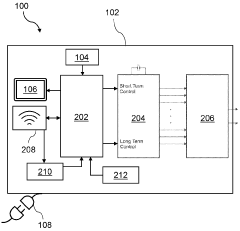
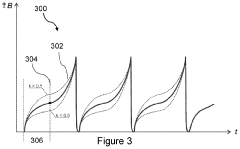
Evaluating Alternative Methods for PEMF Therapy Validation
AUG 11, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
PEMF Therapy Background and Objectives
Pulsed Electromagnetic Field (PEMF) therapy has emerged as a promising non-invasive treatment modality in recent years. This technology harnesses the power of electromagnetic fields to stimulate cellular repair and regeneration, offering potential benefits across a wide range of medical conditions. The evolution of PEMF therapy can be traced back to the mid-20th century, with significant advancements in understanding and application occurring over the past few decades.
The primary objective of PEMF therapy is to enhance the body's natural healing processes by influencing cellular behavior and metabolism. By generating pulsed electromagnetic fields, PEMF devices aim to improve circulation, reduce inflammation, and promote tissue repair at the cellular level. This approach has garnered attention in various medical fields, including orthopedics, neurology, and pain management.
As the technology has progressed, researchers and clinicians have explored its potential in treating conditions such as chronic pain, osteoarthritis, bone fractures, and even neurological disorders. The non-invasive nature of PEMF therapy, coupled with its potential for minimal side effects, has made it an attractive alternative or complementary treatment option in many cases.
However, despite the growing interest and anecdotal success stories, the scientific community continues to grapple with establishing standardized protocols and robust evidence for PEMF therapy's efficacy across different applications. This challenge has led to an increased focus on developing and evaluating alternative methods for validating PEMF therapy outcomes.
The current technological landscape of PEMF therapy encompasses a diverse range of devices, from portable home-use units to more sophisticated clinical systems. These devices vary in their field strength, frequency, and waveform characteristics, contributing to the complexity of standardizing treatment protocols and comparing results across studies.
As we look towards the future, the primary goals in PEMF therapy research and development include:
1. Establishing standardized protocols for different medical conditions
2. Developing more precise and targeted PEMF delivery systems
3. Enhancing our understanding of the cellular and molecular mechanisms underlying PEMF effects
4. Improving methods for measuring and quantifying PEMF therapy outcomes
5. Exploring potential synergies between PEMF therapy and other treatment modalities
By addressing these objectives, researchers and clinicians aim to solidify PEMF therapy's position in modern medicine and unlock its full potential as a versatile, non-invasive treatment option. The ongoing efforts to evaluate alternative methods for PEMF therapy validation are crucial in this journey, as they will help build a stronger evidence base and guide future innovations in the field.
The primary objective of PEMF therapy is to enhance the body's natural healing processes by influencing cellular behavior and metabolism. By generating pulsed electromagnetic fields, PEMF devices aim to improve circulation, reduce inflammation, and promote tissue repair at the cellular level. This approach has garnered attention in various medical fields, including orthopedics, neurology, and pain management.
As the technology has progressed, researchers and clinicians have explored its potential in treating conditions such as chronic pain, osteoarthritis, bone fractures, and even neurological disorders. The non-invasive nature of PEMF therapy, coupled with its potential for minimal side effects, has made it an attractive alternative or complementary treatment option in many cases.
However, despite the growing interest and anecdotal success stories, the scientific community continues to grapple with establishing standardized protocols and robust evidence for PEMF therapy's efficacy across different applications. This challenge has led to an increased focus on developing and evaluating alternative methods for validating PEMF therapy outcomes.
The current technological landscape of PEMF therapy encompasses a diverse range of devices, from portable home-use units to more sophisticated clinical systems. These devices vary in their field strength, frequency, and waveform characteristics, contributing to the complexity of standardizing treatment protocols and comparing results across studies.
