When evaluating these materials, a comprehensive life cycle assessment is essential to understand their true environmental footprint. Most capture materials require significant energy and resources during production, potentially offsetting some of their climate benefits if not carefully managed.
Amine-based sorbents, while effective for CO₂ capture, present environmental concerns including potential atmospheric degradation that can produce harmful byproducts such as nitrosamines and nitramines. These compounds have been linked to adverse health effects and ecosystem damage when released into the environment. Additionally, the manufacturing process for these materials often involves toxic precursors that require careful handling and disposal protocols.
Metal-organic frameworks (MOFs) and zeolites generally demonstrate lower environmental toxicity compared to amine-based alternatives. However, their synthesis frequently requires organic solvents and metal precursors that can contribute to water pollution if improperly managed. The environmental advantage of these materials lies in their potential for regeneration and reuse across multiple capture cycles, reducing the need for continuous production of new materials.
Solid waste generation represents another significant environmental consideration. As capture materials degrade over time, their disposal must be carefully managed to prevent leaching of potentially harmful components into soil and groundwater systems. Some advanced materials incorporate heavy metals or rare earth elements that pose particular disposal challenges and may require specialized treatment facilities.
Water usage during both the production of capture materials and their operational deployment presents additional environmental concerns. Many capture processes require substantial water resources for cooling or regeneration cycles, potentially creating competition for water in water-stressed regions. This aspect becomes particularly critical when considering large-scale deployment of carbon capture technologies in diverse geographical contexts.
Energy requirements for material regeneration also factor significantly into environmental impact assessments. Materials requiring high-temperature regeneration contribute to indirect emissions unless powered by renewable energy sources. Recent innovations focusing on materials with lower regeneration energy requirements represent a promising direction for minimizing the overall environmental footprint of carbon capture operations.
Biodegradability and end-of-life management strategies are increasingly important considerations in material development. Emerging bio-based sorbents offer potential advantages in this regard, though their capture efficiency and durability often lag behind synthetic alternatives. The ideal capture material would combine high CO₂ affinity with minimal environmental impact throughout its entire lifecycle.
Amine-based sorbents, while effective for CO₂ capture, present environmental concerns including potential atmospheric degradation that can produce harmful byproducts such as nitrosamines and nitramines. These compounds have been linked to adverse health effects and ecosystem damage when released into the environment. Additionally, the manufacturing process for these materials often involves toxic precursors that require careful handling and disposal protocols.
Metal-organic frameworks (MOFs) and zeolites generally demonstrate lower environmental toxicity compared to amine-based alternatives. However, their synthesis frequently requires organic solvents and metal precursors that can contribute to water pollution if improperly managed. The environmental advantage of these materials lies in their potential for regeneration and reuse across multiple capture cycles, reducing the need for continuous production of new materials.
Solid waste generation represents another significant environmental consideration. As capture materials degrade over time, their disposal must be carefully managed to prevent leaching of potentially harmful components into soil and groundwater systems. Some advanced materials incorporate heavy metals or rare earth elements that pose particular disposal challenges and may require specialized treatment facilities.
Water usage during both the production of capture materials and their operational deployment presents additional environmental concerns. Many capture processes require substantial water resources for cooling or regeneration cycles, potentially creating competition for water in water-stressed regions. This aspect becomes particularly critical when considering large-scale deployment of carbon capture technologies in diverse geographical contexts.
Energy requirements for material regeneration also factor significantly into environmental impact assessments. Materials requiring high-temperature regeneration contribute to indirect emissions unless powered by renewable energy sources. Recent innovations focusing on materials with lower regeneration energy requirements represent a promising direction for minimizing the overall environmental footprint of carbon capture operations.
Biodegradability and end-of-life management strategies are increasingly important considerations in material development. Emerging bio-based sorbents offer potential advantages in this regard, though their capture efficiency and durability often lag behind synthetic alternatives. The ideal capture material would combine high CO₂ affinity with minimal environmental impact throughout its entire lifecycle.
