Exploring Biofeedback Enhancements through PEMF Therapy
AUG 11, 20259 MIN READ
Generate Your Research Report Instantly with AI Agent
PatSnap Eureka helps you evaluate technical feasibility & market potential.
PEMF Biofeedback Evolution
The evolution of PEMF (Pulsed Electromagnetic Field) biofeedback represents a significant advancement in the field of non-invasive therapeutic technologies. This innovative approach combines the established benefits of PEMF therapy with the precision of biofeedback systems, creating a synergistic treatment modality that adapts to individual physiological responses in real-time.
PEMF therapy, which has been in use for several decades, traditionally involves the application of electromagnetic fields to stimulate cellular repair and enhance overall well-being. The integration of biofeedback into this process marks a crucial step forward, allowing for more personalized and effective treatments.
The early stages of PEMF biofeedback evolution focused on basic physiological measurements, such as heart rate variability and skin conductance. These initial systems provided rudimentary feedback loops, adjusting PEMF parameters based on simple biological markers. As technology progressed, more sophisticated sensors and data analysis techniques were incorporated, enabling the measurement of more complex physiological responses.
A significant milestone in this evolution was the development of real-time neural feedback systems. These advanced setups could monitor brain wave patterns and adjust PEMF output accordingly, opening new possibilities for treating neurological conditions and optimizing cognitive performance.
The integration of artificial intelligence and machine learning algorithms marked another pivotal point in PEMF biofeedback evolution. These technologies allowed for more nuanced interpretation of physiological data and predictive modeling of individual responses to PEMF therapy. This led to highly personalized treatment protocols that could adapt not just in real-time but also over extended periods, learning from cumulative data to optimize long-term outcomes.
Recent advancements have seen the incorporation of wearable technology and Internet of Things (IoT) devices into PEMF biofeedback systems. This has expanded the scope of data collection beyond clinical settings, allowing for continuous monitoring and adjustment of PEMF parameters in everyday life situations. The result is a more holistic approach to therapy that considers the full spectrum of an individual's physiological states throughout their daily activities.
Looking forward, the trajectory of PEMF biofeedback evolution points towards even greater integration with other emerging technologies. The potential combination with virtual and augmented reality systems could create immersive therapeutic experiences that engage multiple sensory modalities alongside PEMF stimulation. Additionally, the ongoing miniaturization of PEMF devices suggests a future where this technology could be seamlessly incorporated into clothing or even implantable devices, providing constant, adaptive electromagnetic therapy tailored to individual needs.
PEMF therapy, which has been in use for several decades, traditionally involves the application of electromagnetic fields to stimulate cellular repair and enhance overall well-being. The integration of biofeedback into this process marks a crucial step forward, allowing for more personalized and effective treatments.
The early stages of PEMF biofeedback evolution focused on basic physiological measurements, such as heart rate variability and skin conductance. These initial systems provided rudimentary feedback loops, adjusting PEMF parameters based on simple biological markers. As technology progressed, more sophisticated sensors and data analysis techniques were incorporated, enabling the measurement of more complex physiological responses.
A significant milestone in this evolution was the development of real-time neural feedback systems. These advanced setups could monitor brain wave patterns and adjust PEMF output accordingly, opening new possibilities for treating neurological conditions and optimizing cognitive performance.
The integration of artificial intelligence and machine learning algorithms marked another pivotal point in PEMF biofeedback evolution. These technologies allowed for more nuanced interpretation of physiological data and predictive modeling of individual responses to PEMF therapy. This led to highly personalized treatment protocols that could adapt not just in real-time but also over extended periods, learning from cumulative data to optimize long-term outcomes.
Recent advancements have seen the incorporation of wearable technology and Internet of Things (IoT) devices into PEMF biofeedback systems. This has expanded the scope of data collection beyond clinical settings, allowing for continuous monitoring and adjustment of PEMF parameters in everyday life situations. The result is a more holistic approach to therapy that considers the full spectrum of an individual's physiological states throughout their daily activities.
Looking forward, the trajectory of PEMF biofeedback evolution points towards even greater integration with other emerging technologies. The potential combination with virtual and augmented reality systems could create immersive therapeutic experiences that engage multiple sensory modalities alongside PEMF stimulation. Additionally, the ongoing miniaturization of PEMF devices suggests a future where this technology could be seamlessly incorporated into clothing or even implantable devices, providing constant, adaptive electromagnetic therapy tailored to individual needs.
Market Demand Analysis
The market demand for biofeedback enhancements through PEMF (Pulsed Electromagnetic Field) therapy has been steadily growing in recent years, driven by increasing awareness of non-invasive treatment options and a growing emphasis on personalized healthcare. This innovative approach combines the established benefits of PEMF therapy with the precision of biofeedback systems, creating a synergistic effect that addresses a wide range of health concerns.
The global PEMF therapy devices market is experiencing significant expansion, with projections indicating robust growth over the next decade. This growth is fueled by the rising prevalence of chronic diseases, musculoskeletal disorders, and stress-related conditions. The integration of biofeedback technology into PEMF therapy devices is expected to further accelerate market growth by enhancing treatment efficacy and patient engagement.