As we look towards the future, the primary goals in PEMF therapy research and development include:
1. Establishing standardized protocols for different medical conditions
2. Developing more precise and targeted PEMF delivery systems
3. Enhancing our understanding of the cellular and molecular mechanisms underlying PEMF effects
4. Improving methods for measuring and quantifying PEMF therapy outcomes
5. Exploring potential synergies between PEMF therapy and other treatment modalities
By addressing these objectives, researchers and clinicians aim to solidify PEMF therapy's position in modern medicine and unlock its full potential as a versatile, non-invasive treatment option. The ongoing efforts to evaluate alternative methods for PEMF therapy validation are crucial in this journey, as they will help build a stronger evidence base and guide future innovations in the field.
Market Analysis for PEMF Therapy Devices
The PEMF (Pulsed Electromagnetic Field) therapy device market has shown significant growth in recent years, driven by increasing awareness of non-invasive treatment options and the rising prevalence of chronic conditions. The global PEMF therapy device market was valued at approximately $500 million in 2020 and is projected to reach $1.2 billion by 2027, growing at a CAGR of around 12% during the forecast period.
The market demand for PEMF therapy devices is primarily fueled by the growing aging population, rising incidence of musculoskeletal disorders, and increasing adoption of alternative pain management solutions. North America currently holds the largest market share, accounting for about 40% of the global market, followed by Europe and Asia-Pacific regions.
Key market segments include home-use devices, clinical-grade equipment, and portable systems. Home-use devices represent the fastest-growing segment, with a CAGR of 15%, as patients seek convenient and cost-effective treatment options. Clinical-grade equipment remains the largest segment in terms of revenue, driven by increasing adoption in hospitals and specialized pain management clinics.
The competitive landscape of the PEMF therapy device market is characterized by a mix of established medical device manufacturers and innovative startups. Leading players include OMI PEMF Therapy, Bemer Group, and Orthofix Medical, collectively holding about 35% of the market share. These companies are focusing on product innovation, clinical validation, and expanding their distribution networks to maintain their market positions.
Emerging trends in the PEMF therapy device market include the integration of smart technologies, such as mobile apps for treatment monitoring and personalization, as well as the development of wearable PEMF devices for continuous therapy. Additionally, there is a growing interest in combining PEMF therapy with other treatment modalities, such as photobiomodulation and electrostimulation, to enhance therapeutic outcomes.
Challenges facing the market include the need for more robust clinical evidence to support efficacy claims, regulatory hurdles in some regions, and the high cost of advanced PEMF devices, which may limit adoption in price-sensitive markets. However, ongoing research and development efforts are expected to address these challenges and drive further market growth.
In conclusion, the PEMF therapy device market presents significant opportunities for growth and innovation. As the demand for non-pharmacological pain management solutions continues to rise, manufacturers that can demonstrate clinical efficacy, improve user experience, and navigate regulatory landscapes effectively are likely to succeed in this expanding market.
The market demand for PEMF therapy devices is primarily fueled by the growing aging population, rising incidence of musculoskeletal disorders, and increasing adoption of alternative pain management solutions. North America currently holds the largest market share, accounting for about 40% of the global market, followed by Europe and Asia-Pacific regions.
Key market segments include home-use devices, clinical-grade equipment, and portable systems. Home-use devices represent the fastest-growing segment, with a CAGR of 15%, as patients seek convenient and cost-effective treatment options. Clinical-grade equipment remains the largest segment in terms of revenue, driven by increasing adoption in hospitals and specialized pain management clinics.
The competitive landscape of the PEMF therapy device market is characterized by a mix of established medical device manufacturers and innovative startups. Leading players include OMI PEMF Therapy, Bemer Group, and Orthofix Medical, collectively holding about 35% of the market share. These companies are focusing on product innovation, clinical validation, and expanding their distribution networks to maintain their market positions.
