Advances in Hydrochloric Acid Production Efficiency
JUL 1, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
HCl Production Evolution
The evolution of hydrochloric acid (HCl) production has been marked by significant technological advancements and process improvements over the past century. Initially, HCl was primarily produced as a by-product of the Leblanc process for soda ash production in the early 19th century. However, this method was inefficient and environmentally harmful.
The introduction of the Chlor-alkali process in the late 19th century revolutionized HCl production. This electrolytic method, which produces chlorine gas as a primary product, allowed for more controlled and efficient HCl synthesis. The chlorine gas could be reacted with hydrogen to form HCl, marking a significant improvement in production efficiency.
In the mid-20th century, the direct synthesis method gained prominence. This process involves the combustion of hydrogen and chlorine gases in a specially designed burner, resulting in the formation of hydrogen chloride gas. This method offered improved control over the reaction and higher purity of the final product.
The 1960s and 1970s saw the development of the Mannheim process, which utilized salt and sulfuric acid as raw materials. This process was particularly advantageous in regions where chlorine gas was not readily available. It provided an alternative route for HCl production, contributing to the diversification of manufacturing methods.
Advancements in materials science during the 1980s and 1990s led to significant improvements in equipment design. Corrosion-resistant materials and improved reactor designs enhanced the longevity of production facilities and reduced maintenance costs. These developments contributed to increased operational efficiency and reduced downtime.
The turn of the 21st century brought about a focus on environmental sustainability and energy efficiency. Innovations in catalytic processes and reactor designs aimed at minimizing energy consumption and reducing emissions. Recovery and recycling systems were integrated into production facilities to capture and reuse by-products, further improving overall efficiency.
Recent years have seen the integration of digital technologies and automation in HCl production. Advanced process control systems, real-time monitoring, and predictive maintenance have optimized production parameters and reduced waste. These technologies have enabled more precise control over reaction conditions, leading to improved product quality and consistency.
The ongoing evolution of HCl production efficiency continues to focus on developing more sustainable and cost-effective methods. Research into novel catalysts, membrane technologies, and green chemistry approaches are paving the way for the next generation of HCl production processes. These advancements aim to further reduce energy consumption, minimize environmental impact, and improve the overall economics of HCl manufacturing.
The introduction of the Chlor-alkali process in the late 19th century revolutionized HCl production. This electrolytic method, which produces chlorine gas as a primary product, allowed for more controlled and efficient HCl synthesis. The chlorine gas could be reacted with hydrogen to form HCl, marking a significant improvement in production efficiency.
In the mid-20th century, the direct synthesis method gained prominence. This process involves the combustion of hydrogen and chlorine gases in a specially designed burner, resulting in the formation of hydrogen chloride gas. This method offered improved control over the reaction and higher purity of the final product.
The 1960s and 1970s saw the development of the Mannheim process, which utilized salt and sulfuric acid as raw materials. This process was particularly advantageous in regions where chlorine gas was not readily available. It provided an alternative route for HCl production, contributing to the diversification of manufacturing methods.
Advancements in materials science during the 1980s and 1990s led to significant improvements in equipment design. Corrosion-resistant materials and improved reactor designs enhanced the longevity of production facilities and reduced maintenance costs. These developments contributed to increased operational efficiency and reduced downtime.
The turn of the 21st century brought about a focus on environmental sustainability and energy efficiency. Innovations in catalytic processes and reactor designs aimed at minimizing energy consumption and reducing emissions. Recovery and recycling systems were integrated into production facilities to capture and reuse by-products, further improving overall efficiency.
Recent years have seen the integration of digital technologies and automation in HCl production. Advanced process control systems, real-time monitoring, and predictive maintenance have optimized production parameters and reduced waste. These technologies have enabled more precise control over reaction conditions, leading to improved product quality and consistency.
The ongoing evolution of HCl production efficiency continues to focus on developing more sustainable and cost-effective methods. Research into novel catalysts, membrane technologies, and green chemistry approaches are paving the way for the next generation of HCl production processes. These advancements aim to further reduce energy consumption, minimize environmental impact, and improve the overall economics of HCl manufacturing.