One of the key drivers of market demand is the increasing adoption of complementary and alternative medicine (CAM) practices. Consumers are increasingly seeking non-pharmacological approaches to manage pain, improve sleep quality, and enhance overall well-being. The combination of PEMF therapy with biofeedback offers a compelling solution that aligns with this trend, providing real-time physiological data to optimize treatment parameters and outcomes.
The healthcare industry's shift towards preventive and personalized medicine is another significant factor contributing to the growing demand for biofeedback-enhanced PEMF therapy. As healthcare providers and patients alike recognize the importance of tailored treatment approaches, technologies that offer customizable and data-driven solutions are gaining traction. This shift is particularly evident in the management of chronic conditions, where long-term, personalized care strategies are essential.
Sports medicine and athletic performance enhancement represent another burgeoning market segment for biofeedback-enhanced PEMF therapy. Professional athletes and sports teams are increasingly incorporating these technologies into their training and recovery regimens, driving demand in the high-performance sports sector. This trend is gradually trickling down to amateur athletes and fitness enthusiasts, expanding the potential consumer base.
The aging population in many developed countries is also contributing to market growth. As the incidence of age-related conditions such as arthritis, osteoporosis, and cognitive decline increases, there is a growing demand for non-invasive, home-based therapies that can improve quality of life and maintain independence. Biofeedback-enhanced PEMF therapy offers a promising solution for this demographic, with its potential to address multiple health concerns simultaneously.
Despite the positive market outlook, challenges remain in terms of regulatory approval processes and insurance coverage for these emerging therapies. However, ongoing clinical research and technological advancements are expected to address these barriers, potentially leading to broader acceptance and integration into mainstream healthcare practices. As the body of evidence supporting the efficacy of biofeedback-enhanced PEMF therapy continues to grow, it is likely to drive further market expansion and innovation in the coming years.
The global PEMF therapy devices market is experiencing significant expansion, with projections indicating robust growth over the next decade. This growth is fueled by the rising prevalence of chronic diseases, musculoskeletal disorders, and stress-related conditions. The integration of biofeedback technology into PEMF therapy devices is expected to further accelerate market growth by enhancing treatment efficacy and patient engagement.
One of the key drivers of market demand is the increasing adoption of complementary and alternative medicine (CAM) practices. Consumers are increasingly seeking non-pharmacological approaches to manage pain, improve sleep quality, and enhance overall well-being. The combination of PEMF therapy with biofeedback offers a compelling solution that aligns with this trend, providing real-time physiological data to optimize treatment parameters and outcomes.
The healthcare industry's shift towards preventive and personalized medicine is another significant factor contributing to the growing demand for biofeedback-enhanced PEMF therapy. As healthcare providers and patients alike recognize the importance of tailored treatment approaches, technologies that offer customizable and data-driven solutions are gaining traction. This shift is particularly evident in the management of chronic conditions, where long-term, personalized care strategies are essential.
Sports medicine and athletic performance enhancement represent another burgeoning market segment for biofeedback-enhanced PEMF therapy. Professional athletes and sports teams are increasingly incorporating these technologies into their training and recovery regimens, driving demand in the high-performance sports sector. This trend is gradually trickling down to amateur athletes and fitness enthusiasts, expanding the potential consumer base.
The aging population in many developed countries is also contributing to market growth. As the incidence of age-related conditions such as arthritis, osteoporosis, and cognitive decline increases, there is a growing demand for non-invasive, home-based therapies that can improve quality of life and maintain independence. Biofeedback-enhanced PEMF therapy offers a promising solution for this demographic, with its potential to address multiple health concerns simultaneously.
Despite the positive market outlook, challenges remain in terms of regulatory approval processes and insurance coverage for these emerging therapies. However, ongoing clinical research and technological advancements are expected to address these barriers, potentially leading to broader acceptance and integration into mainstream healthcare practices. As the body of evidence supporting the efficacy of biofeedback-enhanced PEMF therapy continues to grow, it is likely to drive further market expansion and innovation in the coming years.
PEMF Tech Challenges
Pulsed Electromagnetic Field (PEMF) therapy, while promising, faces several technological challenges that hinder its widespread adoption and efficacy in biofeedback enhancement. One of the primary obstacles is the lack of standardization in PEMF devices and protocols. The wide variety of frequencies, intensities, and waveforms used in different PEMF systems makes it difficult to compare results across studies and establish optimal treatment parameters.
Another significant challenge is the limited understanding of the precise mechanisms by which PEMF interacts with biological tissues. While theories exist, such as the ion cyclotron resonance model and the stochastic resonance hypothesis, more research is needed to fully elucidate how PEMF influences cellular processes and physiological responses. This knowledge gap impedes the development of more targeted and effective PEMF therapies.
The issue of penetration depth also presents a technical hurdle. PEMF fields attenuate rapidly as they pass through tissue, limiting their effectiveness in treating deep-seated conditions. Developing methods to enhance field penetration without increasing the risk of adverse effects is an ongoing challenge for researchers and engineers in the field.