Emerging trends in the PEMF therapy device market include the integration of smart technologies, such as mobile apps for treatment monitoring and personalization, as well as the development of wearable PEMF devices for continuous therapy. Additionally, there is a growing interest in combining PEMF therapy with other treatment modalities, such as photobiomodulation and electrostimulation, to enhance therapeutic outcomes.
Challenges facing the market include the need for more robust clinical evidence to support efficacy claims, regulatory hurdles in some regions, and the high cost of advanced PEMF devices, which may limit adoption in price-sensitive markets. However, ongoing research and development efforts are expected to address these challenges and drive further market growth.
In conclusion, the PEMF therapy device market presents significant opportunities for growth and innovation. As the demand for non-pharmacological pain management solutions continues to rise, manufacturers that can demonstrate clinical efficacy, improve user experience, and navigate regulatory landscapes effectively are likely to succeed in this expanding market.
Current PEMF Validation Challenges
Pulsed Electromagnetic Field (PEMF) therapy has gained significant attention in recent years for its potential therapeutic benefits. However, validating the efficacy of PEMF devices and treatments presents several challenges that researchers and clinicians must address. One of the primary obstacles is the lack of standardized protocols for PEMF therapy validation, which makes it difficult to compare results across different studies and devices.
The variability in PEMF parameters, such as frequency, intensity, and waveform, further complicates the validation process. Different devices and treatment protocols may use vastly different settings, making it challenging to establish a clear cause-and-effect relationship between specific PEMF parameters and observed therapeutic outcomes. This variability also hinders the development of consistent guidelines for clinical applications.
Another significant challenge is the placebo effect, which is particularly pronounced in PEMF therapy due to its non-invasive nature and the subtle sensations experienced during treatment. Designing robust double-blind studies that can effectively control for placebo effects while maintaining the integrity of the PEMF intervention is a complex task that researchers continue to grapple with.
The long-term effects of PEMF therapy are also difficult to assess, as many studies focus on short-term outcomes. Validating the sustained benefits of PEMF treatments over extended periods requires longitudinal studies, which are often resource-intensive and challenging to conduct. This limitation in long-term data makes it harder to establish the overall efficacy and safety profile of PEMF therapy for chronic conditions.
Biological variability among individuals presents another hurdle in PEMF validation. The response to electromagnetic fields can vary significantly between subjects, influenced by factors such as age, health status, and genetic predisposition. This variability necessitates larger sample sizes and more sophisticated statistical analyses to draw meaningful conclusions about PEMF efficacy across diverse populations.
The lack of standardized measurement tools and biomarkers specific to PEMF therapy effects further complicates validation efforts. While some studies rely on subjective patient-reported outcomes, others attempt to quantify physiological changes. However, there is no consensus on which biomarkers or measurement techniques are most appropriate or reliable for assessing PEMF therapy outcomes.
Lastly, the interdisciplinary nature of PEMF research poses a challenge in itself. Effective validation requires collaboration between physicists, bioengineers, clinicians, and statisticians. Bridging the knowledge gap between these disciplines and developing a common language for PEMF validation is crucial for advancing the field and establishing credible evidence for its therapeutic applications.
The variability in PEMF parameters, such as frequency, intensity, and waveform, further complicates the validation process. Different devices and treatment protocols may use vastly different settings, making it challenging to establish a clear cause-and-effect relationship between specific PEMF parameters and observed therapeutic outcomes. This variability also hinders the development of consistent guidelines for clinical applications.
Another significant challenge is the placebo effect, which is particularly pronounced in PEMF therapy due to its non-invasive nature and the subtle sensations experienced during treatment. Designing robust double-blind studies that can effectively control for placebo effects while maintaining the integrity of the PEMF intervention is a complex task that researchers continue to grapple with.
The long-term effects of PEMF therapy are also difficult to assess, as many studies focus on short-term outcomes. Validating the sustained benefits of PEMF treatments over extended periods requires longitudinal studies, which are often resource-intensive and challenging to conduct. This limitation in long-term data makes it harder to establish the overall efficacy and safety profile of PEMF therapy for chronic conditions.