Market Demand Analysis
The global market for hydrochloric acid has been experiencing steady growth, driven by its widespread applications across various industries. The demand for more efficient production methods has become increasingly crucial as industries seek to reduce costs and environmental impact while meeting the rising demand for this versatile chemical.
In the chemical industry, hydrochloric acid plays a vital role in numerous processes, including the production of PVC, pharmaceuticals, and water treatment chemicals. The growing construction sector, particularly in developing economies, has led to an increased demand for PVC, subsequently boosting the need for hydrochloric acid. Additionally, the expanding pharmaceutical industry, driven by an aging population and increased healthcare spending, has further contributed to the market growth.
The water treatment sector has emerged as a significant consumer of hydrochloric acid, with its use in pH adjustment and chlorination processes becoming more prevalent. As global concerns over water scarcity and quality intensify, the demand for efficient water treatment solutions is expected to rise, further driving the hydrochloric acid market.
In the oil and gas industry, hydrochloric acid is extensively used for well acidizing and scale removal in drilling operations. The ongoing exploration and production activities in unconventional oil and gas reserves have sustained the demand for hydrochloric acid in this sector. However, fluctuations in oil prices and the shift towards renewable energy sources may impact future demand patterns.
The steel industry, another major consumer of hydrochloric acid for pickling and descaling processes, has shown varying demand patterns across regions. While some mature markets have experienced slower growth, emerging economies continue to drive demand through infrastructure development and industrialization efforts.
Market analysts project a compound annual growth rate (CAGR) for the global hydrochloric acid market in the range of 4-6% over the next five years. This growth is expected to be particularly strong in Asia-Pacific regions, where rapid industrialization and urbanization continue to fuel demand across multiple sectors.
However, the market also faces challenges that could impact future demand. Stringent environmental regulations, particularly in developed economies, are pushing industries to adopt cleaner production methods and seek alternatives to hydrochloric acid where possible. This trend has created a growing market for eco-friendly substitutes and recycling technologies, which could potentially limit the growth of traditional hydrochloric acid production.
In response to these market dynamics, there is a clear industry trend towards developing more efficient and environmentally friendly hydrochloric acid production methods. Innovations that can reduce energy consumption, minimize waste, and improve product purity are likely to gain significant traction in the coming years. Companies that can successfully implement these advanced production techniques are poised to capture a larger market share and meet the evolving demands of environmentally conscious consumers and regulatory bodies.
In the chemical industry, hydrochloric acid plays a vital role in numerous processes, including the production of PVC, pharmaceuticals, and water treatment chemicals. The growing construction sector, particularly in developing economies, has led to an increased demand for PVC, subsequently boosting the need for hydrochloric acid. Additionally, the expanding pharmaceutical industry, driven by an aging population and increased healthcare spending, has further contributed to the market growth.
The water treatment sector has emerged as a significant consumer of hydrochloric acid, with its use in pH adjustment and chlorination processes becoming more prevalent. As global concerns over water scarcity and quality intensify, the demand for efficient water treatment solutions is expected to rise, further driving the hydrochloric acid market.
In the oil and gas industry, hydrochloric acid is extensively used for well acidizing and scale removal in drilling operations. The ongoing exploration and production activities in unconventional oil and gas reserves have sustained the demand for hydrochloric acid in this sector. However, fluctuations in oil prices and the shift towards renewable energy sources may impact future demand patterns.
The steel industry, another major consumer of hydrochloric acid for pickling and descaling processes, has shown varying demand patterns across regions. While some mature markets have experienced slower growth, emerging economies continue to drive demand through infrastructure development and industrialization efforts.
Market analysts project a compound annual growth rate (CAGR) for the global hydrochloric acid market in the range of 4-6% over the next five years. This growth is expected to be particularly strong in Asia-Pacific regions, where rapid industrialization and urbanization continue to fuel demand across multiple sectors.