Furthermore, the integration of real-time biofeedback with PEMF therapy poses significant technical difficulties. Creating systems that can accurately measure physiological responses and dynamically adjust PEMF parameters in real-time requires advanced sensors, sophisticated algorithms, and robust data processing capabilities. The complexity of such systems increases both development costs and potential points of failure.
Miniaturization and portability of PEMF devices present another set of challenges. As the demand for wearable and home-use PEMF devices grows, engineers must balance power requirements, field strength, and battery life while maintaining device efficacy and user comfort. This often involves trade-offs between performance and practicality.
Additionally, electromagnetic interference (EMI) with other medical devices and electronic equipment is a concern, particularly in clinical settings. Developing PEMF systems that can operate effectively without disrupting other critical equipment requires careful engineering and shielding techniques.
Lastly, the long-term effects of PEMF exposure, especially with frequent or prolonged use, are not fully understood. This knowledge gap raises safety concerns and complicates the regulatory approval process for new PEMF devices and therapies. Conducting comprehensive long-term studies to establish safety profiles and potential cumulative effects is both time-consuming and resource-intensive.
Another significant challenge is the limited understanding of the precise mechanisms by which PEMF interacts with biological tissues. While theories exist, such as the ion cyclotron resonance model and the stochastic resonance hypothesis, more research is needed to fully elucidate how PEMF influences cellular processes and physiological responses. This knowledge gap impedes the development of more targeted and effective PEMF therapies.
The issue of penetration depth also presents a technical hurdle. PEMF fields attenuate rapidly as they pass through tissue, limiting their effectiveness in treating deep-seated conditions. Developing methods to enhance field penetration without increasing the risk of adverse effects is an ongoing challenge for researchers and engineers in the field.
Furthermore, the integration of real-time biofeedback with PEMF therapy poses significant technical difficulties. Creating systems that can accurately measure physiological responses and dynamically adjust PEMF parameters in real-time requires advanced sensors, sophisticated algorithms, and robust data processing capabilities. The complexity of such systems increases both development costs and potential points of failure.
Miniaturization and portability of PEMF devices present another set of challenges. As the demand for wearable and home-use PEMF devices grows, engineers must balance power requirements, field strength, and battery life while maintaining device efficacy and user comfort. This often involves trade-offs between performance and practicality.
Additionally, electromagnetic interference (EMI) with other medical devices and electronic equipment is a concern, particularly in clinical settings. Developing PEMF systems that can operate effectively without disrupting other critical equipment requires careful engineering and shielding techniques.
Lastly, the long-term effects of PEMF exposure, especially with frequent or prolonged use, are not fully understood. This knowledge gap raises safety concerns and complicates the regulatory approval process for new PEMF devices and therapies. Conducting comprehensive long-term studies to establish safety profiles and potential cumulative effects is both time-consuming and resource-intensive.
Current PEMF Solutions
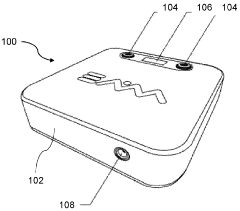
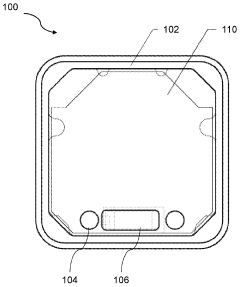
01 PEMF therapy devices with biofeedback capabilities
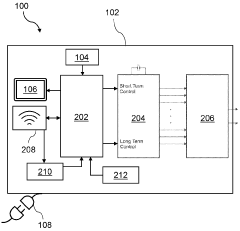
These devices combine Pulsed Electromagnetic Field (PEMF) therapy with biofeedback mechanisms to provide personalized treatment. The systems monitor physiological responses and adjust PEMF parameters accordingly, optimizing therapeutic outcomes. This integration allows for real-time adaptation of the therapy based on the patient's biological signals.- PEMF therapy combined with biofeedback systems: Integration of Pulsed Electromagnetic Field (PEMF) therapy with biofeedback systems for enhanced therapeutic outcomes. This combination allows for real-time monitoring and adjustment of PEMF parameters based on physiological responses, potentially improving treatment efficacy and personalization.
- Wearable PEMF devices with biofeedback capabilities: Development of portable and wearable PEMF devices incorporating biofeedback sensors. These devices enable continuous monitoring of physiological parameters and automatic adjustment of PEMF therapy, allowing for convenient and personalized treatment in various settings.
- PEMF therapy for stress reduction and relaxation: Application of PEMF therapy in combination with biofeedback techniques for stress reduction and relaxation. This approach utilizes physiological indicators such as heart rate variability and skin conductance to optimize PEMF parameters for achieving a relaxed state.
- PEMF and biofeedback for pain management: Utilization of PEMF therapy in conjunction with biofeedback mechanisms for pain management applications. This combination allows for targeted treatment of pain areas with real-time adjustment based on pain perception and physiological responses.
- Advanced control systems for PEMF biofeedback devices: Development of sophisticated control systems and algorithms for PEMF devices with biofeedback capabilities. These systems enable precise modulation of PEMF parameters based on multiple physiological inputs, enhancing the overall effectiveness and adaptability of the therapy.