Biological variability among individuals presents another hurdle in PEMF validation. The response to electromagnetic fields can vary significantly between subjects, influenced by factors such as age, health status, and genetic predisposition. This variability necessitates larger sample sizes and more sophisticated statistical analyses to draw meaningful conclusions about PEMF efficacy across diverse populations.
The lack of standardized measurement tools and biomarkers specific to PEMF therapy effects further complicates validation efforts. While some studies rely on subjective patient-reported outcomes, others attempt to quantify physiological changes. However, there is no consensus on which biomarkers or measurement techniques are most appropriate or reliable for assessing PEMF therapy outcomes.
Lastly, the interdisciplinary nature of PEMF research poses a challenge in itself. Effective validation requires collaboration between physicists, bioengineers, clinicians, and statisticians. Bridging the knowledge gap between these disciplines and developing a common language for PEMF validation is crucial for advancing the field and establishing credible evidence for its therapeutic applications.
Existing PEMF Validation Methods
01 PEMF therapy devices and systems
Various devices and systems have been developed for delivering Pulsed Electromagnetic Field (PEMF) therapy. These include portable devices, wearable applicators, and integrated systems that can generate and control electromagnetic fields for therapeutic purposes. The designs focus on optimizing field strength, frequency, and treatment duration for different medical conditions.- PEMF therapy devices and systems: Various devices and systems have been developed for delivering Pulsed Electromagnetic Field (PEMF) therapy. These include portable devices, wearable applicators, and integrated systems that can generate and control electromagnetic fields for therapeutic purposes. The designs focus on optimizing field strength, frequency, and waveform to achieve desired biological effects.
- Validation methods for PEMF therapy: Validation of PEMF therapy involves various methods to assess its efficacy and safety. These include clinical trials, in vitro studies, and animal models. Researchers use biomarkers, imaging techniques, and patient-reported outcomes to evaluate the therapeutic effects. Validation processes also include standardization of treatment protocols and quality control measures for PEMF devices.
- PEMF therapy applications in medicine: PEMF therapy has been investigated for various medical applications. These include pain management, bone healing, wound healing, and treatment of neurological disorders. Research focuses on optimizing treatment parameters for specific conditions and understanding the underlying mechanisms of action. The therapy is being explored as a non-invasive alternative or complement to conventional treatments.
- Integration of PEMF therapy with other technologies: Efforts are being made to integrate PEMF therapy with other technologies to enhance its effectiveness. This includes combining PEMF with biofeedback systems, incorporating it into smart wearable devices, and using artificial intelligence for personalized treatment optimization. Such integrations aim to improve treatment outcomes and patient compliance.
- Quality control and standardization in PEMF therapy: To ensure the reliability and reproducibility of PEMF therapy, there is a focus on quality control and standardization. This includes developing standardized protocols for device calibration, treatment delivery, and outcome measurement. Efforts are also being made to establish regulatory guidelines and industry standards for PEMF devices and therapies.