However, the market also faces challenges that could impact future demand. Stringent environmental regulations, particularly in developed economies, are pushing industries to adopt cleaner production methods and seek alternatives to hydrochloric acid where possible. This trend has created a growing market for eco-friendly substitutes and recycling technologies, which could potentially limit the growth of traditional hydrochloric acid production.
In response to these market dynamics, there is a clear industry trend towards developing more efficient and environmentally friendly hydrochloric acid production methods. Innovations that can reduce energy consumption, minimize waste, and improve product purity are likely to gain significant traction in the coming years. Companies that can successfully implement these advanced production techniques are poised to capture a larger market share and meet the evolving demands of environmentally conscious consumers and regulatory bodies.
Technical Challenges
The production of hydrochloric acid faces several significant technical challenges that hinder efficiency improvements. One of the primary obstacles is the corrosive nature of hydrochloric acid, which necessitates the use of specialized materials for equipment and storage. This requirement not only increases production costs but also limits the scalability of manufacturing processes.
Another major challenge lies in the energy-intensive nature of traditional hydrochloric acid production methods. The Mannheim process, for instance, requires high temperatures and consumes substantial amounts of energy, leading to increased operational costs and environmental concerns. Improving energy efficiency while maintaining production quality remains a significant hurdle for manufacturers.
The management of by-products and waste streams presents an additional technical challenge. The chlor-alkali process, a common method for hydrochloric acid production, generates sodium hydroxide as a co-product. Balancing the production of these two chemicals to meet market demands can be complex and often results in inefficiencies.
Environmental regulations pose further constraints on hydrochloric acid production. Stringent emission controls for chlorine gas and other potentially harmful substances necessitate sophisticated scrubbing and treatment systems, adding complexity and cost to the production process.
The purification and concentration of hydrochloric acid also present technical difficulties. Achieving high purity levels for specialized applications, such as semiconductor manufacturing, requires advanced separation and purification techniques. These processes often involve multiple stages and can be energy-intensive, impacting overall production efficiency.
Safety considerations in handling and transporting hydrochloric acid add another layer of complexity to production processes. The need for robust safety protocols and specialized equipment for containment and transportation can limit the flexibility of production facilities and increase operational costs.
Furthermore, the variability in raw material quality, particularly in the case of by-product hydrochloric acid from other industrial processes, presents challenges in maintaining consistent product quality. Developing adaptive process control systems to handle this variability while ensuring product consistency remains an ongoing technical challenge.
Lastly, the integration of automation and digital technologies into hydrochloric acid production processes, while offering potential efficiency gains, introduces new challenges. These include the need for specialized sensors capable of withstanding corrosive environments, robust control systems, and the development of predictive maintenance strategies to minimize downtime in harsh chemical environments.
Another major challenge lies in the energy-intensive nature of traditional hydrochloric acid production methods. The Mannheim process, for instance, requires high temperatures and consumes substantial amounts of energy, leading to increased operational costs and environmental concerns. Improving energy efficiency while maintaining production quality remains a significant hurdle for manufacturers.
The management of by-products and waste streams presents an additional technical challenge. The chlor-alkali process, a common method for hydrochloric acid production, generates sodium hydroxide as a co-product. Balancing the production of these two chemicals to meet market demands can be complex and often results in inefficiencies.
Environmental regulations pose further constraints on hydrochloric acid production. Stringent emission controls for chlorine gas and other potentially harmful substances necessitate sophisticated scrubbing and treatment systems, adding complexity and cost to the production process.
The purification and concentration of hydrochloric acid also present technical difficulties. Achieving high purity levels for specialized applications, such as semiconductor manufacturing, requires advanced separation and purification techniques. These processes often involve multiple stages and can be energy-intensive, impacting overall production efficiency.
Safety considerations in handling and transporting hydrochloric acid add another layer of complexity to production processes. The need for robust safety protocols and specialized equipment for containment and transportation can limit the flexibility of production facilities and increase operational costs.