02 Wearable PEMF devices with integrated sensors
Wearable devices that incorporate PEMF technology and various sensors for biofeedback. These devices can be worn on different parts of the body and continuously monitor physiological parameters while delivering PEMF therapy. The integration of sensors allows for ongoing assessment and adjustment of the treatment.Expand Specific Solutions03 PEMF therapy systems with cognitive biofeedback
Systems that combine PEMF therapy with cognitive biofeedback techniques. These devices may include features for monitoring brain activity, stress levels, or cognitive performance. The PEMF therapy is then adjusted based on the cognitive biofeedback data to enhance mental well-being and cognitive function.Expand Specific Solutions04 Multi-modal biofeedback systems incorporating PEMF
Advanced systems that integrate multiple biofeedback modalities with PEMF therapy. These devices may combine various sensing technologies such as EEG, EMG, heart rate variability, and skin conductance with PEMF application. The multi-modal approach allows for a more comprehensive assessment and targeted therapy.Expand Specific Solutions05 PEMF therapy devices with mobile app integration
PEMF devices that connect to mobile applications for enhanced biofeedback and user interaction. These systems allow users to track their therapy progress, view biofeedback data, and adjust treatment parameters through smartphone apps. The integration of mobile technology facilitates remote monitoring and personalized therapy management.Expand Specific Solutions
Key PEMF Players
The field of biofeedback enhancement through PEMF therapy is in a growth stage, with increasing market potential and technological advancements. The global PEMF therapy devices market is expanding, driven by rising awareness of non-invasive treatment options and growing applications in pain management and rehabilitation. Companies like Venus Concept Ltd., SofPulse, Inc., and Regenesis Biomedical, Inc. are at the forefront of developing innovative PEMF technologies, indicating a moderate level of technological maturity. However, the involvement of research institutions such as the National University of Singapore and Swiss Federal Institute of Technology suggests ongoing scientific exploration and potential for further breakthroughs in this field.
Venus Concept Ltd.
Technical Solution: Venus Concept has developed a proprietary PEMF technology called Venus Pulse™, which is integrated into their Venus Heal™ device. This system delivers non-thermal PEMF energy to penetrate deep into soft tissues, promoting cellular health and regeneration. The technology utilizes precise electromagnetic fields to stimulate biological effects at the cellular level, enhancing circulation, reducing inflammation, and accelerating healing processes[1]. Venus Concept's PEMF therapy is designed to be customizable, allowing practitioners to adjust treatment parameters based on individual patient needs and specific conditions being treated[2].
Strengths: Customizable treatment parameters, non-invasive approach, and integration with other therapies. Weaknesses: Limited to specific medical applications and potential for electromagnetic interference with other devices.
SofPulse, Inc.
Technical Solution: SofPulse has developed a patented Electro-Magnetic Pulse Therapy (EMPT) technology that utilizes low-frequency pulsed electromagnetic fields to enhance biofeedback mechanisms. Their PEMF devices are designed to emit specific frequencies that resonate with the body's natural electromagnetic fields, promoting cellular repair and reducing inflammation. SofPulse's technology is particularly focused on post-operative pain management and accelerated wound healing. The company's devices are portable and can be used in both clinical and home settings, making them accessible for continuous treatment[3]. SofPulse's PEMF therapy has shown promising results in reducing the need for post-operative pain medication and improving overall recovery times[4].
Strengths: Portable and user-friendly devices, focus on post-operative care, and potential for reducing medication dependence. Weaknesses: Limited scope of application compared to broader PEMF technologies.