Techno-Economic Analysis of Advanced Sorbents
The techno-economic analysis of advanced sorbents for CO₂ capture reveals significant cost implications across the carbon capture value chain. Current economic assessments indicate that advanced sorbents can potentially reduce the energy penalty associated with CO₂ capture by 30-40% compared to conventional amine scrubbing technologies, translating to operational cost savings of $15-25 per ton of CO₂ captured.
Material costs represent a critical factor in the economic viability of sorbent-based capture systems. Metal-organic frameworks (MOFs), while demonstrating exceptional capture capacities, currently face production costs ranging from $200-1,000/kg, making large-scale deployment economically challenging. In contrast, amine-functionalized silica sorbents present a more economically viable option at $20-50/kg, though with lower capture efficiencies.
Lifecycle economic analysis reveals that sorbent durability significantly impacts long-term operational economics. Advanced sorbents demonstrating stability over 1,000+ capture-regeneration cycles can reduce replacement costs by up to 75% compared to materials requiring frequent regeneration or replacement. This translates to potential savings of $5-8 per ton of CO₂ captured over the operational lifetime of carbon capture facilities.
Energy requirements for sorbent regeneration represent another crucial economic factor. Novel materials with lower regeneration temperatures (60-80°C versus 120-150°C for conventional systems) can reduce energy consumption by 20-35%, significantly improving the overall economics of capture operations. Particularly promising are phase-change sorbents that demonstrate regeneration energy requirements below 2.0 GJ/ton CO₂, compared to 3.5-4.0 GJ/ton for conventional technologies.
Capital expenditure analysis indicates that advanced sorbent systems can potentially reduce equipment sizing by 30-50% compared to liquid amine systems, resulting in CAPEX reductions of $200-400 million for large-scale (1 Mt CO₂/year) capture facilities. However, this advantage is partially offset by the higher initial material costs for specialized sorbents.
Sensitivity analysis demonstrates that sorbent performance improvements yielding 20% higher working capacities can reduce the levelized cost of carbon capture by approximately $7-12 per ton CO₂. This highlights the economic value of continued research into materials with enhanced sorption properties, particularly those maintaining performance under realistic flue gas conditions containing contaminants like SOx and NOx.
Material costs represent a critical factor in the economic viability of sorbent-based capture systems. Metal-organic frameworks (MOFs), while demonstrating exceptional capture capacities, currently face production costs ranging from $200-1,000/kg, making large-scale deployment economically challenging. In contrast, amine-functionalized silica sorbents present a more economically viable option at $20-50/kg, though with lower capture efficiencies.
Lifecycle economic analysis reveals that sorbent durability significantly impacts long-term operational economics. Advanced sorbents demonstrating stability over 1,000+ capture-regeneration cycles can reduce replacement costs by up to 75% compared to materials requiring frequent regeneration or replacement. This translates to potential savings of $5-8 per ton of CO₂ captured over the operational lifetime of carbon capture facilities.
Energy requirements for sorbent regeneration represent another crucial economic factor. Novel materials with lower regeneration temperatures (60-80°C versus 120-150°C for conventional systems) can reduce energy consumption by 20-35%, significantly improving the overall economics of capture operations. Particularly promising are phase-change sorbents that demonstrate regeneration energy requirements below 2.0 GJ/ton CO₂, compared to 3.5-4.0 GJ/ton for conventional technologies.
Capital expenditure analysis indicates that advanced sorbent systems can potentially reduce equipment sizing by 30-50% compared to liquid amine systems, resulting in CAPEX reductions of $200-400 million for large-scale (1 Mt CO₂/year) capture facilities. However, this advantage is partially offset by the higher initial material costs for specialized sorbents.
Sensitivity analysis demonstrates that sorbent performance improvements yielding 20% higher working capacities can reduce the levelized cost of carbon capture by approximately $7-12 per ton CO₂. This highlights the economic value of continued research into materials with enhanced sorption properties, particularly those maintaining performance under realistic flue gas conditions containing contaminants like SOx and NOx.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!