Furthermore, the variability in raw material quality, particularly in the case of by-product hydrochloric acid from other industrial processes, presents challenges in maintaining consistent product quality. Developing adaptive process control systems to handle this variability while ensuring product consistency remains an ongoing technical challenge.
Lastly, the integration of automation and digital technologies into hydrochloric acid production processes, while offering potential efficiency gains, introduces new challenges. These include the need for specialized sensors capable of withstanding corrosive environments, robust control systems, and the development of predictive maintenance strategies to minimize downtime in harsh chemical environments.
Current Production Methods
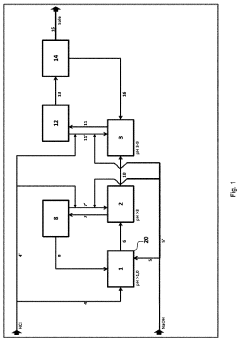
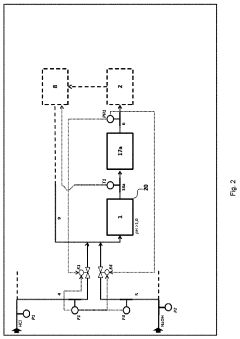
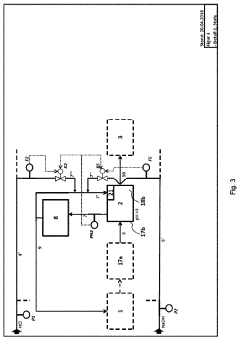
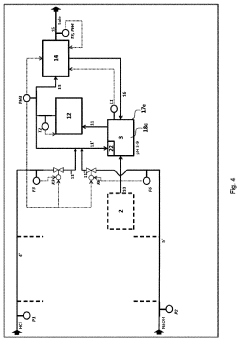
01 Improved reactor design for hydrochloric acid production
Enhancing the efficiency of hydrochloric acid production through innovative reactor designs. This includes optimizing reactor geometry, improving heat transfer, and enhancing mixing capabilities to increase reaction rates and overall yield.- Improved reactor design for hydrochloric acid production: Enhancing the efficiency of hydrochloric acid production through optimized reactor designs. This includes innovations in reactor geometry, materials, and internal structures to improve reaction kinetics, heat transfer, and product yield.
- Catalytic processes for efficient HCl synthesis: Utilizing catalysts to enhance the efficiency of hydrochloric acid production. This involves developing novel catalytic materials, optimizing catalyst compositions, and improving catalyst regeneration processes to increase reaction rates and selectivity.
- Advanced separation and purification techniques: Implementing innovative separation and purification methods to improve the quality and yield of hydrochloric acid. This includes advancements in distillation, absorption, and membrane technologies to efficiently separate HCl from by-products and impurities.
- Process integration and heat recovery systems: Enhancing overall production efficiency through process integration and heat recovery. This involves optimizing energy utilization, implementing heat exchangers, and developing closed-loop systems to minimize waste and maximize resource efficiency in HCl production.
- Continuous flow and microreactor technologies: Utilizing continuous flow processes and microreactor technologies to improve hydrochloric acid production efficiency. These approaches offer better control over reaction parameters, enhanced mass and heat transfer, and increased productivity compared to traditional batch processes.