02 Validation methods for PEMF therapy efficacy
Researchers have developed various methods to validate the efficacy of PEMF therapy. These include clinical trials, in vitro studies, and animal models. Validation techniques often involve measuring physiological changes, such as cell proliferation, inflammation reduction, and pain relief. Advanced imaging and biomarker analysis are also used to assess the therapy's effects on tissue repair and regeneration.Expand Specific Solutions03 PEMF therapy applications in specific medical conditions
PEMF therapy has been investigated for its potential benefits in treating various medical conditions. Studies have explored its use in pain management, bone healing, wound repair, and neurological disorders. Researchers have also examined its effects on cellular processes, including inflammation reduction and tissue regeneration, to validate its therapeutic potential across different medical fields.Expand Specific Solutions04 PEMF therapy dosage and treatment protocols
Researchers have focused on optimizing PEMF therapy dosage and treatment protocols to enhance its effectiveness and safety. This includes determining optimal field strengths, frequencies, pulse shapes, and treatment durations for different conditions. Validation studies have explored the relationship between these parameters and therapeutic outcomes, aiming to establish standardized protocols for clinical use.Expand Specific Solutions05 Integration of PEMF therapy with other treatment modalities
Studies have investigated the combination of PEMF therapy with other treatment modalities to enhance overall therapeutic efficacy. This includes integrating PEMF with physical therapy, pharmacological treatments, and regenerative medicine approaches. Validation efforts have focused on assessing synergistic effects and potential improvements in treatment outcomes when PEMF is used as part of a comprehensive therapeutic strategy.Expand Specific Solutions
Key PEMF Industry Players
The competitive landscape for PEMF therapy validation is evolving rapidly, with the market currently in a growth phase. The global PEMF therapy market is expanding, driven by increasing awareness of non-invasive treatment options and growing applications in pain management and tissue regeneration. Technologically, the field is advancing, with companies like Regenesis Biomedical and SofPulse leading in device development. Established players such as Medtronic and NEC Corp are leveraging their resources to explore PEMF applications, while research institutions like The University of Texas System and Emory University are contributing to the scientific validation of PEMF therapies. The involvement of pharmaceutical giants like Regeneron and Biogen indicates growing interest in integrating PEMF technology with traditional drug therapies, potentially reshaping treatment paradigms in various medical fields.
Regenesis Biomedical, Inc.
Technical Solution: Regenesis Biomedical has developed a proprietary PEMF therapy system called Provant Therapy. Their approach focuses on using specific pulsed electromagnetic field frequencies to stimulate cellular repair and reduce inflammation. The system delivers targeted PEMF therapy to affected areas, with treatment sessions typically lasting 30 minutes[1]. Regenesis has conducted clinical trials to validate the efficacy of their PEMF therapy for various conditions, including post-operative pain and edema reduction. They employ a combination of in vitro studies, animal models, and human clinical trials to evaluate the biological effects and therapeutic outcomes of their PEMF technology[2].
Strengths: Proprietary PEMF technology with specific frequencies, extensive clinical trial data. Weaknesses: Limited to specific medical applications, may require longer treatment times compared to some alternatives.
SofPulse, Inc.
Technical Solution: SofPulse has developed a wearable PEMF therapy device that utilizes a unique low-frequency, low-intensity electromagnetic field. Their validation approach includes both laboratory studies and clinical trials. The company employs in vitro experiments to assess the effects of their PEMF technology on cellular processes such as inflammation and tissue repair. They have also conducted randomized controlled trials to evaluate the efficacy of their device in reducing post-operative pain and accelerating wound healing[3]. SofPulse's validation methods include measuring objective outcomes like analgesic medication usage and wound closure rates, as well as subjective patient-reported pain scores[4].
Strengths: Wearable, portable design for continuous treatment, focus on post-operative care. Weaknesses: Limited to specific frequency range, may not be suitable for all PEMF applications.
Innovative PEMF Validation Techniques
A pulsed electromagnetic field apparatus and method for generating frequencies
PatentWO2024127242A1
Innovation
- A PEMF apparatus with a pulse generator and electromagnetic field generation means that uses modified sawtooth waveforms with pre-stress and relaxation periods, and quasi-sine signals with pulse width modulation, along with a feedback circuit for frequency stability and precision, and a bifilar antenna for scalar wave generation.
Pulsed electromagnetic field (PEMF) stimulation therapy system with bi-phasic coil
PatentInactiveUS6132362A
Innovation
- A PEMF therapy system with a high-efficiency single-coil transducer that recovers flyback energy and uses an energy recovery capacitance circuit to sequence current in both directions, eliminating the need for a secondary coil, thereby reducing weight, power consumption, and manufacturing complexity.