PEMF Biofeedback Patents
Treatment of conditions susceptible to pulsed electromagnetic field therapy
PatentActiveUS20170354830A1
Innovation
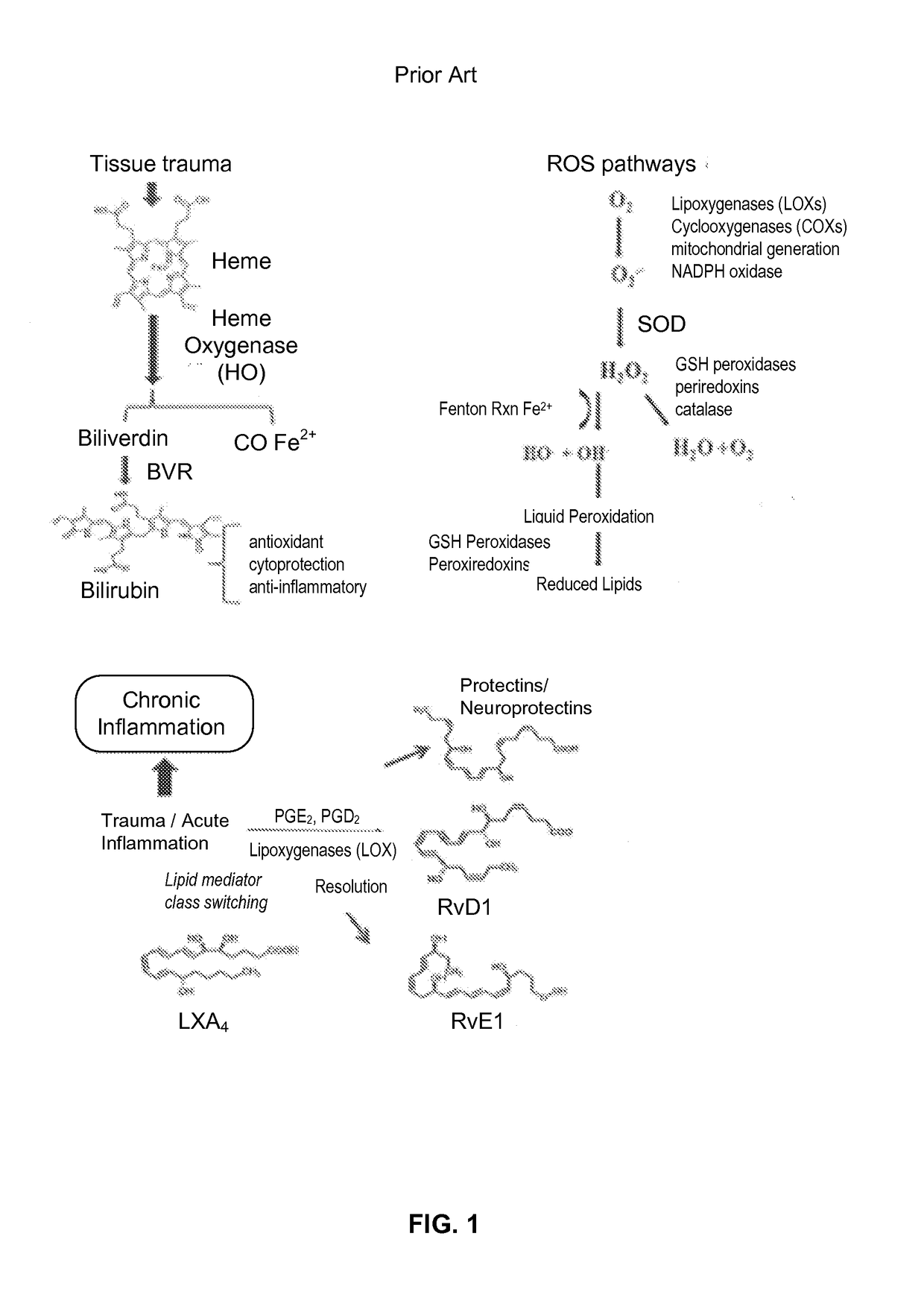
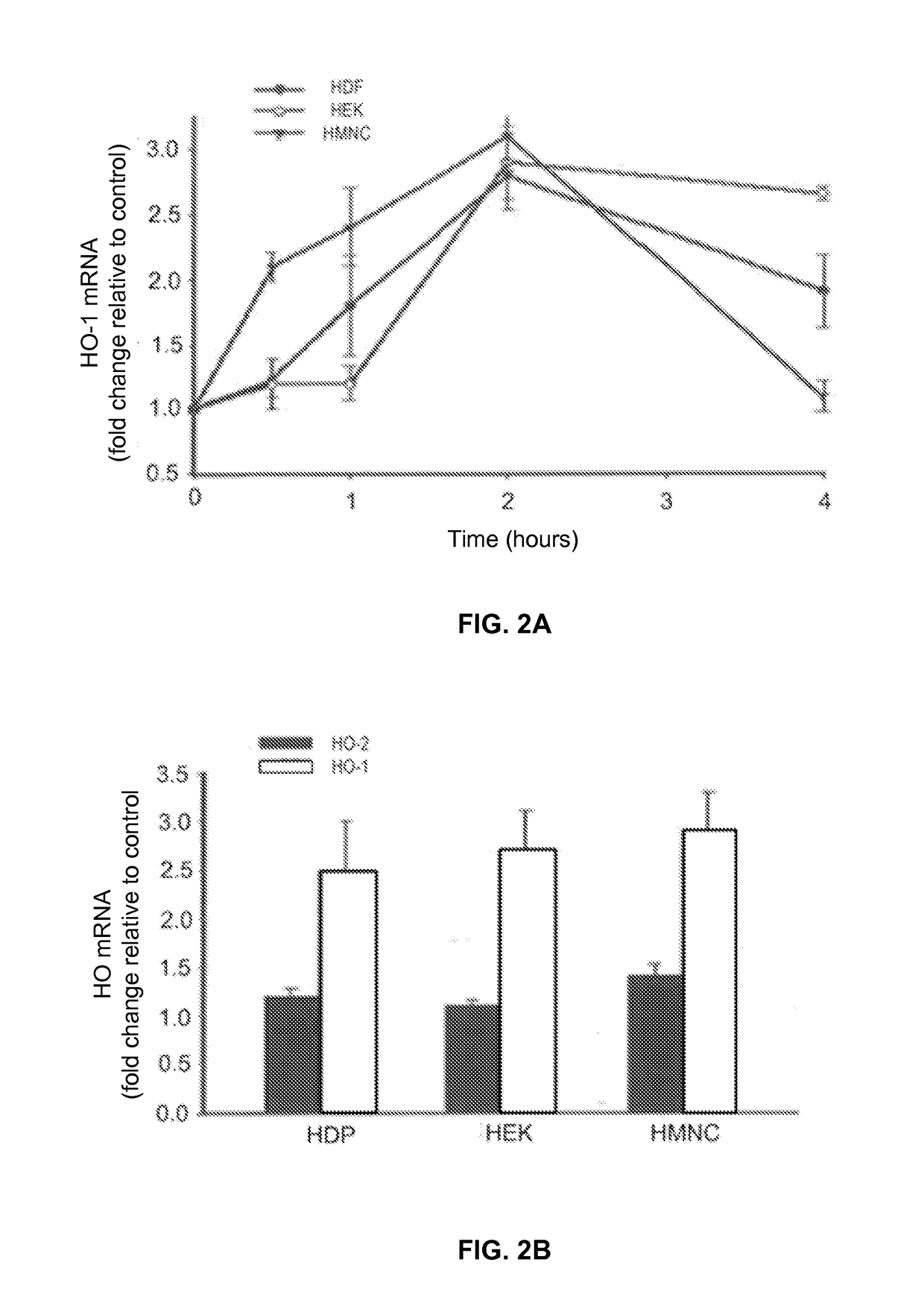
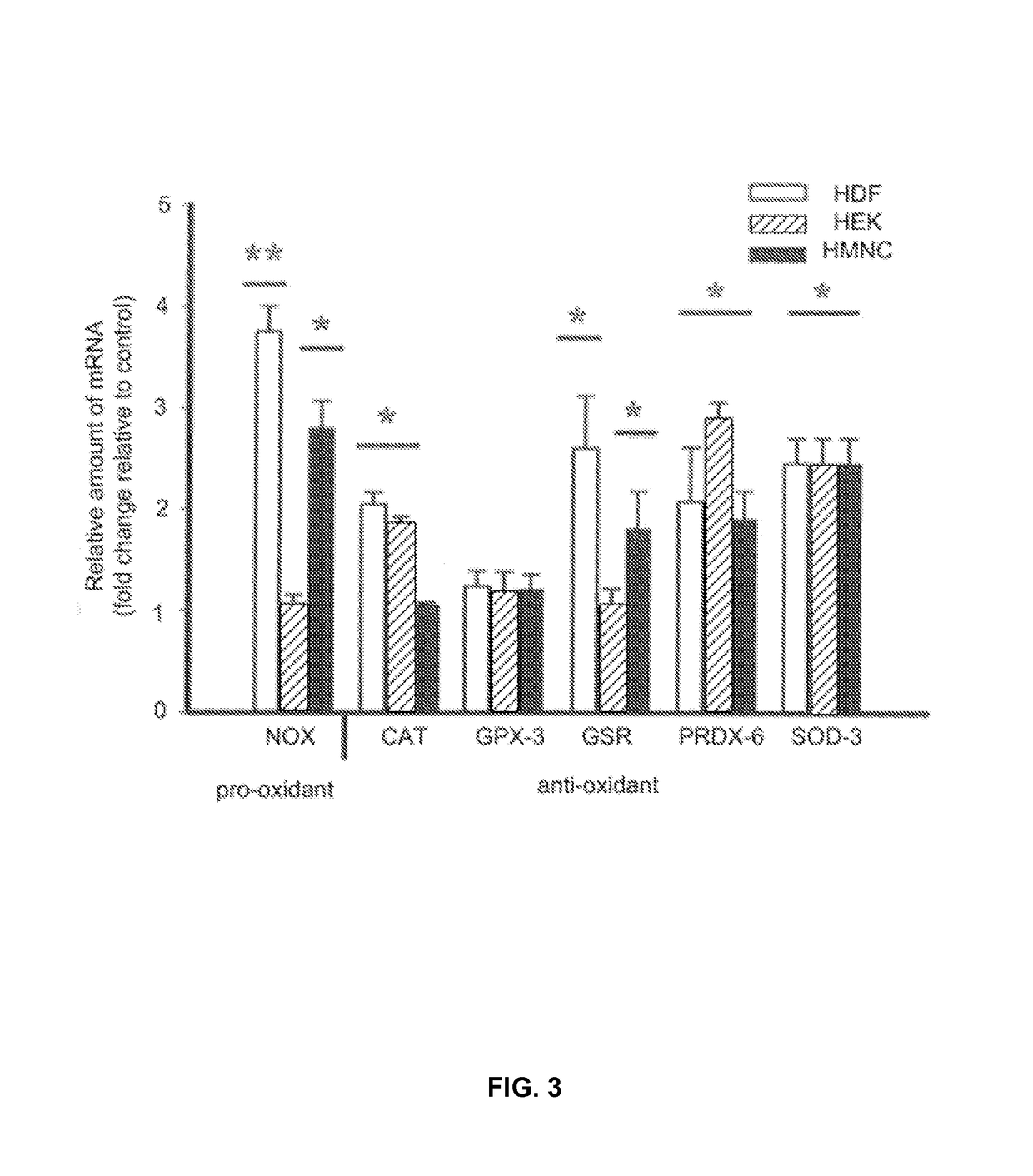
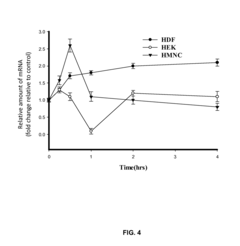
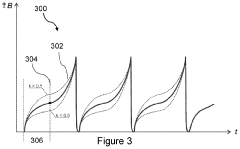
- PEMF therapy is administered to modulate gene expression associated with inflammation pathways, including heme oxygenase, antioxidant enzymes, lipid mediator biosynthesis, and cytokines, using specific parameters such as electric field strength, pulse rate, and duration to produce measurable clinical effects on pain, nerve function, and wound healing.