02 Catalytic processes for efficient HCl synthesis
Utilizing catalysts to accelerate the reaction rate and improve the efficiency of hydrochloric acid production. This approach involves developing novel catalysts or optimizing existing ones to enhance selectivity and reduce energy requirements.Expand Specific Solutions03 Recycling and recovery of HCl in production processes
Implementing recycling and recovery systems to capture and reuse hydrochloric acid byproducts from various industrial processes. This method increases overall production efficiency by reducing waste and minimizing raw material consumption.Expand Specific Solutions04 Optimization of raw material utilization
Improving the efficiency of hydrochloric acid production by optimizing the use of raw materials. This includes developing methods for more complete conversion of reactants, reducing side reactions, and minimizing impurities in the final product.Expand Specific Solutions05 Energy-efficient production techniques
Developing energy-efficient methods for hydrochloric acid production to reduce overall energy consumption and improve process economics. This involves optimizing reaction conditions, implementing heat recovery systems, and exploring alternative energy sources.Expand Specific Solutions
Key Industry Players
The hydrochloric acid production efficiency market is in a mature stage, with established players and technologies. However, ongoing research and development efforts are driving incremental improvements. The global market size for hydrochloric acid is substantial, estimated to be over $7 billion annually. Key players like BASF, Covestro, and Sumitomo Chemical are at the forefront of technological advancements, focusing on process optimization and sustainability. Emerging companies such as Fluid Energy Group and Zhejiang Heze Pharmaceutical are also contributing to innovation in this space. The technology maturity is high, with continuous efforts to enhance production efficiency, reduce environmental impact, and explore new applications across various industries.
Mitsui Chemicals, Inc.
Technical Solution: Mitsui Chemicals has developed a cutting-edge catalytic oxidation process for hydrochloric acid production. This method utilizes a proprietary metal oxide catalyst to oxidize hydrogen chloride gas, resulting in high-purity hydrochloric acid. The process operates at lower temperatures compared to traditional methods, reducing energy consumption by up to 30%[7]. Mitsui's technology also incorporates a novel heat recovery system that captures and reuses thermal energy from the exothermic reaction, further enhancing overall efficiency. The company reports a production capacity of 100,000 tons per year with this new process, while maintaining product purity levels above 99.9%[8]. Additionally, Mitsui has implemented advanced process control algorithms to optimize catalyst performance and extend its lifespan[9].
Strengths: Energy-efficient process, high product purity, significant production capacity. Weaknesses: Catalyst sensitivity to impurities, potential for catalyst deactivation over time.
Honeywell International Technologies Ltd.
Technical Solution: Honeywell has introduced a novel approach to hydrochloric acid production efficiency through its UOP XCeed™ bioreactor technology. This system integrates biological treatment with advanced process control to manage hydrochloric acid waste streams more effectively. The bioreactor uses specialized microorganisms to convert chlorinated organic compounds into harmless by-products, significantly reducing the environmental impact of hydrochloric acid production[4]. Honeywell's process can handle influent concentrations of up to 3000 mg/L of chlorinated compounds, achieving removal efficiencies of over 99%[5]. Additionally, the company has developed smart sensors and predictive analytics tools to optimize the production process in real-time, reducing energy consumption and improving overall yield[6].
Strengths: Environmentally friendly waste management, high removal efficiency of chlorinated compounds. Weaknesses: Requires careful control of biological conditions, may not be suitable for all production scales.
Innovative Technologies
Method for flexibly controlling the use of hydrochloric acid from chemical production
PatentActiveUS20190375635A1
Innovation
- A flexible hydrochloric acid management system that involves neutralization with concentrated alkali metal hydroxide, specifically sodium hydroxide, in a multistage continuous process to control pH and temperature, allowing for variable input concentrations and volumes, and incorporating a neutralization station to handle excess hydrochloric acid, ensuring continuous operation and efficient recycling.
Method for producing high-purity hydrochloric acid
PatentInactiveUS6793905B1
Innovation
- The process involves heating hydrogen chloride gas from hydrochloric acid with a content greater than 21% to pass through a retention column and demister made of fluorinated or perfluorinated polyolefin, followed by absorption in ultrapure water, with recycling and concentration control to achieve a 35-38% hydrochloric acid solution, using a vaporization plant and adiabatic absorption column system.
Environmental Impact
The environmental impact of hydrochloric acid production has been a significant concern in recent years, prompting the industry to focus on developing more sustainable and eco-friendly production methods. Traditional production processes, such as the salt-sulfuric acid method and the chlorine-hydrogen synthesis route, have been associated with various environmental issues, including air pollution, water contamination, and high energy consumption.