Regulatory Framework for PEMF Devices
The regulatory framework for PEMF (Pulsed Electromagnetic Field) devices plays a crucial role in ensuring the safety and efficacy of these therapeutic tools. In the United States, the Food and Drug Administration (FDA) oversees the regulation of PEMF devices, classifying them as Class II medical devices. This classification requires manufacturers to submit a 510(k) premarket notification, demonstrating that their device is substantially equivalent to a legally marketed predicate device in terms of safety and effectiveness.
The FDA's regulatory process for PEMF devices involves several key components. First, manufacturers must provide detailed information about the device's intended use, technical specifications, and performance characteristics. This includes data on the electromagnetic field strength, frequency range, and pulse patterns generated by the device. Additionally, manufacturers are required to submit clinical data supporting the device's safety and efficacy for its intended use.
In Europe, PEMF devices fall under the purview of the Medical Device Regulation (MDR). The MDR establishes a comprehensive framework for the assessment and monitoring of medical devices, including PEMF therapy equipment. Under this regulation, manufacturers must obtain CE marking for their devices, which involves demonstrating compliance with essential safety and performance requirements.
The regulatory landscape for PEMF devices also extends to other regions globally. In Canada, Health Canada regulates these devices under the Medical Devices Regulations, while in Australia, the Therapeutic Goods Administration (TGA) oversees their approval and marketing. These regulatory bodies often have similar requirements to those of the FDA and MDR, emphasizing safety, efficacy, and quality control.
One of the key challenges in the regulatory framework for PEMF devices is the validation of therapeutic claims. Regulatory agencies require robust scientific evidence to support any claims made about the device's effectiveness in treating specific conditions. This often necessitates well-designed clinical trials and peer-reviewed studies, which can be time-consuming and costly for manufacturers.
Furthermore, the regulatory framework addresses the potential risks associated with PEMF therapy, such as electromagnetic interference with other medical devices. Manufacturers must demonstrate that their devices do not pose unacceptable risks to patients or interfere with the function of other critical medical equipment.
As the field of PEMF therapy continues to evolve, regulatory bodies are adapting their frameworks to keep pace with technological advancements. This includes developing new guidelines for emerging PEMF applications and refining the criteria for evaluating the safety and efficacy of these devices. The ongoing dialogue between regulatory agencies, manufacturers, and the scientific community is essential in shaping a regulatory environment that promotes innovation while safeguarding public health.
The FDA's regulatory process for PEMF devices involves several key components. First, manufacturers must provide detailed information about the device's intended use, technical specifications, and performance characteristics. This includes data on the electromagnetic field strength, frequency range, and pulse patterns generated by the device. Additionally, manufacturers are required to submit clinical data supporting the device's safety and efficacy for its intended use.
In Europe, PEMF devices fall under the purview of the Medical Device Regulation (MDR). The MDR establishes a comprehensive framework for the assessment and monitoring of medical devices, including PEMF therapy equipment. Under this regulation, manufacturers must obtain CE marking for their devices, which involves demonstrating compliance with essential safety and performance requirements.
The regulatory landscape for PEMF devices also extends to other regions globally. In Canada, Health Canada regulates these devices under the Medical Devices Regulations, while in Australia, the Therapeutic Goods Administration (TGA) oversees their approval and marketing. These regulatory bodies often have similar requirements to those of the FDA and MDR, emphasizing safety, efficacy, and quality control.
One of the key challenges in the regulatory framework for PEMF devices is the validation of therapeutic claims. Regulatory agencies require robust scientific evidence to support any claims made about the device's effectiveness in treating specific conditions. This often necessitates well-designed clinical trials and peer-reviewed studies, which can be time-consuming and costly for manufacturers.
Furthermore, the regulatory framework addresses the potential risks associated with PEMF therapy, such as electromagnetic interference with other medical devices. Manufacturers must demonstrate that their devices do not pose unacceptable risks to patients or interfere with the function of other critical medical equipment.