A pulsed electromagnetic field apparatus and method for generating frequencies
PatentWO2024127242A1
Innovation
- A PEMF apparatus with a pulse generator and electromagnetic field generation means that uses modified sawtooth waveforms with pre-stress and relaxation periods, and quasi-sine signals with pulse width modulation, along with a feedback circuit for frequency stability and precision, and a bifilar antenna for scalar wave generation.
Regulatory Framework
The regulatory framework surrounding Pulsed Electromagnetic Field (PEMF) therapy and biofeedback technologies is complex and evolving, reflecting the growing interest in these innovative therapeutic approaches. In the United States, the Food and Drug Administration (FDA) plays a crucial role in overseeing PEMF devices and biofeedback systems. PEMF devices are generally classified as Class II medical devices, requiring premarket notification (510(k)) clearance before they can be legally marketed.
The FDA has approved several PEMF devices for specific indications, such as bone healing and pain management. However, the regulatory landscape for biofeedback-enhanced PEMF therapy is still developing, as this combination represents a novel approach. Manufacturers seeking to integrate biofeedback capabilities into PEMF devices may need to navigate additional regulatory hurdles, potentially requiring clinical trials to demonstrate safety and efficacy.
In the European Union, PEMF devices and biofeedback systems fall under the Medical Device Regulation (MDR), which came into full effect in May 2021. The MDR imposes stricter requirements on manufacturers, including enhanced clinical evaluation and post-market surveillance. Devices incorporating both PEMF and biofeedback technologies may be classified in higher risk categories, necessitating more rigorous conformity assessment procedures.
Globally, regulatory bodies are grappling with the rapid advancement of medical technologies, including those combining multiple modalities like PEMF and biofeedback. Many countries are updating their regulatory frameworks to address these innovations while ensuring patient safety. For instance, Health Canada has established specific guidelines for electromagnetic therapy devices, which would encompass PEMF systems.
As research into biofeedback-enhanced PEMF therapy progresses, regulatory agencies may need to develop new guidelines or adapt existing ones to address the unique characteristics of this combined approach. This could include considerations for data privacy and security, given the potential for these devices to collect and process sensitive biometric information.
Manufacturers and researchers in this field must stay abreast of regulatory developments across different jurisdictions. Compliance with international standards, such as those set by the International Electrotechnical Commission (IEC) for medical electrical equipment, will be crucial for global market access. Additionally, as the therapeutic applications of biofeedback-enhanced PEMF therapy expand, regulatory bodies may require more extensive clinical evidence to support claims of efficacy and safety.
The regulatory landscape for this emerging technology is likely to continue evolving, with potential changes in classification, testing requirements, and approval processes. Stakeholders in the field should engage proactively with regulatory authorities to help shape appropriate guidelines that foster innovation while safeguarding public health.
The FDA has approved several PEMF devices for specific indications, such as bone healing and pain management. However, the regulatory landscape for biofeedback-enhanced PEMF therapy is still developing, as this combination represents a novel approach. Manufacturers seeking to integrate biofeedback capabilities into PEMF devices may need to navigate additional regulatory hurdles, potentially requiring clinical trials to demonstrate safety and efficacy.
In the European Union, PEMF devices and biofeedback systems fall under the Medical Device Regulation (MDR), which came into full effect in May 2021. The MDR imposes stricter requirements on manufacturers, including enhanced clinical evaluation and post-market surveillance. Devices incorporating both PEMF and biofeedback technologies may be classified in higher risk categories, necessitating more rigorous conformity assessment procedures.
Globally, regulatory bodies are grappling with the rapid advancement of medical technologies, including those combining multiple modalities like PEMF and biofeedback. Many countries are updating their regulatory frameworks to address these innovations while ensuring patient safety. For instance, Health Canada has established specific guidelines for electromagnetic therapy devices, which would encompass PEMF systems.
As research into biofeedback-enhanced PEMF therapy progresses, regulatory agencies may need to develop new guidelines or adapt existing ones to address the unique characteristics of this combined approach. This could include considerations for data privacy and security, given the potential for these devices to collect and process sensitive biometric information.