One of the primary environmental challenges in hydrochloric acid production is the emission of chlorine gas and other volatile compounds. These emissions can contribute to air pollution and pose health risks to workers and nearby communities. To address this issue, advanced scrubbing systems and closed-loop production processes have been implemented, significantly reducing atmospheric emissions and improving air quality in production facilities and surrounding areas.
Water pollution has also been a major concern, particularly in facilities using the salt-sulfuric acid method. The discharge of contaminated wastewater containing high levels of dissolved salts and heavy metals can have detrimental effects on aquatic ecosystems. In response, innovative wastewater treatment technologies, such as membrane filtration and advanced oxidation processes, have been developed to minimize the environmental impact of effluents.
Energy consumption in hydrochloric acid production has been another area of focus for environmental improvements. The industry has made significant strides in enhancing energy efficiency through process optimization, heat recovery systems, and the integration of renewable energy sources. These efforts have not only reduced the carbon footprint of production facilities but also contributed to overall cost reduction and improved competitiveness.
The shift towards more sustainable raw materials has also played a crucial role in mitigating the environmental impact of hydrochloric acid production. For instance, the utilization of by-product hydrogen chloride from other industrial processes has gained traction, reducing the need for primary production and minimizing waste. Additionally, the development of bio-based precursors and green chemistry approaches has opened up new avenues for environmentally friendly hydrochloric acid production.
Waste reduction and recycling initiatives have further contributed to improving the environmental profile of hydrochloric acid production. Advanced separation techniques and process intensification have enabled the recovery and reuse of valuable components, minimizing the generation of hazardous waste and reducing the demand for raw materials.
As regulatory pressures and public awareness of environmental issues continue to grow, the hydrochloric acid industry is increasingly adopting life cycle assessment (LCA) approaches to evaluate and minimize the overall environmental impact of production processes. This holistic approach considers all stages of the product lifecycle, from raw material extraction to end-of-life disposal, enabling more comprehensive and sustainable decision-making in process design and optimization.
One of the primary environmental challenges in hydrochloric acid production is the emission of chlorine gas and other volatile compounds. These emissions can contribute to air pollution and pose health risks to workers and nearby communities. To address this issue, advanced scrubbing systems and closed-loop production processes have been implemented, significantly reducing atmospheric emissions and improving air quality in production facilities and surrounding areas.
Water pollution has also been a major concern, particularly in facilities using the salt-sulfuric acid method. The discharge of contaminated wastewater containing high levels of dissolved salts and heavy metals can have detrimental effects on aquatic ecosystems. In response, innovative wastewater treatment technologies, such as membrane filtration and advanced oxidation processes, have been developed to minimize the environmental impact of effluents.
Energy consumption in hydrochloric acid production has been another area of focus for environmental improvements. The industry has made significant strides in enhancing energy efficiency through process optimization, heat recovery systems, and the integration of renewable energy sources. These efforts have not only reduced the carbon footprint of production facilities but also contributed to overall cost reduction and improved competitiveness.
The shift towards more sustainable raw materials has also played a crucial role in mitigating the environmental impact of hydrochloric acid production. For instance, the utilization of by-product hydrogen chloride from other industrial processes has gained traction, reducing the need for primary production and minimizing waste. Additionally, the development of bio-based precursors and green chemistry approaches has opened up new avenues for environmentally friendly hydrochloric acid production.
Waste reduction and recycling initiatives have further contributed to improving the environmental profile of hydrochloric acid production. Advanced separation techniques and process intensification have enabled the recovery and reuse of valuable components, minimizing the generation of hazardous waste and reducing the demand for raw materials.
As regulatory pressures and public awareness of environmental issues continue to grow, the hydrochloric acid industry is increasingly adopting life cycle assessment (LCA) approaches to evaluate and minimize the overall environmental impact of production processes. This holistic approach considers all stages of the product lifecycle, from raw material extraction to end-of-life disposal, enabling more comprehensive and sustainable decision-making in process design and optimization.