As the field of PEMF therapy continues to evolve, regulatory bodies are adapting their frameworks to keep pace with technological advancements. This includes developing new guidelines for emerging PEMF applications and refining the criteria for evaluating the safety and efficacy of these devices. The ongoing dialogue between regulatory agencies, manufacturers, and the scientific community is essential in shaping a regulatory environment that promotes innovation while safeguarding public health.
PEMF Therapy Safety Considerations
Safety considerations are paramount when evaluating alternative methods for PEMF therapy validation. The primary concern is ensuring that the electromagnetic fields generated during therapy do not pose any health risks to patients or operators. This requires a comprehensive assessment of potential biological effects, both short-term and long-term, associated with exposure to pulsed electromagnetic fields.
One crucial aspect of safety evaluation is determining the appropriate intensity and frequency ranges for PEMF therapy. Different tissues and conditions may require varying field strengths and pulse patterns, necessitating careful calibration and monitoring of device output. Researchers must establish clear safety thresholds based on extensive preclinical and clinical studies, taking into account factors such as exposure duration, target tissue characteristics, and individual patient sensitivities.
Another important safety consideration is the potential for electromagnetic interference with other medical devices. PEMF therapy devices must be designed and tested to ensure compatibility with implanted electronic devices, such as pacemakers or insulin pumps, as well as other diagnostic and therapeutic equipment commonly found in healthcare settings. This requires rigorous electromagnetic compatibility (EMC) testing and adherence to relevant regulatory standards.
The long-term effects of repeated PEMF exposure must also be carefully evaluated. While short-term studies have generally shown PEMF therapy to be safe, more research is needed to assess potential cumulative effects over extended periods. This includes investigating any possible impact on cellular processes, DNA integrity, and tissue function, particularly in sensitive populations such as pregnant women, children, and the elderly.
Quality control and device reliability are critical components of PEMF therapy safety. Validation methods must include thorough testing of device components, including coils, power supplies, and control systems, to ensure consistent and accurate field generation. Regular calibration and maintenance protocols should be established to maintain device performance and safety over time.
Lastly, proper training and education for healthcare providers and patients are essential for safe PEMF therapy implementation. Clear guidelines on device operation, treatment protocols, and potential contraindications must be developed and communicated effectively. This includes providing information on recognizing and reporting any adverse effects, as well as instructions for proper device handling and maintenance.
One crucial aspect of safety evaluation is determining the appropriate intensity and frequency ranges for PEMF therapy. Different tissues and conditions may require varying field strengths and pulse patterns, necessitating careful calibration and monitoring of device output. Researchers must establish clear safety thresholds based on extensive preclinical and clinical studies, taking into account factors such as exposure duration, target tissue characteristics, and individual patient sensitivities.
Another important safety consideration is the potential for electromagnetic interference with other medical devices. PEMF therapy devices must be designed and tested to ensure compatibility with implanted electronic devices, such as pacemakers or insulin pumps, as well as other diagnostic and therapeutic equipment commonly found in healthcare settings. This requires rigorous electromagnetic compatibility (EMC) testing and adherence to relevant regulatory standards.
The long-term effects of repeated PEMF exposure must also be carefully evaluated. While short-term studies have generally shown PEMF therapy to be safe, more research is needed to assess potential cumulative effects over extended periods. This includes investigating any possible impact on cellular processes, DNA integrity, and tissue function, particularly in sensitive populations such as pregnant women, children, and the elderly.
Quality control and device reliability are critical components of PEMF therapy safety. Validation methods must include thorough testing of device components, including coils, power supplies, and control systems, to ensure consistent and accurate field generation. Regular calibration and maintenance protocols should be established to maintain device performance and safety over time.
Lastly, proper training and education for healthcare providers and patients are essential for safe PEMF therapy implementation. Clear guidelines on device operation, treatment protocols, and potential contraindications must be developed and communicated effectively. This includes providing information on recognizing and reporting any adverse effects, as well as instructions for proper device handling and maintenance.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!