Manufacturers and researchers in this field must stay abreast of regulatory developments across different jurisdictions. Compliance with international standards, such as those set by the International Electrotechnical Commission (IEC) for medical electrical equipment, will be crucial for global market access. Additionally, as the therapeutic applications of biofeedback-enhanced PEMF therapy expand, regulatory bodies may require more extensive clinical evidence to support claims of efficacy and safety.
The regulatory landscape for this emerging technology is likely to continue evolving, with potential changes in classification, testing requirements, and approval processes. Stakeholders in the field should engage proactively with regulatory authorities to help shape appropriate guidelines that foster innovation while safeguarding public health.
Safety Considerations
Safety considerations are paramount when exploring biofeedback enhancements through Pulsed Electromagnetic Field (PEMF) therapy. While PEMF therapy has shown promising results in various medical applications, it is crucial to address potential risks and establish robust safety protocols to ensure patient well-being.
One of the primary safety concerns in PEMF therapy is the potential for electromagnetic interference with implanted medical devices, such as pacemakers or insulin pumps. Patients with such devices should undergo thorough screening and consultation with their healthcare providers before engaging in PEMF therapy. Additionally, manufacturers of PEMF devices must adhere to strict electromagnetic compatibility standards to minimize the risk of interference with other electronic equipment.
The intensity and frequency of electromagnetic fields used in PEMF therapy require careful calibration to avoid adverse effects. Excessive exposure to high-intensity fields may lead to tissue heating, which could potentially cause discomfort or, in extreme cases, tissue damage. Therefore, it is essential to establish clear guidelines for treatment duration, field strength, and frequency based on extensive clinical research and regulatory standards.
Pregnant women and individuals with certain medical conditions, such as epilepsy or active bleeding disorders, may be at higher risk for complications from PEMF therapy. Comprehensive pre-treatment assessments and individualized treatment plans are necessary to mitigate these risks. Furthermore, long-term effects of PEMF therapy on cellular and genetic structures require ongoing research to ensure the therapy's safety over extended periods.
Quality control and regular maintenance of PEMF devices are critical to maintaining safety standards. Malfunctioning equipment or improper application techniques could lead to ineffective treatment or potential harm. Implementing rigorous quality assurance processes, including regular device calibration and operator training, is essential for minimizing these risks.
As PEMF therapy integrates with biofeedback systems, the complexity of the combined technologies introduces new safety considerations. Data security and privacy become paramount, as biofeedback information is sensitive and must be protected from unauthorized access or manipulation. Robust encryption protocols and secure data storage systems are necessary to safeguard patient information.
Regulatory compliance is a crucial aspect of safety in PEMF therapy. Adhering to guidelines set by organizations such as the FDA and the International Commission on Non-Ionizing Radiation Protection (ICNIRP) ensures that PEMF devices meet established safety standards. Regular updates to these guidelines based on emerging research findings are essential to maintain the highest level of patient safety.
One of the primary safety concerns in PEMF therapy is the potential for electromagnetic interference with implanted medical devices, such as pacemakers or insulin pumps. Patients with such devices should undergo thorough screening and consultation with their healthcare providers before engaging in PEMF therapy. Additionally, manufacturers of PEMF devices must adhere to strict electromagnetic compatibility standards to minimize the risk of interference with other electronic equipment.
The intensity and frequency of electromagnetic fields used in PEMF therapy require careful calibration to avoid adverse effects. Excessive exposure to high-intensity fields may lead to tissue heating, which could potentially cause discomfort or, in extreme cases, tissue damage. Therefore, it is essential to establish clear guidelines for treatment duration, field strength, and frequency based on extensive clinical research and regulatory standards.
Pregnant women and individuals with certain medical conditions, such as epilepsy or active bleeding disorders, may be at higher risk for complications from PEMF therapy. Comprehensive pre-treatment assessments and individualized treatment plans are necessary to mitigate these risks. Furthermore, long-term effects of PEMF therapy on cellular and genetic structures require ongoing research to ensure the therapy's safety over extended periods.
Quality control and regular maintenance of PEMF devices are critical to maintaining safety standards. Malfunctioning equipment or improper application techniques could lead to ineffective treatment or potential harm. Implementing rigorous quality assurance processes, including regular device calibration and operator training, is essential for minimizing these risks.
As PEMF therapy integrates with biofeedback systems, the complexity of the combined technologies introduces new safety considerations. Data security and privacy become paramount, as biofeedback information is sensitive and must be protected from unauthorized access or manipulation. Robust encryption protocols and secure data storage systems are necessary to safeguard patient information.
Regulatory compliance is a crucial aspect of safety in PEMF therapy. Adhering to guidelines set by organizations such as the FDA and the International Commission on Non-Ionizing Radiation Protection (ICNIRP) ensures that PEMF devices meet established safety standards. Regular updates to these guidelines based on emerging research findings are essential to maintain the highest level of patient safety.
Unlock deeper insights with PatSnap Eureka Quick Research — get a full tech report to explore trends and direct your research. Try now!
Generate Your Research Report Instantly with AI Agent
Supercharge your innovation with PatSnap Eureka AI Agent Platform!