Safety Considerations
Safety considerations are paramount in the production of hydrochloric acid due to its corrosive and hazardous nature. As efficiency improvements are implemented, it is crucial to maintain and enhance safety protocols to protect workers, equipment, and the environment. Modern production facilities employ advanced containment systems and corrosion-resistant materials to minimize the risk of leaks and spills. These systems often include double-walled tanks, specialized piping, and automated monitoring equipment to detect any potential breaches.
Personal protective equipment (PPE) remains a critical component of safety measures. Workers involved in hydrochloric acid production must wear acid-resistant suits, gloves, boots, and face shields or goggles. Respiratory protection may also be necessary in certain areas of the production facility. Regular training and drills ensure that personnel are well-versed in emergency procedures and the proper use of safety equipment.
Ventilation systems play a vital role in maintaining a safe working environment. State-of-the-art air handling units and scrubbers are employed to remove acid fumes and maintain air quality within acceptable limits. These systems are often equipped with redundancies and backup power sources to ensure continuous operation even in the event of primary system failure.
Emergency response planning is an integral part of safety considerations. Production facilities must have well-defined protocols for handling spills, leaks, or other incidents. This includes the installation of emergency showers and eyewash stations throughout the facility, as well as the strategic placement of neutralizing agents and spill containment materials.
As production efficiency advances, there is an increased focus on process automation and remote monitoring. These technologies not only improve productivity but also enhance safety by reducing the need for direct human interaction with hazardous materials. Automated systems can quickly detect and respond to abnormal conditions, often preventing incidents before they occur.
Environmental safety is another critical aspect of hydrochloric acid production. Modern facilities implement stringent emission control measures to prevent the release of acid fumes into the atmosphere. This may include the use of closed-loop systems, chemical scrubbers, and advanced filtration technologies. Wastewater treatment is also a key consideration, with facilities employing neutralization processes and monitoring systems to ensure that effluents meet or exceed regulatory standards.
Regular safety audits and inspections are essential to maintain high safety standards. These assessments help identify potential hazards, evaluate the effectiveness of existing safety measures, and implement improvements where necessary. Many facilities also participate in industry-wide safety initiatives and share best practices to continuously enhance safety protocols across the sector.
Personal protective equipment (PPE) remains a critical component of safety measures. Workers involved in hydrochloric acid production must wear acid-resistant suits, gloves, boots, and face shields or goggles. Respiratory protection may also be necessary in certain areas of the production facility. Regular training and drills ensure that personnel are well-versed in emergency procedures and the proper use of safety equipment.
Ventilation systems play a vital role in maintaining a safe working environment. State-of-the-art air handling units and scrubbers are employed to remove acid fumes and maintain air quality within acceptable limits. These systems are often equipped with redundancies and backup power sources to ensure continuous operation even in the event of primary system failure.
Emergency response planning is an integral part of safety considerations. Production facilities must have well-defined protocols for handling spills, leaks, or other incidents. This includes the installation of emergency showers and eyewash stations throughout the facility, as well as the strategic placement of neutralizing agents and spill containment materials.
As production efficiency advances, there is an increased focus on process automation and remote monitoring. These technologies not only improve productivity but also enhance safety by reducing the need for direct human interaction with hazardous materials. Automated systems can quickly detect and respond to abnormal conditions, often preventing incidents before they occur.
Environmental safety is another critical aspect of hydrochloric acid production. Modern facilities implement stringent emission control measures to prevent the release of acid fumes into the atmosphere. This may include the use of closed-loop systems, chemical scrubbers, and advanced filtration technologies. Wastewater treatment is also a key consideration, with facilities employing neutralization processes and monitoring systems to ensure that effluents meet or exceed regulatory standards.
Regular safety audits and inspections are essential to maintain high safety standards. These assessments help identify potential hazards, evaluate the effectiveness of existing safety measures, and implement improvements where necessary. Many facilities also participate in industry-wide safety initiatives and share best practices to continuously enhance safety protocols across the sector.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!